Abstract
Purpose
Oral alterations are frequently observed in patients undergoing palliative care and are linked to the direct or indirect effects of the primary medical condition, comorbidities and medical management, leading to oral pain, impacting oral intake, and affecting quality of life. This systematic review aims to assess the prevalence of oral disease in palliative care patients.
Methods
The protocol was registered at the PROSPERO database, and a systematic review of the literature was performed based on the PRISMA statement. A thorough evaluation of studies from five databases and gray literature was conducted. The risk of bias in each study was assessed using the Joanna Briggs Institute checklist for cross-sectional and case-control studies. A quantitative analysis was conducted on five studies using meta-analysis, and the degree of certainty in the evidence was determined using the GRADE tool.
Results
The sample consisted of 2,502 patients, with a slight male predominance (50.43%). The average age was 66.92 years. The prevalence of oral diseases among palliative care patients was as follows: caries 32% (95% CI, 0.11-0.56; I2 = 93%), and oral candidiasis 17% (95% CI,0.11-0.25; I2 = 74%). Gingivitis and stomatitis were also reported, but with less frequency.
Conclusion
Dental intervention should take place as early as possible, ideally from the time of the patient's initial admission to palliative care, with regular monitoring of oral health. This approach can enhance the patient's comfort and quality of life and help prevent more severe complications in the future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Palliative care is defined by the World Health Organization as care that enhances the quality of life for patients (adults and children) and their families who deal with issues related to life-threatening diseases [1]. Palliative care aims to promote early diagnosis and treatment of suffering from a biological, psychological, and spiritual perspective, avoiding and alleviating pain [1, 2]. The involvement of a multidisciplinary team is essential to providing prevention and adequate treatment of the symptoms presented by patients with a life-threatening illness. The purpose of palliative care is to improve the quality of life of the patient and their caregivers and significant others, including family [2, 3]. Patients with serious medical conditions, such as advanced organ failure (e.g., cardiomyopathy and chronic obstructive pulmonary disease (COPD)), neurological or other health conditions, and terminal cancer, are among the patients need of this type of care [4, 5].
Oral alterations are frequently observed in patients undergoing palliative care, and they are associated with the direct or indirect effects of the primary medical condition [5, 6]. Many therapies are utilized to treat the primary diseases that lead these patients to palliative care, and these oral alterations can be, direct or indirect, related to these therapies [6,7,8]. For example, some patients are treated with chemotherapy and/or radiotherapy for their primary neoplastic disease. Additionally, the management of patients who are on palliative care due to other diseases, such as is based on medications, such as opioids or antibiotics [6,7,8]. These therapies used for the primary disease can lead to the onset of a plethora of oral conditions, such as xerostomia, oral candidiasis, caries, mucositis, stomatitis, ulceration, impaired chewing function, taste disturbances, sore and dry lips, oral pain, and dysgeusia [9, 10]. Importantly, these conditions can affect the overall quality of life for these individuals, which may result in multidimensional (biological, psychological, and spiritual) suffering [9, 10].
Disease can be defined as a state of maladaptation to the physical, psychological, or social environment in which the individual feels unwell (has symptoms) and/or has objectively evident organic changes (clinical signs) [11]. However, the definition of disease can vary [12, 13]. Oral manifestations are symptoms or conditions in the oral cavity that can arise due to disease affecting other organs [14, 15]. Few studies in the literature have mapped the epidemiological profile and prevalence of these oral alterations in palliative care patients. Although there is a systematic review [16] that synthesizes the evidence on oral conditions in palliative care patients, this systematic review with meta-analysis aims to assess the prevalence of oral disease in palliative care patients.
Methods
Eligibility criteria
The PECOS acronym (Population, Exposure, Comparison, Outcomes, Studies), in accordance with Preferred Reporting Items for Systematic Reviews and Meta Analyses (PRISMA) recommendations [17] (Supplemental Table 1), was employed to formulate a focused research question and establish the inclusion criteria for this study, wherein: (P) patients under palliative care; (E) palliative care; (C) not applicable; (O) prevalence of oral diseases in a patient undergoing palliative care; (S) case–control, cross-sectional and cohort. Studies that did not align with the PECOS approach were excluded from the review. The following studies were excluded: (i) did not describe oral diseases in palliative care patients; (ii) reviews; (iii) clinical trials; (iv) case reports; (v) protocols; (vi) brief communications; (vii) personal opinions; (viii) letters; (ix) conference abstracts; (x) laboratory research; (xi) full texts were not available even after contacting the corresponding author, and (xii) published in a language other than English, Spanish or Portuguese.
Information sources and search strategy
The date of the electronic search was February 9, 2023, and updated on January 19, 2024. Five electronic databases were used in the search strategy: PubMed (via MEDLINE), Scopus, LILACS, Web of Science, and Embase. Additionally, ProQuest Dissertation & Theses Global and Google Scholar were reviewed as gray literature (Supplemental Table 2). To avoid overlooking studies during the initial search, an additional query was conducted on the list of references.
Selection process
Reference management software (Rayyan QCRIR) [18] was used to eliminate any duplicate articles after database scanning. The selection process consisted of two phases. During the initial phase, two reviewers (M.S. and E.S.S) independently evaluated the titles and abstracts of all articles identified from databases using Rayyan software. During the second phase, the authors read the full text of the articles and excluded those that did not meet the inclusion criteria. Exclusion reasons were recorded and can be found in Supplemental Table 3.
Data collection process and data items
The two independent authors (M.S. and E.S.S) collected the data. Authors, publication year, country, study type, total number of patients, underlying disease, proportion of women, mean age, age range, oral diseases, and conclusions were among the data extracted from the included studies. In case of disagreement, a third author was consulted, and all disagreements were resolved by consensus.
Study risk of bias assessment
The methodological quality and risk of bias of the included research were assessed using two critical appraisal checklists from the Joanna Briggs Institute. These checklists corresponded to analytical cross-sectional studies and case–control studies [19]. Two independent authors (MS and ESS) completed this analysis. If there was any dispute, the third author (ARSS) was consulted. The risk of bias was determined based on the number of 'yes' responses to each question. Studies with more than 70% 'yes' were classified as having a low risk of bias. Studies with 50%-69% 'yes' were considered to have a moderate risk of bias, while those with less than 49% 'yes' were classified as having a high risk of bias.
Effect measures
The primary objective was to evaluate the prevalence of oral diseases among cancer patients under palliative care. Furthermore, an analysis of the demographic profile and clinical characteristics of these individuals was completed.
Synthesis methods
The data reported in the included studies on the outcomes assessed were grouped and compared for qualitative synthesis. The meta-analyses of proportions were performed using the MetaXL 5.3 (EpiGear International, Queensland, Australia) add-in for Microsoft Excel software. The random effects model was employed due to anticipated heterogeneity (I2). Heterogeneity is considered high when (> 50%) and low when (≤ 50%) [20].
Certainty assessment
The quality of evidence was assessed using GRADE (Grading of Recommendation, Assessment, Development, and Evaluation). All studies included in the meta-analysis were evaluated based on study design, risk of bias, inconsistency, indirectness, imprecision, and other considerations. The evidence was characterized as moderate or very low [21].
Results
Study selection
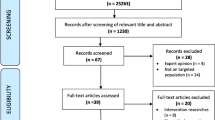
The literature search initially identified 1,098 records in the five databases. After removing duplicates, 974 records were screened for title and abstract, and 91 studies were selected for full-text reading. In the other sources, 107 studies were identified and 17 were evaluated for full-text reading.
Thirteen studies were selected for qualitative and quantitative synthesis after applying the inclusion and exclusion criteria. Figure 1 illustrates the study selection process.
Study characteristics
The studies covered four continents: North America [5, 22, 23] (n = 3), South America [24] (n = 1), Europe [25,26,27,28,29,30] (n = 6), and Asia [31,32,33] (n = 3) (Fig. 2). The data collection period for the studies was from 1989 to 2022. They were characterized as retrospective studies [24, 31, 33] (n = 3) and prospective studies [5, 22, 23, 25,26,27,28,29,30, 32] (n = 10), being case–control [31, 32] (n = 2) and cross-sectional design [5, 22,23,24,25,26,27,28,29,30, 32] (n = 11). The clinical and epidemiological characteristics of the included studies are described in Table 1.
Risk of bias in studies
Most studies had a low risk of bias. Among the eleven cross-sectional studies, two had a moderate risk of bias [26, 29] and nine had a low risk of bias [5, 22,23,24,25, 27, 28, 30, 32] (Supplemental Figure S1A). One case–control study had a low risk of bias [31], while the other had a moderate risk of bias [33] (Supplemental Figure S1B). Supplemental Table 4 provides a complete assessment of the risk of bias.
Results of individual studies
Cancer was the disease most often described as a factor inducing the palliative state. The six most frequent cancer were lung [22, 23, 26,27,28,29, 31, 33] with 428 patients, breast with 179 patients [22, 24,25,26,27,28, 31, 33], gastrointestinal with 766 patients [22, 24,25,26,27,28,29, 33], prostate [24, 28, 29, 33] with 42 patients, head and neck [24, 25, 27, 29, 31] with 69 patients, and hematological [25, 28, 33] with 88 patients. Non-cancer was reported in 12 patients with organ failure (e.g., cardiomyopathic) [5], 8 patients with neurological or other health conditions [5], 16 patients with COPD [26], 9 patients with end stage organ failure [30]. One study used the term without specifying what type of cancer it is [5], eight studies [5, 22, 24, 26,27,28, 31, 33] described “other” in 193 patients without specifying what type of cancer it is. Only one study [24] reported “not informed” for 1 patient.
Results of syntheses
The total sample size for this systematic review comprised 2,502 patients, with a range from 49 [5] to 669 [28]. Notably, there was a minor preference for males, constituting 1,262 of the sample, while the age range spanned from 21 to 112 years, with a mean age of 66.92 years.
The oral diseases reported in this review were oral candidiasis, which was reported in four studies [24, 29, 31, 33] with 99 patients. Three studies [5, 29, 33] reported the presence of caries in 78 patients. One study [29] reported gingivitis in 11 patients. Only one study [22] described "oral diseases" in 10 patients without specifying the disease. Nakajima [31] described the presence of stomatitis in his study with a total of 38 patients. The prevalence of each disease was individually analyzed. A meta-analysis of three studies [5, 29, 33] revealed that caries showed a prevalence of 32% in three studies [5, 29, 33] (95% CI, 0.11, 0.56; I2 = 93%), and oral candidiasis had a prevalence of 17% in four studies [24, 29, 31, 33] (95% CI, 0.11, 0.25; I2 = 74%) (Fig. 3).
The oral manifestations reported in the reviewed studies encompass a range of oral conditions and orofacial symptoms. Xerostomia, or dry mouth, was documented in ten studies, involving a total of 1,253 patients [5, 23, 25,26,27,28,29,30,31, 33]. Mucositis was observed in two studies [24, 28], affecting 157 patients. Dental pain/toothache pain/pain in teeth was reported in three studies [5, 25, 32] among 41 patients. Taste changes/dysgeusia were noted in seven studies [5, 23,24,25,26, 29, 30], involving 416 patients. Oral pain [5], mouth pain [29], intraoral pain [23], mouth discomfort/pain [25] without a precisely specified origin, was described in four studies [5, 23, 25, 29] with 222 patients. Periodontal abscess was mentioned in one study [5], affecting a single patient. A burning sensation in the mouth was reported in one study [25] with 24 patients, and tooth sensitivity was noted in 52 patients. Gingival inflammation was identified in one study [33] involving 48 patients. One study described teeth (caries, fracture, broken root stumps) in 41 patients [32] and one study teeth problems (plaque or debris) in 42 patients [30].
Lesions on the lips, characterized as dry, red, swollen, ulcerated, cracked, or fissured, were detailed in four studies [23, 25, 30, 32], involving 424 patients. Lesions on the tongue, including coated, red and/or white patches, loss of papillae with a shiny appearance, ulcerated, sloughing, or inflamed, were described in four studies [25, 30, 32, 33], affecting 300 patients. Gingival lesions, such as swelling, bleeding, white/red patches, ulcers, and redness under dentures, were reported in three studies [5, 32, 33] with 35 patients. Additionally, one study [25] noted the presence of oral ulcers in 42 patients, bleeding from mouth in 19 patients, bad breath in 41 patients, and bleeding spots in 25 patients (Table 2).
Certainty of evidence (by Grading of Recommendations Assessment, Development, and Evaluation)
In observational studies, the level of certainty can be classified as "low", "very low", "moderate" or "high". Due to the high degree of heterogeneity among studies and small sample sizes, the level of certainty of evidence regarding the prevalence of oral diseases, caries and gingivitis was very low. However, moderate certainty was observed for the prevalence of oral candidiasis (Supplemental Table 5).
Discussion
This systematic review was conducted to evaluate the prevalence of oral diseases in patients receiving palliative care. A total of 2,502 patients were included, with an average age of 66.9 years. Cancer was the most common condition observed among the palliative care patients included in this study. The prevalence of dental caries was 32%, and oral candidiasis was 17%. Additionally, the most common oral manifestation was xerostomia, present in 1,253 patients. It is of utmost importance that the care team remains vigilant for any oral manifestations to ensure early intervention and thus prevent future complications.
Palliative care aims to improve the quality of life for patients and their families who are facing serious diseases such as terminal cancer, neurological disorders, heart disease or other health problems that are life-threatening [14, 34]. Palliative care has experienced significant growth and change in recent decades, including new models of care and increased public and professional consciousness [14, 34]. It is not uncommon to find descriptions of oral manifestations in palliative care patients in the literature, so understanding the prevalence of oral disease is essential to improve dissemination of information, especially as the oral condition of many patients is often neglected by their care teams [7, 35].
The treatment of patients in palliative care should be based on an integrated and holistic approach, considering the needs of both patients and their families throughout the course of the illness [36]. Radiotherapy and chemotherapy are commonly employed in cancer treatment and can lead to side effects in the oral cavity [37, 38]. One of the side effects is mucositis, an acute complication characterized by ulceration, severe pain, burning sensation and discomfort, often resulting in challenges for the patient in consuming food and liquids. In addition, inflammation, bleeding and infections can occur, with oral candidiasis, caused by Candida albicans, being one of the most common infections that can appear in and coexist with mucositis lesions. Loss or alteration of taste is another frequent manifestation after this type of treatment [28, 37,38,39,40]. Chronic complications can also arise or persist for months and years after the end of treatment, including xerostomia (dry mouth) and tooth decay [37,38,39,40]. It is important to remember that, although cancer is the most common disease in palliative care patients, not all patients will receive radiotherapy or chemotherapy, as treatment will be based on the individual needs of each patient [41].
There is a growing recognition of the significance of palliative care not just in oncology but across various medical fields. This is especially clear in the treatment of many neurological diseases, which are often chronic, incurable, and autonomy-impairing [42]. Patients with heart failure, pulmonary diseases, nephrological disorders, and other conditions can also require palliative care [43, 44]. Various medications, including anticholinergics, antihypertensives, antiparkinsonians, bronchodilators, and diuretics, used to treat these underlying conditions, can lead to oral manifestations, with xerostomia being common. These drugs primarily work by inhibiting signaling pathways within salivary tissues and reducing the fluid output of the glands [43, 45]. According to the National Cancer Institute at the National Institutes of Health in the United States, 80% of patients undergoing myeloablative chemotherapy experience oral complications, and palliative drugs such as bisphosphonates and analgesics are linked to oral mucositis and taste disturbances [14].
Approximately 69% of patients in palliative care corresponds to older adults [46]. Towards end of life, approximately 50% to 76% of individuals become unable to perform oral self-care [30, 47]. Overall, the oral hygiene of these patients tends to be suboptimal, marked by high rates of plaque on the teeth, thereby elevating the risk of caries and periodontal disease and impacting quality of life [30, 48].
Xerostomia is a common condition, characterized by the dryness of the oral cavity due to inadequate saliva secretion or a complete absence of saliva, is associated with various causes such as the use of medications, cancer treatments, autoimmune diseases, dehydration, advanced age, among other factors [7, 49]. A decrease in saliva production associated with xerostomia increases the risk of developing diseases such as oral candidiasis and tooth decay, which can make it difficult to chew and swallow, and can lead to changes in taste and other conditions [4, 50, 51]. The general prevalence of dry mouth in the population varies from 5.5% to 46%, depending on gender and age, with some studies indicating a higher prevalence of 78% or 81% in terminal cancer patients [29, 52].
The host's overall health is generally considered the etiological factor in the development of oral candidiasis [53]. Hyposalivation, diabetes, specific medications, and reduced nutritional status are strongly linked to the occurrence of this disease [54]. More than 17 species of Candida can cause infections in the oral mucosa, with Candida albicans being the most frequently identified pathogen [53]. Clinically, oral candidiasis can be characterized as pseudomembranous, atrophic (erythematous), or hyperplastic. The erythematous type is the most common, causing a reddish lesion that mainly affects the tongue and palate, although other areas of the oral mucosa can also be affected. Burning in the mouth is one of the symptoms reported by patients with erythematous candidiasis [9]. Oral candidiasis in our study had a prevalence of 17%, falling within the reported range in the literature, which varies between 8 and 94% [55, 56].
Caries is a multifactorial disease and can be related to xerostomia, radiation, poor hygiene and cariogenic diet [57]. Caries is a bacterial disease characterized by demineralization of the enamel and dentin. Initially asymptomatic, if left untreated, caries can develop into reversible or irreversible pulpitis, which causes severe pain. Untreated pulpitis can develop into an abscess, which can lead to more serious complications [57]. Differently, radiation caries is mainly due to damage to the salivary glands, resulting in hyposalivation and changes in the oral microflora. This disease becomes evident three months after irradiation and can have a rapid progression and usually no acute pain [58]. Hong et al. [59] reported a prevalence of dental caries of 21% in patients treated with chemotherapy and radiotherapy, while our study showed a prevalence of 32% for caries.
Described in 11 patients in this study, gingivitis can be divided into plaque-induced and non-plaque-induced gingivitis. Plaque-induced gingivitis is one of the most common inflammatory diseases in humans and is characterized by erythema, gingival swelling, and absence of periodontal loss. It is usually painless, rarely associated with spontaneous bleeding and often goes unnoticed by patients who are unaware of the disease. Gingivitis is considered a precursor to periodontitis, which is why it is so important to treat it [60, 61]. The literature reports indicating a prevalence of 20% of gingivitis in patients undergoing chemotherapy and radiotherapy [59, 62].
It is crucial to recognize limitations in this systematic review. Firstly, there is a discrepancy among studies in describing the manifestations of oral diseases, often without exploring into the specific type of disease or condition to which the reported lesion may refer. Secondly, some studies lacked detailed information about the pre-existing disease. In addition, the heterogeneity of the studies, in terms of variations in individual criteria utilized to describe the lesions precluded an extension of the analysis through meta-analysis.
Conclusions
Individuals receiving palliative care often experience oral diseases that significantly compromise their physical, psychological, and spiritual well-being. Comprehensive dental assessment and timely intervention should take place as early as possible, ideally from the patient's initial admission to palliative care, along with regular monitoring of oral health. This strategy will enhance the patient's comfort and quality of life, as well as prevent potential complications. It would be important to conduct studies with larger sample sizes that thoroughly document and standardize oral manifestations in palliative care patients would be crucial, enabling the development of more precise approaches for these individuals.
Other information
Protocol and registration
This systematic review is registered with PROSPERO (International Prospective Register of Systematic Reviews) under registration number CRD42023395301. PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) was used as the foundation for the review.
Large Language Models
The content of this article is entirely original. ChatGPT was used to assist in organizing the text's structure in English.
Data availability
No datasets were generated or analysed during the current study.
References
World Health Organization (2002) National cancer control programmes: policies and managerial guidelines. https://www.who.int/publications/i/item/national-cancer-control-programmes Accessed August 28, 2023
O'Neill B, Fallon M (1997) ABC of palliative care. Principles of palliative care and pain control. BMJ 315(7111):801–4. https://doi.org/10.1136/bmj.315.7111.801
Rome RB, Luminais HH, Bourgeois DA, Blais CM (2011) The role of palliative care at the end of life. Ochsner J Winter 11(4):348–352
Agarwal R, Epstein AS (2017) The Role of Palliative Care in Oncology. Semin Intervent Radiol 34(4):307–312. https://doi.org/10.1055/s-0037-1608702
Chen X, D’Souza V, Thomsen TA, Gilbertson-White S, Madiloggovit J, Pendleton C, Munjal A, Xianjin X (2021) Oral health in adult patients receiving palliative care: a Mixed Method Study. Am J Hosp Palliat Med 38(12):1516–1525. https://doi.org/10.1177/10499091211007449
Saini R, Pp M, Shete S et al (2009) Dental expression and role in palliative treatment. Indian J Palliat Care 15(1):26–29. https://doi.org/10.4103/0973-1075.53508
Kvalheim SF, Marthinussen IMC, Berg E, Strand GV (2022) Dry mouth in the seriously ill and dying. Tidsskr Nor Laegeforen 142(3). https://doi.org/10.4045/tidsskr.21.0232
World Health Organization (2018) Integrating palliative care and symptom relief into primary health care: a WHO guide for planners, implementers and managers. https://iris.who.int/handle/10665/274559. Accessed 28 Aug 2023
Silva ARP, Bodanezi AV, Chrun ES, Lisboa ML, de Camargo AR, Munhoz EA (2023) Palliative oral care in terminal cancer patients: Integrated review. World J Clin Cases 11(13):2966–2980. https://doi.org/10.12998/wjcc.v11.i13.2966
Meneguin S, Matos TDS, Ferreira MLDSM (2018) Perception of cancer patients in palliative care about quality of life. Rev Bras Enferm 71(4):1998–2004. https://doi.org/10.1590/0034-7167-2017-0360
Funkhouser WK (2009) Pathology: The Clinical Description of Human Disease. Molecular Pathology. pp 197–207. https://doi.org/10.1016/B978-0-12-374419-7.00011-1
Kottow MH (1980) A medical definition of disease. Med Hypotheses 6(2):209–213. https://doi.org/10.1016/0306-9877(80)90085-7
Eisenberg L (1977) Disease and illness. Distinctions between professional and popular ideas of sickness. Cult Med Psychiatry 1(1):9–23. https://doi.org/10.1007/BF00114808
https://www.nature.com/subjects/oral-manifestations. Accessed June 15, 2023
Porter S, Mercadente V, Fedele S (2018) Oral manifestations of systemic disease. BDJ Team 5:18012. https://doi.org/10.1038/bdjteam.2018.12
Venkatasalu MR, Murang ZR, Ramasamy DTR, Dhaliwal JS (2020) Oral health problems among palliative and terminally ill patients: an integrated systematic review. BMC Oral Health 20(1):79. https://doi.org/10.1186/s12903-020-01075-w
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A (2016) Rayyan-a web and mobile app for systematic reviews. Syst Rev 5(1):210. https://doi.org/10.1186/s13643-016-0384-4
Moola S, Munn Z, Sears K, Sfetcu R, Currie M, Lisy K, Tufanaru C, Qureshi R, Mattis P, Mu P (2015) Conducting systematic reviews of association (etiology): The Joanna Briggs Institute’s approach. Int J Evid Based Health 13(3):163–169. https://doi.org/10.1097/XEB.0000000000000064
Migliavaca CB, Stein C, Colpani V, Barker TH, Ziegelmann PK, Munn Z, Falavigna M (2022) Meta-analysis of prevalence: I2 statistic and how to deal with heterogeneity. Res Synth Methods 13(3):363–367. https://doi.org/10.1002/jrsm.1547
Balshem H, Helfand M, Schünemann HJ et al (2011) GRADE guidelines: 3 Rating the quality of evidence. J Clin Epidemiol 64(4):401–406. https://doi.org/10.1016/j.jclinepi.2010.07.015
Burge FI (1993) Dehydration symptoms of palliative care cancer patients. J Pain Symptom Manage 8(7):454–464. https://doi.org/10.1016/0885-3924(93)90188-2
Fischer DJ, Epstein JB, Yao Y, Wilkie DJ (2014) Oral health conditions affect functional and social activities of terminally ill cancer patients. Support Care Cancer 22(3):803–810. https://doi.org/10.1007/s00520-013-2037-7
da Fonseca Orcina B, Jaccottet CMG, Savian MCB (2021) Prevalence of Oral Manifestations in Cancer Patients Enrolled in a Home Care Program in the City of Pelotas-RS. Revista Brasileira de Cancerologia 67(2):081184. https://doi.org/10.32635/2176-9745.RBC.2021v67n2.1184
Davies A, Buchanan A, Todd J, Gregory A, Batsari KM (2021) Oral symptoms in patients with advanced cancer: an observational study using a novel oral symptom assessment scale. Support Care Cancer 29(8):4357–4364. https://doi.org/10.1007/s00520-020-05903-1
Fleming M, Craigs CL, Bennett MI (2020) Palliative care assessment of dry mouth: what matters most to patients with advanced disease? Support Care Cancer 28(3):1121–1129. https://doi.org/10.1007/s00520-019-04908-9
Maltoni M, Pirovano M, Scarpi E, Marinari M, Indelli M, Arnoldi E, Amadori D (1995) Prediction of survival of patients terminally III with cancer. Results of an Italian prospective multicentric study. Cancer 75(10):2613–2622. https://doi.org/10.1002/1097-0142(19950515)75:10%3c2613::aid-cncr2820751032%3e3.0.co;2-1
Mercadante S, Aielli F, Adile C, Ferrera P, Valle A, Fusco F, Porzio G (2015) Prevalence of oral mucositis, dry mouth, and dysphagia in advanced cancer patients. Support Care Cancer 23(11):3249–3255. https://doi.org/10.1007/s00520-015-2720-y
Wilberg P, Hjermstad MJ, Ottesen S, Herlofson BB (2012) Oral health is an important issue in end-of-life cancer care. Support Care Cancer 20(12):3115–3122. https://doi.org/10.1007/s00520-012-1441-8
Magnani C, Mastroianni C, Giannarelli D, Stefanelli MC, Di Cienzo V, Valerioti T, Casale G (2019) Oral hygiene care in patients with advanced disease: an essential measure to improve oral cavity conditions and symptom management. Am J Hosp Palliat Care 36(9):815–819. https://doi.org/10.1177/1049909119829411
Nakajima N (2017) Characteristics of oral problems and effects of oral care in terminally ill patients with cancer. American Journal of Hospice and Palliative Medicine® 34(5):430–434. https://doi.org/10.1177/1049909116633063
Amran NAH, Rahman HA, Awang AMZBH, Murang ZR, Nara A, Teo SP, Dhaliwal JS (2022) Assessment of oral health problems and oral hygiene practices among palliative care patients using a new tool: A pilot study. Palliative Medicine in Practice 16(1):49–58. https://doi.org/10.5603/PMPI.2021.0030
Matsuo K, Watanabe R, Kanamori D et al (2016) Associations between oral complications and days to death in palliative care patients. Support Care Cancer 24(1):157–161. https://doi.org/10.1007/s00520-015-2759-9
Kelley AS, Morrison RS (2015) Palliative Care for the Seriously Ill. N Engl J Med 373(8):747–755. https://doi.org/10.1056/NEJMra1404684
Wiseman MA (2000) Palliative care dentistry. Gerodontology 17(1):49–51. https://doi.org/10.1111/j.1741-2358.2000.00049.x
Henson LA, Maddocks M, Evans C, Davidson M, Hicks S, Higginson IJ (2020Mar 20) Palliative Care and the Management of Common Distressing Symptoms in Advanced Cancer: Pain, Breathlessness, Nausea and Vomiting, and Fatigue. J Clin Oncol 38(9):905–914. https://doi.org/10.1200/JCO.19.00470
Soysa NS, Samaranayake LP, Ellepola NA (2004) Cytotoxic drugs, radiotherapy and oral candidiasis. Oral Oncol 40(10):971–978. https://doi.org/10.1016/j.oraloncology.2003.12.013
Specht L (2002) Oral complications in the head and neck radiation patient. Introduction and scope of the problem. Support Care Cancer 10(1):36–9. https://doi.org/10.1007/s005200100283
de Oliveira Lula EC, de Oliveira Lula CE, Alves CM, Lopes FF, Pereira AL (2007) Chemotherapy-induced oral complications in leukemic patients. Int J Pediatr Otorhinolaryngol 71(11):1681–1685. https://doi.org/10.1016/j.ijporl.2007.07.006
McCarthy GM, Skillings JR (1992) Orofacial complications of chemotherapy for breast cancer. Oral Surg Oral Med Oral Pathol 74(2):172–178. https://doi.org/10.1016/0030-4220(92)90378-4
Siouta N, Beek K, Preston N, Hasselaar J, Hughes S, Payne S et al (2016) Towards integration of palliative care in patients with chronic heart failure and chronic obstructive pulmonary disease: a systematic literature review of European guidelines and pathways. BMC Palliat Care. https://doi.org/10.1186/s12904-016-0089-4
Provinciali L, Carlini G, Tarquini D et al (2016) Need for palliative care for neurological diseases. Neurol Sci 37(10):1581–1587. https://doi.org/10.1007/s10072-016-2614-x
Pugian Jardim P, Dantas Cavalcanti AC, Santos Borges A, Peclat Flores PV, Achão Rosa C (2022) Signs and symptoms of patients with heart failure in palliative care: scoping review. Anna Nery School Journal of Nursing 26:20220064. https://doi.org/10.1590/2177-9465-EAN-2022-0064en
Voltz R, Borasio G (1997) Palliative therapy in the terminal stage of neurological disease. J Neurol 244(Suppl 4):S2–S10. https://doi.org/10.1007/PL00007721
Cassolato S, Turnbull R (2003) Xerostomia: clinical aspects and treatment. Gerodontology 20(2):64–77. https://doi.org/10.1111/J.1741-2358.2003.00064.X
Connor SR (2020) Global Atlas of Palliative Care, 2nd edn. World Health Organization, London, UK
Chen X, Chen H, Douglas C, Preisser JS, Shuman SK (2013) Dental treatment intensity in frail older adults in the last year of life. J Am Dent Assoc 144(11):1234–1242. https://doi.org/10.14219/jada.archive.2013.0051
Vargas J, Hernandez M, Silvestri C, Jiménez O, Guevara N, Carballo M et al (2006) Brain abscess due to Arcanobacterium haemolyticum after dental extraction. Clin Infect Dis 42:1810–1811. https://doi.org/10.1086/504436
Plemons JM, Al-Hashimi I, Marek CL (2014) American Dental Association Council on Scientific Affairs. Managing xerostomia and salivary gland hypofunction: executive summary of a report from the American Dental Association Council on Scientific Affairs. J Am Dent Assoc 145(8):867–73. https://doi.org/10.14219/jada.2014.44
Phongtankuel V, Meador L, Adelman RD et al (2018) Multicomponent Palliative Care Interventions in Advanced Chronic Diseases: A Systematic Review. Am J Hosp Palliat Med 35(1):173–183. https://doi.org/10.1177/1049909116674669
Guggenheimer J, Moore PA (2003) Xerostomia: etiology, recognition and treatment. J Am Dent Assoc 134(1):61–69. https://doi.org/10.14219/jada.archive.2003.0018
Temel JS, Greer JA, Muzikansky A, Gallagher ER, Admane S, Jackson VA, Dahlin CM, Blinderman CD, Jacobsen J, Pirl WF, Billings JA, Lynch TJ (2010) Early palliative care for patients with metastatic non-small-cell lung cancer. N Engl J Med 363:733–742. https://doi.org/10.1056/NEJMoa1000678
Lu SY (2021) Oral Candidosis: Pathophysiology and Best Practice for Diagnosis, Classification, and Successful Management. J Fungi (Basel) 7(7):555. https://doi.org/10.3390/jof7070555
Pedersen AML, Sørensen CE, Proctor GB, Carpenter GH, Ekström J (2018) Salivary secretion in health and disease. J Oral Rehabil 45(9):730–746. https://doi.org/10.1111/joor.12664
Davies AN (1997) The management of xerostomia: a review. Eur J Cancer Care (Engl) 6(3):209–214. https://doi.org/10.1046/j.1365-2354.1997.00036.x
Davies AN, Brailsford SR, Beighton D (2006) Oral candidosis in patients with advanced cancer. Oral Oncol 42(7):698–702. https://doi.org/10.1016/j.oraloncology.2005.11.010
Douglass AB, Douglass JM (2003) Common dental emergencies. Am Fam Physician 67(3):511–516
Vissink A, Jansma J, Spijkervet FK, Burlage FR, Coppes RP (2003) Oral sequelae of head and neck radiotherapy. Crit Rev Oral Biol Med 14(3):199–212. https://doi.org/10.1177/154411130301400305
Hong CHL, Napeñas JJ, Hodgson BD et al (2010) A systematic review of dental disease in patients undergoing cancer therapy. Support Care Cancer 18(8):1007–1021. https://doi.org/10.1007/s00520-010-0873-2
Holmstrup P, Plemons J, Meyle J (2018) Non-plaque-induced gingival diseases. J Clin Periodontol 45(Suppl 20):S28–S43. https://doi.org/10.1111/jcpe.12938
Trombelli L, Farina R, Silva CO, Tatakis DN (2018) Plaque-induced gingivitis: Case definition and diagnostic considerations. J Clin Periodontol 45(Suppl 20):S44–S67. https://doi.org/10.1111/jcpe.12939
Navarrete-Reyes AP et al (2023) Oral health in older adults with cancer. Geriatr, Gerontol Aging 17:1–12. https://doi.org/10.53886/gga.e0230016
Funding
This work was supported by Coordination for the Improvement of Higher Education Personnel (CAPES/PROEX) with finance code 001 and received support from the Sao Paulo Research Foundation (FAPESP, Brazil) under grant number 2019/09692–9.
Author information
Authors and Affiliations
Contributions
M.S – methodology, formal analysis, investigation, data curation, writing – original draft, writing—review and editing, visualization, project administration.
E.S.S – conceptualization, methodology, formal analysis, investigation, data curation, writing – review and editing, visualization, supervision, project administration.
C.M.P – formal analysis, investigation, writing—review and editing, visualization.
J.B.E – writing—review and editing, visualization, validation.
A.R.S–S – conceptualization, methodology, project administration, writing—review and editing, visualization, supervision, project administration, resources, validation.
L.P.K – writing—review and editing, visualization, supervision, validation.
All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study represents a systematic review of the literature. The Research Ethics Committee from Piracicaba Dental School, University of Campinas, Brazil, has confirmed that ethical approval is unnecessary for this investigation.
Consent to participate
As this study is a systematic review of the literature, it did not involve the inclusion of human subjects. Consequently, the absence of informed consent from patients for study participation is justified.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Silva, M., Santos, E.S., Pedroso, C.M. et al. Prevalence of oral diseases in patients under palliative care: a systematic review and meta-analysis. Support Care Cancer 32, 607 (2024). https://doi.org/10.1007/s00520-024-08723-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-024-08723-9