Abstract
Background
Colonoscopic perforation (CP) has a low incidence rate. However, with the extensive use of colonoscopy, even low incidence rates should be evaluated to identify and address risks. Information on CP is quite limited in China.
Objective
Our study aimed to determine the frequency of CP in colonoscopies performed by surgeons at a large teaching hospital in China over a 12-year period.
Methods
A retrospective review of medical records was performed for all patients who had CPs from 1 January 2000 to 31 December 2012. Iatrogenic perforations were identified mainly by abdominal X-ray or computed tomography scan. Follow-up information of adverse events post-colonoscopy was identified from the colorectal surgery database of our hospital. Patients’ demographic data, colonoscopy procedure information, location of perforation, treatment, and outcome were recorded.
Results
A total of 110,785 diagnostic and therapeutic colonoscopy procedures were performed (86,800 diagnostic cases and 23,985 therapeutic cases) within the 12-year study period. A total of 14 incidents (0.012 %) of CP were reported (seven males and seven females), of which nine cases occurred during diagnostic colonoscopy (0.01 %) and five after therapeutic colonoscopy (three polypectomy cases, one endoscopic mucosal resection, and one endoscopic mucosal dissection). Mean patient age was 67.14 years. One case of CP (7.14 %) after colonoscopy polypectomy was treated using curative colonoscopy endoclips. Other patients underwent operations: six cases (46.15 %) of primary repair, four cases (28.57 %) of resection with anastomosis, and two cases (15.38 %) of resection without anastomosis. No obvious perforation was found in one patient (7.69 %). Surgeons attempted to treat one case laparoscopically but eventually resorted to open surgery. The postoperative course was uncomplicated in eight cases (57.14 %) and complicated in six cases (42.86 %) but without mortality.
Conclusion
CP is a serious but rare complication of colonoscopy. A perforation risk of 0.012 % was found in our study. The optimal management of CP remains controversial. Treatment for CP should be individualized according to the patient’s condition, related devices, and surgical skills of endoscopists or surgeons. Selective measures such as colonoscopy without intravenous sedation and decrease of loop formation can effectively reduce rates of perforation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
-
What is current knowledge?
-
The usual rate, risk factors and treatment of CP.
-
What is new here?
-
The recommended methods and new concepts about CP.
Flexible colonoscopy was introduced by the Department of Surgery at Beth Israel Medical Center in New York City, USA, in 1969 [1]. Colonoscopy has become a safe and cost-effective diagnostic and therapeutic procedure [2]. Colonoscopy is widely used in the diagnosis of colorectal cancer (CRC) as well as in the detection and removal of adenomatous polyps. Optical colonoscopy has been endorsed as the preferred CRC screening strategy for CRC prevention, beginning at the age of 50 years [3, 4]. The number of colonoscopic procedures is expected to continually increase in the aging population in China as a result of increasing lifespans. The safety of the procedure must be considered when a screening strategy is applied to a large population. The most serious complication of colonoscopy is perforation [5–10]. The frequency of perforations after colonoscopy is estimated to be 0.03–0.8 % for diagnostic colonoscopy and 0.15–3 % for therapeutic colonoscopy [11]. With the increasing frequency of colonoscopies performed for screening purposes, the small possibility of perforations may still result in a high number of clinical problems.
The US Multi-Society Task Force on Colorectal Cancer has set targets for colonoscopic perforation (CP) at ≤1 in 1,000 for all colonoscopies, ≤1 in 2,000 for screening colonoscopies, [12] and 1 in 100 as target for postpolypectomy bleeding. We designed our current study to determine the incidence of CP in a large teaching hospital in China over a 12-year period and compare our data with those presented in previous studies. Additionally, we hope to share with gastroenterologists, endoscopists, and surgeons who perform colonoscopy worldwide the factors behind the lower rate of CPs in China.
Patients and methods
We performed a retrospective study of iatrogenic perforations caused by colonoscopies at our center between 1 January 2000 and 31 December 2012. Iatrogenic perforations were identified mainly by abdominal X-ray or computed tomography (CT) scan. We obtained the information for all patients from the colorectal surgery database. Our study was approved by The Committee on Ethics of Biomedicine Research of the Second Military Medical University, Shanghai, China. We compiled and analyzed the following variables: demographic characteristics of patients, type and indication of colonoscopy, clinical manifestations during perforation and diagnosis, type of treatment applied, intraoperative findings, duration of hospital confinement, and complications encountered during confinement.
Results
Patient demography
A total of 110,785 recorded colonoscopies were performed by surgeons in the colorectal department over a 12-year period from 1 January 2000 to 31 December 2012. Nearly all (98.44 %) patients who underwent colonoscopic examination received mechanical bowel preparation using either polyethylene glycol or 120 ml 50 % magnesium sulfate with 4 liters of water. Nearly all (99.35 %) procedures were performed without sedation. Patients were informed of possible complications associated with the procedure prior to signing appropriate informed consent. Over the 12-year period, 14 patients (seven males and seven females) were found to have CP (0.012 %). However, the percentage of male and female patients who underwent colonoscopy was unknown; thus, relative risk based on gender cannot be calculated. Patient age ranged from 32 to 85 years, with a median age of 76.14 years (Table 1).
Indications and type of colonoscopic procedure
Indications for colonoscopy in patients with perforation were polyps (N = 5), obstructive tumor (N = 3), constipation (N = 3), bowel obstruction (N = 2), and unknown cause (N = 1). A diagnostic procedure was performed for nine cases and a therapeutic procedure for five cases. All therapeutic procedures consisted of polypectomies, including one endoscopic mucosal resection (EMR) and one endoscopic submucosal dissection (ESD). Our hospital is a teaching hospital, thus, all procedures were performed by or under the supervision of experienced colorectal surgeons. Nearly all (99.35 %) procedures were performed without sedation. Patients cooperated with examiners during the whole examination. During examination, the procedure is halted if the patient is in obvious discomfort or in case of loop formation. Prompt and effective release of the colon loop, as well as careful maneuvering of the instrument, was performed.
Symptoms and diagnosis
CP during colonoscopy may result from visualization of extra-intestinal tissue during the procedure. Early symptoms of perforation include persistent abdominal pain and distention. Later symptoms are the result of peritonitis, which includes fever and leukocytosis. Plain radiographs of the chest and abdomen may demonstrate free air. However, we concur with previous studies that an abdominal CT scan should be considered for patients with unremarkable plain film results in cases of highly probable perforation [13].
Perforations
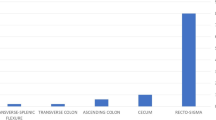
Perforation was observed in one case (6.7 %) by an examiner through visualization of extra-intestinal tissue during the procedure, prompting the immediate endoscopic endoclip closure. Most perforations were diagnosed shortly after the procedure because of indicative signs and symptoms. A total of three patients (21.4 %) underwent surgery on the same day as the colonoscopy, five (35.7 %) on the second day, and five (35.7 %) more than 2 days after colonoscopy. Four perforations (28.57 %) occurred in the sigmoid colon, two perforations each (14.28 %) occurred in the conjunction of sigmoid and descending colon or descending colon or cecum, one case each occurred (7.14 %) in the rectum, recto sigmoid, splenic flexure of colon, and ileum. Only one patient did not undergo surgery. Serosal tears were seen with no actual perforation in one case. Four types of surgical correction were performed: primary repair, resection with anastomosis, primary repair with colonic diversion, and resection with colonic diversion. Primary repair was performed on six of the actual perforations (42.86 %). Resection with anastomosis was performed on four patients (28.57 %). Primary repair with colonic diversion was performed on one patient (7.14 %), and resection with colonic diversion was performed on two patients (14.28 %). The CP rate for diagnostic colonoscopy is 0.01 %, and for therapeutic colonoscopy is 0.021 %.
Complications
Of the 14 patients, eight (57.14 %) had no complications and fast postoperative recovery. The remaining six patients (42.86 %) experienced complications: three had wound infection, one had both wound and urinary infection, one patient had pneumonia, and one patient had wound infection and cardiovascular accident (CVA). No patient died after surgery. The duration of hospital confinement ranged from 3 to 45 days, with a mean of 21.93 days.
Discussion
Our department was one of the pioneers of colonoscopy in China; it has been performed in our colorectal department since the early 1980s. Our academic leader, Professor Yu, spearheaded colonoscopy at our institution. Professor Yu trained at St. Mark’s Hospital in London and he has accomplished nearly 60,000 cases, which includes diagnosis and therapy, with no incidence of perforation in diagnostic cases and only two cases reported during therapeutic colonoscopy. The present study evaluated CP in a large teaching hospital in China over a 12-year period. Results showed that perforation occurred in 0.012 %. Nine perforations occurred during a diagnostic colonoscopy (0.01 %), and the others occurred after therapeutic colonoscopy (0.021 %, including three cases for polypectomy and one case each for EMR and ESD). The frequency of CP in our present study seems lower than that previously reported in the literature. No death was directly associated with the procedure, and a very low rate of adverse effects was observed. The frequency of CP in some larger series (sample size >30,000 cases) published from 2000 onwards are shown in Table 2 [1, 14–25]. Sieg et al. [15] prospectively evaluated 82,416 colonoscopies using a mailed questionnaire and found a very low incidence estimated at 0.005 %. This phenomenon is most likely explained by selection bias because of the physician self-reported cases of perforations. No significant lowering of CP perforation rate over a decade (2000–2010) was found. More studies are needed to evaluate this phenomenon because learning curves, performance of a colonoscopy by trainees, and performance by inadequately trained endoscopists are insufficient to elucidate this phenomenon. Therefore, with the extensive use of colonoscopy worldwide, the safety, learning curve, risk factor, and possible complications of colonoscopy should be evaluated. Our paper fills a gap created by the lack of data regarding the perforation rates of diagnostic and therapeutic colonoscopy performed by surgeons in a teaching hospital in China. Moreover, given the limited data available from China, our study contributes important information for the geographically adjacent areas from East Asia.
The present study is retrospective and reports perforation rates that are lower than previously reported rates, which may cause anxiety among experts fearing that the risk of CP may be underestimated. There is concern that patients who sustain complications may be hospitalized at other facilities, leading to imprecise recording of CP incidents. There are three reasons to eliminate those concerns. First, patients will be outpatients, followed-up within 2 weeks after the colonoscopy inspection or treatment. Second, in China, the majority of patients are of poor economic status, leading to difficulties obtaining and paying for medical treatment. The doctor–patient relationship is usually tense. If a patient feels uncomfortable after any outpatient examination, he/she must return to the primary hospital whenever possible. The patient automatically assumes a problem was encountered during his examination. Thus, we can confirm the presence of CP via information held for the relevant operation and records of recent colonoscopy. Third, to ensure accuracy of the study, all relevant data were kept, and none were omitted.
Rabeneck et al. [25] found that after outpatient colonoscopy, the use of a 7-day period for reporting would capture all perforations requiring hospital admission. In the present study, the last report of the occurrence of perforation happened 6 days after diagnostic colonoscopy. This patient only had a minor serosal tear without transmural perforation. Perforations after therapeutic procedures are often diagnosed late, probably because of different pathophysiologic mechanisms [14, 26]. In diagnostic procedures, perforations most often result from pressure applied to the colonic wall and can be noticed immediately via visualization of extra-intestinal tissue by examination. In therapeutic procedures, ischemia of the colonic wall caused by electrical or thermal injury after electrocoagulation or laser can cause delayed perforation [11]. Patients with CP caused by therapeutic colonoscopies also tend to have a smaller-sized perforation and delayed presentation and diagnosis than do those caused by diagnostic colonoscopy [17, 22]. In this report, we compared the time of perforation diagnosis after diagnostic and therapeutic colonoscopy between nonparametric Wilcoxon test. No significant difference was found between CP diagnosis times after diagnostic and therapeutic colonoscopies. These results may still have errors due to bias or a low number of cases, therefore, further reevaluation and study are recommended.
The most common site of colonic perforation is in the sigmoid or rectosigmoid junction [11, 17, 21, 22, 27–29]. In our report, 50 % of perforations happened in the sigmoid. Redundant colon and more tight curves are more common in this bowel segment. Diverticular disease is also common in this bowel segment [17].The muscular layer of the bowel wall may be thin or fragile because of previous inflammation (diverticulitis), although this condition is usually uncommon in Chinese patients. This bowel segment is vulnerable to injury because of several factors such as a sharp angulation at either the recto-sigmoid junction or the sigmoid-descending colon junction and the great mobility of the sigmoid colon. Perforation can occur when maneuvering the scope around tight curves by applying pressure that may create a kink in the scope [16, 27, 30].
Many studies have shown that some CP risk factors may be associated with demography. Old age was independently associated with CP. Patients over 75 years of age also have an approximately four to sixfold increase in the CP rate compared with younger patients [20, 29]. A newly published meta-analysis showed that elderly patients, especially octogenarians, appear to have higher risk of CP [31]. A possible explanation for this finding is that the elderly might have declining colonic wall mechanical strength. Moreover, a greater frequency of abnormal colorectal findings that require endoscopic intervention has been detected in the elderly [32]. Although the ages of the majority of our CP patients were known, not all ages of subjects in our study were known. Thus, we cannot verify whether age is a risk factor for CP. Several studies have suggested that more perforations occur in female patients [6, 11, 14, 17, 19]. There was also adverse results in the study of Arora et al. [23]. A previous study has also reported differences between males and females in the anatomy of the large intestine [33]. Typically, females have a more mobile transverse colon and longer colon. In our study, nine cases of CP occurred during diagnostic colonoscopy, of which six patients were females (66.67 %). However, the overall gender ratio in our study is unknown. Therefore, we cannot verify whether sex is a risk factor for CP. According to previous studies, other risk factors for CP include previous abdominal surgery, colonic obstruction, previous polypectomy, and complications from previous procedures carried out by inexperienced endoscopists. [17, 20, 23, 34].
Numerous studies have elucidated the risk factors for CP. Only a few clinical studies have discussed practical colonoscopy procedures in an attempt to decrease the incidence of CP. In this paper, we present our experiential knowledge.
Colonoscopy may be performed without using intravenous sedation and analgesia, although several studies have shown that use of sedation improved the overall success of colonoscopy [35, 36]. We surmise that sedation posts more disadvantages than advantages in colonoscopy [37]. The ability of the patient to cooperate with the examiner is reduced if the patient is sedated, which is disadvantageous when a change in position is required during examination. Analgesia and loss of pain sensation will also hamper the recognition of excessive pressure on the bowel wall. Hence, the possibility of mechanical injury to the bowel wall will be higher in colonoscopy under sedated conditions. Non-sedated patients undergoing colonoscopy will be able to fully interact with the examiner and immediately provide crucial feedback of discomfort, which can direct the examiner to maneuver the scope gently, reducing the chances for trauma or perforation [29].
Loop formation may be decreased, and advancing the scope may be performed more carefully. Generally, perforation occurs in three principal mechanisms, i.e., perforation by the tip or shaft of the endoscope, barotraumas of overinsufflation, and therapeutic procedures such as polypectomy and stricture expansion. Lohsiriwat et al. [29] stated that 40 % of CPs were due to direct trauma from the endoscope shaft and 33 % by direct trauma from the endoscope tip. We consider that most perforations caused by the shaft of an endoscope were caused by colon loop formation. Excessive axial pressure of colonoscopy will lead to lacerated wound of the colon wall and develop into CP. By contrast, CP caused by the tip of an endoscope happens because of crudely advancing colonoscopy during examination. Therefore, prompt and effective release of the colon loop during colonoscopy can obviously improve the successful rate of cecal intubation, reduce procedural time, decrease pressure on the colon wall, enhance patient comfort, and more importantly reduce the CP rate.
The optimal management of CP remains a controversial program. Treatment for CP should be individualized. CP patients may be managed conservatively, by colonoscopic closure, or by surgical operation. We believe that conservative management is only suitable for patients with no obvious perforation, such as transmutable bowel injury, which is common after polypectomy and more commonly known as postpolypectomy coagulation syndrome or transmural burn syndrome [38, 39]. Patients with transmural burn syndrome will have abdominal pain, fever, local peritonitis, and leukocytosis. Plain abdominal radiograph and CT scan are often negative in this condition [40, 41]. We agree that conservative management should be very carefully selected only for patients with very few latent perforations and transmural burn syndrome. The first successful colonoscopic clip closure of CP was reported in 1997 [42]. In our study, one patient underwent this procedure. We consider the following preconditions necessary for this procedure: good medical conditions, clean bowel preparation, small perforation size, an examiner with high endoscopic skills, appropriate endoscopic devices, broad-spectrum antibiotics, and nutritional support. Patients with perforations of less than 10 mm are suitable for this procedure and should be placed under careful observation after treatment [43, 44]. Surgical intervention is the most commonly used treatment for CP, as shown in our study. Selection of surgical method depends on the size of perforation, patient condition, quality of bowel preparation, pathology of damaged intestine, time between injury and operation, and experience of surgeon [45]. Operations have increasingly been performed laparoscopically for patients with CP [46–49]. We attempted to perform laparoscopic repair of perforation in one of the cases in this study. However, it was later converted to open surgery because of apprehension surrounding the safety of the repair. In our opinion, laparoscopic surgery is not suitable for situations in which there is a long delay between injury and surgery, severe stool contamination of the peritoneum, diffuse peritonitis, and lack of advanced laparoscopic surgery skills.
Our study presents several potential limitations. First, it is a retrospective study. Second, some demographic data were not included, such as ages and sex of all patients, prior surgical history, and use of drugs, which include non-steroidal anti-inflammatory and antiplatelet agents. Thus, effective prediction of risk factors for CP is quite difficult. Third, patients were not identified as either outpatient or inpatient. Finally, a very few patients who suffered perforation may not have sought treatment at our hospital; thus, they would not have been included in our study.
Conclusion
Colonoscopy is a remarkably useful procedure for CRC surveillance and treatment of other related diseases. CP is a severe complication of colonoscopy. Examiners should be thoroughly aware of the usual risk factors and related inherent characteristics or presentations indicative of CP. Patient safety takes precedence in all management strategies for CP. The optimal management of CP remains controversial. Implementation of methods and skills such as withholding administration of intravenous sedation, decreasing loop formation, and careful maneuvering of the endoscope can effectively improve the safety of colonoscopy and decrease complications such as perforations.
References
Wolff WI, Shinya H (1971) Colonofiberoscopy. JAMA 217:1509–1512
Avgerinos DV, Liaguna OH, Lo AY et al (2008) Evolving management of colonoscopic perforations. J Gastrointest Surg 12:1783–1789
US Preventive Services Task Force (2008) Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med 149:627–637
McFarland EG, Levin B, Lieberman DA et al (2008) Revised colorectal screening guidelines: joint effort of the American Cancer Society, US Multisociety Task Force on Colorectal Cancer, and American College of Radiology. Radiology 248:717–720
Gerstenberger PD, Plumeri PA (1993) Malpractice claims in gastrointestinal endoscopy: analysis of an insurance industry data base. Gastrointest Endosc 39:132–138
Anderson ML, Pasha TM, Leighton JA (2000) Endoscopic perforation of the colon: lessons from a 10-year study. Am J Gastroenterol 95:3418–3422
Nelson DB, McQuaid KR, Bond JH et al (2002) Procedural success and complications of large-scale screening colonoscopy. Gastrointest Endosc 55:307–314
Garbay JR, Suc B, Rotman N et al (1996) Multicentre study of surgical complications of colonoscopy. Br J Surg 83:42–44
Jentschura D, Raute M, Winter J et al (1994) Complications in endoscopy of the lower gastrointestinal tract. Therapy and prognosis. Surg Endosc 8:672–676
Tran DQ, Rosen L, Kim R et al (2001) Actual colonoscopy: what are the risks of perforation? Am Surg 67:845–847
Lüning TH, Keemers ME, Barendregt WB et al (2007) Colonoscopic perforations: a review of 30,366 patients. Surg Endosc 21:994–997
Rex DK, Bond JH, Winawer S et al (2002) Quality in the technical performance of colonoscopy and the continuous quality improvement process for colonoscopy: recommendations of the U.S. Multi-Society Task Force on Colorectal Cancer. Am J Gastroenterol 97:1296–1308
American Society for Gastrointestinal Endoscopy (2003) Complications of colonoscopy. Gastrointest Endosc 57:441–445
Araghizadeh FY, Timmcke AE, Opelka FG et al (2001) Colonoscopic perforations. Dis Colon Rectum 44:713–716
Sieg A, Hachmoeller EU, Eisenbach T (2001) Prospective evaluation of complications in outpatient GI endoscopy: a survey among German gastroenterologists. Gastrointest Endosc 53:620–627
Gatto NM, Frucht H, Sundararajan V et al (2003) Risk of perforation after colonoscopy and sigmoidoscopy: a population-based study. J Natl Cancer Inst 95:230–236
Korman LY, Overholt BF, Box T et al (2003) Perforation during colonoscopy in endoscopic ambulatory surgical centers. Gastrointest Endosc 58:554–557
Cobb WS, Heniford BT, Sigmon LB et al (2004) Colonoscopic perforations: incidence, management, and outcomes. Am Surg 70:750–757 discussion 757–758
Iqbal CW, Chun YS, Farley DR (2005) Colonoscopic perforations: a retrospective review. J Gastrointest Surg 9:1229–1235
Rabeneck L, Paszat LF, Hilsden RJ et al (2008) Bleeding and perforation after outpatient colonoscopy and their risk factors in usual clinical practice. Gastroenterology 135:1899–1906
Iqbal CW, Cullinane DC, Schiller HJ et al (2008) Surgical management and outcomes of 165 colonoscopic perforations from a single institution. Arch Surg 143:701–706 discussion 706–707
Teoh AY, Poon CM, Lee JF et al (2009) Outcomes and predictors of mortality and stoma formation in surgical management of colonoscopic perforations: a multicenter review. Arch Surg 144:9–13
Arora G, Mannalithara A, Singh G et al (2009) Risk of perforation from a colonoscopy in adults: a large population-based study. Gastrointest Endosc 69:654–664
Mai CM, Wen CC, Wen SH et al (2010) Iatrogenic colonic perforation by colonoscopy: a fatal complication for patients with a high anesthetic risk. Int J Colorectal Dis 25:449–454
Rabeneck L, Saskin R, Paszat LF (2011) Onset and clinical course of bleeding and perforation after outpatient colonoscopy: a population-based study. Gastrointest Endosc 73:520–523
Dafnis G, Ekbom A, Pahlman L et al (2001) Complications of diagnostic and therapeutic colonoscopy within a defined population in Sweden. Gastrointest Endosc 54:302–309
Lohsiriwat V, Sujarittanakarn S, Akaraviputh T et al (2008) Colonoscopic perforation: a report from World Gastroenterology Organization endoscopy training center in Thailand. World J Gastroenterol 14:6722–6725
Gedebou TM, Wong RA, Rappaport WD et al (1996) Clinical presentation and management of iatrogenic colon perforations. Am J Surg 172:454–457 discussion 457–458
Lohsiriwat V, Sujarittanakarn S, Akaraviputh T et al (2009) What are the risk factors of colonoscopic perforation? BMC Gastroenterol 9:71
Trecca A, Gaj F, Gagliardi G (2008) Our experience with endoscopic repair of large colonoscopic perforations and review of the literature. Tech Coloproctol 12:315–321
Day LW, Kwon A, Inadomi JM et al (2011) Adverse events in older patients undergoing colonoscopy: a systematic review and meta-analysis. Gastrointest Endosc 74:885–896
Karajeh MA, Sanders DS, Hurlstone DP (2006) Colonoscopy in elderly people is a safe procedure with a high diagnostic yield: a prospective comparative study of 2000 patients. Endoscopy 38:226–230
Saunders BP, Fukumoto M, Halligan S et al (1996) Why is colonoscopy more difficult in women? Gastrointest Endosc 43:124–126
Ramirez FC (2012) Re-learning colonoscopy: just a matter of time. J Interv Gastroenterol 2(3):133–134
Rodney WM, Dabov G, Orientale E et al (1993) Sedation associated with a more complete colonoscopy. J Fam Pract 36:394–400
Radaelli F, Meucci G, Sgroi G, Italian Association of Hospital Gastroenterologist (AIGO) (2008) Technical performance of colonoscopy: the key role of sedation/analgesia and other quality indicators. Am J Gastroenterol 103:1122–1130
Johannes L (2011) The patient’s perspective: patients should be made aware of the options of sedation or no sedation and have a choice in screening colonoscopy. J Interv Gastroenterol 1(1):42–44
Putcha RV, Burdick JS (2003) Management of iatrogenic perforation. Gastroenterol Clin N Am 32:1289–1309
Choo WK, Subhani J (2012) Complication rates of colonic polypectomy in relation to polyp characteristics and techniques: a district hospital experience. J Interv Gastroenterol 2(1):8–11
Zissin R, Konikoff F, Gayer G (2006) CT findings of latrogenic complications following gastrointestinal endoluminal procedures. Semin Ultrasound CT MR 27:126–138
Kim DH, Pickhardt PJ, Taylor AJ et al (2008) Imaging evaluation of complications at optical colonoscopy. Curr Probl Diagn Radiol 37:165–177
Yoshikane H, Hidano H, Sakakibara A et al (1997) Endoscopic repair by clipping of iatrogenic colonic perforation. Gastrointest Endosc 46:464–466
Lohsiriwat V (2010) Colonoscopic perforation: incidence, risk factors, management and outcome. World J Gastroenterol 16:425–430
Jovanovic I, Zimmermann L, Fry LC et al (2011) Feasibility of endoscopic closure of an iatrogenic colon perforation occurring during colonoscopy. Gastrointest Endosc 73:550–555
Siegel D (2013) Changes in colonoscopy: new tricks for an old dog. J Interv Gastroenterol 3(2):57–58
Wullstein C, Koppen M, Gross E (1999) Laparoscopic treatment of colonic perforations related to colonoscopy. Surg Endosc 13:484–487
Hansen AJ, Tessier DJ, Anderson ML et al (2007) Laparoscopic repair of colonoscopic perforations: indications and guidelines. J Gastrointest Surg 11:655–659
Alonso S, Dorcaratto D, Pera M et al (2010) Incidence of iatrogenic perforation during colonoscopy and their treatment in a university hospital. Cir Esp 88:41–45
Busić Z, Lovrić Z, Busić V et al (2007) Laparoscopic treatment of iatrogenic endoscopic sigmoid colon perforation: a case report and literature review. J Laparoendosc Adv Surg Tech A 17:324–325
Disclosure
Xiaohui Shi, Yongqi Shan, Enda Yu, Ronggui Meng, Miao Lin, Honglian Xu, Hantao Wang, Lianjie Liu, Liqiang Hao, Wei Zhang, Chuangang Fu, Hao Wang, Xiaodong Xu, Haifeng Gong, Zheng Lou, Junjie Xing, Xianhua Gao, Beili Cai and Haiyan He have no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
Xiaohui Shi and Yongqi Shan contributed equally to the paper.
Rights and permissions
About this article
Cite this article
Shi, X., Shan, Y., Yu, E. et al. Lower rate of colonoscopic perforation: 110,785 patients of colonoscopy performed by colorectal surgeons in a large teaching hospital in China. Surg Endosc 28, 2309–2316 (2014). https://doi.org/10.1007/s00464-014-3458-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-014-3458-1