Abstract
Oil palm fronds are the most abundant lignocellulosic biomass in Malaysia. In this study, fronds were tested as the potential renewable biomass for ethanol production. The soaking in aqueous ammonia pretreatment was applied, and the fermentability of pretreated fronds was evaluated using simultaneous saccharification and fermentation. The optimal pretreatment conditions were 7 % (w/w) ammonia, 80 °C, 20 h of pretreatment, and 1:12 S/L ratio, where the enzymatic digestibility was 41.4 % with cellulase of 60 FPU/g-glucan. When increasing the cellulase loading in the hydrolysis of pretreated fronds, the enzymatic digestibility increased until the enzyme loading reached 60 FPU/g-glucan. With 3 % glucan loading in the SSF of pretreated fronds, the ethanol concentration and yield based on the theoretical maximum after 12 and 48 h of the SSF were 7.5 and 9.7 g/L and 43.8 and 56.8 %, respectively. The ethanol productivities found at 12 and 24 h from pretreated fronds were 0.62 and 0.36 g/L/h, respectively.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Since oil palm trees (Elaeis guineensis Jacq.) were introduced as a major cash crop from West Africa to South-East Asian countries including Malaysia, Indonesia, and India, the increasing land area of oil palm plantation has been generating massive amount of oil palm wastes including trunks, empty fruit bunches (EFB), and fronds in harvesting places [1]. The quantity of oil palm residue production is estimated to be 115 million tons per year based on the 4.7 million hectares in Malaysia alone, and fronds represent approximately 83 million tons per year in available biomass from Malaysian plantations [2]. This production quantity of fronds is 5.5 and 4.7 times larger than those of trunks and EFB in Malaysia, respectively [2]. The vast amount of fronds provides high potential as a biomass feedstock for ethanol production.
Due to the natural recalcitrance and rigidity of lignocellulose, without appropriate physicochemical pretreatment, the enzymatic saccharification yield of glucose is only 20 % of theoretical maximum yield [3, 4]. For instance, the aqueous ammonia pretreatment was found to be effective on trunks of oil palm residues, and 95.4 % of enzymatic digestibility was obtained from ammonia-pretreated trunks [3]. Therefore, search of biomass-specific pretreatment is necessary to establish an effective biomass to biofuel conversion process.
In this study, we tested the feasibility of applying soaking in aqueous ammonia (SAA) pretreatment to the fronds from oil palm for ethanol production. The important pretreatment parameters such as pretreatment temperature, pretreatment time, solid-to-liquid (S/L) ratio, and ammonia concentration were evaluated based on the enzymatic digestibility of cellulose in pretreated biomass. Also, the fermentability of pretreated fronds was evaluated by ethanol yield and productivity from the simultaneous saccharification and fermentation (SSF). This is the first report on the systematic lignocellulosic biomass feedstock evaluation of oil palm fronds in terms of ethanol production.
Materials and methods
Oil palm fronds
Oil palm fronds were kindly supplied from Tropical Chase (Kuala Lumpur, Malaysia). They were ground with a high-speed rotary cutting mill (MF 10, IKA) and screened with sieves (Chung Gye Sang Gong Sa, Seoul, Korea) to give particle sizes in the range 125–706 μm. The initial biomass composition of fronds on a dry weight basis was 39.4 % (w/w) of glucan, 18.6 % of xylan, 1.6 % of galactan, 6.2 % of arabinan, 4.5 % of mannan, 28.5 % of lignin, and 3.2 % of ash. Avicel (Sigma-Aldrich, St. Louis, MO) was used as the control cellulose.
Compositional analysis of fronds
Compositional analysis of pretreated or untreated oil palm fronds was carried out following the National Renewable Energy Laboratory (NREL) procedure [5]. In brief, 0.3 g of fronds was hydrolyzed in 3 ml of 72 % (w/w) sulfuric acid at 30 °C for 1 h, diluted to 4 % and autoclaved at 121 °C for 1 h. The hydrolysate was then filtered through a crucible. The flow through liquid was neutralized with calcium carbonate, and the solid residues on the crucible were dried at 105 °C for 4 h, and the crucible containing the residues was transferred into a furnace (Daihan Labtech, Namyangju, Korea) at 575 °C for 3 h. Acid-insoluble lignin was determined by measuring the weight difference before and after burning in the furnace. Carbohydrate contents were determined by high-performance liquid chromatography equipped with refractive index detector (HPLC-RID; Agilent 1100, Agilent Technologies, Waldbronn, Germany) and SP0810 column (Pb2+ form, 300 mm × 8.0 mm × 7 μm; Shodex, Showa Denko, Kawasaki, Japan). All analyses were conducted in triplicate.
SAA pretreatment of biomass
Ammonia solution (28 %, w/w) (Junsei, Tokyo, Japan) diluted to desired concentrations to be used in SAA pretreatment. Ten grams of fronds was soaked in various concentrations of aqueous ammonia solutions contained in screw-capped laboratory bottles at different S/L ratios. After pretreatment, solid residues of the slurry were filtered through a filtration cloth (pore sizes of 22–25 μm, Calbiochem, La Jolla, CA) and washed with 2 L of distilled water until the wash liquor pH was measured at 6–7. The pretreated insoluble solids were dried in a vacuum-drying oven (BioFree, Seoul, Korea) at 45 °C for more than 3 days until the weight of dry solids became constant, and the samples were stored at −20 °C until further use. To find statistical significances between the differences of overall glucose yields obtained by enzymatic saccharification (i.e., insoluble glucan yield × enzymatic digestibility using 60 FPU/g-glucan) of fronds pretreated at different conditions, a one-way analysis of variance (ANOVA) with post-hoc and least significant difference (LSD) tests was performed using Statistica software (Version 7.1, StatSoft, Tulsa, OK).
Enzymatic digestibility of biomass
Enzymatic digestibility (%) of theoretical maximum yield of glucose from cellulose in pretreated lignocellulose was measured by hydrolyzing 1 % (w/v) glucan of pretreated fronds with various amounts of cellulase and β-glucosidase following the NREL procedure [6]. The cellulase and β-glucosidase that were used in this study were Accellerase 1000 (Genencor, Rochester, NY) and Novozyme 188 (Novozymes, Bagsvaerd, Denmark), respectively. Their activities were measured to be 66.8 FPU/ml and 812.8 CBU/ml, respectively. Cellulase loadings of 15, 30, 60, 100, and 150 FPU of Accellerase 1000/g-glucan were used for the evaluation of enzymatic digestibility of pretreated fronds. The reaction mixtures containing pretreated and washed fronds, untreated fronds, or Avicel in 0.05 M sodium citrate buffer (pH 4.8) were incubated in a shaking incubator (Labcamp, Seoul, Korea) at 50 °C with agitation at 170 rpm for 96 h. The enzymatic hydrolysis experiments were conducted in duplicate.
SSF
SSF was performed at 38 °C, pH 4.8 in a 200-rpm shaking incubator for 48 h. Each flask was equipped with a needle-pierced silicone stopper to vent CO2 produced during fermentation. For SSF, 1 % yeast extract, 2 % peptone, and 0.05 M citrate buffer were added to pretreated and washed fronds, untreated fronds, or Avicel at a 3 % (w/v) of the final glucan concentration in total 100 mL of the SSF medium containing Accellerase 1000 (60 FPU/g-glucan) with β-glucosidase (30 CBU/g-glucan). 10 mL culture of Saccharomyces cerevisiae D5A (ATCC 200062) grown to an absorbance of 5.0 at 600 nm at 30 °C for 12 h was added to the SSF medium as an inoculum.
Quantitative analysis of ethanol
Ethanol was analyzed by gas chromatography equipped with a flame ionization detector (GC-FID; Agilent 7890, Agilent Technologies, Wilmington, DE) with a wax column (DB WAXetr, 30 m × 250 μm × 0.25 μm, Agilent Technologies, Wilmington, DE). The GC was operated in splitless mode with an inlet temperature of 175 °C, an oven temperature of 155 °C, and a detector temperature of 250 °C.
Results and discussion
Effects of pretreatment temperature and S/L ratio
The effects of temperature and S/L ratio on the ammonia pretreatment of oil palm fronds were investigated at temperatures of 40, 60, and 80 °C and S/L ratios of 1:6 and 1:12 (Table 1). In these experiments, pretreatment time and ammonia concentration were fixed at 12 h and 14 % (w/w), respectively. The insoluble solids recovery yield was not affected by changing temperature and remained slightly over 80 % at all the three temperatures. The solid recovery yields of fronds in SAA pretreatment in this study were higher than those of 70 % shown in other SAA pretreatment studies using rice straw and corn stover [4, 7]. From the SAA pretreatment runs at two S/L ratios and three temperatures, the insoluble glucan recovery yield was in the range 84.3–90.6 %, and the xylan recovery yield was 47.3–59.1 %. The highest yields of insoluble glucan and xylan, 90.6 and 59.1 %, were shown at 40 °C and 1:12 of S/L ratio, respectively. Therefore, glucan was much more resistant to solubilization by ammonia than xylan. The glucan recovery yields were much higher than those of SAA-pretreated oil palm trunks and EFB, of which the maximal values were 70.7 and 89.6, respectively [3, 8]. These results imply that lower loss of glucan in fronds than in trunks and EFB after SAA pretreatment can be related to the high recovery yield of insoluble solids and the low lignin removal percentage.
Lignin removal is the major target of aqueous ammonia pretreatment [4, 7] since lignin provides protective shielding of cellulose [9] and enzymes nonspecifically bind to lignin [10, 11]. In the previous studies on SAA for pretreating oil palm trunks [3], rice straw [4], and corn stover [7], the maximal lignin removal percentages were 56.5, 60.6, and 77.3, respectively. However, the lignin removal rates by SAA were only 17.8–19.4 % in oil palm fronds in this study, of which values were even lower than 43.6 % in EFB reported to show relatively low enzymatic digestibility compared to other SAA-pretreated lignocellulose [8]. These low lignin removal percentages may be the reason for the low enzymatic digestibilities that showed the maximal values of 33.7 and 19.5 % with 60 and 15 FPU/g-glucan, respectively. In a previous study of SAA pretreatment of rice straw, the lignin removal percentage was strongly proportional to the enzymatic digestibility of SAA-pretreated rice straw, showing 70.4 % of the maximal enzymatic digestibility for pretreated rice straw with 60.6 % of lignin removal [4]. In the present study, the S/L ratio of 1:12 and temperature of 80 °C were selected as the optimal S/L ratio and temperature for further experiments since the glucose yield from enzymatic hydrolysis using 60 FPU/g-glucan (i.e., insoluble glucan yield × enzymatic digestibility) was significantly higher than those at other pretreatment conditions (p < 0.05).
Effect of pretreatment time
The effects of pretreatment time was studied in the range 4–24 h at the fixed values of S/L ratio of 1:12, temperature of 80 °C, and ammonia concentration of 14 % (w/w) as seen in Table 2. When the cellulase loading was increased to 60 FPU/g-glucan, the enzymatic digestibility increased from 27.6 to 37.0 % by increasing pretreatment time from 4 to 20 h. However, the enzymatic digestibility rather decreased to 31.4% when further extending pretreatment time to 24 h. The lignin removal percentage was in the range 17.0–19.4 when pretreatment times were 4, 8, and 12 h. When the pretreatment time was increased to 16, 20, and 24 h, the lignin removal percentage was in the range 21.0–22.7. The pretreatment time (20 h) that exhibited the highest lignin removal (i.e., 22.7 %) showed the maximal enzymatic digestibility, 37.0 %. The lignin removal levels were much lower than those of other studies using SAA for other lignocellulose [3, 4, 7], and even lower than EFB, which were reported to show relatively low enzymatic digestibility compared to other SAA-pretreated lignocellulose [8]. Therefore, the low enzymatic digestibility of fronds was presumed to be primarily due to the low lignin removal in the present study.
The insoluble xylan recovery yields in the insoluble solids after SAA pretreatment ranged from 47.3 to 63.3 %. The insoluble glucan recovery yields were in the range 89.4–93.2 %, which were much higher than those of SAA-pretreated oil palm trunks and EFB [3, 8]. These results imply that the impact of SAA on the loss of insoluble glucan due to its solubilization into the liquid phase or degradation was weak in the SAA pretreatment of fronds in this study. Possibly due to the high glucan recovery yield and the low lignin removal percentage, the recovery yield of insoluble solids remained over 80 %. The pretreatment time of 20 h was selected as the optimum value, which gave the highest enzymatic digestibility of 37.0 % when cellulase of 60 FPU/g-glucan was used, and the overall glucose yield encompassing the insoluble glucan recovery yield and enzymatic digestibility was significantly higher at 20 h than at other pretreatment times (p < 0.01).
Effect of ammonia concentration
The effects of ammonia concentration of the aqueous solution used in the pretreatment were investigated in the range 2–28 % (w/w) as seen in Table 3. The enzymatic digestibility ranged from 17.8 to 21.9 % and from 28.5 to 41.4 % with cellulase loadings of 15 and 60 FPU/g-glucan, respectively. There was no particular correlation between the enzymatic digestibility and ammonia concentration in this study. The lignin removal percentages were 8.4–24.5 %, and the recovery yields of xylan in the insoluble solids were 59.3–68.4 %. The glucan was highly conserved after SAA pretreatment showing 86.8–94.4 % of insoluble glucan recovery yields. The insoluble solids’ recovery yields remained 81.2–81.7 % and did not significantly change depending on the ammonia concentration. Due to the significantly higher overall glucose yield from the saccharification of 7 % ammonia-pretreated fronds using 60 FPU/g-glucan than those with other ammonia concentrations (p < 0.01), 7 % was chosen as the optimal ammonia concentration.
Effect of cellulase loading on hydrolysis of pretreated fronds
As shown in Fig. 1, fronds pretreated at the optimal pretreatment conditions such as 80 °C, 20 h, an S/L ratio of 1:12, and 7 % of ammonia concentration were hydrolyzed with different loadings of Accellerase 1000. As a control, untreated fronds were hydrolyzed, and the final glucose yields after 96 h were 6.2 and 16.6 % with cellulase loading of 15 and 150 FPU/g-glucan, respectively. The enzymatic digestibility of pretreated fronds significantly increased with an increase of cellulase loading in the range 15–60 FPU/g-glucan showing the glucose yield of 41.4 % after 96 h with 60 FPU/g-glucan. However, further increases of cellulase loading to 100 or 150 FPU/g-glucan did not significantly increase the enzymatic digestibility.
In this study, there was no specific relationship between enzymatic digestibility and pretreatment severity at the enzyme loading of 15 FPU. However at 60 FPU, the enzymatic digestibility increased as the lignin removal percentage increased in all ranges of pretreatment except 2 % ammonia concentration shown by a correlation coefficient of 0.74 (data not shown). In other studies using different lignocellulosic feedstocks with low lignin contents, lignin removal of 39–56 % from oil palm trunks, 33–61 % from rice straw, and 38–77 % from corn stover by SAA were observed [3, 4, 7]. Therefore, the relatively low enzymatic digestibility of SAA-pretreated fronds shown in this study was probably attributed to the low lignin removal percentages, which was similar to the SAA pretreatment of oil palm EFB in a previous study [8].
SSF of pretreated oil palm fronds
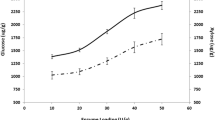
SSF was carried out using pretreated fronds using Accellerase 1000 (60 FPU/g-glucan) and Novozyme 188 (30 CBU/g-glucan) and S. cerevisiae D5A. When untreated fronds were used as shown in Fig. 2a, the final ethanol concentration and yield were only 1.9 g/L and 11.2 % of theoretical maximum ethanol. Xylose was also produced during the SSF due to the possible presence of xylanases in the commercial cellulase preparation. In the SSF of the pretreated fronds (Fig. 2b), glucose increased in the initial SSF period (i.e., 6 h) but immediately decreased and remained <0.5 g/L in the later period. The ethanol concentration and yield at 48 h were 9.7 g/L and 56.8 % of theoretical maximum, respectively. The ethanol productivity until 12 and 24 h from the SSF of pretreated fronds was 0.62 and 0.36 g/L/h, which were comparable to the reported values such as 0.7 g/L/h from steam exploded wheat straw during the initial 3 h of SSF [12] and 0.694 g/L/h from industrial wastes such as old corrugated cardboard during the initial 5 h of SSF [13]. These results indicate that SAA-pretreated fronds can be a candidate biomass feedstock for ethanol production although they showed relatively low enzymatic digestibility (i.e., approximately 50 %) after SAA pretreatment. After 48 h of SSF, 7.6 g/L of xylose was produced, which was equivalent to 62.4 % of theoretical maximum yield of xylose that could be obtained by the hydrolysis of xylan in the pretreated fronds. If a xylose-fermenting strain is applied to the SSF of SAA-pretreated fronds, a much higher yield of ethanol is anticipated.
In conclusion, oil palm fronds are found to be a feasible biomass feedstock for ethanol production. The amounts of sugars and ethanol produced from this process using oil palm fronds are summarized in Fig. 3. In the SSF of the pretreated fronds with 3 % (w/v) glucan loading, the ethanol fermentability comparable to the results of other studies, such as ethanol productivities of 0.62 and 0.36 g/L/h at 12 and 24 h, respectively, were exhibited. Therefore, fronds could be used as biomass for ethanol production.
Mass balance of the overall process including pretreatment, wash, enzymatic hydrolysis, and SSF. a Fronds were pretreated by 7 % (w/w) aqueous ammonia with a S/L ratio of 1:12 at 80 °C for 20 h. b Pretreated fronds were washed with water until neutral pH and hydrolyzed with 60 FPU of Accellerase 1000/g-glucan at 50 °C and 170 rpm for 96 h. c SSF of fronds was conducted using S. cerevisiae D5A and 60 FPU of Accellerase 1000 with 30 CBU of β-glucosidase/g-glucan at 38 °C and 200 rpm for 48 h
References
Sun RC, Fang JM, Mott L, Bolton J (1999) Fractional isolation and characterization of polysaccharides from oil palm trunk and empty fruit bunch fibres. Holzforschung 53:253–260
MPOC (2010) Palm oil: a success story in green technology innovations. http://www.akademisains.gov.my/download/asmic/asmic2010/Plenary12.pdf. Accessed 10 July 2011
Jung YH, Kim IJ, Kim JJ, Oh KK, Han J-I, Choi I-G, Kim KH (2011) Ethanol production from oil palm trunks treated with aqueous ammonia and cellulase. Bioresour Technol 102:7307–7312
Ko JK, Bak JS, Jung MW, Lee HJ, Choi I-G, Kim TH, Kim KH (2009) Ethanol production from rice straw using optimized aqueous-ammonia soaking pretreatment and simultaneous saccharification and fermentation processes. Bioresour Technol 100:4374–4380
Sluiter A, Hames B, Ruiz R, Scarlata C, Sluiter J, Templeton D, Crocker D (2008) Laboratory analytical procedure: determination of structural carbohydrates and lignin in biomass. National Renewable Energy Laboratory, Golden
Selig M, Weiss N, Ji Y (2008) Laboratory analytical procedure: enzymatic saccharification of lignocellulosic biomass. National Renewable Energy Laboratory, Golden
Kim TH, Lee YY (2007) Pretreatment of corn stover by soaking in aqueous ammonia at moderate temperatures. Appl Biochem Biotechnol 136–140:81–92
Jung YH, Kim IJ, Han J-I, Choi I-G, Kim KH (2011) Aqueous ammonia pretreatment of oil palm empty fruit bunches for ethanol production. Bioresour Technol 102:9806–9809
Lipinsky ES (1979) Perspectives on preparation of cellulose for hydrolysis. In: Brown RD Jr, Jurasek L (eds) Hydrolysis of cellulose: mechanisms of enzymatic and acid catalysis. American Chemical Society, Washington DC
Tu M, Pan X, Saddler JN (2009) Adsorption of cellulase on cellulolytic enzyme lignin from lodgepole pine. J Agric Food Chem 57:7771–7778
Yang B, Wyman CE (2006) BSA treatment to enhance enzymatic hydrolysis of cellulose in lignin containing substrates. Biotechnol Bioeng 94:611–617
Tomas-Pejo E, Oliva JM, Ballesteros M, Olsson L (2008) Comparison of SHF and SSF processes from steam-exploded wheat straw for ethanol production by xylose-fermenting and robust glucose-fermenting Saccharomyces cerevisiae strains. Biotechnol Bioeng 100:1122–1131
Kádár Z, Szengyel Z, Réczey K (2004) Simultaneous saccharification and fermentation (SSF) of industrial wastes for the production of ethanol. Ind Crop Prod 20:103–110
Acknowledgments
This work was supported by a grant from the Advanced Biomass R&D Center of Korea (2011-0031353) funded by MEST and also financially supported by the Ministry for Food, Agriculture, Forestry and Fisheries of Korea. Facility support at Korea University Food Safety Hall for the Institute of Biomedical Science and Food Safety is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jung, Y.H., Kim, S., Yang, T.H. et al. Aqueous ammonia pretreatment, saccharification, and fermentation evaluation of oil palm fronds for ethanol production. Bioprocess Biosyst Eng 35, 1497–1503 (2012). https://doi.org/10.1007/s00449-012-0739-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-012-0739-8