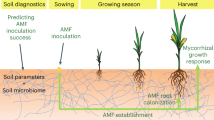
Abstract
Arbuscular mycorrhizal (AM) fungi are considered to be a key group of soil organisms for assessments of soil biological properties and developing relationships among crop production management practices, soil properties, crop performance, and ecosystem services. In a field study of cover crop treatments established during the transition from small grains to corn (Zea mays L), we assessed multiple measures of AM fungal responses to the management treatments: soil propagule numbers, biomass via lipid biomarkers, and root colonization extent. Our objectives were to determine response variables that reliably distinguished cover crop treatments and formed consistent relationships with grain yield, plant biomass, and mineral nutrient concentrations of the following corn crop. The number of soil AM fungal propagules and amount of the NLFA biomarker C16:1cis11 measured on fall-collected soils most consistently and significantly responded to fall cover crop treatments. Neither of these measures of soil inoculum potential was strongly related to measures of crop performance. The PLFA biomarker C16:1cis11 was marginally responsive to cover crop but did not strongly relate to crop performance parameters. Corn root colonization by AM fungi was not significantly affected by cover crop treatment, but significant negative relationships were found between root colonization and grain N concentration and plant biomass at maturity. In contrast, a significant positive relationship between root colonization and plant N concentration at the 6-leaf stage was found. Understanding the relative effectiveness and limitations of AM fungal response variables will inform their application in field studies of agricultural management practices.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Obligate plant symbiotic arbuscular mycorrhizal (AM) fungi have a 460-million-year-old evolutionary history of partnership with many terrestrial plants. Research has documented the frequent benefits of this symbiosis to contemporary plant species, including most major crops (Lekberg and Koide 2005; Rillig 2004; Smith and Read 2008). Consequently, AM fungi are considered to be a key group of soil organisms for monitoring in agricultural production systems and developing relationships between management practices, soil properties, crop performance, and ecosystem services (Creamer et al. 2016; Dai et al. 2014; de Vries et al. 2013; Dias et al. 2014; Gianinazzi et al. 2010; Gottshall et al. 2017; Hamel 1996; Jeffries et al. 2003; Miller 2000; Soliveres et al. 2016; Verbruggen et al. 2010; Wagg et al. 2014).
While there are multiple approaches for measuring properties of AM fungi (Sylvia 1994), each measure presents limitations in practice and interpretation that must be considered with respect to the question being investigated. In general, many common crop production management practices such as no-till, fallow, and monocropping often negatively affect some measurement of AM fungi such as spore counts, spore diversity, or root colonization (Jansa et al. 2006; Mader et al. 2000). But, the results can also be contradictory between root colonization and various measures of soil inoculum potential such as spores, viable propagules, hyphal density, and fatty acid biomarkers (Douds Jr. et al. 1993; Gosling et al. 2010; Martinez and Johnson 2010; Sharma and Buyer 2015). Many studies of agricultural management practices have found varying degrees of association between AM fungi soil inoculum potential or root colonization with plant performance indicators such as biomass and nutrient uptake (Douds Jr. et al. 2011; Galvez et al. 2001; Gosling et al. 2016; Johnson et al. 1992; Ryan and Graham 2018; Smith and Smith 2011; Treseder 2013). But, definitive conclusions on these relationships are elusive because experimental design, selected methods, methodological details, and degree of methodological replication differ widely from study to study.
Our study objectives were to examine multiple AM fungal response variables using a fully replicated methodology in multiyear, replicated agronomic field trials to determine which AM fungal response variables: (1) were most reliable in distinguishing agricultural management treatments; and (2) were related to crop yield, plant biomass, and mineral nutrient concentrations. We evaluated the effect of fall cover cropping treatments imposed during the transition from small grains to corn (Zea mays L) on measures of soil AM fungal inoculum potential, corn root colonization, and corn yield, biomass, and mineral nutrient concentrations. For AM fungal soil inoculum potential, we enumerated viable propagules that include spores which alone do not represent full inoculum potential (Gosling et al. 2010; Hamel 1996; Martinez and Johnson 2010) and quantified fatty acid biomarkers in both the phospholipid and neutral lipid fractions because their responses differ (Sharma and Buyer 2015). We have previously published some of the soil inoculum data from this field experiment as part of a multilocation study on fall cover cropping effects on soil inoculum potential of AM fungi (Lehman et al. 2012). In the current paper, our objectives are the comparative treatment effects on AM fungal response variables and their relationship with plant performance. We use 3 years of previously unreported data for root colonization, phospholipid fatty acid biomarkers, grain yield, plant biomass, and mineral nutrient concentrations and an additional year of soil propagule data.
Materials and methods
Field plots
Soil and plant samples were collected in the performance of a three-year field study at the 65 ha Eastern South Dakota Soil and Water Research Farm in Brookings, South Dakota (44° 19’ N latitude; 96° 46’ W longitude). The research farm is located in the Big Sioux Basin of the northern glaciated plains at 500 m elevation with 58 cm mean annual precipitation (MAP) and mean annual temperature (MAT) of 8 °C (Bryce et al. 1996). The Mollisol soils are Barnes sandy clay loam (fine-loamy, mixed, superactive, frigid Calcic Hapludoll) that are moderately drained, with a clay content of about 280 g kg−1 (Pikul et al. 2007). Pre-experiment soil analyses showed circumneutral soils with relatively high soil organic matter (ca. 3–4%) and low (< 10 mg kg−1) levels of extractable N and P (Lehman et al. 2012). Eight cover crop treatments were replicated four times in a randomized complete block design: no cover crop, forage oats (Avena sativa (L.) Hausskn.), hairy vetch (Vicia villosa Roth), winter canola (Brassica napus L.), and all possible combinations of the three cover crops. Starting in 2009, fall cover crop treatments were direct seeded in no-till plots (3 m × 16 m) following small grain harvest in August; the cover crops were chemically terminated the following May, and the plots were planted to corn. These experimental design and management sequence were repeated in 2010 and 2011 at adjacent sites. The small grain was spring wheat (Triticum aestivum) in 2009 and oat (Avena sativa L.) in 2010 and 2011. Corn received 165 kg N ha−1 at the 6-leaf vegetative (V6) growth stage, following plant and root sampling. No phosphorus was applied. Six randomly-selected plants per plot were collected for above-ground plant biomass (shoots plus leaves), and mineral nutrient concentrations (N, P) at three corn growth stages were as follows: V6, R1 (silking), and R6 (physiological maturity). Annual corn grain yields and plant biomass were measured by standard methods; mineral nutrient concentrations in dried, milled grain, and above-ground plant biomass were measured by inductively coupled plasma emission spectrometry.
Soil and root sampling for AM fungi analyses
Six 30-mm diameter cores, 0–15 cm depth, were mixed to form a composite soil sample for each field plot in November of 2009, 2010, and 2011 following the fall cover crop growth period in May of 2010, 2011, and 2012 following cover crop termination. Roots from the subsequent corn crop were collected between 3 and 4 weeks post-emergence from six randomly selected plants per plot in 2010, 2011, and 2012. Root sampling timing corresponded to 35 (± 1.5) days after planting when the majority of corn was in the V5 or V6 growth stage.
AM fungal soil propagule numbers
Arbuscular mycorrhizal propagules which include spores, infected root pieces, and vegetative hyphal fragments that together represent AM fungal soil inoculum potential were measured on soils collected in late November following the fall cover crop growth period. Propagule numbers were measured for each plot using the most-probable-number (MPN) assay (Porter 1979) by serially diluting the soils in a sterile 1:1:1:1 (v/v) mix of quartz sand (4030 silica sand, 0.45–0.55 mm diameter, Unimin Minnesota Corp, Le Sueur, MN), vermiculite (coarse, grade 2A, Therm-O-Rock, New Eagle PA), calcined clay (Turface All Sport Pro, Profile Products, Buffalo Grove, IL), and sterilized site soil (Douds Jr. et al. 2011). Dilution soils were sterilized by autoclaving twice for 60 min. at 121 °C with a 24 h break in between. Five levels of serial dilution were performed in triplicate to create a five by three MPN matrix for each soil sample (Woomer 1994). Bahiagrass (Paspalum notatum Flugge) was the host species for the MPN assay and was planted into 65 cm3 pots of the diluted test soils, grown in the greenhouse (day/night 16/8 h, 25/18 °C) with weekly application of Hoagland’s nutrient solution without P (Hoagland and Arnon 1950) and harvested after four weeks of growth. Pots with dilution soils alone were planted for negative controls. Washed roots were cleared and stained with trypan blue (Philips and Hayman 1970) in lactoglycerol and scored for presence/absence of AMF structures (arbuscles, vesicles, intraradical hyphae) in the root cortex under × 25 to × 40 magnification with a stereomicroscope (Leica M-32). Presumptive positive infections were confirmed by viewing these root fragments mounted on a glass slide at × 200 with a Leica DM LB2 (Leica Microsystems, Buffalo Grove, IL) compound microscope. The method detection limit for AMF propagules in an individual soil sample was 0.06 propagules g−1 for the 2009 samples using tenfold soil dilutions and 0.02 propagules g−1 for 2010 and 2011 using fourfold soil dilutions. All soil samples that were below the detection limit were considered to have zero propagules for treatment mean calculation.
AM fungal soil biomass by fatty acid biomarker concentration
The soil biomass of AM fungi includes the aggregate mass of extraradical hyphae as well as spores and infected root fragments and should be related to soil inoculum potential. The measurement of extracted and fractionated lipids, a cellular component of organisms, has long been used as a biochemical proxy for biomass of soil microorganisms (Pinkart et al. 2002). For AM fungi, the fatty acid methyl ester C16:1cis11 is commonly used as a biomarker with the expectation that the amount of this biomarker is proportional to biomass (Olsson 1999; Sharma and Buyer 2015). We measured the amounts (nmol g−1) of the C16:1cis11 biomarker in both neutral and phospholipid fractions for both fall- and spring-collected soils. Lipids were extracted from soil samples and partitioned into neutral, polar, and phospholipid fractions as described in Buyer et al. (2010). The neutral lipid fraction was evaporated under nitrogen. Transesterification, cleanup, and gas chromatography of the neutral lipid fatty acids (NLFA) were accomplished exactly as described for the phospholipid fatty acids (PLFA) (Buyer et al. 2010). Amounts (nmol g−1) of the AM fungal biomarker, C16:1cis11, were reported relative to an external standard of hexadecanoic acid methyl ester. The absence of NLFA data for fall of 2011 and spring of 2011 and 2012 was due to an unfortunate freezer failure.
AM fungal root colonization (arbuscular and total)
Washed corn roots were cleared and stained with trypan blue (Philips and Hayman 1970) in lactoglycerol. Randomly selected portions of the fine roots from each plant were placed in individual biopsy cassettes for clearing and staining. Stained roots were stored in 1:1 (w:w) glycerol/deionized water at 4 °C prior to evaluation. Ten root segments (2.5 cm) from each plant were trimmed of root hairs using a steel scalpel and mounted on glass slides in 1:1 (vol:vol) glycerol/deionized water solution. A Leica DM LB2 compound microscope was used at × 200 magnification to score roots for colonization using the magnified, gridline-intersect method (McGonigle et al. 1990a). An average of 198 ± 25 (mean ± one standard deviation) fields for each plant was scored for the presence of AM fungal structures. Arbuscular colonization rates are reported as the most conservative estimate of AM fungal colonization; total colonization (arbuscle, vesicle, intraradical hyphae) rates are also reported for comparability with other studies.
Statistics
Propagule numbers plus one were square root transformed and root colonization percentages were arcsine transformed for statistical analyses. Homogeneity of variances was tested with Bartlett’s chi-square statistic. Means for AM fungal response variables were tested for statistically significant differences among cover crop treatments at the p ≤ 0.05 level by analysis of variance procedures with block and year as random factors. Mean separations were analyzed using a protected PDIFF option (t test) within the LSMEANS statement when F tests indicated that significant differences existed (p ≤ 0.05). Relationships among dependent variables for all treatments were initially assessed using Pearson’s correlation coefficient which was tested for statistical significance with the Dunn–Sidak corrected probability. Relationships among variable pairs that exhibited strong correlations were quantified by least squares linear regression analysis of variance. Principal components analysis (PCA) was conducted to examine the relative contributions of AM fungal response variables and plant performance variables to the variation within the dataset. For that analysis, NLFA measures were excluded because there was no data from some years, and only the plant performance parameters measured at plant maturity were used for clarity and their significance. The PCA was performed on the correlation matrix with no factor rotation; variable loadings were retained for factors with eigenvalues ≥ 1.
Results
There were seven AM fungal response variables measured for the fall cover crop treatments: soil AM fungal propagules (fall soils), total AM fungal colonization of subsequent corn roots, arbuscular AM fungal colonization of subsequent corn roots, NLFA biomarker on fall soil, NLFA biomarker on spring soils, PLFA biomarker on fall soils, and PLFA biomarker on spring soils. The number of soil AM fungal propagules significantly (p = 0.0010) responded to cover crop treatment (Table 1). Among the treatments, oat and the three-way cover crop treatments had the highest average numbers of propagules (Fig. 1). In all 3 years, every cover crop treatment had AM propagule numbers that were numerically greater or equal to the no cover crop control. There was no obvious negative effect of the non-AM fungal host canola on soil propagule numbers.
Number of soil AM fungal propagules (mean ± one standard error; n = 4 plots) measured in cover crop treatments for each year. No visible bar in 2009 for “none” and “canola” means that propagule numbers were below the detection limit. Bars topped by the same letter do not differ significantly at P ≤ 0.05 by protected t test; pairwise treatment comparisons are within each year. The combined data for the first 2 years were previously published (Lehman et al. 2012)
The NLFA biomarker C16:1cis11 measured on fall soils was affected by cover crop treatments (p < 0.0001) in the 2 years with data. There was a significant effect of year (< 0.0001) because individual cover crop treatments responded differently in the 2 years. Cover crop treatments containing oat had significantly more NLFA biomarker than the no cover crop controls in both years (Fig. S1a). Cover crop combinations containing vetch also had significantly higher values in 2010. The PLFA data on the same biomarker (C16:1cis11) for fall soils was not significantly (p = 0.2153) affected by cover crop treatment (Table 2). In the single year where there was NLFA biomarker data on spring soils (2010), the effect of cover crop was not significant at the p ≤ 0.05 level (p = 0.0623); vetch-containing treatments had the highest values (Fig. S1b). The PLFA biomarker for spring soil was significantly affected (p = 0.005) by cover crop treatment with the oat/vetch and oat/vetch/canola treatments having the highest values in 2011 and 2012 (Table 2).
There were no significant effects of cover crop treatments on arbuscular root colonization of the subsequent corn crop, but there was a significant (p = 0.0325) main treatment effect on total root colonization (Fig. 2, Table 1). There was a strong (p < 0.0001) effect of year on both arbuscular and total root colonization. In 2011, total colonization was highest for the three-way cover crop mix, and in 2012, several cover crop treatments were significantly higher than canola. Corn root colonization in 2010 ranged from 18 to 31% based on arbuscular occurrence and 70 to 79% based on all structures. Arbuscular corn root colonization in 2011 ranged from 58 to 79% and in 2012 from 68 to 81%. In these 2 years, total colonization was about 10–15% higher than arbuscular colonization.
Percent colonization (mean ± one standard error; n = 4 plots) of corn roots measured by the occurrence of arbuscules (a) and all intraradical structures (b) for the eight cover crop treatments for all 3 years of the study. There were no significant main effects of treatments on arbuscules; therefore, no pairwise comparisons were performed (a). Bars topped by the same letter do not differ significantly at P ≤ 0.05 by protected t test; pairwise treatment comparison are within each year (b)
We evaluated simple correlations between the AM fungal biomass response variables and corn grain yield and mineral nutrient concentrations (N and P), and corn plant biomass and nutrient concentrations at V6, R1, and physiological maturity (R6) for each plot for all 3 years (Table S1). Of these 82 correlations, only eight correlations had a Dunn–Sidak-corrected probability < 0.1. The strongest correlations all involved arbuscular colonization which was negatively related to grain N concentration (r = − 0.640; p < 0.001) and plant biomass at maturity (r = −0.736; p < 0.001), and positively related to V6 plant N concentration (r = 0.684; p < 0.001). Total colonization was also negatively correlated with grain N concentration and plant biomass at maturity. Weaker correlations were observed between fall NLFA and V6 plant biomass (r = −0.462, p = 0.034) and plant biomass at maturity (r = 0.432; p = 0.082). A significant negative correlation (r = −0.430; p = 0.014) was observed between spring NLFA and V6 plant biomass. A separate correlation analysis was performed on the seven AM fungal response variables (Table S2). As expected, total and arbuscular colonization were strongly correlated. The only other significant correlation was between fall NLFA and arbuscular colonization (r = − 0.454; p = 0.004).
A least squares linear regression fit to the scatterplot of arbuscular colonization and V6 plant N concentration demonstrated a significant positive relationship (Fig. 3a). In contrast, arbuscular colonization was negatively related to grain N concentration (Fig. 3b) and plant biomass at maturity (Fig. 3c).
Scatterplots of root colonization (arbuscular) and corn grain properties, n = 93. a Plant (shoot and leaves) N concentration (g kg−1) at the V6 growth stage with linear regression fitted to the data. b Grain N concentration (g kg−1) with linear regression fitted to the data. c Plant biomass (shoots and leaves) at maturity (kg ha−1) with linear regression fitted to the data
The PCA resulted in four factors with eigenvalues > 1 (Table S3); the first two factors accounted for 54.4% of the variance in the dataset. To illustrate the relative contributions of the variables and their interrelationships within the structure of the dataset, factor loadings for 11 variables were plotted for factor 1 (33.6%) and factor 2 (20.8%) (Fig. 4). Factor 1 had positive loadings for plant performance parameters and negative loadings for root colonization and spring PLFA. Factor 2 showed positive loadings for root colonization and P concentration of grain and plant tissue, which were both opposed by plant biomass.
Principal components factor loadings for AM fungal response variables and plant performance variables plotted for factors one and two. Plant biomass includes shoots and leaves, but not grain. The data for plant biomass, N, and P were measured at physiological maturity. Fall and spring NLFA are not included in this analyses because there were no data for one and 2 years, respectively
Discussion
The number of soil AM fungal propagules responded to the treatments in a consistent manner over each of the 3 years but were not related to any plant performance indicators. These soil samples were collected in late November, following the fall-seeded cover crop growth period and prior to freeze-up. It is generally acknowledged that AMF propagule numbers represent an ecologically relevant pool (Rillig 2004) that represents inoculum potential better than spore counts (Gosling et al. 2010; Hamel 1996; Martinez and Johnson 2010). Further, while measuring soil propagules using the MPN procedure is laborious, it is not technically difficult and does not require the level of experience required to distinguish viable AM fungal spores among other structures found in the soil.
The NLFA biomarker C16:1cis11 measured on fall-collected soils was also responsive to treatment in the 2 years with data, while the single year of data on spring-collected soils was not significant at the 0.05 alpha level (p = 0.0623). The NLFA data reinforced the treatment results from the propagule numbers wherein cover crop treatments-containing oat elevated the amount of AM fungi in the soils and also showed a response to some vetch treatments. The PLFA biomarker was only responsive to treatment in spring-collected soils for 2 years. The highest PLFA biomarker values were recorded in two oat-containing cover crop combinations; however, the numerical range in PLFA values across all treatments was limited. In a study of field soils, the AM fungal biomarker analyzed with the ester-linked fatty acid methyl ester (EL-FAME) procedure showed a stronger response to agricultural management practice than the same biomarker analyzed with the PLFA procedure (Drijber et al. 2000). The EL-FAME analysis reports the AM fungal biomarker combined from both the neutral- and phospholipid fractions. In a greenhouse study, C16:1cis11 analyzed by both NLFA and EL-FAME was found to be a superior biomarker for AM fungi compared with the same in the phospholipid fraction (PFLA) (Sharma and Buyer 2015). Our current data on field trials also indicate that NLFA produces a more reliable AM fungal biomarker than PLFA. The combined NLFA and PLFA biomarkers in EL-FAME may be considered advantageous for quantifying AM fungi in soils, but also may dilute more true NLFA values. Notably, EL-FAME protocols utilize stronger extraction conditions that produce larger amounts of non-target lipids and thus the signal-to-noise ratio may be reduced for specific fatty acids.
Root colonization of the corn at 3 to 4 weeks post-emergence, or V5/V6 growth stage, was not an effective measure of AM fungal response to the cover crop treatments. Colonization measured at an earlier plant growth stage may have produced different results, although similar and later plant growth stages have been used in other studies (Bowles et al. 2017; McGonigle et al. 1990b). Root colonization in the first year was much lower than the second year and may be related to winter wheat instead of oat as the small grain that preceded the cover crop in the first year. A positive relationship was observed between arbuscular colonization and plant N concentration at the V6 growth stage, suggesting AM fungal contributions to early N uptake by the plant. But, most unexpected were the negative relationships observed between arbuscular colonization and grain N concentration and plant biomass at maturity that were based on 96 plot-years, with root data from 6 plants per plot and 200 microscope fields per plant. Grain yield, N concentration, and mature plant biomass all shared positive but not significant correlations (data not shown), and grain yield had a non-significant negative correlation with arbuscular colonization. Colonization and plant P concentrations had positive scores on factor 2, suggesting a link between these measures within the dataset, yet the linear regression analysis of these variable pairs was not significant (data not shown). It is likely that this association only occurred in 1 or 2 years of the 3-year study and is dependent on unknown, annual-specific conditions.
Root colonization is probably the most frequently measured AM fungal response variable; however, methodological details vary considerably. In a survey of studies where AM fungi were inoculated to crop or forage plots, inoculation produced elevated root colonization and higher yields, but there was no relationship between the extent of colonization and yield magnitude (McGonigle 1988). In meta-analyses of cropping systems, elevated rates of root colonization with inoculation have been linked with better crop performance (Lekberg and Koide 2005), particularly with wheat (Pellegrino et al. 2015). A more recent meta-analysis of studies using AM fungal inocula that includes natural systems and excludes fertilized treatments also found positive relationships between colonization and plant biomass and P concentration (Treseder 2013). Most interesting is that the strengths of these relationships varied with the specific fungal taxa in the inoculum.
A meta-analysis of agricultural management effects on native AM fungi found that AM fungal root colonization of annual crops was increased by cover cropping and reduced tillage, but no plant performance data were evaluated (Bowles et al. 2017). Studies of agricultural management treatments on native AM fungi often find no positive relationship between AM fungal root colonization and crop performance parameters (yield, biomass, nutrient uptake) (Barber et al. 2013; Galvez et al. 2001; Köhl et al. 2014; McGonigle et al. 1990b; Ryan and Graham 2018; Ryan et al. 2005; Verbruggen et al. 2012). It appears that for non-inoculated cropping systems, relationships between root colonization and plant performance are less predictable than inoculated systems and may depend on the identity of the specific fungal taxa that are colonizing the crop roots (Dai et al. 2014). Individual plant cultivars also differ in their growth responsiveness to AM fungi; therefore, root colonization may not always be a suitable indicator for benefits delivered to the plant (Ryan and Graham 2018; Smith and Smith 2011).
Given the enduring and often beneficial partnership between plant and mycorrhizae and the documented reduction of AM fungi by many conventional cropping practices, it seems justified to identify practices that may increase the native soil inoculum potential, even when a growth response is not always observed. It has been pointed out that there are other potential benefits of increasing soil AM fungal inoculum in cropping systems such as promoting soil aggregation, carbon sequestration, and plant resilience (Smith and Smith 2011). The overall goal of determining the appropriate AM fungal response variable is to promote adoption of resource-efficient cropping systems and to understand the basis of their function. Our results indicate that inoculum potential in terms of number of AM fungal propagules with the MPN assay or as biomass according to the NLFA biomarker C16:1cis11 were effective at distinguishing our agricultural management treatments, although these measures did not correlate with plant performance indicators. In contrast, corn root colonization by AM fungi was not affected by cover crop treatment, and extensive root colonization was associated with lower grain N concentration and lower plant biomass at maturity.
References
Barber NA, Kiers ET, Theis N, Hazzard RV, Adler LS (2013) Linking agricultural practices, mycorrhizal fungi, and traits mediating plant–insect interactions. Ecol Appl 23:1519–1530
Bowles TM, Jackson LE, Loeher M, Cavagnaro TR (2017) Ecological intensification and arbuscular mycorrhizas: a meta-analysis of tillage and cover crop effects. J Appl Ecol 54:1785–1793
Bryce SA, Omernik JM, Pater DE, Ulmer M, Schaar J, Freeouf J, Johnson R, Kuck P, Azevedo SH (1996) Ecoregions of North Dakota and South Dakota (map scale 1:1,500,000). U.S. Geological Survey, Reston
Buyer JS, Teasdale JR, Roberts DP, Zasada IA, Maul JE (2010) Factors affecting soil microbial community structure in tomato cropping systems. Soil Biol Biochem 42:831–841
Creamer R, Hannula S, Van Leeuwen J, Stone D, Rutgers M, Schmelz R, De Ruiter P, Hendriksen NB, Bolger T, Bouffaud M-L (2016) Ecological network analysis reveals the inter-connection between soil biodiversity and ecosystem function as affected by land use across Europe. Appl Soil Ecol 97:112–124
Dai M, Hamel C, Bainard LD, Arnaud MS, Grant CA, Lupwayi NZ, Malhi SS, Lemke R (2014) Negative and positive contributions of arbuscular mycorrhizal fungal taxa to wheat production and nutrient uptake efficiency in organic and conventional systems in the Canadian prairie. Soil Biol Biochem 74:156–166
de Vries FT, Thébault E, Liiri M, Birkhofer K, Tsiafouli MA, Bjørnlund L, Jørgensen HB, Brady MV, Christensen S, de Ruiter PC (2013) Soil food web properties explain ecosystem services across European land use systems. Proc Natl Acad Sci 110:14296–14301
Dias T, Dukes A, Antunes PM (2014) Accounting for soil biotic effects on soil health and crop productivity in the design of crop rotations. J Sci Food Agric 95:447–454
Douds DD Jr, Janke RR, Peters SE (1993) VAM fungus spore populations and colonization of roots of maize and soybean under conventional and low-input sustainable agriculture. Agric Ecosyst Environ 43:325–335
Douds DD Jr, Nagahashi G, Wilson DO, Moyer J (2011) Monitoring the decline in AM fungus populations and efficacy during a long term bare fallow. Plant Soil 342:319–326
Drijber RA, Doran JW, Parkhurst AM, Lyon DJ (2000) Changes in soil microbial community structure with tillage under long-term wheat-fallow management. Soil Biol Biochem 32:1419–1430
Galvez L, Douds DD Jr, Drinkwater LE, Wagoner P (2001) Effect of tillage and farming system upon VAM fungus populations and mycorrhizas and nutrient uptake of maize. Plant Soil 228:299–308
Gianinazzi S, Gollotte A, Binet MN, van Tuinen D, Redecker D, Wipf D (2010) Agroecology: the key role of arbuscular mycorrhizas in ecosystem services. Mycorrhiza 20:519–530
Gosling P, Ozaki A, Jones J, Turner M, Rayns F, Bending GD (2010) Organic management of tilled agricultural soils results in a rapid increase in colonisation potential and spore populations of arbuscular mycorrhizal fungi. Agric Ecosyst Environ 139:273–279
Gosling P, Jones J, Bending GD (2016) Evidence for functional redundancy in arbuscular mycorrhizal fungi and implications for agroecosystem management. Mycorrhiza 26:77–83
Gottshall CB, Cooper M, Emery SM (2017) Activity, diversity and function of arbuscular mycorrhizae vary with changes in agricultural management intensity. Agric Ecosyst Environ 241:142–149
Hamel C (1996) Prospects and problems pertaining to the management of arbuscular mycorrhizae in agriculture. Agric Ecosyst Environ 60:197–2010
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Circular 347 (2nd edition). California Agricultural Experimental Station, Berkeley
Jansa J, Wiemken A, Frossard E (2006) The effects of agricultural practices on arbuscular mycorrhizal fungi. Geol Soc Lond Spec Publ 266:89–115
Jeffries P, Gianinazzi S, Perotto S, Turnau K, Barea J-M (2003) The contribution of arbuscular mycorrhizal fungi in sustainable maintenance of plant health and soil fertility. Biol Fertil Soils 37:1–16
Johnson NC, Copeland PJ, Crookston RK, Pfleger FL (1992) Mycorrhizae: possible explanation for yield decline with continuous corn and soybean. Agron J 84:387–390
Köhl L, Oehl F, van der Heijden MGA (2014) Agricultural practices indirectly influence plant productivity and ecosystem services through effects on soil biota. Ecol Appl 24:1842–1853
Lehman RM, Taheri WI, Osborne SL, Buyer JS, Douds DD Jr (2012) Fall cover cropping can increase arbuscular mycorrhizae in soils supporting intensive agricultural production. Appl Soil Ecol 61:300–304
Lekberg Y, Koide RT (2005) Is plant performance limited by abundance of arbuscular mycorrhizal fungi? A meta-analysis of studies published between 1988 and 2003. New Phytol 168:189–204
Mader P, Edenhofer S, Boller T, Wiemken A, Niggli U (2000) Arbuscular mycorrhizae in a long-term field trial comparing low-input (organic, biological) and high-input (conventional) farming systems in a crop rotation. Biol Fertil Soils 31:150–156
Martinez TN, Johnson NC (2010) Agricultural management influences propagule densities and functioning of arbuscular mycorrhizas in low- and high-input agroecosystems in arid environments. Appl Soil Ecol 46:300–306
McGonigle T (1988) A numerical analysis of published field trials with vesicular-arbuscular mycorrhizal fungi. Funct Ecol 2:473–478
McGonigle T, Miller M, Evans D, Fairchild G, Swan J (1990a) A new method which gives an objective measure of colonization of roots by vesicular—arbuscular mycorrhizal fungi. New Phytol 115:495–501
McGonigle TP, Evans DG, Miller MH (1990b) Effect of degree of soil disturbance on mycorrhizal colonization and phosphorus absorption by maize in growth chamber and field experiments. New Phytol 116:629–636
Miller MH (2000) Arbuscular mycorrhizae and the phosphorous nutrition of maize: a review of Guelph studies. Can J Plant Sci 80:47–52
Olsson PA (1999) Signature fatty acids provide tools for determination of the distribution and interactions of mycorrhizal fungi in soils. FEMS Microbiol Ecol 29:303–310
Pellegrino E, Öpik M, Bonari E, Ercoli L (2015) Responses of wheat to arbuscular mycorrhizal fungi: a meta-analysis of field studies from 1975 to 2013. Soil Biol Biochem 84:210–217
Philips JM, Hayman DS (1970) Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans Br Mycol Soc 55:158–160
Pikul JL, Osborne S, Ellsbury M, Riedell W (2007) Particulate organic matter and water-stable aggregation of soil under contrasting management. Soil Sci Soc Am J 71:766–776
Pinkart HC, Ringelberg DB, Piceno YM, MacNaughton SJ, White DC (2002) Biochemical approaches to biomass measurements and community structure analysis. In: Hurst CJ, Crawford RL, Knudsen GR, McInerney MJ, Stetzenbach LD (eds) Manual of environmental microbiology. ASM Press, Washington, D.C., pp 101–113
Porter WM (1979) The "most probable number" method for enumerating infective propagules of vesicular-arbuscular mycorrhizal fungi in soil. Austr J Soil Res 17:515–519
Rillig MC (2004) Arbuscular mycorrhizae and terrestrial ecosystems processes. Ecol Lett 7:740–754
Ryan MH, Graham JH (2018) Little evidence that farmers should consider abundance or diversity of arbuscular mycorrhizal fungi when managing crops. New Phytol 220:1092–1107. https://doi.org/10.1111/nph.15308
Ryan MH, Van Herwaarden AF, Angus JF, Kirkegaard JA (2005) Reduced growth of autumn-sown wheat in a low-P soil is associated with high colonisation by arbuscular mycorrhizal fungi. Plant Soil 270:275–286
Sharma MP, Buyer JS (2015) Comparison of biochemical and microscopic methods for quantification of arbuscular mycorrhizal fungi in soil and roots. Appl Soil Ecol 95:86–89
Smith SE, Read DJ (2008) Mycorrhizal Symbiosis, 3rd edn. Academic, London
Smith FA, Smith SE (2011) What is the significance of the arbuscular mycorrhizal colonization of many economically important crop plants. Plant Soil 348:63–79
Soliveres S, Van Der Plas F, Manning P, Prati D, Gossner MM, Renner SC, Alt F, Arndt H, Baumgartner V, Binkenstein J (2016) Biodiversity at multiple trophic levels is needed for ecosystem multifunctionality. Nature 536:456–459
Sylvia DM (1994) Vesicular-arbuscular mycorrhizal fungi. In: Weaver RW, Angle S, Bottomley P, Bezdicek D, Smith S, Tabatabai A, Wollum A (eds) Methods of soil analysis, part 2. Microbiological and biochemical properties, vol 5. Soil Science Society of America, Madison, pp 351–378
Treseder KK (2013) The extent of mycorrhizal colonization of roots and its influence on plant growth and phosphorus content. Plant Soil 371:1–13
Verbruggen E, Röling WFM, Gamper HA, Kowalchuk GA, Verhoef HA, van der Heijden MGA (2010) Positive effects of organic farming on below-ground mutualists: large-scale comparison of mycorrhizal fungal communities in agricultural soils. New Phytol 186:968–979
Verbruggen E, Kiers ET, Bakelaar PC, Röling WM, van der Heijden MGA (2012) Provision of contrasting ecosystem services by soil communities from different agricultural fields. Plant Soil 350:43–55
Wagg C, Bender SF, Widmer F, van der Heijden MGA (2014) Soil biodiversity and soil community composition determine ecosystem multifunctionality. Proc Natl Acad Sci 111:5266–5270
Woomer PL (1994) Most probable number counts. In: Weaver RW, Angle S, Bottomley P, Bezdicek D, Smith S, Tabatabai A, Wollum A (eds) Methods of soil analysis, part 2. Microbiological and biochemical properties, vol 5. Soil Science Society of America, Madison, pp 59–80
Acknowledgments
The research was conducted with the technical support of Sharon Nichols, Amy Christie, Kurt Dagel, and Chris Nelson.
Funding
This work was partially funded by the South Dakota Corn Utilization Council.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclaimer
Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Figure S1
Amounts (nmol g−1) of the NLFA biomarker, C16:1cis11, for the eight cover crop treatments (mean ± one standard error; n = 4 plots). A. Fall-collected soils in 2009 and 2010. Bars topped by the same letter do not differ significantly at P ≤ 0.05 by protected t-test; pairwise comparisons are indicated per year with 2009 in lower case and 2010 in upper case letters. Data in panel A were previously published (Lehman et al. 2012). B. Spring-collected soils (2010) following first fall cover crop planting. There were no significant main effects of treatments, therefore no pairwise comparisons were performed. (PPTX 230 kb)
Table S1
(DOCX 14.1 kb)
Table S2
(DOCX 13 kb)
Table S3
(DOCX 12 kb)
Rights and permissions
About this article
Cite this article
Lehman, R.M., Osborne, S.L., Taheri, W.I. et al. Comparative measurements of arbuscular mycorrhizal fungal responses to agricultural management practices. Mycorrhiza 29, 227–235 (2019). https://doi.org/10.1007/s00572-019-00884-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-019-00884-4