Abstract
The expression pattern of mitotic Ki-67 and anti-apoptotic bcl-2 proteins, as well as apoptotic caspase-3 and p53 proteins, were investigated in the human mesonephros and metanephros of 5–9 week-old human conceptuses. Apoptotic cells were additionally detected using the terminal deoxynucleotidyl transferase (TdT) nick-end labelling (TUNEL) method. Between the 5th and 7th developmental weeks Ki-67, caspase-3 and TUNEL-positive cells characterized all mesonephric structures, indicating importance of cell proliferation in the growth of the mesonephros and role of apoptosis in nephrogenesis. From the 7th week on, p53 and bcl-2 positive cells appeared in the mesonephros as well. Regressive changes in the mesonephros could be regulated by activation of p53, while bcl-2 could contribute to selective survival of some tubules giving rise to adult structures. In the early human metanephros (5–7 weeks), Ki-67 positive cells characterized all metanephric structures, indicating a role of cell proliferation in branching of the ureteric bud and in nephron formation. During the same period bcl-2, caspase-3 and TUNEL-positive cells were found only in the metanephric mesenchyme and nephrons. Bcl-2 protein probably protected nephrons from apoptosis, while caspase-3 protein controlled cell death in the mesenchyme. At later stages (7–9-weeks), appearance of p53-expressing cells could participate in further morphogenesis of the metanephric collecting system. The factors investigated had a spatially and temporally restricted pattern of appearance in developing kidneys. Changes in that pattern might lead to serious disturbances of kidney formation and function in early childhood.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
During the human embryonic period, three excretory kidney systems develop: the pronephros, the mesonephros and the metanephros. The pronephros is a non-functional and transitory structure by the end of the fourth week of development; it degenerates leaving behind only its caudal part, which will become a primary ureter used by mesonephros. The mesonephros develops from nephrogenic cord caudal to the pronephros late in the fourth developmental week. It is a large ovoid organ, situated laterally to the developing gonad and formed of large numbers of vesicles that differentiate into S-shaped loops. The lateral ends of these loops enter the primary ureter, now called the mesonephric (Wolffian) duct, while the medial end forms the Bowman’s capsule surrounding the glomerulus, thus forming the mesonephric corpuscle. The mesonephros is a temporary functional kidney, which subsequently degenerates. However, in the male embryo, some parts of the Wolffian duct and mesonephric tubules persist as genital ducts. Formation of the permanent kidney, the metanephros, begins in the fifth week of development, when a dorsal outgrowth of Wolffian duct called the ureteric bud penetrates the metanephric mesenchyme. The metanephric mesenchyme develops from the sacral portion of the nephrogenic cord and differentiates into renal vesicles and S-shaped tubules (nephrons) under inductive influence of the terminal branches (ampullae) of the ureteric bud. Later on, more mature nephrons develop, consisting of the Bowman’s capsule and glomerulus, proximal and distal convoluted tubule, and the loop of Henle. Collecting tubules, renal calyces, renal pelvis and ureter are derivates of the ureteric bud [1, 2].
Development of all three excretory systems depends on reciprocal tissue interactions and many transcription factors, growth factors, signalling molecules, their receptors and other proteins. Knowledge about the role of those factors and genes is based on investigations on transgenic animals showing different kidney abnormalities, such as absence of kidney morphogenesis (Pax-2 and Lim-1 knockout), defects in epithelial transformation of mesenchyme (Wnt-4 and Bmp-7 knockout) and reduced numbers of nephrons (FGF-7 knockout) [3, 4].
Among those factors, distribution and intensity of programmed cell death and cell proliferation are considered as important and often combined processes in nephrogenesis. Apoptosis, a programmed cell death, occurs at precise stages of kidney development and has an important role in the normal development and regression of all three forms of embryonic kidneys [5]. During degeneration of the pronephros and mesonephros in rat embryos, cell death follows a strict temporo-spatial pattern [6]. In metanephric development of the rat embryo, evidence of apoptosis, such as condensed nuclei, fragmented cytoplasm and cell shrinking, were found in cells surrounding the new epithelium induced by the ureteric bud [7].
Apoptosis may be executed through the caspase protein family. Over a dozen caspases have been found to operate in humans [8]. Caspases have an important function in cell cycle regulation [9], especially caspase-3 which is the key effector caspase in apoptotic process [10, 11]. In mouse embryos, general caspase inhibition was shown to prevent ureteric bud branching and further development of the metanephros [12].
Bcl-2 is an integral mitochondrial, nuclear and endoplasmic reticulum membrane protein. It was shown that bcl-2 over-expression protects cell from apoptotic death [13, 14]. During human kidney development, bcl-2 gene expression may be necessary for the differentiation of uninduced mesenchyme into the mature nephrons [13]. In bcl-2 deficient mice, renal hypoplasia was found associated with abnormal renal growth, cyst formation and decrease in number of nephrons [13, 15–18].
Proliferation is another important factor involved in kidney development and differentiation. Ki-67 nuclear protein is often used as a proliferation marker because of its expression in all phases of the cell cycle except the resting phase (G0) [19, 20]. In human nephrogenesis, Ki-67 expression was found to be the most pronounced in the early stages of metanephric glomeruli differentiation, in the differentiating vesicles and folding glomeruli. Ki-67 expression was found to decrease with glomerular maturation [21].
p53 is a negative regulator of the G1-S phase transition in the cell cycle. In the case of DNA damage it induces growth arrest or apoptosis in the damaged cells [14]. In p53 transgenic mouse, the kidneys are smaller and have lower numbers of compensatory hypertrophic glomeruli, owing to altered differentiation of the ureteric bud [22]. In human embryos aged 4 to 8 weeks, p53 is expressed in the nuclei of the mesonephric secretory canal epithelium and focally in the cells of the mesonephric glomeruli [14]. In the metanephros, p53 expression is found only during the foetal period [23].
Numerous studies on kidney development, dealing with cell proliferation, apoptosis and their genetic background and control, have been done on experimental animals [3, 12, 13, 15–18, 22]. Only few studies of their expression pattern in the mesonephros and metanephros have been performed in human embryos [13, 14], some of them done on developmental stages different from ours [24–26].
Aberrations of proliferation and cell death in the human metanephros often appear in association with lower urinary tract obstructions. Even changes in environment, such as altered maternal diet, cause nephron defects accompanied by enhanced apoptosis [27].
Therefore, the aim of this study was to analyse the spatial and temporal expression pattern of factors involved in cell proliferation and programmed cell death during the mesonephric and early metanephric development of the human kidney. The consequences of disturbed cell proliferation and cell death are associated with several kidney abnormalities (agenesis, dysplasia, hypoplasia), thus accounting for the great number of young children with chronic renal failure.
Materials and methods
Human material
A total of six normal human conceptuses between their 5th and 9th week of development was collected after spontaneous abortions from the Department of Gynaecology and Obstetrics, Clinical Hospital of Split, Croatia, and after tubal pregnancies from the Department of Pathology, Clinical Hospital of Split. The embryos and foetuses were examined macroscopically and measured. Only normal conceptuses, without any sign of abnormality, signs of intrauterine death or maceration, were used in our study. The embryonic tissues were treated as postmortem material with permission of the Ethics and Drug Committee of the Clinical Hospital of Split in accordance with the 1964 Helsinki Declaration. The post-ovulatory age was estimated on the basis of menstrual data and correlated with crown–rump length (CRL) and Carnegie stages [28].
Immunohistochemical staining
Caudal parts of embryos containing developing kidneys were dissected. Tissue samples were fixed in 4% paraformaldehyde in phosphate buffer and dehydrated in 100% ethanol. They were embedded in paraffin wax, serially sectioned at 4–6 μm, mounted on glass slides, and examined with an Olympus BX-40 light microscope (Olympus, Tokyo, Japan). The paraffin was removed with xylene, and the sections were rehydrated in ethanol and water. In order to quench endogenous peroxidase activity, we incubated the sections for 30 min in 0.1% H2O2.
For Ki-67 staining, after incubation in H2O2, paraffin-embedded sections were washed with phosphate-buffered saline (PBS) and then incubated in EDTA (pH 8.0) for 10 min at 95°C. They were than cooled to room temperature and incubated with monoclonal mouse anti-human Ki-67 antigen (M 7240, DAKO, Glostrup, Denmark) for 30 min. After being washed with PBS, the binding was visualized by incubating the sections with Envision+ single reagent visualization system, which contains peroxidase-conjugated anti-mouse secondary antibody (K 4001, DAKO) for 30 min. Afterwards, sections were washed with PBS and then stained with diaminobenzidine tetrahydrochloride (DAB) solution. Finally, the sections were rinsed in distilled water, counter-stained with haematoxylin, and dehydrated in ethanol and xylol [29].
Sections for immunohistochemical staining of bcl-2, p53 and caspase-3 antigens were treated with H2O2, washed in PBS and then incubated in sodium citrate buffer for 10 min at 95°C. After being cooled to room temperature, they were incubated with rabbit anti-human/mouse active caspase-3 primary antibody (AF835, R&D Systems, Minneapolis, Minn., USA) and mouse anti-human Bcl-2 oncoprotein primary antibody (M 0887, DAKO) overnight at 4°C in a humidified chamber. The rest of the paraffin sections were incubated with mouse anti-human p53 protein (M 7001, DAKO) for 45 min. After being washed with PBS, the bcl-2 and caspase-3 sections were incubated with biotinylated secondary antibody (mouse and rabbit UniTect ABC Kit, Oncogene, Boston, Mass., USA) for 30 min at room temperature. They were then washed again in PBS and incubated with avidin biotinylated horseradish peroxidase complex (ABC) for 30 min, washed again with PBS, and then stained with DAB. Finally, the sections were rinsed in distilled water, counter-stained with haematoxylin, and dehydrated in ethanol and xylol.
Additionally, we pre-incubated some sections with immunizing peptide for 30 min in order to remove the bcl-2 signal.
For the p53 sections, primary antibodies were detected using a streptavidin–biotin peroxidase system (K0690, DakoCytomation, Carpinteria, Calif., USA) as recommended by the manufacturer. The p53 sections were later washed with PBS, stained, counter-stained and dehydrated as described above [30, 31].
Detection of apoptotic cells by the terminal deoxynucleotidyl transferase-mediated nick-end labelling method
DNA fragmentation in apoptotic cells was examined using the terminal deoxynucleotidyl transferase (TdT)-mediated nick-end labelling (TUNEL) method. DNA fragmentation in apoptotic cells was detected with TdT-mediated nick-end binding of fluorescein-labelled and unlabelled deoxynucleotides using the Fluorescein FragEL DNA Fragmentation Detection Kit (Calbiochem, USA). Sections with the paraffin removed were rehydrated and pretreated with proteinase K for 20 min and then treated with equilibration buffer for 30 min, all at room temperature. The sections were later covered with working TdT-labelling reaction mixture and incubated in a humidified chamber at 37°C for 1.5 h. After the incubation, the sections were briefly stained with haematoxylin and mounted with the mounting media provided in the TUNEL kit. Labelled nuclei were examined with a standard fluorescein filter (465–495 nm). Microphotographs were captured with a SPOT Insight QE camera (Diagnostic Instruments, USA) mounted on an Olympus BX51 microscope using the SPOT software.
Quantification of Ki-67 positive cells
Six conceptuses of different developmental ages (5–9 weeks) were examined. The positively labelled and unlabelled cells were counted in three non-adjacent sections taken from each conceptus. For Ki-67 positivity, any level of nuclear positivity was considered, regardless of the intensity of the staining. Counts were made over the total area of the kidney (mesonephric or metanephric). In the mesonephros, as well as in the metanephros, three distinct areas of 50 μm×50 μm at 200× magnification were counted over each chosen structure. Thus, in the mesonephros, three collecting nephrons and three mesenchymal areas were counted in each section. In the metanephros, three nephrons, three tubules and three mesenchymal (interstitial) areas were counted per section. The examination was performed on an Olympus BX-51 microscope equipped with a DMP digital camera and using DP-SOFT version 3.1 software. In each area (50 μm×50 μm), the percentage of Ki-67-positive cells was calculated.
Quantitative analysis was performed for three groups (5 and 6, 7, 8 and 9 weeks) according to the corresponding age of the embryo. The percentage of Ki-67 positive cells was expressed as mean ± SD. Data were analysed by the Kruskal–Wallis analysis of variance (ANOVA) test followed by Dunn’s post-hoc test.
Results
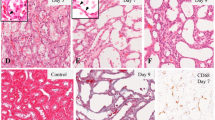
During the 5th and 6th week of development, both the mesonephros and the metanephros are present in the human embryo. The mesonephros forms the large ovoid organ on each side of the midline, on the lateral side of the developing gonad. It consists of renal glomerules and tubules opening into the mesonephritic duct (Wolffian duct) at the lateral side and forming the Bowman’s capsule at its medial extremity (Fig. 1a). Behind the lower end of the mesonephros, the metanephros develops as well. The collecting system of the mesonephros develops from the ureteric duct undergoing multiple divisions. The anterior, actively growing, portion of the bud is the ampulla, which induces surrounding cells of the metanephric mesoderm to proliferate, condense and form the metanephric cup (Fig. 1b).
Transverse sections through the developing mesonephros and metanephros in the 5th and 6th developmental weeks. A Mesonephros (5 weeks): primitive nephrons (n), mesonephric duct (d), mesonephric mesenchyme (m), the gonad (g) primordium. Haematoxylin and eosin, ×10. B Metanephros (5 weeks): the ureteric bud (b), ampullae (a), metanephric mesenchyme (mm), the mesenchymal metanephric cup (c). Haematoxylin and eosin, ×10. C+ Metanephros (5 weeks): note moderately bcl-2 positive cells in the metanephric cup (c), very mildly positive cells in the metanephric mesenchyme (mm) and non-reactive cells in the ampullae (a). Immunostaining to bcl-2 protein, ×40. C− Absence of bcl-2 positivity in all metanephric structures. Immunostaining to bcl-2 protein pre-incubated with immunizing peptide, ×40. D Mesonephros (5 weeks): three caspase-3 positive cells with dark brown-stained nuclei (arrow) are seen among the vital euchromatic nuclei of mesenchymal cells (m). Mesonephric duct (d) and coelomic epithelium (ce) are seen as well. Immunostaining to active caspase-3, ×40. E Metanephros (5 weeks): only one caspase-3 positive cell nucleus (arrow) is seen among the euchromatic cell nuclei of the metanephric cup mesenchyme (c). Ampulla (a) is missing caspase-3 positive cells. Immunostaining to active caspase-3, ×100. F Mesonephros (6 weeks): nuclei of Ki-67 positive cells (arrow) are seen in the mesonephric duct (d), glomeruli (g) and Bowman’s capsule (bc), mesonephric tubules (t) and mesonephric mesenchyme (m). Immunostaining to Ki-67 protein, ×20. G Metanephros (5 weeks): numerous brown-stained nuclei of Ki-67 positive cells (arrow) are seen in the metanephric cup (c), adjacent ureteric bud (b) and ampullae (a), and in the metanephric mesenchyme (mm) Immunostaining to Ki-67 protein, ×10
During the developmental period described, bcl-2 positive cells are not present in the human mesonephros, while, in the metanephros, mildly expressing bcl-2 cells can be seen in the metanephric mesenchyme, and moderately expressing bcl-2 cells in the metanephric cup. Ampullae do not contain bcl-2 positive cells. In the control sections, pre-incubated with immunizing peptide, the bcl-2 signal was gone from metanephric mesenchyme and metanephric cup. (Table 1, Fig. 1c+,c-).
Caspase-3 positive cells are present in all structures forming the human mesonephros: the Wolffian duct, mesonephric mesenchyme, glomeruli and Bowman’s capsule, mesonephric tubules and coelomic epithelium (Table 1). In comparison with the euchromatic nuclei of vital cells, caspase-3 positive cells have characteristic condensed nuclei or dark-stained nuclear fragments (Fig. 1d). Contrary to the mesonephric structures, in the metanephros rare caspase-3 positive cells can be seen only in the metanephric mesenchyme and in its condensation forming the metanephric cup. Uretheric bud and ampullae are completely devoid of caspase-3 positive cells (Table 1, Fig. 1e).
In the 5th and 6th weeks of development, both mesonephros and metanephros are missing p53 positive cells, but they can be found in the coelomic epithelium (Table 1).
During the same developmental period, all structures of the human mesonephros show Ki-67 positive cells: the Wolffian duct and surrounding mesenchyme, mesonephric glomeruli and Bowman’s capsule and mesonephric tubules. Ki-67 positive cells are also seen among the cells of the coelomic epithelium (Table 1, Fig. 1f).
In the metanephros, the number of Ki-67 positive cells is even higher than in the mesonephros, particularly in the ampullae and metanephric cup mesenchyme (Table 1, Fig. 1g).
During the 7th, 8th and 9th weeks of development, the caudal parts of the mesonephros still differentiate, while its cranial parts already show signs of degenerative changes.
In the same developmental period, the metanephros undergoes characteristic developmental changes: the ureteric bud dilates to form the primordium of the collecting system, including collecting tubules and developing ureters. Further differentiation of metanephric cup mesenchyme leads to formation of metanephric vesicles, S-shaped nephrons and more mature nephrons, consisting of Bowman’s capsule and renal glomerulus. Other parts of the metanephric mesenchyme between the developing nephrons give rise to interstital connective tissue (Table 2, Fig. 2b).
Transverse sections through the 7th and 8th week human mesonephros. A Mesonephros (7 weeks): bcl-2 positive cells (arrow) are seen in metanephric tubules (t), glomeruli (g) and mesonephric mesenchyme (m). Immunostaining to bcl-2 protein, ×40. B Metanephros (8 weeks): bcl-2 positive cells (arrow) are seen in the metanephric cup (c), comma-shaped bodies (cb), S-shaped nephrons (sn) and renal corpuscles (rc), while they are missing in the collecting tubules (ct). Some metanephric mesenchymal cells (mm) are bcl-2 positive as well. Immunostaining to bcl-2 protein, ×10. Insert: detail showing the strongest bcl-2 positivity in the metanephric cup, less strong positivity in the comma-shaped bodies (cb) and mesenchyme (mm) and absence of positivity in the collecting tubuli and ampullae (a), ×40. C Mesonephros (7 weeks): caspase-3 positive cells (arrows) are seen in the mesonephric tubuli (t) and glomeruli (g) as brown-stained nuclei among the blue-stained nuclei of vital cells. Immunostaining to active caspase-3, ×40. D Metanephros (8 weeks): nuclei of caspase-3 positive cells (arrows) are seen in the developing S-shaped nephrons (sn) and in the nearby cells of the metanephric mesenchyme (mm). Immunostaining to active caspase-3, ×40. E Metanephros (8 weeks): p53-positive cells (arrows) are found in the ampullae (a), renal vesicles (v) and metanephric mesenchyme (mm). Immunostaining to p53 nuclear marker, ×40. F Metanephros (8 weeks): numerous Ki-67 positive cells (arrows) are seen in the collecting tubuli (ct) and ampullae (a), and they are less numerous in the proximal divisions of the ureteric bud (b). The metanephric cup (c) and comma-shaped bodies (cb) contain extensive numbers of Ki-67 positive cells, while their numbers are lower in the metanephric mesenchyme (mm). Immunostaining to Ki-67 protein, ×10
During the described developmental period, bcl-2 positive cells appear in all structures forming the disappearing mesonephros (Table 2, Fig. 2a).
In the methanephros, the bcl-2 positive cells are missing in the whole collecting system, while they are numerous in developing nephrons, particularly in the less mature forms (Table 2, Fig. 2b).
In the mesonephros, caspase-3 positive cells are still present in all structures forming the mesonephros, but their numbers decrease in the 8th and 9th developmental weeks. (Table 2, Fig. 2c).
Compared with earlier developmental stages, the number of caspase-3 positive cells increases in the metanephros: in the 8th and 9th developmental weeks, caspase-3 positive cells appear in ampullae and collecting tubuli for the first time. They are also present in all parts of developing nephrons, particularly in the more mature forms of renal bodies, and in interstitial cells (Table 2, Fig. 2d).
The p53-positive cells appear in the mesonephros for the first time at the end of the 6th developmental week, except in the Wolffian duct and Bowman’s capsule. In the 8th and 9th weeks, some p53-positive cells are detected in the Bowman’s capsule as well (Table 2).
During the same developmental period, p53-positive cells are present in all structures of the developing metanephros, being the most numerous in the ampullae (Table 2, Fig. 2e).
All structures of the mesonephros show Ki-67 positive cells from the 7th to 9th developmental week (Table 2).
In the metanephros, the collecting system displays numerous Ki-67 positive cells, particularly in its ampullar part. The interstitial cells and differentiating nephrons contain large numbers of Ki-67 positive cells; their numbers increase with maturation of nephrons and advanced developmental stage (Table 2, Fig. 2f).
In the mesonephros, TUNEL-positive cells are seen in the same areas as caspase-3 positive cells (Fig. 3a–c). In the metanephros, TUNEL-positive cells are found in all parts of the developing kidney, having the same distribution as caspase-positive cells (Fig. 3d–f).
Transverse sections through the 5th week mesonephros and 8th week metanephros. A Mesonephros: mesonephric glomerulus (g), Bowman’s capsule (bc), mesonephric tubule (t), and mesonephric mesenchyme (m). Haematoxylin, ×40. B Several fluorescent TUNEL-positive cells are seen. TUNEL method, ×40. C TUNEL-positive cells are seen in areas corresponding to the glomerulus and mesenchyme. Merging of A+B, ×40. D Metanephros: renal vesicle (v), comma-shaped bodies (cb), collecting tubuli (ct) and metanephric mesenchyme (mm). Haematoxylin, ×40. E Several fluorescent TUNEL-positive cells are seen. TUNEL method, ×40. F Position of TUNEL-positive cells corresponds to areas of renal vesicle and metanephric mesenchyme. Merging of D+E, ×40
Quantitative analysis of the proliferation activity
Quantification was done only with Ki-67 nuclear marker, because it is the only antibody used in our study that gives very clear and distinct brown nuclear staining of reactive cells.
The percentage of Ki-67 positive cells increased significantly in both the mesonephric mesenchyme and nephrons comparing 5- and 6-week with 8- and 9-week old conceptuses (Fig. 4a).
Percentage of Ki-67 positive cells in A the mesonephros and B the metanephros from the 5th to 9th gestational weeks. Diamonds nephrons, squares mesenchyme, triangles collecting tubuli. *Significant difference (P<0.05, Dunn’s post-hoc test) between the first and the third group, #significant difference (P<0.05, Dunn’s post-hoc test) between the first and the second group
The activity of the proliferation marker Ki-67 in the metanephric mesenchyme (Fig. 4b) decreased significantly in embryos aged 7 weeks compared with embryos aged 5 and 6 weeks. Significant increase of Ki-67 expression in the nephrons was found between 5- and 6-week and 8- and 9-week-old conceptuses (Fig. 4b). In the metanephric collecting tubules, significant decrease of Ki-67 activity was found between 5- and 6-week and 7-week-old embryos as well as between 5- and 6-week and 8- and 9-week-old embryos (Fig. 4b).
Discussion
The mesonephros
The mesonephric human kidney consists of transient glomeruli and tubules, which mostly degenerate except for the part that becomes the efferent ductules of the testes [32, 33].
Apoptosis seems to be important in the normal regression of this primitive kidney system that usually ends in the 16th developmental week [6, 27]. Indeed, in our study, caspase-3 and TUNEL-positive cells were already permanently present in all parts of the developing mesonephros in 5–8 week embryos, while their number slightly decreased with the beginning of the foetal period. According to Pole et al. [6], apoptosis commences in the mid-to-caudal region of the human mesonephros. We found cells expressing the p53 gene in all mesonephric structures, except for the mesonephric duct, for the first time in the 7th developmental week, while Lichnovsky et al. [24] found the same expression already in the 4-week human embryos.
Owing to the temporal and spatial appearance of those two factors in our study, caspase-3 mediated apoptosis might be associated with early morphogenesis of the mesonephros, while p53 expression appeared later in development, parallel to regressive processes in the primitive kidneys. Proliferation marker Ki-67 was expressed in all parts of the mesonephros and increased significantly at later embryonic stages, both in the mesenchymal and in the epithelial tissue components (nephrons). This is in line with the peak of mesonephric size and development being in the 8th developmental week, following the described intense cell proliferation and differentiation of all tissue components [6]. The anti-apoptotic bcl-2 protein appeared for the first time in the 7th developmental week. Its expression increased in some of the metanephric tubules in 8–9 week conceptuses, probably due to their survival and transformation into efferent ductules of a male adult.
The metanephros
During the developmental period investigated in our study, the human metanephros underwent several characteristic developmental steps, from invasion of the ureteric bud into the metanephric mesenchyme to induction and gradual differentiation of nephrons.
In the early metanephric development, we found caspase-3 positive and TUNEL-cells only in the metanephric mesenchyme and its condensation forming the metanephric cup. At later developmental stages, the number of apoptotic cells increased in the developing nephrons, particularly in the more mature forms and in the part of the metanephric mesenchyme giving rise to the interstitium. Apoptotic cells were detected in all parts of the metanephric collecting system for the first time in the 9th developmental week. These data indicate the important involvement of caspase-3 mediated apoptosis in early nephrogenesis and, later on, in development of the collecting system as well. This is in line with experimental data showing that administration of caspase-3 and caspase-9 inhibitors led to disturbances of both ureteric branching and nephrogenesis [11], while excessive apoptosis occurred in polycystic kidney disease where expression of various caspases, bax and bcl-2 was upregulated [9]. Similar to our results, in rat kidneys, dying cells formed 60% of metanephric mesenchymal cells and 40% of developing nephrons, while they were rarely found in branches of the ureteric bud [34]. The peak of apoptotic index was 3% in rat kidneys [34, 35]. In the early foetal metanephros (8–9th week), p53-positive cells appeared for the first time in both collecting systems and developing nephrons, being more numerous in the ampullae. Miosge et al. [23] found the first p53 expression later in development (10th–15th week) than we did in our study, while Prochazkova et al. [36] found p53-positive cells only sporadically and not in all metanephric structures. The appearance of p53-positive cells in all parts of the metanephros temporally coincided with the intense differentiation of the kidney collecting system. This might indicate importance of both apoptotic pathways, caspase-3 and p53 mediated, for the formation of the metanephric collecting system. Experiments with p53 transgenic mice showed defective differentiation of the ureteric bud and hypoplastic kidneys due to increased apoptosis in the undifferentiated mesenchyme [22]. It was also shown that p53 over-expression could cause defects in human kidney development [14]. The process of apoptosis might be regulated through extrinsic factors, such as trophic hormones and growth factors [13]. Thus, administration of epidermal growth factor can reduce the number of apoptotic cell to 50% [35]. An intrinsic regulatory factor that can protect cells from apoptosis is bcl-2 protein. In the 5th developmental week, bcl-2 positive cells appeared in the metanephros, later on increasing in the nephrons, particularly in less mature forms, as previously shown by Prochazkova et al. [36]. Throughout the developmental stages investigated, bcl-2 positive cells were hardly or not at all detectable in derivatives of the ureteric bud, as earlier described in human tissue [24–26, 36]. Experiments with bcl-2 deficient mice showed kidney abnormalities, including hypoplasia with fulminant apoptosis within the metanephric mesenchyme [15, 16]. In the early development, only metanephric mesenchymal cells expressing bcl-2 differentiate into renal epithelium [13], as bcl-2 inhibits apoptosis in those cells [16]. Most of the remaining mesenchymal cells differentiate into stromal (interstitial) cells, some (cortical cells) of them giving rise to renin-producing cells, and some (medullary cells) differentiating into nephrons or undergoing apoptosis [7, 37]. In bcl-2 deficient mice renal failure results from severe polycystic kidney disease [17, 18].
A proliferation marker, Ki-67, was detected in all structures of the developing human metanephros throughout the developmental period investigated. Our quantitative analysis showed a significant decrease of proliferation of the metanephric mesenchyme between the 5th and 9th developmental weeks and a decrease of proliferation activity in the collecting system (50% – 30%) with advancing development. On the other hand, intense nephrogenesis was accompanied by significantly increased proliferation activity (from 50% to 80%). Previous investigations on human foetal kidneys of older developmental stages found a decrease of Ki-67 expression with progressive glomerular maturation, while Ki-67 expression was undetectable in terminally differentiated glomeruli [21].
In conclusion, in the human mesonephros, up to the 7th developmental week, the parallel presence of proliferation Ki-67 and apoptotic caspase-3 and TUNEL-positive cells in all mesonephric structures might be explained by initial growth of the mesonephros by mitosis and simultaneous nephron formation due to apoptosis. From the 7th week on, the appearance of p53 expression coincided with the regressive changes in the mesonephros, while bcl-2 positive cells enabled selective survival of some tubules giving rise to adult efferent ductules. In the early human metanephros (5–7 weeks), only Ki-67 positive cells were present in all metanephric structures, indicating the importance of this gene for the intense branching of the ureteric bud and induction of distinct numbers of nephrons. During the same period, both bcl-2 and caspase-3 expressing and TUNEL-positive cells were seen only in the metanephric mesenchyme and in differentiating nephrons, protecting nephrons from apoptosis on one hand and enabling selective cell death in the metanephric mesenchymal cells on the other hand. At later stages (from the 7th week on), increased proliferation activity accompanied advancing nephrogenesis. More numerous apoptotic cells, and the appearance of p53-expressing cells, probably contributed to the morphogenesis of both the collecting system and the nephrons. Cells expressing bcl-2 remained present only in the developing nephrons, thus saving them from apoptosis. All the mitotic, pro-apoptotic and anti-apoptotic factors described had spatially and temporally restricted patterns of appearance in developing human kidneys. Their balance and strict time course seem to be important for normal kidney development. Changes in their expression pattern might be associated with serious disturbances of kidney morphogenesis and function, often leading to chronic renal failure.
References
Saxen L (1987) Organogenesis of the kidney. Cambridge University press, Cambridge
Sadler TW (1985) Langman’s medical embryology, 5th edn. Williams & Wilkins, Baltimore, pp 247–280
Kuure S, Vuolteenaho R, Vainio S (2000) Kidney morphogenesis: cellular and molecular regulation. Mech Dev 92:31–45
Davies JA, Fisher CE (2002) Genes and proteins in renal development. Exp Nephrol 10:102–113
Alison MR, Sarraf CE (1992) Apoptosis: a gene-directed programme of cell death. J R Coll Phys Lond 26:25–35
Pole RJ, Qi BQ, Beasly SW (2002) Patterns of apoptosis during degeneration of the pronephros and mesonephros. J Urol 167:269–271
Koseki C, Herzlinger D, Al-Awqati Q (1992) Apoptosis in metanephric development. J Cell Biol 119:1327–1333
Hengartner MO (2000) The biochemistry of apoptosis. Nature 407:770–776
Ali SM, Wong V, Kikly K, Fredrickson TA, Keller PM, DeWolf WE Jr., Lee D, Brooks DP (2000) Apoptosis in polycystic kidney disease: involvement of caspases. Am J Physiol Integr Comp Physiol 278:763–769
Hayashi M, Araki T (2002) Caspase in renal development. Nephrol Dial Transplant 17:8–10
Araki T, Hayashi M, Nakanishi K, Morishima N, Saruta T (2003) Caspase-9 takes part in programmed cell death in developing mouse kidney. Nephron Exp Nephrol 93:e117–e124
Araki T, Saruta T, Okano H, Miura M (1999) Caspase activity is required for nephrogenesis in the developing mouse metanephros. Exp Cell Res 248:423–429
Hammerman MR (1998) Regulation of cell survival during renal development. Pediatr Nephrol 12:596–602
Lichnovský V, Koláø Z, Murray P, Hlobilková A, Èernochová D, Pospíšilová E, Vojtìšek B, Nenutil R (1998) Differences in p53 and bcl-2 expression in relation to cell proliferation during the development of human embryos. Mol Pathol 51:131–137
Sorenson CM, Rogers SA, Korsmeyer SJ, Hammerman MR (1995). Fulminant metanephric apoptosis and abnormal kidney development in bcl-2 deficient mice. Am J Physiol 268:F73–F81
Nagata M, Nakauchi H, Nakayama K, Nakayama K, Loh D, Watanabe T (1996) Apoptosis during an early stage of nephrogenesis induces renal hypoplasia in bcl-2-deficient mice. Am J Pathol 148:1601–1611
Kamada S, Shimono A, Shinto Y, Tsujimura T, Takahashi T, Noda T, Kitamura Y, Kondoh H, Tsujimoto Y (1995) Bcl-2 deficiency in mice leads to pleiotropic abnormalities: accelerated lymphoid cell death in thymus and spleen, polycystic kidney, hair hypopigmentation, and distorted small intestine. Cancer Res 55:354–359
Veis DJ, Sorenson CM, Shutter JR, Korsmeyer SJ (1993) Bcl-2-deficient mice demonstrate fulminant lymphoid apoptosis, polycystic kidneys, and hypopigmented hair. Cell 75:229–240
Klein CL, Wagner M, Kirkpatrick CJ, Van Kooten TG (2000) A new quantitative test method for cell proliferation based on detection of the Ki-67 protein. J Mater Sci Mater Med 11:125–132
Kee N, Sivalingam S, Boonstra R, Wojtowicz JM (2002) The utility of Ki-67 and BrdU as proliferative markers of adult neurogenesis. J Neurosci Methods 115:97–105
Combs HL, Shankland SJ, Setzer SV, Hudkins KL, Alpers CE (1998) Expression of the cyclin kinase inhibitor, p27kip1 in developing and mature human kidney. Kidney Int 53:892–896
Godley LA, Kopp JB, Eckhaus M, Paglino JJ, Owens J, Varmus HE (1996) Wild-type p53 transgenic mice exhibit altered differentiation of the ureteric bud and possess small kidneys. Genes Dev 10:836–850
Miosge N, Schneider W, Gotz W, Herken R (1997) The oncoproteins c-erb-B2, c-fos and the tumor suppressor protein p53 in human embryos and fetuses. Anat Embryol 195:345–352
Lichnovsky V, Erdosova B, Punkt K, Zapletal M (1999) Expression of bcl-2 in the developing kidney of human embryos and fetuses: qualitative and quantitative study. Acta Univ Palacki Olomuc Fac Med 142:61–64
Erdosova B, Hlavkova L, Prochazkova J, Lichnovsky V (2002) Part of CD68+ macrophages in the clearance of apoptotic bodies in human metanephros. Biomed Pap Med Fac Univ Palacky Olomuc Czech Repub 146:41–45
Erdosova B, Wagner F, Kylarova D (2004) The detection of Myc proteins in the developing human kidney. Biomed Pap Med Fac Univ Palacky Olomuc Czech Repub 148:205–207
Woolf AS, Welham SJM (2002) Cell turnover in normal and abnormal kidney development. Nephrol Dial Transplant 17 [Suppl 9]:2–4
O’Rahilly R, Gardner R (1971) The timing and sequence of events in the development of the human nervous system during the embryonic period proper. Z Anat Entwicklungsgesch 134:1–12
Cattoretti G, Becker MHG, Key G, Duchrow M, Schlüter C, Galle J, Gerdes J (1992) Monoclonal antibodies against recombinant parts of the Ki-67 antigen (MIB 1 and MIB 3) detect proliferating cells in microwave-processed formalin-fixed paraffin sections. J Pathol 168:357–363
Huppertz B, Frank HG, Kaufmann P (1999) The apoptosis cascade—morphological and immunohistochemical methods for its visualization. Anat Embryol 200:1–18
Vojtesek B, Bartek J, Midgley CA, Lane DP (1992) An immunochemical analysis of the human nuclear phosphoprotein p53. New monoclonal antibodies and epitope mapping using recombinant p53. J Immunol Methods 151:237–244
Potter EL (1972) Normal and abnormal development of the kidney. Year Book Medical Publishers, Chicago, pp 3–79
Moore KL (1989) Before we are born: basic embryology and birth defects, 3rd edn. Saunders, Philadelphia, pp 180–199
Coles HS, Burne JF, Raff MC (1993) Large-scale normal cell death in the developing rat kidney and its reduction by epidermal growth factor. Development 118:777–784
Savill J (1994) Apoptosis and the kidney. J Am Soc Nephrol 5:12–21
Prochazkova J, Lichnovsky V, Kylarova D, Erdosova B, Vranka P (2004) Involvement of p53 and bcl-2 family proteins in regulating programmed cell death and proliferation in human embryogenesis. Gen Physiol Biophys 23:209–229
Sainio K, Nonclercq D, Saarma M, Palgi J, Saxen L, Sariola H (1994) Neuronal characteristics in embryonic renal stroma. Int J Dev Biol 38:77–84
Acknowledgements
We are grateful to Mrs. Asja Miletiæ for her skilful technical assistance. This work was supported by the Ministry of Science, Education and Sports of the Republic of Croatia (grants no. 0216002 and 0216001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carev, D., Krni, D., Saraga, M. et al. Role of mitotic, pro-apoptotic and anti-apoptotic factors in human kidney development. Pediatr Nephrol 21, 627–636 (2006). https://doi.org/10.1007/s00467-006-0057-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-006-0057-y