Abstract
Purpose
Patients with spinal muscular atrophy (SMA) are often treated with growth friendly devices such as vertical expandable prosthetic titanium rib(VEPTR) and magnetically controlled growing rods(MCGR) to correct spinal deformity and improve pulmonary function. There is limited data on this topic, and the purpose of this study was to assess the effect of these constructs and the addition of chest wall support (CWS) on spinal deformity, thorax morphology and pulmonary outcomes.
Methods
This is a retrospective analysis of prospectively collected data. We included patients with chest wall deformity and scoliosis secondary to SMA who were treated with growth friendly interventions and had two-year follow-up. Descriptive statistics and univariate analyses were performed.
Results
This study included 66 patients (25% MCGR, 73% VEPTR, 2% unknown). Approximately 23% of constructs included CWS. The average Cobb angle improved from 67° (SD: 27°) to 50° (SD: 26°) at 2 years in patients with CWS (p = 0.02), and from 59° (SD: 20°) to 46° (SD: 15°) at 2 years in patients without CWS (p < 0.01). Hemithorax height improved in patients treated with and without CWS (p = 0.01), but hemithorax width only improved in patients with CWS (p = 0.01). One patient with CWS and two patients without CWS required additional respiratory support at 2 years. The rates of postoperative complications were not significantly different in patients treated with and without CWS (p = 0.31).
Conclusions
Growth friendly constructs improve spinal deformity and may be effective in altering the progression toward respiratory failure in patients with SMA. Patients treated with CWS have significant improvements in thorax morphology compared to patients without CWS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spinal muscular atrophy (SMA) is an autosomal recessive genetic disorder caused by a deletion of the SMN1 gene [1]. The incidence of SMA has been noted to be approximately 10 in 100,000 live births, and its prevalence is approximately 1 to 2 per 100,000 persons [2]. There are four clinical subtypes based on age of onset, achievement of motor milestones and prognosis with type 1 being the most severe and associated with a significantly shortened life expectancy and type 4 being the mildest form and associated with a normal life expectancy [3]. In general, SMA is characterized by loss of anterior horn cells in the spinal cord, and orthopaedic manifestations include hip subluxation and scoliosis [1, 4]. Scoliosis can significantly affect respiratory mechanics, and severe cases are associated with significant morbidity and pulmonary failure [5]. Additionally, thoracic insufficiency can result from primary rib collapse not exclusively due to spinal rotation, which Campbell and Smith termed “collapsing parasol deformity” [6, 7]. Historically, intermediate and severe cases of SMA have resulted in poor outcomes and eventually death, but modern advances in medical management have allowed patients to live longer [8, 9]. For example, a disease-modifying treatment named nusinersen has been shown to halt disease progression and improve motor function, and other gene-replacing therapies are being tested [10]. As a result, more patients are requiring management of their orthopaedic issues.
Management of scoliosis in patients with SMA is integral in maintaining balance and preventing pulmonary failure. Spinal fusion has been found to result in maintained pulmonary function at long-term follow-up [11, 12]. However, definitive spinal fusion may be detrimental in young children with significant potential for growth and pulmonary development. Growth friendly interventions have recently being shown to control spinal deformity and allow lung volumes to increase over time in patients with SMA [13]. Magnetically controlled growing rods (MCGR), traditional growing rods and vertical expandable prosthetic titanium rib (VEPTR) devices have been shown to be effective in stabilizing and in some cases, decreasing the need for pulmonary support in patients with SMA [13,14,15,16]. However, previous studies have been limited by small sample sizes and short-term follow-up.
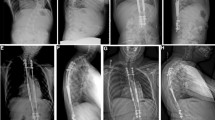
Additionally, lateral chest wall support (CWS) has also been shown to be effective in patients with thoracic insufficiency syndrome (TIS) since it allows the rib cage to expand and address the collapsing parasol deformity (Fig. 1) [6]. This construct allows for lateral expansion of the thorax as well as direct distraction over time with the use of transverse bars and rib-cradles [17]. It has been suggested that the CWS will be effective in patients with SMA, but it has not been specifically studied in this population. In this study, we aimed to assess the effect of growth friendly constructs and CWS on spinal deformity correction and pulmonary function in patients with SMA.
Methods
We performed a retrospective study of prospectively collected data through the Pediatric Spine Study Group (PSSG). PSSG is a multicenter study group that focuses on early-onset scoliosis (EOS) and pediatric spinal deformity. In our analysis, we included patients with a diagnosis of SMA treated with growth friendly constructs and had a minimum of 2-year postoperative follow-up. The PSSG review board and research committee, as well as the IRB at each institution, approved this study.
Data collection
Data were collected by participating surgeons and surgical sites. Available data included patient demographics, surgical details, baseline respiratory and radiographic data and 2-year respiratory and radiographic data. Respiratory data included respiratory support information and pulmonary function testing data. Types of respiratory support include full-time and part-time ventilator assistance and CPAP/BiPAP. Spinal deformity data included major Cobb angles and measures of hemithorax height and width. All measurements were standardized and provided by the study group.
Statistical analysis
Descriptive statistics were used to summarize patient demographics, respiratory data (assisted ventilation rating, AVR) and radiographic measures. Paired t-test was used to compare differences in baseline and 2-year Cobb angles and hemithorax size. Student’s t-test was used to compare differences in continuous variables between groups, and Z test was used to compare proportions. All analyses were performed in Microsoft Excel (v14, 2010).
Results
This study included 66 patients with SMA. Patients were treated at 15 centers by 16 surgeons. Approximately, 45% of patients were female, 55% of patients were male, and the majority of patients were Caucasian (80%). Most patients had SMA type II (68%) followed by SMA type I (21%) and SMA type III (5%). Approximately 42% of patients were treated with nusinersen over the course of enrollment. The average age at time of index surgery was 7.3 years (range: 1.9–12.8 years). Approximately 50% of cases had initial implantation between the ages of 6 and 8 inclusive. At baseline, 47% of patients required no respiratory assistance while 53% of patients required part-time or full-time respiratory assistance (n = 51) (Table 1).
In total, 26% of patients were treated with a MCGR (n = 17) and 71% of patients were treated with a VEPTR device (n = 47). MCGRs were used starting in 2013, and since that time, 61% of patients were treated with a MCGR (n = 17) and 39% of patients were treated with a VEPTR device (n = 11). Approximately 95% of cases were treated with bilateral devices, and most patients had proximal rib fixation (78%) and distal pelvic fixation (74%). The average major Cobb angle before treatment was 61° (SD: 22°, n = 63) and 47° (SD: 18°, n = 50) at two-year follow-up (p < 0.01). All curves were thoracic or lumbar in location. Approximately, 45% of patients required no respiratory assistance and 55% of patients required part-time or full-time respiratory assistance at two-year follow-up (n = 33). Specifically, 15 patients required ventilator or supplemental oxygen, and 3 patients required BiPAP/CPAP. Only one patient that required no respiratory support at baseline needed respiratory support at follow-up, and three patients in total required higher levels of respiratory support at 2-year follow-up (3/32 patients). Complete preoperative and postoperative pulmonary function data were available for 11 patients. The percentage of predicted FVC decreased for all patients by approximately 4% each year and approximately 24% over an average of 5 years (Range: 3–12 years).
Lateral chest wall support
In this cohort, 15 patients were treated with CWS (23%) and 51 patients were treated without CWS (77%). The choice of treatment was determined by the treating surgeon. Hemithorax height improved in patients treated with and without CWS (p = 0.01), but hemithorax width only improved in patients with lateral chest wall support (p = 0.01) (Table 2). There was no difference in improvement in Cobb angle at 2-year follow-up between patients with and without CWS (19.7° vs. 15°, p = 0.67). One patient with CWS and two patients without CWS required additional respiratory support at two-year follow-up. Predicted FVC decreased by 24% in patients with CWS (n = 2) and 26% in patients without CWS (n = 9) (p = 0.83).
Complications
The overall complication rate during treatment was 38% (Table 3). More specifically, the complication rate was 24% in patients treated with magnetically controlled growing rods and 45% in patients treated with a VEPTR device (p = 0.13), and several patients had multiple complications. The complication rate was 27% in patients with CWS and 41% in patients without CWS (p = 0.31). The average time from index surgery to complication was approximately 1 year (SD: 1.7 years). The most common complication in either group was surgical site infection (20% in patients with CWS vs. 25% in patients without CWS). Superficial and deep infections were grouped together as surgical site infection in this database.
Discussion
SMA has traditionally been associated with a poor prognosis; however, recent medical advances have slowed disease progression and improved patient function [8,9,10]. Scoliosis, collapsing parasol deformity of the thorax and worsening pulmonary function are usually associated with SMA, but growth friendly interventions are being used to control spinal deformity and slow the decline of pulmonary function in these patients. In this study, we found that growth friendly interventions are effective in improving and sustaining spinal deformity in patients with SMA at 2-year follow-up. Additionally, very few patients require additional respiratory support at follow-up even though respiratory function continues to deteriorate with time. Spinal deformity improves with and without CWS, but CWS may be more effective in improving thoracic morphology and preventing parasol rib deformity.
Previous studies have assessed correction of spinal deformity and pulmonary function with the use of growth friendly constructs in patients with SMA [11,12,13,14,15, 18,19,20]. However, these have been limited by small sample sizes, focused on different diagnoses, and none of these studies have compared different growth friendly devices or the use of CWS. Several studies have noted an improvement in spinal deformity with the use of growth friendly interventions in patients with SMA, and our results support these previous findings. For example, Lenhart et al. found an improvement in curve magnitude with the use of growing rods in patients with SMA, but their study was limited due to variable follow-up [13]. Similarly, McElroy et al. noted improvement in curve magnitude with growing rod constructs, but their study included only 15 patients with SMA [19]; and Colombo et al. reported an improvement in curve magnitude with MCGR, but this study was limited to four patients with SMA and included patients with other neuromuscular diagnoses [15].
Previous studies have also studied the effect of growth friendly constructs on PFT and respiratory support, and the results have been variable. For example, Colombo et al. noted no significant difference in PFTs through 2 years in patients with SMA [15]. In comparison, Lenhart et al. found an improvement in thorax height and width; no changes in need for respiratory support; and diminished predicted FVC over time in SMA patients [13]. However, PFT data were only available for six children in this previous study. Similar to the findings of Lenhart et al., we found a decrease in predicted FVC over time, but a significant improvement in thorax width was only noted in patients with CWS. Other studies have focused on PFTs in patients with SMA, but these parameters have been studied in the setting of definitive spinal fusion [11, 12]. To our knowledge, no studies have determined the effect of VEPTR devices or CWS on spinal deformity and pulmonary function specifically in patients with SMA. Livingston et al. reported the effects of VEPTR device on parasol rib deformity in a range of patients with hypotonic neuromuscular scoliosis and found no significant improvements [21].
In this study, approximately 40% of patients had a postoperative complication with surgical site infection being the most common complication. Additionally, we did not find any statistically significant difference in complications with MCGR or VEPTR devices, but our study may be underpowered for this comparison. Complications with growth friendly interventions have been widely reported in the literature, and complication rates of up to 84% have been reported in patients with early-onset scoliosis. In patients with SMA, Chandran et al. reported no surgical complications or unplanned return to surgery in 11 patients treated with growing rods [16]. However, Lorenz et al. noted complications in approximately 4 out of 21 patients (20%) with SMA that were treated with MCGR [20], which is similar to the rate of complications in patients managed with MCGRs in our study (24%). In addition, previous studies have noted issues with wound healing in patients with SMA, which was the most common complication noted in our study as well, and it is an issue with constructs that need repeat surgeries for lengthening [22].
This study has several strengths. To our knowledge, this is the largest study focusing on the effect of growth friendly constructs in patients with SMA. Additionally, we have been able to assess the effect of other growth friendly devices such as the VEPTR and the use of CWS on radiographic and thorax anatomy. We have also included data from multiple centers and populations, which improves the generalizability of our findings. However, variability in practice may also affect the results of this study and lead to potential biases.
On the other hand, limitations of this study include incomplete follow-up, which subjects our findings to selection bias. Specifically, radiographic follow-up was available for 76% of patients but AVR data were available for 50% of patients and PFT data were available for only 17% of patients. Additionally, we are unable to comment on the effects of growth friendly interventions and CWS on ribs morphology and orientation or the clinical implications of improving thoracic volume [21]. Our findings are limited to radiographic measurements of the thorax (height and width) and pulmonary outcomes measured by AVR and PFTs. Additional studies are needed to describe and quantify parasol rib deformity and define the effects of growth friendly interventions and CWS on pulmonary function. Larger, long-term, multi-center studies are needed to compare pulmonary outcomes and postoperative complications in patients with SMA, as well as determine any differences between ambulatory and non-ambulatory patients with SMA or patients being treated with or without disease-modifying agents such as nusinersen. A post-hoc power analysis revealed that this study had only 30% power to assess differences in complications between patients managed with MCGR and VEPTR devices, and additional studies are needed to assess the complication profiles for these devices and CWS.
In conclusion, growth friendly constructs such as MCGR and VEPTR device have the ability to improve and control spinal deformity in patients with SMA. These devices seem to maintain the need for respiratory support, and despite decreases in pulmonary function over time, they may decrease the progression toward respiratory failure in patients with SMA. Patients treated with CWS have significantly better outcomes in thorax width without increased complications, and additional study is needed to determine the effects of this improvement on pulmonary function and rib deformity. In conjunction with medical management, patients with SMA and spinal deformity may be managed with growth friendly constructs to improve their spinal deformity, maintain pulmonary function and slow respiratory decline.
Availability of data and material
The authors grant permission to reproduce copyrighted materials or signed patient consent forms IRB approval.
References
Mesfin A, Sponseller PD, Leet AI (2012) Spinal muscular atrophy: manifestations and management. JAAOS J Am Acad Orthop Surg 20(6):393–401
Verhaart IEC, Robertson A, Wilson IJ, Aartsma-Rus A, Cameron S, Jones CC, Cook SF, Lochmüller H (2017) Prevalence, incidence and carrier frequency of 5q-linked spinal muscular atrophy—a literature review. Orphanet J Rare Dis 12(1):124. https://doi.org/10.1186/s13023-017-0671-8
CADTH Common Drug Reviews (2018). In: Clinical review report: Nusinersen (Spinraza): (Biogen Canada Inc.): indication: treatment of patients with 5q SMA. Canadian agency for drugs and technologies in health copyright© 2018. Canadian agency for drugs and technologies in health, Ottawa (ON)
Granata C, Merlini L, Magni E, Marini ML, Stagni SB (1989) Spinal muscular atrophy: natural history and orthopaedic treatment of scoliosis. Spine 14(7):760–762
Mayer OH (2015) Scoliosis and the impact in neuromuscular disease. Paediatr Respir Rev 16(1):35–42
Campbell RM (2013) VEPTR: past experience and the future of VEPTR principles. Eur Spine J 22(2):106–117
Campbell RM Jr, Smith MD (2007) Thoracic insufficiency syndrome and exotic scoliosis. JBJS 89:108–122
Finkel RS, Mercuri E, Darras BT, Connolly AM, Kuntz NL, Kirschner J, Chiriboga CA, Saito K, Servais L, Tizzano E (2017) Nusinersen versus sham control in infantile-onset spinal muscular atrophy. N Engl J Med 377(18):1723–1732
Mendell JR, Al-Zaidy S, Shell R, Arnold WD, Rodino-Klapac LR, Prior TW, Lowes L, Alfano L, Berry K, Church K (2017) Single-dose gene-replacement therapy for spinal muscular atrophy. N Engl J Med 377(18):1713–1722
Wurster CD, Ludolph AC (2018) Nusinersen for spinal muscular atrophy. Ther Adv Neurol Disord. https://doi.org/10.1177/1756285618754459
Chou S-H, Lin G-T, Shen P-C, Lue Y-J, Lu C-C, Tien Y-C, Lu Y-M (2017) The effect of scoliosis surgery on pulmonary function in spinal muscular atrophy type II patients. Eur Spine J 26(6):1721–1731
Chua K, Tan CY, Chen Z, Wong HK, Lee EH, Tay SK, Ong HT, Goh DY, Hui JH (2016) Long-term follow-up of pulmonary function and scoliosis in patients with Duchenne’s muscular dystrophy and spinal muscular atrophy. J Pediatr Orthop 36(1):63–69
Lenhart RL, Youlo S, Schroth MK, Noonan KJ, McCarthy J, Mann D, Hetzel S, Sund SA, Halanski MA (2017) Radiographic and respiratory effects of growing rods in children with spinal muscular atrophy. J Pediatr Orthop 37(8):e500–e504
Nossov SB, Curatolo E, Campbell RM, Mayer OH, Garg S (2019) VEPTR: Are we reducing respiratory assistance requirements? J Pediatr Orthop 39(1):28–32
Colombo L, Martini C, Bersanini C, Izzo F, Villafañe JH, Berjano P, Lamartina C (2017) Effects of magnetically controlled growing rods surgery on pulmonary function in young subjects with spinal muscular atrophy type 2 and other neuromuscular scoliosis. J Neurosurg Sci 64(3):253–257
Chandran S, McCarthy J, Noonan K, Mann D, Nemeth B, Guiliani T (2011) Early treatment of scoliosis with growing rods in children with severe spinal muscular atrophy: a preliminary report. J Pediatr Orthop 31(4):450–454
Campbell RM (2016) VEPTR expansion thoracoplasty. The growing spine. Springer, Berlin, pp 669–690
Modi HN, Suh S-W, Hong J-Y, Park Y-H, Yang J-H (2011) Surgical correction of paralytic neuromuscular scoliosis with poor pulmonary functions. Clin Spine Surg 24(5):325–333
McElroy MJ, Shaner AC, Crawford TO, Thompson GH, Kadakia RV, Akbarnia BA, Skaggs DL, Emans JB, Sponseller PD (2011) Growing rods for scoliosis in spinal muscular atrophy: structural effects, complications, and hospital stays. Spine 36(16):1305–1311
Lorenz HM, Badwan B, Hecker MM, Tsaknakis K, Groenefeld K, Braunschweig L, Hell AK (2017) Magnetically controlled devices parallel to the spine in children with spinal muscular atrophy. JBJS Open Access 2(4):e0036
Livingston K, Zurakowski D, Snyder B (2015) Parasol rib deformity in hypotonic neuromuscular scoliosis. Spine 40(13):E780–E786
Burow M, Forst R, Forst J, Hofner B, Fujak A (2017) Perioperative complications of scoliosis surgery in patients with Duchenne muscular dystrophy and spinal muscular atrophy, focussing on wound healing disorders. Int J Neurosci 127(6):479–485
Funding
The authors did not receive funding for this study.
Author information
Authors and Affiliations
Consortia
Contributions
IS: study conceptualization and design, data gathering, statistical analysis, primary manuscript preparation, final manuscript preparation/approval, accountability for all aspects of submitted work, EMM: data gathering, data analysis, manuscript preparation, final manuscript preparation/approval, accountability for all aspects of submitted work, OHM: study conceptualization and design, final manuscript preparation/approval, accountability for all aspects of submitted work, RKL: study conceptualization and design, final manuscript preparation/approval, accountability for all aspects of submitted work, JTS: study conceptualization and design, final manuscript preparation/approval, accountability for all aspects of submitted work, MGV: study conceptualization and design, final manuscript preparation/approval, accountability for all aspects of submitted work, JMF: study conceptualization and design, final manuscript preparation/approval, accountability for all aspects of submitted work, JBA: study conceptualization and design, final manuscript preparation/approval, accountability for all aspects of submitted work, PSSG: study conceptualization and design, data gathering, final manuscript preparation/approval, PJC: final manuscript preparation/approval, study conceptualization and design, development of research methodologies, study guidance, accountability for all aspects of submitted work.
Corresponding author
Ethics declarations
Conflict of interest
The authors of this work have no disclosures related to this study.
Ethics approval
This study was given Ethical Approval by the Institutional Review Board.
Consent to participate and publication
Informed consent was obtained.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Swarup, I., MacAlpine, E.M., Mayer, O.H. et al. Impact of growth friendly interventions on spine and pulmonary outcomes of patients with spinal muscular atrophy. Eur Spine J 30, 768–774 (2021). https://doi.org/10.1007/s00586-020-06564-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-020-06564-8