Abstract
Purpose
The purpose of this study is to evaluate the impact of fecal extended-spectrum β-lactamase-producing Escherichia coli (ESBL-EC) colonization for bloodstream infection (BSI), clinical outcome, and costs in patients with hematologic malignancies (HM) and severe neutropenia.
Methods
This is a cohort study, carried out at a cancer-referral hospital. The study population comprises patients with HM, hospitalized prior to administration of the first chemotherapy cycle. A stool culture was taken during the first 48 h; they were grouped as colonized by ESBL-EC or non-ESBL-EC. Patients were followed upon completion of chemotherapy or death. The sum of the days of antibiotics and the length of stay of all hospitalizations in the different cycles of chemotherapy were recorded.
Results
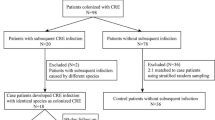
We included 126 patients with a recent diagnosis of HM, grouped as 63 patients colonized by ESBL-EC and 63 colonized by non-ESBL-EC, aged 42 ± 16 years old, 78 males (62 %). BSI by ESBL-EC developed in 14 patients (22.2 %) colonized by the same strain and in 5 (7.9 %) in the group colonized with non-ESBL-EC. BSI by non-ESBL-EC was observed in 3 patients (4.7 %) colonized by ESBL-EC and in 17 (26.9 %) patients colonized by non-ESBL-EC. Colonization with ESBL-EC increased the risk of BSI by the same strain (relative risk (RR) = 3.4, 95 % confidence interval (95 % CI) 1.5–7.8, p = 0.001), shorter time to death (74 ± 62 vs. 95 ± 83 days, p < 0.001), longer hospital stay (64 ± 39 vs. 48 ± 32 days, p = 0.01), and higher infection-related costs ($6528 ± $4348 vs. $4722 ± $3173, p = 0.01). There was no difference in overall mortality between both groups.
Conclusions
Fecal colonization by ESBL-EC is associated with increased risk of BSI by this strain, longer hospital stay, and higher related costs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Escherichia coli (EC) is a Gram-negative bacterium that acquires resistance to β-lactam antibiotics mainly through the production of extended-spectrum beta-lactamases (ESBL), while ESBL-encoding plasmids frequently bear resistance genes for other antibiotic classes [1, 2].
Early recognition of patients who are at risk for infection with ESBL-producing bacteria is necessary in order to guide empirical treatment for diminishing mortality through early therapy and applying preventive measures [3].
Patients with cancer on chemotherapy with neutropenia have an increased risk for developing bloodstream infections (BSI), frequently associated with Gram-negative bacteria, particularly EC [1]. Prevalence of fecal colonization by ESBL-EC is observed at around 10 and 30 % of community and hospitalized patients with hematologic malignancies (HM) and neutropenia, respectively [1]. Previous studies on immunosuppressed patients support the role of ESBL-EC colonization as a risk factor for BSI development and higher mortality, as compared with non-ESBL-EC [4]. The relationship between colonization and development of BSI is explained by intestinal translocation during neutropenia episodes. Mortality increase is probably related with the presence of ESBL virulence factors, ESBL patients’ greater vulnerability (resulting in more hospital admissions, frequent exposure to antibiotics, and increased likelihood of colonization and infection with ESBL-EC), and a delay in administering an appropriate antibiotic due to unanticipated resistance [4].
The aims of the present study were to assess the impact of fecal colonization with ESBL-EC on the development of BSI by the same strain and to investigate the type of ESBLs circulating in the hospital, hospitalization-time length, mortality, and therapy-related costs.
Methods
An observational prospective cohort study was conducted between November 1, 2012, and January 31, 2014, at the Instituto Nacional de Cancerología (INCan) in Mexico City, Mexico. The study was approved by the Institutional Review Board (013/005/INI) (CEI/824). INCan is a teaching referral hospital for adult patients with cancer, with 135 beds and a mean of 7500 hospital discharges per year. Each year, a mean of 80 patients with acute leukemia and 350 patients with lymphoma are seen for the first time at INCan to confirm their diagnosis and to initiate treatment. The standard care of these patients does not include antimicrobial prophylaxis, because the resistance of Enterobacteriaceae group to fluoroquinolones is about 60 %. Patients with fever and severe neutropenia receive as initial treatment ceftazidime plus amikacin, unless they have severe sepsis or septic shock in whom meropenem plus amikacin is started.
Patients.
The study population comprised adult patients with HM (acute myeloid leukemia, acute lymphoblastic leukemia, non-Hodgkin lymphoma, Hodgkin lymphoma, and myelodysplastic syndrome), hospitalized prior to the initiation of the first chemotherapy cycle. Eligible patients showed intestinal colonization by ESBL-EC or non-ESBL-EC in stool culture collected upon hospitalization; in the absence of previous enteric disease or medical condition that could modify intestinal permeability and/or immunity, as well as antimicrobial therapy during the last 30 days. Consecutive recruitment, regardless of the type of ESBL at intestinal colonization, was performed. Patients were further assigned to ESBL-EC group or non-ESBL-EC group, up to completion of 63 patients in each group. Demographic and clinical data were acquired.
During follow-up, blood cultures were obtained on a regular basis from patients with fever and severe neutropenia (<500 cells/mm3). All patients were followed upon completion of chemotherapy or death. The sum of the days of antibiotics and the length of stay of all hospitalizations in the different cycles of chemotherapy were recorded. Thirty-day survival from the last chemotherapy cycle was recorded, along with cause of death (infectious and non-infectious).
Health-related costs were calculated based on the days of antimicrobial treatment time (calculated by each antibiotic used by number of vials used per day), surveillance stool cultures, blood cultures, and imaging studies performed (X-ray, computed tomography, ultrasound, magnetic resonance) of all hospitalizations during different chemotherapy cycles. The administrative office of costs at the National Institute of Cancer has a catalogue with prices: for hospital day (general ward or intensive care unit), all procedures, laboratory and image studies, and all medications; these are updated annually. These fees were used to estimate the cost for each patient whether classified as ESBL-EC or non-ESBL-EC. The exchange rate was 9.06 pesos per dollar, on the date on which calculations were performed.
Microbiology analysis.
Stool samples were cultured on three plates: MacConkey; Sabouraud, and Salmonella-Shigella agar. Blood samples were processed by the automated blood culture BACTEC 9240 System (Becton-Dickinson Microbiology Systems, USA). EC identification and susceptibility were recognized by Phoenix BD™. EC ATCC 25922 was utilized for quality control according to Clinical and Laboratory Standards Institute (CLSI) guidelines [5]. Identification of the type of ESBL enzyme was performed by the double-disk synergism test following CLSI recommendations [5].
Genetic characterization and ESBL determination.
All EC strains were stored at −70 °C. Molecular analysis was performed at Instituto Nacional de Salud Pública (INSP), Mexico. For EC-ESBL determination, a randomized sample 1:2 was selected; for phylogenetic group, a randomized sample 1:3 was chosen. Genomic DNA genotyping was done by pulsed-field gel electrophoresis (PFGE). For ESBL determination, whole-cell DNA was obtained according to the method described by Kaufman [6, 7]. Gels were stained and analyzed according to the criteria of Tenover et al. [8] and Gel Compar II software (Applied Maths). Phylogenetic EC group was obtained by PCR for obtaining fragments of the genes Chua, yjaA, and TSPE4 [9].
Plasmids were extracted from clinical isolates by the method of Kieser [10]. PCR amplification for each gene was detected using the specific oligonucleotides bla CTX-M [11], bla SHV [12], and bla TLA-1 [13], which have been reported previously. In all cases, the resulting PCR products were analyzed using an automatic sequencer (ABI PRISM 377-18 kit EL: Taq FS Dye Terminator Cycle Sequencing Fluorescence-Based Sequencing). Amino acid sequences were obtained using the translation tool available at ExPASy (http://www.expasy.ch/tools/dna). Multiple alignments of nucleotide and amino acid sequences were performed with ClustalW (http://clustalw.genome.jp/) software, and the sequences were compared with the CTX-M-15 (AY044436) and SHV-1 (AF148850) genes. New genomic data was not generated in this study.
Statistical analysis
Clinical and demographic characteristics of patients colonized with ESBL-EC vs. non-ESBL-EC were compared. Categorical variables were analyzed by chi-square test or Fisher exact test, as appropriate, while continuous variable analysis was performed by Student t test or Wilcoxon rank test. Risk of BSI associated with ESBL-EC vs. non-ESBL-EC was calculated through the relative risk (RR) and 95 % confidence interval (95 % CI). Survival was assessed by Kaplan-Meier curves and log-rank test. Therapy-related costs were analyzed calculated from a costs of illness (COI) perspective. Software used for statistical analysis was STATA (ver. 12; Stata, College Station, TX, USA). p values ≤0.05 were considered statistically significant.
Results
We included 126 patients: 63 colonized by ESBL-EC and 63 by non-ESBL-EC. Mean age for the study group was 42 ± 17 years; 78 patients (62 %) were male. Relevant demographic and clinical characteristics are shown in Table 1.
Fecal colonization
Modification of gut colonization status by ESBL-EC or non-ESBL-EC was observed during follow-up. Seventeen patients (27 %) initially colonized with non-ESBL-EC strains ended up with positive ESBL-EC stool cultures during subsequent hospitalizations (follow-up). No patient colonized with ESBL-EC changed to a non-resistant strain. There were significant differences in antibiotic exposure time between patients who shifted from a non-resistant to a resistant strain (median, 60 days; range, 19–100 days), as compared with patients who remained with a non-ESBL-producing strain (median, 30.5 days; range, 0–159 days; p = 0.02). Length of hospital stay was 61 ± 27 vs. 42 ± 32 days, respectively (p = 0.03).
Risk for bloodstream infection
Fourteen patients in the ESBL-EC-colonized group (22.2 %) and five patients (8 %) who switched from non-ESBL-EC into ESBL-EC developed ESBL-EC bacteremia. Seventeen (27 %) patients colonized with non-ESBL-EC developed bacteremia by this strain, whereas three patients (4.7 %) colonized by ESBL-EC developed bacteremia by non-ESBL-EC. There were significant differences in hospital length, days of antibiotic exposure, and use of third-generation cephalosporins, when patients who developed BSI by E. coli were compared with those who did not (Table 2).
Mean time between gut colonization and development of ESBL-EC BSI was 66 ± 47 days and 108 ± 72 days for non-ESBL-EC patients (p = 0.09), respectively. Relative risk for ESBL-EC BSI in patients colonized with ESBL-EC was 3.4 (95 % CI, 1.5–7.8; p < 0.001).
Survival
Follow-up time was 159 ± 94 and 182 ± 54 days for ESBL-EC and non-ESBL-EC groups, respectively (p = 0.892). Mortality was similar in both groups: 10 (15.9 %) vs. 11 (17.5 %) patients died in the ESBL-EC and non-ESBL-EC colonization groups, with significant difference in time to death: 74 ± 62 days vs. 95 ± 83 days (p < 0.008), respectively.
Molecular analysis
Molecular analysis was performed in 20 random stool-blood culture pairs. Phylogenetic concordance was found in eight of these pairs (40 %): five of these belonged to the A phylogenetic group and three to the D phylogenetic group. PFGE and phylogroup analysis demonstrated polyclonal strain distribution.
ESBL enzyme was analyzed in 38 randomized strains: CTX-M was found in 30 (79 %), TEM in seven (18 %), and SHV and CTX-M in one (3 %). A representative sample of 23 isolates (60 %) was sequenced and analyzed to determine the corresponding allelic variant: 22 (95.7 %) had CTX-M-15 and one (4.3 %) CTX-M-15/SHV-12. The eight pairs with phylogenetic agreement had also the same ESBL.
Cost analysis
Mean global costs for all 126 patients were $5625 ± $3761. Mean costs for antibiotics were $2374 ± $1979; for hospitalization, $2338 ± $1487; and for laboratory/imaging studies, $913 ± $645. Total costs for patients colonized with ESBL-EC were significantly higher compared with those of patients colonized with non-ESBL-EC, in each of the three categories analyzed (antibiotics, hospitalization, and laboratory/imaging). Costs for both groups are depicted in Table 3.
Discussion
In immunocompromised patients with cancer, the severity and duration of neutropenia is one of the main risk factors for BSI [14]. Patients analyzed in this study comprise a representative group of adult patients with HM, with no significant differences in their baseline characteristics or clinical features, other than their intestinal colonization status (ESBL-EC and non-ESBL-EC). The sample was selected in a consecutive way including all HM patients prior to the first chemotherapy cycle.
In recent years, a significant increase in the incidence of ESBL-EC fecal carriage has been detected in a variety of settings. Extensive use of broad-spectrum antibiotics and prolonged hospitalization have been found as risk factors for ESBL-EC colonization in several studies [15, 16]. We do not have data on the prevalence of colonization by E. coli, but the sample of non-ESBL-EC was completed 2 months earlier than the resistant patients, which orients indirectly that sensitive strains predominate in the community.
The predominant clone of endogenous members of Enterobacteriaceae is the most likely to translocate and cause an intestinal bacterial overgrowth and an increased bacterial translocation of gut flora from the intestinal lumen, predisposing patients to bacterial infections [17–20]. This phenomenon has been reported to occur in between 7 and 15 % in different settings, such as older patients with intestinal obstruction, chronic liver disease, or after abdominal surgery [20–22], but there have been scarce reports in patients with neutropenia. In a previous report on 63 (29 %) patients colonized with ESBL-EC of 217 patients with HM and neutropenia, only one developed BSI by ESBL-EC and eight by non-ESBL-EC [23].
We found 17 patients initially classified as colonized by non-ESBL-EC who switched during follow-up (mean days, 102 days) to an ESBL-EC strain. These patients had a significantly longer hospital stay and received more days of broad-spectrum antibiotics compared with those who did not switch their basal colonization status.
Our data show that fecal colonization with ESBL-EC (63 patients initially classified and 17 patients who switched during follow-up) increased nearly 3.5 times in the probability of developing a BSI by an ESBL-EC strain. This is consistent with a previous study that found an increased risk of bacteremia by ESBL-EC in patients colonized with ESBL-EC with a RR = 4.5 (95 % CI 2.89–7.04) [1]. The higher risk to BSI by ESBL-EC of the same colonizing strain may be related with HM type; thus, therapy for primary disease: aggressive myelosuppressive chemotherapy; profound neutropenia, and the use of broad-spectrum antibiotics, consistent with previous findings [15, 16, 23, 24]. It has been reported that acute myelogenous leukemia and prior treatment with platinum analogues or quinolones are independent risk factors for ESBL-EC BSI in colonized patients [25].
We found a trend toward earlier development of BSI caused by ESBL-EC in patients colonized by an ESBL strain (66 ± 47 days to BSI) as compared with those colonized with non-ESBL-EC (108 ± 72 days to BSI; p = 0.09). In the former group, time to death was also shorter; however, development of BSI by ESBL-EC had no effect on mortality [15, 16]. This finding can be partially explained by differences in the EC strain, population characteristics, study design, or sample size.
Increase in the healthcare costs for infection by multidrug-resistant strains are usually related with the use of more expensive antibiotics, longer hospital stay, and laboratory and diagnostic resources such as imaging studies, blood cultures, etc. performed to study the infection episode. In this study, patients colonized with ESBL-EC generated higher costs in all three categories analyzed. Antimicrobial resistance has been associated with higher health-related costs, longer length of hospital stay, and higher death rates [16, 23]. Mortality increase is related to many risk factors such as greater severity of illness, a delay in administering an effective antibacterial therapy, and limitation of therapeutic interventions associated with higher risk of infections (chemotherapy, surgeries, or transplantation) [4, 26, 27].
Our results suggest the usefulness of fecal colonization screening in high-risk populations, such as severely immunocompromised patients in whom severe neutropenia will develop. Strategies to prevent the development and spread of antimicrobial-resistant organisms through antibiotic stewardship, active surveillance, and screening for resistance in high-risk patients are essential. Future studies will determine with greater certainty whether to use antibiotics with activity against ESBL in patients colonized by these strains may reduce mortality related to infection by these germs.
Molecular characterization was conducted in a subset of samples, exhibiting high clonal diversity among isolates. This finding supports polyclonal distribution of our strains, not related with a unique source, as demonstrated in some other studies on resistant Gram-negative bacteria. CTX-M enzyme was the main ESBL found in our setting. In previous studies, CTX-M enzymes are replacing SHV and TEM enzymes [28]. The emergence of CTX-M-type ESBLs has spread globally, particularly among E. coli and has modified the epidemiology of ESBLs, since dissemination of these enzymes is not restricted to the healthcare setting, but also involves the community [29].
In our study, the phylogenetic correlation between stool and blood samples was 40 % (not has high as expected), probably because of the diversity of strains that colonized the gut and the fact that these might change; thus, the strains are not exactly the same weeks later, when the patient develops bacteremia. In addition, subtypes A and D were the phylogroups identified and not the B2 described with major extraintestinal virulence [30].
The most frequent ESBL identified was CTX-M-15, consistent with previous reports from our hospital and from the same geographical area [24, 31, 32]. This enzyme has been related with third-generation cephalosporin resistance [33], antimicrobials extensively used in our setting. CTX-M-15 in particular has been related with increased mortality [30, 34].
There are some limitations in this study. The results shown correspond to a single institution in Mexico, so it cannot be generalized until other studies confirm these findings. The low rate of bacteremia does not allow us to have a more precise relationship between the status of colonization and its negative or positive predictive value for developing BSI, neither to establish a significant difference in mortality. Likewise, we need to further assess the risk factors for progression from colonization to BSI by ESBL-EC.
In conclusion, fecal colonization of patients with HM by ESBL-EC increased risk of BSI by the same strain, length of hospital stay, and overall healthcare-related costs.
References
Arnan M, Gudiol C, Calatayud L, Liñares J, Dominguez MA, Batlle M et al (2011) Risk factors for, and clinical relevance of, faecal extended-spectrum β-lactamase producing Escherichia coli (ESBL-EC) carriage in neutropenic patients with haematological malignancies. Eur J Clin Microbiol Dis 30:355–360. doi:10.1007/s10096-010-1093-x
Yu WL, Chuang YC, Walther-Rasmussen J (2006) Extended-spectrum beta-lactamases in Taiwan: epidemiology, detection, treatment and infection control. J Microbiol Immunol Infect 39:264–277
Ben-Ami R, Rodríguez-Baño J, Arslan H, Pitout JD, Quentin C, Calbo ES et al (2009) A multinational survey of risk factors for infection with extended-spectrum beta-lactamase-producing enterobacteriaceae in nonhospitalized patients. Clin Infect Dis 49:682–690. doi:10.1086/604713
Melzer M, Petersen I (2007) Mortality following bacteraemic infection caused by extended spectrum beta-lactamase (ESBL) producing Escherichia coli compared to non-ESBL producing E. coli. J Infection 55:254–259
Clinical and Laboratory Standards Institute (2011) Performance standards for antimicrobial susceptibility testing. 21st Informational Supplement, M-100-S21. Wayne, PA, USA: CLSI
Kaufmann ME (1988) Pulse-field gel electrophoresis. Methods Mol Med 15:17–31
Akpaka PE, Legall B, Padman J (2010) Molecular detection and epidemiology of extended-spectrum beta-lactamase genes prevalent in clinical isolates of Klebsiella pneumoniae and E. coli from Trinidad and Tobago. West Indian Med J 59:591–596
Tenover FC, Arbeit RD, Goering RV, Mickelsen PA, Murray BE, Persing DH, Swaminathan B (1995) Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J Clin Microbiol 33:2233–2239
Clermont O, Bonacorsi S, Bingen E (2000) Rapid and simple determination of the Escherichia coli phylogenetic group. Appl Environ Microbiol 66:4555–4558
Kieser T (1984) Factors affecting the isolation of CCC DNA from Streptomyces lividans and Escherichia coli. Plasmid 12:19–36
Espinal P, Garza-Ramos U, Reyna F, Rojas-Moreno T, Sanchez-Perez A, Carrillo B et al (2010) Identification of SHV-type and CTX-M-12 extended-spectrum beta-lactamases (ESBLs) in multiresistant Enterobacteriaceae from Colombian Caribbean hospitals. Chemotherapy 22:160–164
Prodinger WM, Fille M, Bauernfeind A, Stemplinger I, Amann S, Pfausler B et al (1996) Molecular epidemiology of Klebsiella pneumoniae producing SHV-5 beta-lactamase: parallel outbreaks due to multiple plasmid transfer. J Clin Microbiol 34:564–568
Berçot B, Poierel L, Silva-Sánchez J, Nordmann P (2010) Association of the extended-spectrum beta-lactamase gene blaTLA-1 with a novel ISCR element, ISCR20. Antimicrob Agents Chemother 54:4026–4028. doi:10.1128/AAC.00075-10
Chen CY, Tsay W, Tang JL, Tien HF, Chen YC, Chang SC, Hsueh PR (2009) Epidemiology of bloodstream infections in patients with haematological malignancies with and without neutropenia. Epidemiol Infect 27:1–8. doi:10.1017/S0950268809991208
Levin BR (2001) Minimizing potential resistance: a population dynamics view. Clin Infect Dis 33:S161–S169
Smith DL, Dushoff J, Perencevich EN, Harris AD, Levin SA (2004) Persistent colonization and the spread of antibiotic resistance in nosocomial pathogens: resistance is a regional problem. Proc Natl Acad Sci U S A 101:3709–3714
Tancrede CH, Andremont AO (1985) Bacterial translocation and Gram-negative bacteremia in patients with hematological malignancies. J Infect Dis 152:99–103
Yu WL, Chuang YCH, Rasmussen JW (2006) Extended-spectrum beta-lactamases in Taiwan: epidemiology, detection, treatment and infection control. J Microbiol Immunol Infect 39:264–277
Ellis M (2008) Febrile neutropenia. Evolving strategies. Ann N Y Acad Sci 1138:329–350
Ilan Y (2012) Leaky gut and the liver: a role for bacterial translocation in nonalcoholic steatohepatitis. World J Gastroenterol 18:2609–2618. doi:10.3748/wjg.v18.i21.2609
Van Leeuwen PAM, Boermeester MA, Houdijk APJ, Ferwerda CC, Cuesta MA, Meyer S, Wesdorp RIC (1994) Clinical significance of translocation. Gut Suppl 1:S28–S34
Hu B, Ye H, Xu Y, Ni Y, Hu Y, Yu Y et al (2010) Clinical and economic outcomes associated with community-acquired intra-abdominal infections caused by extended spectrum beta-lactamase (ESBL) producing bacteria in China. Curr Med Res Opin 26:1443–1449. doi:10.1185/03007991003769068
Cornejo-Juárez P, Pérez-Jiménez C, Silva-Sánchez J, Velázquez-Acosta C, González-Lara F, Reyna-Flores F et al (2012) Molecular analysis and risk factors for Escherichia coli producing extended-spectrum β-lactamase bloodstream infection in hematological malignancies. PLoS One 7(4):e35780
Hayakawa K, Gattu S, Marchaim D, Bhargava A, Palla M, Alshabani K et al (2013) Epidemiology and risk factors for isolation of Escherichia coli producing CTX-M-type extended-spectrum β-lactamase in a large U.S. medical center. Antimicrob Agents 57:4010–4018. doi:10.1128/AAC.02516-12
Liss BJ, Vehreschild JJ, Cornely OA, Hallek M, Fätkenheuer G, Wisplinghoff H et al (2012) Intestinal colonisation and blood stream infections due to vancomycin-resistant enterococci (VRE) and extended-spectrum beta-lactamase-producing Enterobacteriaceae (ESBL) in patients with haematological and oncological malignancies. Infection 40:613–619. doi:10.1007/s15010-012-0269-y
Schwaber MJ, Carmeli Y (2008) Carbapenem-resistant Enterobacteriaceae: a potential threat. JAMA 300:2911–2913. doi:10.1001/jama.2008.896
Kang CL, Chung DR, Ko KS, Peck KR, Song JH, Korean Network for study of infectious diseases (2012) Risk factors for infection and treatment outcome of extended-spectrum β-lactamase-producing Escherichia coli and Klebsiella pneumoniae bacteremia in patients with hematologic malignancy. Ann Hematol 91:115–121. doi:10.1007/s00277-011-1247-7
Falagas ME, Karaeorgopoulos DE (2009) Extended-spectrum β-lactamase-producing organisms. J Hosp Infect 73:345–354
Wu UI, Wang JL, Chen WC, Chang SC, Chen YC (2011) Risk factors and outcomes of Escherichia coli bacteremia caused by strains that produce CTX-M or non-CTX-M extended-spectrum-beta-lactamases. Eur J Clin Microbiol Infect Dis 30:33–39
Clermont O, Lavollay M, Vimont S, Deschamps C, Forestier C, Branger C et al (2008) The CTX-M-15 producing Escherichia coli diffusing clone belongs to a highly virulent B2 phylogenetic subgroup. J Antimicrob Chemother 61:1024–1028. doi:10.1093/jac/dkn084
Reyna-Flores F, Barrios H, Garza-Ramos U, Sánchez-Pérez A, Rojas-Moreno T, Uribe-Salas FJ et al (2013) Molecular epidemiology of Escherichia coli O25b-ST131 isolates causing community-acquired UTIs in Mexico. Diagn Microbiol Infect Dis 76:396–398. doi:10.1016/j.diagmicrobio.2013.03.026
Morfín-Otero R, Mendoza-Olazarán S, Silva-Sánchez J, Rodríguez-Noriega E, Laca-Díaz J, Tinoco-Carrillo P et al (2013) Characterization of Enterobacteriaceae isolates obtained from a tertiary care hospital in Mexico, which produces extended-spectrum β-lactamase. Microb Drug Resist 19:378–383. doi:10.1089/mdr.2012.0263
Paterson DL, Bonomo RA (2005) Extended-spectrum β-lactamases: a clinical update. Clin Microbiol Rev 18:657–686
Clermont O, Dhanji H, Upton M, Gribeel T, Fox A, Boyd D et al (2009) Rapid detection of the O25b-ST131 clone of Escherichia coli encompassing the CTX-M-15-producing strains. J Antimicrob Chemother 64:274–277. doi:10.1093/jac/dkp194
Acknowledgments
We would like to thank the Microbiology Laboratory for the facilities provided in processing and storing the samples for this research.
Financial support
This project had partial support from CONACYT (Consejo Nacional de Ciencia y Tecnología) no. 136339 for the phylogenetic analysis and the molecular characterization of ESBL-EC producers. No additional external funding was received for this study. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of interest
The authors declare no conflict of interest. This study did not receive any funding. The study investigators planned, conducted, and analyzed all data. All of the authors reviewed and accepted the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cornejo-Juárez, P., Suárez-Cuenca, J.A., Volkow-Fernández, P. et al. Fecal ESBL Escherichia coli carriage as a risk factor for bacteremia in patients with hematological malignancies. Support Care Cancer 24, 253–259 (2016). https://doi.org/10.1007/s00520-015-2772-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-015-2772-z