Abstract
Purpose
Spinal teratomas are rare tumours noted in adults and are commonly located in the thoracolumbar region. Currently, there appears to be a lack of clear consensus regarding the management and prognosis of these lesions. A comprehensive review along with an illustrative case managed at the author’s institute has been presented.
Materials and methods
Online database search was performed for literature review.
Results
A 26-year-old male presented with acute onset neurological deficits and imaging revealed features of conus teratoma. Subtotal resection was performed and patient achieved fair recovery. Including the present one, a total of 146 cases have been reported and were analysed. Mean age was 39.6 years (range 18–85 years) and males predominated. Limb weakness and backache were the commonest symptoms. Majority of the lesions were intramedullary and located in the thoracolumbar region. Complete resection (CR) was achieved in 45% of cases. All, except one, were mature teratomas. Recurrences were noted in nine (6.1%) cases. Outcome was good/excellent in 86 (60%) and fair/poor in 26 (18%) cases. The presence of pain, absence of limb weakness and CR were significantly associated with good outcomes. Furthermore, subtotal resection (STR) had significantly higher recurrence rates than CR. Four deaths (2.7%) were noted.
Conclusions
Total surgical resection is the standard treatment and appears to be beneficial both in terms of outcomes and recurrences. Overall, recurrences are rare and may be managed by re-surgery. In addition, the present report is the eighth case of adult spinal teratoma with an acute onset presentation.
Graphic abstract
These slides can be retrieved under Electronic Supplementary Material.

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spinal teratomas (ST) are rare tumours, constituting less than 1% of central nervous system tumours [1,2,3]. They are mostly noted in the paediatric age group and most commonly in sacrococcygeal spine [1, 2]. However, the thoracolumbar (TL) region is the most frequent location in adults and spinal teratomas presenting in adults are a rare entity. Till date, less than 150 cases of adult spinal teratomas have been reported in the literature [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100]. The most common symptoms include pain, progressive limb weakness and bowel/bladder disturbances. An acute onset presentation has been rarely described in earlier reports [22, 28, 32, 33, 40, 82, 98]. In the present report, a rare case of a mature cystic teratoma of the conus–cauda region in a 26-year-old male is described. The patient presented with an acute onset paraplegia and bowel/bladder dysfunction and was managed by surgical resection. In addition, a thorough literature review has been presented on this topic of adult spinal teratomas reported in the world literature till date.
Materials and methods
All previous published reports (case reports, letters to editor, review articles and original articles) of adult onset spinal teratomas (with no language restriction) were retrieved and analysed in the current paper. Adult onset was defined as age ≥ 18 years. The first case that was reported in the year 1888 and all the cases that have reported till the present date were included in the analysis. Cases in which no information could be gathered (because of language barriers or missing data) were excluded. Sacrococcygeal teratomas and paediatric onset teratomas were excluded from the analyses. The following keywords were searched using online databases (Pubmed, Index Scopus, EMBASE, Google Scholar): ‘teratoma’, ‘spinal’, ‘adult’, ‘spinal teratoma’, ‘intramedullary’, ‘intradural’, ‘extradural’, ‘mature teratoma’, ‘cystic teratoma’ and ‘review’. Hospital records were retrieved for our case report. The following characteristics were analysed: demographics (age, gender), presenting features, duration of symptoms, spinal level of the lesion (cervical/thoracic/thoracolumbar/lumbar/lumbosacral/sacral), axial level of the lesion [extradural (ED)/extramedullary (EM)/intramedullary (IM)], the presence of associated spinal anomalies, radiological and histopathological features, type of surgical procedures, completeness of resection, recurrences, mean time to recurrence (TTR) and outcomes at last follow-up (FU). Acute/rapid progression of deficits was taken as those occurring within 2 weeks of presentation. Outcome was considered good/excellent if functional or complete motor recovery was achieved, while outcome was considered fair/poor when the motor recovery was non-functional or there was non-improvement of symptoms or in cases of neurological deterioration after surgery. Chi-square test was applied to predict the correlation of the above-mentioned characteristics with outcomes and recurrences. A p value of < 0.05 was taken as significant.
Results
Case illustration
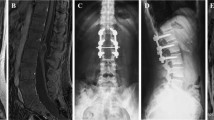
A 26-year-old male presented with acute onset weakness (over 48 h) of both lower limbs 1½-months back, associated with bladder and bowel dysfunction. Neurological examination revealed flaccidity in both lower limbs and complete paraplegia (MRC grade 0/5). Lower limb deep reflexes were non-elicitable. There were absent and reduced tactile sensations, respectively, on the left and right lower limbs below the L1 dermatomes. No cutaneous stigmata of spinal dysraphism were evident on examination. Magnetic resonance imaging (MRI) of the spine showed a heterogeneous lesion consisting of 2 components (cranial and caudal) extending from L1 to L2 vertebrae at the level of conus–cauda region. There was no evidence of associated spinal anomalies. The cranial part was intramedullary and appeared hyperintense on T1 and profoundly hypointense on T2 sequences. The caudal part was extramedullary and was isointense and hyperintense on T1 and T2 sequences, respectively. On fat-saturated sequences, the cranial component showed suppression, suggestive of a fat containing lesion, while both the components showed mild enhancement on contrast administration (Fig. 1a–f). Radiological diagnosis was a mature cystic teratoma. A L1-L2 laminectomy and tumour resection were performed. Intraoperatively, the spinous elements were normal. Caudally, an EM cystic tumour was noted attached to the conus containing calcium deposits and straw-coloured fluid. The cyst could be easily separated from the cauda equina roots and was totally removed. There was no breach in the pia over the nerve roots. The cranial part was intramedullary and contained predominantly fat and blood vessels. An ultrasonic aspirator was used for debulking the lesion. Since it was relatively difficult to differentiate from the conus, subtotal resection was performed. Intraoperative electrophysiological studies were not available and hence were not utilized in this case. Later, a watertight dural closure was performed. Post-operatively, patient had improvement in his lower limb tone and could appreciate tactile sensations from around the second week of surgery and also had mild improvement in his weakness (wiggle his toes and hold his limbs on the bed when kept in a flexed posture) when discharged from hospital. Histopathological examination showed pseudostratified ciliated columnar epithelium overlying fibrocollagenous stroma containing sebaceous glands, smooth muscle, nerve fibres, lobules of adipocytes and congested vessels—overall features suggestive of a mature teratoma. At 15-month follow-up, he had modest improvement in his power and was able to stand with support, but was mostly dependent on others for his daily activities. However, his urinary problem did not recover and was on a catheter for voiding that was changed to clean intermittent catheterization (CIC) few months later. A follow-up MRI could have been helpful in determining the extent of resection, but could not be done because of non-willingness of the patient.
Pre-operative MRI of lumbosarcal spine showing the heterogeneously appearing conus–cauda lesion with cranial and caudal components. Sagittal and axial T1 WI (a, b), sagittal and axial T2 WI (c, d) and STIR images (e) showing the cranial component as hyperintense on T1, hypointense on T2 WI and suppressing on STIR images, while the caudal cystic part is hypointense and hyperintense on T1 and T2 sequences and does not show fat suppression. The entire lesion is showing heterogeneous enhancement (f). The cranial part is intramedullary (arrow in b), and caudal one is extramedullary (long arrow in d). The cord is seen to be compressed and displaced anteriorly (short arrow in d)
Including ours, a total of 146 cases of adult spinal teratomas have been reported till the present date and have been analysed in this present report. The mean age of the cohort was 39.6 years (range 18–85 years), and the median age was 36 years. The male/female ratio was 1.33:1. The mean duration of symptoms was 46.7 months (3 days to 20 years). Limb weakness and pain were seen in 76 subjects, bladder/bowel disturbances in 61 subjects and meningitic symptoms in three cases. Of those presenting with pain, axial pain (neck pain/backache) was seen in 47 cases and radiculopathy in 24 cases and site of pain was not mentioned for remaining 18 cases. History of minor trauma was reported in two cases. Acute/rapidly progressive symptoms were noted in eight cases (including ours) [22, 28, 32, 33, 40, 82, 98]. Details of clinical features were not available (NA) for a total of 28 cases.
With regard to the spinal level, the locations of the tumour were as follows: cervical: n = 15, thoracic: n = 15, thoracolumbar: n = 72, lumbar: n = 38, lumbosacral: n = 6 and NA: n = 6 cases. With regard to the axial location, the teratoma was located as follows: IM: n = 69, EM: n = 60, combined IM + EM: n = 8, ED: n = 4 and NA: n = 5 cases.
Associated cutaneous abnormalities and spinal anomalies such as spina bifida, split cord malformations and scoliosis were present in 55 cases, absent in 84 cases and details NA for 6 cases. Of those, SCM-1 and SCM-2 were seen in 7 cases each and were noted in all levels, except the cervical spine. Complete resection (CR) was achieved in 65 (45%) cases, while 54 (37%) cases underwent subtotal resection (STR). Details of resection were NA for 26 (18%) cases. In intramedullary teratomas, CR was achieved in 37.6% (n = 26), STR in 46.3% (n = 32) and details NA in 16% (n = 11) cases. All, except one, were mature teratomas on histopathological examinations.
The mean FU duration was 37 months (2 weeks–208 months). Nine recurrences were noted, and the overall recurrence rate was 6.1%. One patient had six recurrences in 6 years [39]. Overall, the mean time to recurrence (TTR) was 88.2 months (4–180 months). With the exclusion of the single case of immature teratoma that recurred at 4 months, the mean TTR for mature teratomas was 98.8 months (12–180 months). With regard to the extent of surgical resection, there were one and five recurrences, noted for CR and STR, respectively. On statistical analysis, this difference was significant (p = 0.029). Details of surgical resection were not reported (NR) for the remaining three recurred cases. No malignant changes were evident in the recurred cases.
Outcomes at last FU were as follows: good/excellent in 86 (60%) cases, fair/poor outcome in 26 (18%) cases and NA for 33 (22%) cases. Four deaths (2.7%) were reported, and one case was diagnosed at autopsy. Of the four deaths, two cases each were in the cervical and conus regions. Three cases were IM in location, and the reported case of teratoma with CSF leak/meningitis was EM. The causes of death were intracranial fat dissemination in two cases, CSF leak/meningitis and respiratory failure in one case each. On applying Chi-square test, the presence of pain (p = 0.019), absence of limb weakness (p = 0.001) and CR (p = 0.015) were significantly associated with good outcomes. Also, EM tumours had higher rates of CR as compared to IM tumours (p = 0.025). The remaining variables such as age (< 40 years and > 40 years, mean age being 39.6 years), duration of symptoms (< 46 months, > 46 months, mean duration being 46.7 months), gender, bowel/bladder involvement, location of tumour and associated anomalies did not have a significant correlation with outcomes or recurrence rates on statistical analyses. Table 1 summarizes all the reported cases of adult onset spinal teratomas.
Discussion
Epidemiology and clinical features
In general terms, teratomas are classically defined as neoplasms consisting of multipotential cells of all the three germ cell layers (according to Russell and Rubinstein) or as neoplasms with the power of autonomous growth (according to Wills) [1, 38, 78, 82, 101,102,103]. Spinal teratoma is an entirely separate entity from that of the intracranial and sacrococcygeal teratomas [39]. Excluding sacrococcygeal teratomas that are predominantly found in neonates and young children, STs are very rare tumours [1, 2]. They account for only 0.2–0.5% of all spinal cord tumours, and only 2% of all teratomas are found to occur in the neuraxis as reported in various studies [2, 3, 48, 72, 73, 104]. Sloof and Kernohan [15] noted only two cases of teratomas in their cohort of 1322 intraspinal tumours. In one previous report of 256 spinal tumours by Tapper and Lack [105], only four cases were teratomas. Sawamura [106] analysed 34 cases of CNS teratomas, and none were intraspinal.
Gowers and Horsley [4] described the first case of a ST in 1888. Majority of the STs are located in the TL region involving the conus, while thoracic and cervical ones are relatively uncommon [1, 48]. Adult onset presentation is rare, and only 145 cases have been reported till date [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100]. Among adults, STs are most frequently seen in the 4–5th decades. They are more common in males; however, there appears to be a female preponderance in children [1, 40]. Spinal teratomas may remain asymptomatic or may present with non-specific features similar to that of any spinal mass lesion [1, 40, 54]. The most common presenting features include back pain, limb weakness and bowel/bladder disturbances. Except eight cases, all cases have reported a gradual progression of symptoms with the mean symptom duration being 46.7 months. There may be periods of remissions and exacerbations in the symptomatology, as noted by few authors [40]. However, in this present review, none of the cases have reported this type of presentation. With regard to the axial location in the spine, they can either be IM, EM or ED [1, 3, 40, 82, 104]. Intramedullary location appears to be the rarest, and conus medullaris appears to be the most frequent site for IM teratomas [49, 58]. Park et al. [104] compared ED versus ID teratomas and concluded that there were no differences between them in terms of male preponderance, lumbar location, young age, associated anomalies and pathogenic mechanisms.
Imaging
Plain radiographs have no significant role in the diagnosis of these lesions and are limited to detecting vertebral changes such as erosion or widening of the interpedicular space and other vertebral anomalies [49, 60, 107]. There are no characteristic features that can differentiate ST from other spinal neoplasms. Computed tomography (CT) is a sensitive method for differentiation based on the heterogeneity of the teratoma contents such as fat, calcification and cystic areas [40, 60]. MRI is the most sensitive imaging modality; however, in many cases, the diagnosis can be made only after histopathological examination. Heterogeneous solid-cystic masses consisting of fat and calcium with or without haemorrhage are the major radiological clues for a ST on MRI. Contrast enhancement is not a typical feature of mature teratomas; however, they may show marginal rim enhancement or a nodular enhancement [43, 46, 47, 54, 56, 66, 67, 69, 70, 72, 73, 78, 80, 82, 86,87,88, 90, 92, 100, 102].
In the present review, majority were heterogeneous lesions, but a few homogeneous lesions have also been reported [49, 66, 69, 78, 81,82,83, 86, 93, 99]. Characteristically, these lesions appear hyperintense on T1 sequences indicating the presence of fat. However, lesions that are predominantly T1 hypointense and isointense (with peripheral fat in a few cases) have also been reported [2, 28, 33, 45, 48, 54, 63, 65,66,67, 69, 70, 76, 78, 83, 86, 87, 89, 91, 93, 96, 100]. Associated congenital abnormalities may or may not be evident [60, 72, 73, 102, 108]. In our case, the lesion had similar characteristics that pointed towards a possible diagnosis of a teratoma preoperatively. Intradural teratomas are commonly oval or lobulated heterogeneous masses in contrast to the ED teratomas that are more often dumb-bell shaped [3, 104]. Although Sharma and coworkers and Moon and associates pointed out that STs frequently co-exist with dysraphic congenital spinal malformations, most authors have noted that simultaneous presentation of occult spinal dysraphic lesions and a ST appear to be a rare phenomenon in adults [1, 3, 47, 49, 72, 109]. The associated congenital anomalies may include spina bifida (SB), myelomeningocele (MMC), dermal sinus (DS) and split cord malformations (SCM) [102]. These anomalies are mostly associated with IM tumours, and the presence of such congenital abnormalities should raise the suspicion of a teratoma [73, 102]. In the present analysis, these anomalies were present in 55 cases (38%). The occurrence of ST with SCM is extremely rare, especially in adult population, and the split cord might exist at a remote location from the tumour. Hence, in such cases, it has been suggested that the entire spine need to be imaged because of the possibility of dysraphic lesions being present at non-contiguous sites [3, 47, 58]. There were 14 (9.6%) cases of associated SCM (7 cases each of types I and II) noted in the present review.
Embryology and pathogenesis
Several theories have been proposed to account for the embryogenesis of these tumours. Kubie and Fulton [6] believed this tumour to be an ependymal diverticulum. Ugarte and associates proposed that persistence of neurenteric canal resulted in a teratoma [107]. Bucy and Buchanan supported the theory of germinal cell aberration [32, 110]. However, none of the above theories are accepted now. According to the dysembryogenic theory, a teratoma is formed by chaotic differentiation of pluripotent cells in locally disturbed developmental environment in primitive streak or caudal cell mass [1, 3, 73, 82, 104]. The misplaced germ cell theory suggests that certain pluripotent primordial germ cells of the neural tube are misplaced during migration from the yolk sac to the gonad, thus resulting in a teratoma formation [1, 16, 73, 82, 104]. The presence of Barr bodies in teratomas in male patients has been suggested as an evidence of germ cell origin of such tumours [38]. This theory is more likely to be feasible in adult intraspinal teratomas due to the absence of significant dysraphism [3, 104]. However, despite all these theories, the exact pathogenesis of STs remains unclear.
Histopathology
Teratomas are histologically characterized by the presence of tissues derived from all the three germ layers [1, 48, 82]. Although, the presence of derivatives of two germ layers does not exclude the lesion as a teratoma, they are more aptly termed as teratoid tumours [38, 49, 102]. Walter and Kleinert classified teratomas into mature, immature and malignant based on the degrees of differentiation [1, 2, 39, 111]. Mature teratomas are composed exclusively of the fully differentiated ‘adult type’ tissue elements such as mature cartilage, squamous epithelium, skin appendages, columnar mucosa and nervous tissue and are considered as benign forms. Immature teratomas are aggressive tumours having primitive, undifferentiated components resembling ‘‘foetal’’ tissues and tend to recur frequently. Malignant teratomas are derived from the yolk sac or endodermal sinus and are associated with high levels of serum alpha-fetoprotein (AFP) and are associated with very poor prognosis [1, 40, 66, 73, 105, 112]. Histology of a ST shows mature type in most cases, while immature type STs are rarely reported in adults. Nephrogenic and pulmonary differentiations and carcinoid tumour arising in teratomas are very rare [1, 46]. In our present review, all except one were mature teratomas on histopathology. These teratomas may be hormonally responsive as noted in one report, where a sudden worsening of symptoms was noted during pregnancy [54]. Furthermore, because of the heterogeneity of the tissues, few authors have opined that the entire specimen should be thoroughly sampled to look for the components of a teratoma and other immature elements. This is because of the reason that these components may not be evident in the initial sections and are likely to go undetected which might have important prognostic values [1].
Treatment and outcome
Surgical resection is the treatment of choice for STs, and early surgery is recommended before irreversible neurological damage ensues. Complete resection should be aimed on all occasions and whenever possible, they should be excised with the capsule intact in order to reduce the risk of postoperative complications such as meningitis or myelitis [1, 40, 48, 60, 72]. This is generally possible in EM tumours; however, it might not be feasible in an IM tumours. In such cases, STR with preservation of functioning neural tissue seems to be a feasible option [1, 2, 48, 49, 58, 72]. Intraoperative use of electrophysiological monitoring may help to achieve greater tumour resection with preservation of neural function. In one of the previous reviews, IM tumours could be completely removed in 61.8% of the reported cases and improvement of symptoms occurred in 45.5% of them [40]. However, in this present review, CR was noted in only 37.6% cases of IM teratomas while STR was reported in 46.3% cases. It was noted that 66% of EM tumours underwent CR while 55% of IM tumours had STR, and this difference was statistically significant. Recurrences are uncommon and are mainly seen in the immature forms [40]. Because of the slow growth of these tumours, symptomatic recurrences usually occur after very long periods despite STR [49, 54]. However, few authors have noted STR not to be associated with poor outcomes [1, 49]. Hejazi et al. reported no difference in recurrence rates with CR versus STR (9% and 11%, respectively) at a mean follow-up of 25 months (43, 49). However, as noted in our results section, the overall recurrence rate was 6.1% (n = 9) at a mean follow-up of 37 months. Subtotal resection and CR were performed for 54 (37%) and 65 (45%) cases. There were one and five recurrences in CR and STR, respectively, and this difference was statistically significant (p = 0.029). Also, no malignant changes were evident in the recurred cases.
Several serum markers such as β-human chorionic gonadotropin (β-hCG) and AFP may be monitored for detection of recurrences of sacrococcygeal teratomas; however, this is limited in STs, as recurrences may originate from the non-secreting parts of the tumours [1, 3, 43]. Recurrences may be seen after a prolonged latency with one report documenting recurrence as late as 96 months after surgery, thus highlighting the extremely slow growth of these tumours [43]. In this present review, the mean TTR was 98.8 months (12–180 months) for mature teratomas. Hence, long-term clinical and imaging follow-up is recommended, especially after STR. Re-exploration and excision is advisable for symptomatic recurrent cases [1, 43, 49, 72]. Outcome is good to excellent in majority of cases [1, 39]. Immature and malignant forms are uniformly associated with poor outcomes and high recurrence rates. Poeze and associates pointed out that in benign teratomas, 26.3% died during a mean FU period of 38 months, while patients with malignant or immature IM teratomas died within 1 year after surgery [40]. As noted in the results section, there were four deaths reported and one case was diagnosed at autopsy [15, 56, 71, 78, 90]. Adjuvant radiotherapy (RT) is of no utility even in recurrent tumours, and it should be reserved only for teratomas with an immature or malignant component [1, 43, 49].
Limitations
Although this paper is the most extensive review till date on adult onset STs and including even those cases reported in non-English literature, cases reported in other languages are surely to be missed due to indexing problems and technical difficulties in accessing those articles.
Conclusions
This present paper provides a very comprehensive and in-depth analysis of all the adult onset spinal teratomas reported till date and analyses all the parameters possibly related to outcomes and recurrences. Spinal teratomas are uncommon neoplasms and are rarely encountered in the adult population. Majority of them are located in the thoracolumbar region as opposed to sacrococcygeal location in neonates and young children. Total surgical resection is the treatment of choice; however, this might not be possible in intramedullary cases for which subtotal resection might be a good option. Recurrences are uncommon and are mainly seen in immature teratomas and subtotal resections. Outcomes are good/excellent in over two-thirds of cases. Results of this paper show that the presence of pain, absence of limb weakness and complete tumour resection were significantly associated with good outcomes. Furthermore, subtotal resections had significantly higher recurrence rates than complete resections. Adjuvant radiotherapy is of no utility even in recurrent tumours, and hence, it should be reserved only for teratomas with immature or malignant components. This paper also highlights the importance of performing surgery even in those with late clinical presentations and complete neurological deficits, especially if an extramedullary component is present.
References
Sharma MC, Jain D, Sarkar C, Suri V, Garg A, Singh M, Mahapatra AK, Sharma BS (2009) Spinal teratomas: a clinico-pathological study of 27 patients. Acta Neurochir (Wien) 151:245–252
Ak H, Ulu MO, Sar M, Albayram S, Aydin S, Uzan M (2006) Adult intramedullary mature teratoma of the spinal cord: review of the literature illustrated with an unusual example. Acta Neurochir (Wien) 148:663–669
Li Y, Yang B, Song L, Yan D (2013) Mature teratoma of the spinal cord in adults: an unusual case. Oncol Lett 6:942–946
Gowers WR, Horsley V (1888) A case of tumour of the spinal cord. Removal; Recovery. Med Chir Trans 71(377–430):11
Frick K (1911) Ube rein Teratoma des Ruckenmarks. Frankfurt Z Path 7:127–134
Kubie LS, Fulton JF (1928) A clinical and pathological study of two teratomatous cysts of the spinal cord, containing mucous and ciliated cells. Surg Gynec Obstet 47:297–311
Hosoi K (1931) Intradural teratoid tumors of the spinal cord: report of a case. Arch Pathol 11:875–883
Naffiziger HG, Jones OW (1935) Dermoid tumors of the spinal cord. Report of four cases with observations on a clinical test for the differentiation of the source of radicular pains. Arch Neurol Psychiat 33:941–958
Adams RD, Wegener W (1947) Congenital cyst of the spinal meninges as cause of intermittent compression of the spinal cord. Arch Neurol Psychiat 58:57–69
Sullivan BH (1948) Intraspinal teratoma with report of a case. Brooklyn Hosp 6:142–145
Furtado D, Marques V (1951) Spinal teratoma. J Neuropathol Clin Neurol 10:384–393
Dereymacker A (1954) Medullar compression by congenital tumors or cysts. Acta Neurol Belg 54:874–889
Bakay L (1956) Case records of the Massachusetts general hospital: case 42502. N Engl J Med 266:1153–1157
Teng P, Gordon J (1958) Teratoma of the conus medullaris: report of a case. J Neurosurg 15:569–571
Sloof JL, Kernohan JW, MacCarty CS (1964) Primary intramedullary tumors of the spinal cord and filum terminale. Saunders, Philadelphia, pp 1–20
Rewcastle NB, Francoeur J (1964) Teratomatous cysts of the spinal canal; with “sex chromatin” studies. Arch Neurol 11:91–99
Hansebout RR, Bertrand G (1965) Intraspinal teratoma simulating protruded intervertebral disc. J Neurosurg 22:374–379
Reddy DR, Prabhakar V, Rao BD (1971) Intraspinal teratoma. Neurol India 19:45–47
Eneström S, von Essen C (1977) Spinal teratoma. Acta Neurochir (Wien) 39:121–126
Rosenbaum TJ, Soule EH, Onofrio BM (1978) Teratomatous cyst of the spinal canal: case report. J Neurosurg 49:292–297
Garrison JE, Kasdon DL (1980) Intramedullary spinal teratoma: case report and review of the literature. Neurosurgery 7:509–512
Padovani R, Tognetti F, Sanpaolo P, Pozzati E, Gaist G, Kuba I (1982) Intramedullary cystic teratoma. Acta Neurochir (Wien) 62:101–108
Padovani R, Tognetti F, Laudadio S, Manetto V (1983) Teratoid cyst of the spinal cord. Neurosurgery 13:74–77
Garza-Mercado R (1983) Diastematomyelia and intramedullary epidermoid spinal cord tumor combined with extradural teratoma in an adult: case report. J Neurosurg 58:954–958
Nakayama K, Miyasaka Y, Matsumori K, Beppu T, Asao T (1983) Spinal teratoma. Report of an elderly case. Neurol Med Chir (Tokyo) 23:963–967
Conti P, Conti R, De Luca G (1984) Observations on some rare cases of vertebro-medullar malformations associated with tumors. J Neurosurg Sci 28:81–87
Ironside JW, Jefferson AA, Royds JA, Taylor CB, Timperley WR (1984) Carcinoid tumour arising in a recurrent intradural spinal teratoma. Neuropathol Appl Neurobiol 10:479–489
Smoker WR, Biller J, Moore SA, Beck DW, Hart MN (1986) Intradural spinal teratoma: case report and review of the literature. AJNR Am J Neuroradiol 7:905–910
Giacomini P, Lunardi P, Liccardo G, Paris L, Sancesario G (1986) Cystic cervico-dorso-lumbar teratoma. Riv Neurol 56:1–8
Monajati A, Spitzer RM, Wiley JL, Heggeness L (1986) MR imaging of a spinal teratoma. J Comput Assist Tomogr 10:307–310
Pelissou-Guyotat I, Sindou M, Pialat J, Goutelle A (1988) Intramedullary mature teratoma associated with an attached cord and an intradural lipoma. Apropos of a surgically treated case. Review of the literature. Neurochirurgie 34:205–209
Hamabuchi M, Hasegawa R, Murase T (1989) Teratoma of the spinal cord. A case report with CT scans. J Bone Joint Surg Br 71:390–392
Üstün ME, Erdoğan A, Avunduk MC (1992) A case of cervical spinal teratoma associated with dermal sinus tract: MRI and surgical findings. Turk Neurosurg 2:194–198
Kaji S (1993) An elderly case of spinal teratoma in conus medullaris region. Seikeigeka 44:1359–1361 (in Japanese)
Nicoletti GF, Passanisi M, Platania N, Lanzafame S, Albanese V (1994) Intramedullary spinal cystic teratoma of the conus medullaris with caudal exophytic development: case report. Surg Neurol 41:106–111
Eide PK, Skullerud K (1994) Teratoma of the medullary cone as a cause of peripheral polyneuropathy. Tidsskr Nor Laegeforen 114:1185–1186 (in Norwegian)
Caruso R, Antonelli M, Cervoni L, Salvati M (1996) Intramedullary teratoma: case report and review of the literature. Tumori 82:616–620
Koen JL, McLendon RE, George TM (1998) Intradural spinal teratoma: evidence for a dysembryogenic origin. Report of four cases. J Neurosurg 89:844–851
Al-Sarraj ST, Parmar D, Dean AF, Phookun G, Bridges LR (1998) Clinicopathological study of seven cases of spinal cord teratoma: a possible germ cell origin. Histopathology 32:51–56
Poeze M, Herpers MJ, Tjandra B, Freling G, Beuls EA (1999) Intramedullary spinal teratoma presenting with urinary retention: case report and review of the literature. Neurosurgery 45:379–385
Natarajan M (1999) Conus medullaris teratoma presenting as myokymia. Neurol India 47:251
Bloch I, Varga Z, Benini A (1999) Teratoma of the conus medullaris. J Clin Neurosci 6:249–252
Allsopp G, Sgouros S, Barber P, Walsh AR (2000) Spinal teratoma: is there a place for adjuvant treatment? Two cases and a review of the literature. Br J Neurosurg 14:482–488
Okuyama K, Abe E, Hoshi N, Misawa A, Tamura Y, Chiba M (2000) Dumb-bell-type teratoma in the lumbar spine. Skelet Radiol 29:104–108
Arai Y, Takahashi M, Takeda K, Shitoto K (2000) Adult-onset intradural spinal teratoma in the lumbar spine: a case report. J Orthop Surg (Hong Kong) 8:69–74
Fan X, Turner JE, Turner TM, Elrod JP, Clough JA, Howell EI, Johnson MD (2001) Carcinoid tumor development in an intramedullary spinal cord mature teratoma. AJNR Am J Neuroradiol 22:1778–1781
Elmaci I, Dagcinar A, Ozgen S, Ekinci G, Pamir MN (2001) Diastematomyelia and spinal teratoma in an adult: case report. Neurosurg Focus 10:ecp2
Nonomura Y, Miyamoto K, Wada E, Hosoe H, Nishimoto H, Ogura H, Shimizu K (2002) Intramedullary teratoma of the spine: report of two adult cases. Spinal Cord 40:40–43
Hejazi N, Witzmann A (2003) Spinal intramedullary teratoma with exophytic components: report of two cases and review of the literature. Neurosurg Rev 26:113–116
Maiuri F, Gangemi M, Cavallo LM, De Divitiis E (2003) Dysembryogenetic spinal tumours in adults without dysraphism. Br J Neurosurg 17:234–238
Fernandez-Cornejo VJ, Martinez-Perez M, Polo-Garcia LA, Martinez-Lage JF, Poza M (2004) Cystic mature teratoma of the filum terminale in an adult. Case report and review of the literature. Neurocirugia (Astur) 15:290–293
Ates O, Cayli SR, Koçak A, Alkan A, Onal C, Usta U (2005) Mature spinal teratoma associated with thickened filum terminale. Neurol Med Chir (Tokyo) 45:375–378
Kao TH, Shen CC, Chen CC, Kwan PH (2005) Primary Benign retroperitoneal and intraspinal dumbbell-shaped cystic teratoma. Spine (Phila Pa 1976) 30:E439–E443
Kumar V, Peng EW, Kurian KM, Smith C, Fitzpatrick MO, Whittle IR (2006) An unusual progression of benign thoracic spinal cord teratoma in pregnancy: a hormonally-mediated pathway? Br J Neurosurg 20:106–108
Stevens QE, Kattner KA, Chen YH, Rahman MA (2006) Intradural extramedullary mature cystic teratoma: not only a childhood disease. J Spinal Disord Tech 19:213–216
Paterakis KN, Karantanas AH, Barbanis S, Hadjigeorgiou GM, Karavelis A (2006) Cervical spinal cord intramedullary teratoma. Clin Neurol Neurosurg 108:514–517
Kahilogullari G, Erdem A, Heper AO, Erden E (2006) Intramedullary mature cystic teratoma of the conus medullaris. A case report. J Neurosurg Sci 50:55–58
Tsitsopoulos P, Rizos C, Isaakidis D, Liapi G, Zymaris S (2006) Coexistence of spinal intramedullary teratoma and diastematomyelia in an adult. Spinal Cord 44:632–635
Caruso R, Colonnese C (2006) Intramedullary teratomas: two case reports and a review of the literature. Zentralbl Neurochir 67:213–218
Makary R, Wolfson D, Dasilva V, Mohammadi A, Shuja S (2007) Intramedullary mature teratoma of the cervical spinal cord at C1-2 associated with occult spinal dysraphism in an adult. Case report and review of the literature. J Neurosurg Spine 6:579–584
Mut M, Shaffrey ME, Bourne TD, Jagannathan J, Shaffrey CI (2007) Unusual presentation of an adult intramedullary spinal teratoma with diplomyelia. Surg Neurol 67:190–194
Sung KS, Sung SK, Choi HJ, Song YJ (2008) Spinal intradural extramedullary mature cystic teratoma in an adult. J Korean Neurosurg Soc 44:334–337
Mohindra S, Chhabra R, Gupta R, Dass Radotra B (2008) Cystic, exophytic teratoma of conus medullaris presenting with chronic renal failure. Surg Neurol 69:81–83
Han IH, Kuh SU, Chin DK, Kim KS, Jin BH, Cho YE (2008) Surgical treatment of primary spinal tumors in the conus medullaris. J Korean Neurosurg Soc 44:72–77
Benes V III, Barsa P, Mikulastik J, Suchomel P (2009) Exophytic intramedullary mature teratoma of the conus medullaris: case report and review of the literature. Cent Eur Neurosurg 70:154–160
Ijiri K, Hida K, Yano S, Iwasaki Y (2009) Huge intradural ossification caused by a mature spinal teratoma: case report. Neurosurgery 64:E1200–E1201
Ghostine S, Perry E, Vaynman S, Raghavan R, Tong KA, Samudrala S, Johnson JP, Colohan A (2009) The rare case of an intramedullary cervical spinal cord teratoma in an elderly adult: case report and literature review. Spine (Phila Pa) 34:E973–E978
Gu W, Shang H, Jin X, Xie J, Zhao W (2010) Intradural lumbar mature teratoma with neuronal and glial tissue component in an adult. Case report. Neurol Med Chir (Tokyo) 50:1112–1115
Arvin B, Pohl U, David K (2009) Intramedullary cervical teratoma in an adult. Spine J 9:e14–e18
Jeong SJ, Youm JY, Choi SW, Kim SH (2009) Intradural mature teratoma in the lumbar spine found in adult. Korean J Spine 6:211–213
Oh JS, Im SB, Kim BT, Shin WH (2009) Surgical findings of a lumbar mature teratoma accompanying the preoperative intracranial dissemination of Fatty droplets. Korean Neurosurg Soc 46:409–412
Moon HJ, Shin BK, Kim JH, Kim JH, Kwon TH, Chung HS, Park YK (2010) Adult cervical intramedullary teratoma: first reported immature case. J Neurosurg Spine 13:283–287
Agrawal M, Uppin MS, Patibandla MR, Bhattacharjee S, Panigrahi MK, Saradhi V, Rani JY, Purohit AK, Challa S (2010) Teratomas in central nervous system: a clinico-morphological study with review of literature. Neurol India 58:841–846
Jian W, Ying W, Chao Y (2010) Intramedullary spinal teratoma of the conus medullaris: report of two cases. Acta Neurochir (Wien) 152:553–554
Yu J, Qu LM, Li Y, Huang H (2012) Coexistence of spinal teratoma of the conus medullaris and arteriovenous malformation in an adult: a case report. Turk Neurosurg 22:510–514
Maiti T, Bhat DI, Devi BI, Sampath S, Mahadevan A, Shankar SK (2010) Teratoma in split cord malformation: an unusual association: a report of two cases with a review of the literature. Pediatr Neurosurg 46:238–241
Conti P, Tenenbaum R, Capozza M, Mouchaty H, Conti R (2010) Diastematomyelia and tumor in adults: report of two cases and literature review. Spine (Phila Pa 1976) 35:E1438–E1443
Bouaziz M, Haouam K, Laouar O, Lankar A (2011) A case of cervical intradural extramedullary mature cystic teratoma: diagnosis and management. Neurochirurgie 57:88–91
Musil J, Mrlian A, Duba M, Kren L, Smrcka M (2011) Probably the oldest patient with the diagnosis of medullar conus teratoma. Bratisl Lek Listy 112:357–359
Ben Nsir A, Hammouda KB, Said IB, Kassar AZ, Kchir N, Jemel H (2015) Spinal intradural mature teratoma in an elderly patient. J Cancer Metastasis Treat 1:106–110
Garg A, Arora V, Vaishy S, Sinha LK (2012) Chemical meningitis caused by spontaneous rupture of spinal teratoma. J Assoc Phys India 60:54–56
Kalani MY, Iyer S, Coons SW, Smith KA (2012) Spinal intradural teratomas: developmental programs gone awry? Neurosurg Focus 33:E1
Jiang H, Xiao Z, Zhan X, Chen Q, He M, Lu L (2013) Unusual association of intraspinal extramedullary teratoma with congenital scoliosis in an elderly adult: case report and literature review. Eur Spine J 22(Suppl 3):S306–S310
Vanguardia MK, Honeybul S, Robbins P (2014) Subtotal resection of an intradural mature teratoma in an adult presenting with difficulty initiating micturition. Surg Neurol Int 25(5):23
Babu R, Reynolds R, Moreno JR, Cummings TJ, Bagley CA (2014) Concurrent split cord malformation and teratoma: dysembryology, presentation, and treatment. J Clin Neurosci 21:212–216
Pandey S, Sharma V, Shinde N, Ghosh A (2015) Spinal intradural extramedullary mature cystic teratoma in an adult: a rare tumor with review of literature. Asian J Neurosurg 10:133–137
Alkherayf F, Arab AF, Tsai E (2015) Conus medullaris teratoma with utilization of fiber tractography: case report. J Neurol Surg Rep 76:e183–e187
Gunbey HP, Aslan K, Barıs YS, Incesu L (2015) Unusual presentation of a adult filum terminale teratoma associated with diplomyelia. Spine J 15:e49–e50
Yılmaz B, Demir MK, Yapıcıer O, Toktaş ZO, Akakın A, Konya D (2015) Mature cystic teratoma of the lumbar spine without fat in the cystic cavity. Spine J 15:e43–e44
Oh HS, Kim TW, Park KH (2015) Spinal teratoma concomitant with intracranial lipid droplet dissemination. Korean J Spine 12:15–18
Turan N, Halani SH, Baum GR, Neill SG, Hadjipanayis CG (2016) Adult intramedullary teratoma of the spinal cord: a case report and review of literature. World Neurosurg 87:661.e23–661.e30
Asan Z, Kaymaz H, Kilitci A (2017) Spinal intramedullary mature cystic teratoma in an adult. Br J Neurosurg 31:489–491
Agay AK, Garg S, Hedaoo K (2016) Spinal intradural extramedullary mature cystic teratoma in young adult: a rare tumor with review of literature. Int J Res Med Sci 4:5481–5483
Ariñez Barahona E, Navarro Olvera JL, Esqueda Liquidano MA, Muñoz Cobos A, Gonzalez Echeverria KE, Rivera Arroyo AD et al (2016) A special case of intramedullary teratoma in an adult. Literature review. Rev Med Hosp Gen Mex. https://doi.org/10.1016/j.hgmx.2016.09.001
Khazendar A, Hama Ameen HM, Jabbar NI, Hasan SO, Ahmed TS, Ali AA (2016) Upper lumbar mature cystic teratoma: a case report. World Neurosurg 96(609):e7–e11
Wang H, Huang Y, Nie P, Dong C, Hou F, Hao D, Xu W (2016) MRI findings in intraspinal mature teratoma. Clin Radiol 71:717.e1–717.e8
Schmidt RF, Casey JP, Gandhe AR, Curtis MT, Heller JE (2017) Teratoma of the spinal cord in an adult: report of a rare case and review of the literature. J Clin Neurosci 36:59–63
Wan W, Yang C, Yan W, Liu T, Yang X, Song D, Xiao J (2017) Adult-onset intradural spinal teratoma: report of 18 consecutive cases and outcomes in a single center. Eur Spine J 26:1917–1928
Mohammadi A, Fereydouni T, Rahbari A, Mokarian P, Khademi Z (2017) Intradural intramedullary teratoma presenting in the lumbar spine: report of a rare case. World Neurosurg 106(1051):e5–e8
Lekgabe E, Rogers TW, Laidlaw J, Lui E, Gaillard F (2018) A case of conus medullaris teratoma. J Clin Neurosci 47:123–126
Russell DS, Rubinstein LJ (1989) Pathology of tumors of the nervous system, 5th edn. Williams & Wilkins, Baltimore, pp 664–750
Ebner FH, Roser F, Acioly MA, Schoeber W, Tatagiba M (2009) Intramedullary lesions of the conus medullaris: differential diagnosis and surgical management. Neurosurg 32:287–301
Willis RA (1948) Pathology of tumours. Butterworth, London, pp 940–984
Park SC, Kim KJ, Wang KC, Choe G, Kim HJ (2010) Spinal epidural teratoma: review of spinal teratoma with consideration on pathogenesis: case report. Neurosurgery 67:E1818–E1825
Tapper D, Lack E (1983) Teratomas in infancy and childhood. Ann Surg 198:388–410
Sawamura Y, Kato T, Ikeda J, Murata J, Tada M, Shirato H (1998) Teratomas of the central nervous system: treatment considerations based on 34 cases. J Neurosurg 89:728–737
Ugarte N, Gonzalez-Crussi F, Sotelo-Avila C (1980) Diastematomyelia associated with teratomas. Report of two cases. J Neurosurg 53:720–725
Norman D, Mills CM, Brant-Zawadzki M, Yeates A, Crooks LE, Kaufman L (1983) Magnetic resonance imaging of the spinal cord and canal: potentials and limitations. AJR Am J Roentgenol 141:1147–1152
Daszkiewicz P, Roszkowski M, Przasnek S, Grajkowska W, Jurkiewicz E (2006) Teratoma or enterogenous cyst? The histopathological and clinical dilemma in co-existing occult neural tube dysraphism. Folia Neuropathol 44:24–33
Bucy PC, Buchanan DN (1935) Teratoma of spinal cord. Surg Gynecol Obstet 60:1137–1144
Walter GF, Kleinert R (1987) Dysontogenetic brain tumours—proposal for an improved classification. Neuropathol Appl Neurobiol 13:273–287
Valdiserri RO, Yunis EJ (1981) Sacrococcygeal teratomas. Cancer 48:217–221
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no declarations of interest
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Prasad, G.L., Divya, S. A comprehensive review of adult onset spinal teratomas: analysis of factors related to outcomes and recurrences. Eur Spine J 29, 221–237 (2020). https://doi.org/10.1007/s00586-019-06037-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-019-06037-7