Abstract
Introduction
Impaired muscle function and lumbar proprioception have been observed in lumbar spinal stenosis (LSS) but those have not been studied in LSS patients with age-matched controls. We assessed lumbar movement perception and paraspinal and biceps brachii (BB) muscle responses during sudden upper limb loading in age-matched healthy subjects and patients with LSS.
Methods
The study included 30 patients selected for an operation due to LSS and 30 age-matched controls without chronic back pain. The paraspinal and BB muscle responses for upper limb loading during unexpected and expected conditions were measured by surface EMG. The ability to sense lumbar rotation was assessed in a previously validated motorized trunk rotation unit in a seated position. Pain, disability and depression scores were recorded.
Results
Patients had poorer lumbar perception (mean difference 2.3 ± 0.6°, P < 0.001) and longer paraspinal muscle response latencies [mean difference 4.6 ± 0.6 ms (P = 0.033)] than age-matched healthy controls. Anticipation increased paraspinal and BB muscle activation prior to the load perturbation (P < 0.001) but less in LSS patients than in controls [9 vs. 30 %, P = 0.016 (paraspinals); 68 vs. 118 %, P = 0.047 (BB)].
Conclusions
The observed impairments in lumbar proprioception and activation of paraspinal and upper limb muscles indicate an extensive loss of both sensory and motor functions in LSS. The main new finding was decreased anticipatory muscle activation during expected upper limb loading reflecting involvement of central movement control mechanisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Appropriate movement sensation and muscular control are essential in preventing low back injury and falls, especially among the elderly. Protection from injury requires an ability to anticipate events and to make suitable muscular responses. Paraspinal muscle denervation and atrophy have been previously reported in patients with lumbar disc herniation [22] and lumbar spinal stenosis (LSS) [16]. It has also been shown that paraspinal muscle density decreases and the proportion of fat in the paraspinal muscles increases with age [9]. These changes may have impact on the muscle receptors and subsequently on trunk proprioception as well as reflex activation.
Voluntary upper [6, 8] limb movements cause trunk muscle activation via a feed-forward mechanism. This refers to the activity of the central movement control system, which maintains postural stability and prepares the trunk to bear a potentially increasing load by activating certain trunk muscles [1, 6, 8]. Impairment of feed-forward control of the trunk muscles in low back pain (LBP) has been observed previously [14]. In addition, abnormal trunk muscle activation has been observed in LBP patients during sudden upper limb and trunk loading [21, 27] and delayed reflex responses are considered as a risk factor for low back injury [4]. Active physical rehabilitation seems to improve these functions [17, 27].
Impaired lumbar proprioception has been observed previously in chronic non-specific LBP [3, 25], sciatica [14], and LSS patients [16]. However, the paraspinal muscle responses during sudden upper limb loading and lumbar proprioception have not been evaluated previously in LSS with age-matched controls.
We assessed lumbar movement perception and paraspinal and biceps brachii (BB) muscle activation during sudden upper limb loading in LSS patients and healthy controls of the same age.
Materials and methods
The study included 30 age-matched healthy controls (14 males and 16 females) and 30 patients with LSS (12 males and 18 females). The sample size was powered to 30 subjects in each group according to a previous study [11]. The study was approved by the Kuopio University Hospital Research Ethics Board and it was performed according to the Declaration of Helsinki. The independently living healthy control subjects were recruited from local pensioner organizations. The main inclusion criterion was a good general health. They all were personally interviewed and were confirmed not to have illness or disability affecting functionality. None of them used medication affecting central nervous system. The exclusion criteria were spinal surgery or notable spinal pain in history, other significant pain during past year and severe illnesses, such as diabetes, neurological or cardiovascular disease or cancer. Dropout percent was zero since all the pensioner organizations were given these criteria before candidate enrolment. The LSS patients were recruited from the Departments of Orthopaedic Surgery and Neurosurgery of the Kuopio University Hospital, where they were selected for decompression surgery due to LSS. All patients had both clinically and radiologically diagnosed LSS. Surgeon, in consensus with patient, made the decision to treat the patient operatively. The use of prescribed drugs was registered, but substances like caffeine were not. 28 patients used either NSAIDs or paracetamol, or both of them. 18 patients used tramadol, and two patients used neuropathic pain medication such as gabapentin or amitriptyline. None of the patients used narcotic alkaloids. Patients with clinically symptomatic spondylolysis or spondylolisthesis were excluded.
Pain intensity was determined by Visual Analogue Scale (0–10 VAS) [23]. Functional disability was assessed by the Oswestry disability index (OSW) [5]. Depressive symptoms were evaluated by using the 21-item Beck Depression Inventory (BDI-21) [2] (Table 1).
Lumbar movement perception
Lumbar proprioception was assessed in an earlier demonstrated trunk rotation measurement unit (DBC International Ltd, Helsinki, Finland) [25], which targeted the rotation on the lumbar/thoracic spine. The test was identical with the previous studies [13, 16]. In the test, the subject was placed in the device in a seated position, ears and eyes covered, while the seat was rotated with an angular velocity of 1°/s and the subject indicated the initiation of the movement by releasing a finger switch. The magnitude of the lumbar rotation was recorded. In addition, the subject was asked to indicate the direction of the movement. The results of five consecutive trials were pooled.
Muscle responses
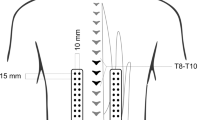
Paraspinal and BB muscle responses for unexpected (eyes closed) and expected (eyes open) upper limb loading were measured by a previously described protocol [12, 14]. During the procedure, the subject was standing and holding a box in the hands while a weight of 1.0 kg was suddenly dropped from the height of the subject’s eyes into the box equipped with a marker switch indicating the impact moment. Twelve consecutive measurements were performed in sequences of three trials with eyes open and three with eyes closed in supported (first six) and unsupported (last six) standing positions. Bipolar surface electromyography (EMG) was recorded bilaterally over the paraspinal muscles at L5–S1 levels by a four-channel ME 3000P EMG system (Mega Electronics Ltd, Kuopio, Finland) with disposable Ag/AgCl surface electrodes (Medicotest, Olstykke, Denmark). The electrode placements at paraspinal muscles (MF, L5–S1 level) were similar to the previous studies [12, 14]. A pair of 10-cm long cables connected the electrodes to the preamplifier in each EMG channel. The preamplifier was secured by attaching it to the corresponding reference electrode. A pair of 2.5-m long cables connected the preamplifier to the amplifier box. The raw EMG signal was recorded at the sampling rate of 2 kHz and band-pass filtered between 7 and 500 Hz with an analogue filter, amplified (differential amplifier, CMRR >110 dB, gain 1000, noise <1 V), analogue-to-digital converted (12-bit), and stored in a personal computer for later analysis.
The muscle activation onsets and offsets were determined visually from the rectified EMG. The determination was made without reference points in order to exclude observer bias. The latencies are presented with respect to the trigger in the bottom of the box. In addition, the BB and paraspinal muscle activity levels were obtained from 200 ms duration prior to the impact of the load. The activity level was normalized according to the activity of the first trial. The results from three repeated trials were pooled for statistical analysis.
Statistical analysis
In this study the statistical significance was set at P < 0.05 and statistical power calculations were done with 80 % power to determine group sizes. The degree of lumbar rotation was compared in patients and controls by independent samples t test. A repeated measures analysis of variance with three within factors (control of position, level and expectance) and grouping factor (patient vs. control) was used to analyse the effects of supported position, anticipation and LSS for the short latency responses of paraspinal muscles and preparatory muscle activation. Pearson’s correlation coefficients were calculated to assess the associations between questionnaire data, degree of lumbar rotation and paraspinal reflex latencies in patients. The statistical analyses were performed with SPSS 19.0 software (SPSS, Chicago, IL) and statistical significance was set as P < 0.05.
Results
The average threshold to detect a change in the position during lumbar rotation was approximately 2.3 ± 0.6 and 4.6 ± 0.6° in controls and in patients, respectively (mean difference 2.3 ± 0.6°, P < 0.001, Fig. 1).
The patients had longer paraspinal muscle response latencies for unexpected and expected upper limb loading, mean difference 4.9 ± 1.4 and 4.3 ± 1.0 ms, respectively (P = 0.033, Fig. 2). Expectation decreased the latency (P < 0.001) similarly in patients and controls.
Anticipation increased paraspinal and BB muscle activation prior to the load perturbation (P < 0.001) but less in LSS patients than controls [9 ± 2.9 vs. 30 ± 10.3 %, P = 0.016 (paraspinals, Fig. 3a) and 68 ± 18 % vs. 118 ± 36.2 %, P = 0.047 (BB, Fig. 3b)].
Very low correlations were found between subjective questionnaire data and the degree of lumbar rotation (r’s at or below 0.07) or BB muscle activation (r’s at or below 0.26). The correlation coefficient between unexpected paraspinal reflex latency and the ODI score was 0.46 (P < 0.05) (Table 2). The analgesics used or type of pain medication had no correlation to pain intensity (r’s at or below 0.25), lumbar sensation (r’s at or below 0.14), muscle responses (r’s at or below 0.16), or reflex latency (r’s at or below 0.20).
Discussion
Impaired preparatory muscle activation, paraspinal reflex activation and lumbar movement perception were observed in LSS patients. The increased BB muscle activation prior to load perturbation in expected loading trials is in line with the previous observations seen in similar setting in chronic LBP patients [11] and in self-initiated loading [20]. The preparatory upper limb activation and the increased paraspinal muscle activation are probably induced by the feed-forward mechanism, which prepares trunk for perturbation. The decline in these preparatory muscle activations in LSS patients indicates impaired feed-forward control, which supports the previous findings seen in experimental pain [7] and in chronic LBP patients during sudden upper limb loading [11]. It has also been suggested that during voluntary activation transient experimental muscle pain has a limited effect on central motor pathways [10]. Chronic pain has also been shown to lead to reduction in corticospinal drive to the legs making the patient more vulnerable to physical insult [24].
Impairments in lumbar movement perception have been observed previously in LSS [16]. Current study confirms the previous finding utilizing a different set of patient data with age-matched controls. There are several potential explanations for the differences seen between the current results in LSS and previous results in CLBP-like disruption of local nervous and muscular tissues, which is usually more severe in LSS than CLPB. Also the stenotics have both leg and back pain, while patients with CLBP usually have only back pain.
The impaired lumbar movement perception may be due to a sensory loss causing feedback error, or a deficit in information processing, or a combination of both mechanisms. Muscle spindles seem to be important in ensuring the correct positioning of the lumbosacral spine and muscle spindle input seems to be decreased in lumbar pain [3, 25]. The denervation of lumbar paraspinal muscles and lumbar degeneration probably decrease proprioceptive information [15]. Also fine tuning of the control of postural equilibrium is known to be compromised in people with LBP [19]. It has been indicated previously that the impaired lumbar proprioception was a reversible phenomenon in sciatica patients with 3 months follow-up [13]. The effect of surgery on lumbar perception should be assessed in LSS patients in further studies.
The difference in the trunk muscle reflex latency in anticipated sudden loading between controls and sciatica patients has been observed previously [14]. Anticipation shortened the reflex latency in controls, but not in patients when they were in an unsupported standing position. The reason why anticipation did not speed up postural reflex latencies of lumbar muscles in patients with sciatica is attributed to impaired feed-forward control in their central processing of information, also, this phenomenon was associated with clinically observed motor weakness. In the present study patients had longer paraspinal muscle reflex latencies both in unexpected and expected conditions and thus the results were partly different from those found in patients with disc herniation. The current results in LSS are in line with the results in chronic low back pain (CLBP) patients seen in Wilder et al. [27]. An acute condition such as disc herniation, perhaps in otherwise healthy lumbar spine, does not affect the paraspinal responses but a clearly chronic degenerative condition such as stenosis is affecting the reflexes. The potential effects of surgery and rehabilitation procedures on these reflex latencies should be tested in further studies.
An earlier study hypothesized that the functioning of short-term memory may be impaired in CLBP [18]. The limited effect of anticipation in CLBP patients was hypothesized to be due to impairment in the processing of visual feedback information in the short-term memory [14]. It was suggested that the higher-level information processing could be affected in CLBP patients, indicative of the possible relation with cognitive complaints and chronic pain. This is also supported by the observation of reorganization of motor cortex in recurrent spinal pain [26]. The present results support the hypothesis that also in LSS pain may affect motor behaviour at a higher level, which is involved in anticipatory behaviours. The BB muscle was chosen since its innervation and muscular structures were expected to be unaffected by LSS.
The reliability and validity of the methods used in this study have been demonstrated previously. These results increase our knowledge of the extensive changes in sensory–motor control of lumbar spine in LSS and have directed impact on planning-specific rehabilitation procedures to be tested clinically. However, the differences in paraspinal muscle activation levels and response latencies are rather small and therefore their clinical significance is expected to be rather limited. Also, correlation of lumbar proprioception and reflex control to clinical outcome of these patients seems to be limited and still requires further study.
In conclusion, the results indicate impaired lumbar proprioception and paraspinal muscle reflex activation in LSS patients. The main new finding was decreased anticipatory paraspinal and BB muscle activation during expected upper limb loading in LSS indicating involvement of a central control mechanism.
References
Aruin AS, Latash ML (1995) Directional specificity of postural muscles in feed-forward postural reactions during fast voluntary upper limb movements. Exp Brain Res 103:323–332
Beck AT, Ward CH, Mendelson M et al (1961) An inventory for measuring depression. Arch Gen Psychiatry 4:53–63
Brumagne S, Cordo P, Lysens R et al (2000) The role of paraspinal muscle spindles in lumbosacral position sense in individuals with and without low back pain. Spine 25:989–994
Cholewicki J, Silfies SP, Shah RA et al (2005) Delayed trunk muscle reflex responses increase the risk of low back injuries. Spine 30:2614–2620
Fairbank JCT, Couper J, Davies J, O’Brien JP (1980) The Oswestry low back pain disability questionnaire. Physiotherapy 66:271–273
Hodges P, Cresswell A, Thorstensson A (1999) Preparatory trunk motion accompanies rapid upper limb movement. Exp Brain Res 124:69–79
Hodges PW, Moseley GL, Gabrielsson A, Gandevia SC (2003) Experimental muscle pain changes feedforward postural responses of the trunk muscles. Exp Brain Res 151:262–271
Hodges PW, Richardson CA (1996) Inefficient muscular stabilisation of the lumbar spine associated with low back pain. A motor control evaluation of transversus abdominis. Spine 21:2640–2650
Kalichman L, Hodges P, Li L, Guermazi A, Hunter DJ (2010) Changes in paraspinal muscles and their association with low back pain and spinal degeneration: CT study. Eur Spine J 19(7):1136–1144
Khan SI, McNeil CJ, Gandevia SC, Taylor JL (2011) Effect of experimental muscle pain on maximal voluntary activation of human biceps brachii muscle. J Appl Physiol 111(3):743–750
Leinonen V, Airaksinen M, Taimela S et al (2007) Low back pain suppresses preparatory and triggered upper limb loading. Spine 32:E150–E155
Leinonen V, Kankaanpaa M, Hanninen O et al (2002) Paraspinal muscle responses during sudden upper limb loading. Eur J Appl Physiol 88:42–49
Leinonen V, Kankaanpää M, Luukkonen M et al (2003) Lumbar paraspinal muscle function, perception of lumbar position and postural control in disc herniation-related back pain. Spine 28:842–848
Leinonen V, Kankaanpaa M, Luukkonen M et al (2001) Disc herniation-related back pain impairs feed-forward control of paraspinal muscles. Spine 26:E367–E372
Leinonen V, Maatta S, Taimela S et al (2003) Paraspinal muscle denervation, paradoxically good lumbar endurance, and an abnormal flexion-extension cycle in lumbar spinal stenosis. Spine 28:324–331
Leinonen V, Määttä S, Taimela S et al (2002) Impaired lumbar position sense in association with postural stability and motor and somatosensory evoked potential findings in lumbar spinal stenosis. Spine 27:975–983
Luoto S, Taimela S, Hurri H et al (1996) Psychomotor speed and postural control in chronic low-back pain patients: a controlled follow-up study. Spine 21:2621–2627
Luoto S, Taimela S, Hurri H, Alaranta H (1999) Mechanisms explaining the association between the low back trouble and deficits in information processing. A controlled study with follow-up. Spine 24:255–261
Mok NW, Brauer SG, Hodges PW (2011) Postural recovery following voluntary arm movement is impaired in people with chronic low back pain. Gait Posture 34(1):97–102
Moseley GL, Hodges PW, Gandevia SC (2003) External perturbation of the trunk in standing humans differentially activates components of the medial back muscles. J Physiol 547:581–587
Radebold A, Cholewicki J, Panjabi MM, Patel TC (2000) Muscle response pattern to sudden trunk loading in healthy individuals and in patients with chronic low back pain. Spine 25:947–954
Rantanen J, Hurme M, Falck B et al (1993) The lumbar multifidus muscle five years after surgery for a lumbar intervertebral disc herniation. Spine 18:568–574
Scott J, Huskinsson E (1976) Graphic representation of pain. Pain 2:175–184
Strutton PH, Catley M, McGregor AH, Davey NJ (2003) Corticospinal excitability in patients with unilateral sciatica. Neurosci Lett 353:33–36
Taimela S, Kankaanpää M, Luoto S (1999) The effect of lumbar fatigue on the ability to sense a change in lumbar position—a controlled study in chronic LBP patients and healthy controls. Spine 24:1322–1327
Tsao H, Galea MP, Hodges PW (2008) Reorganization of the motor cortex is associated with postural control deficits in recurrent low back pain. Brain 131:2161–2171
Wilder DG, Aleksiev AR, Magnusson ML et al (1996) Muscular response to sudden load. A tool to evaluate fatigue and rehabilitation. Spine 21:2628–2639
Acknowledgments
We would like to acknowledge Minna Siitari, PT and Paula Heinonen, PT for patient measurements and Vesa Kiviniemi, MSc, for his valuable help in the statistical analysis.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by Kuopio University Hospital EVO Fund.
Rights and permissions
About this article
Cite this article
Kääriäinen, T., Leinonen, V., Taimela, S. et al. Lumbar paraspinal and biceps brachii muscle function and movement perception in lumbar spinal stenosis. Eur Spine J 22, 788–793 (2013). https://doi.org/10.1007/s00586-012-2563-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-012-2563-x