Abstract
Long-distance bird migration is fueled by energy gathered at stopover sites along the migration route. The refueling rate at stopover sites is a determinant of time spent at stopovers and impacts the overall speed of migration. Refueling rate during spring migration may influence the fitness of individuals via changes in the probability of successful migration and reproduction during the subsequent breeding season. We evaluated four plasma lipid metabolites (triglycerides, phospholipids, β-OH-butyrate, and glycerol) as measures of refueling rate in free-living semipalmated sandpipers (Calidris pusilla) captured at non-breeding areas. We described the spatial and temporal variation in metabolite concentrations among one winter site in the Dominican Republic and four stopover sites in the South Atlantic and Mid-Atlantic Coastal Plain regions of North America. Triglycerides and β-OH-butyrate clearly identified spatial variation in refueling rate and stopover habitat quality. Metabolite profiles indicated that birds had higher refueling rates at one site in the Mid-Atlantic Coastal Plain than at three sites on the South Atlantic Coastal Plain and one site in the Dominican Republic. Temporal variation in lipid metabolites during the migration season suggested that male semipalmated sandpipers gained more weight at stopovers on the South Atlantic Coastal Plain than did females, evidence of differential migration strategies for the sexes. Plasma lipid metabolites provide information on migration physiology that may help determine stopover habitat quality and reveal how migratory populations use stopover sites to refuel and successfully complete long-distance migrations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The evolution and maintenance of long-distance migration in birds is conditional on the ability of individuals to store sufficient energy to fuel extended non-stop flights. The primary source of energy for flight phases of bird migration is subcutaneous lipid deposits (Stevens 1996). At the beginning of migration, substantial amounts of lipid are accumulated before the first flight, and then replenished at stopovers en route. The refueling rate at migratory stopovers influences not only the amount of time spent at a stopover, but also may impact individual fitness during the remainder of migration, and during the ensuing breeding season (Alerstam and Lindström 1990). Recent evidence suggests that declines in fuel deposition rate of red knots (Calidris canutus) staging at Delaware Bay, USA may be responsible for apparent declines of this species (Baker et al. 2004).
Energetic condition of migrants can be evaluated in many ways. Size-corrected body mass has been used as a measure of energy reserves for many bird species (Davidson 1983; Summers 1988; Winker et al. 1992). Predictive regression equations derived from chemical analysis of carcasses have also been used to predict lean mass and total lipid stores of live birds (Lyons and Haig 1995). Indices of energetic condition (e.g., estimated percent fat of live birds) provide a static measure of energetic condition, however, and provide no information about the trajectory of body mass change in individuals. Recent work suggests that plasma metabolites provide indices of rate and direction of mass change of refueling individuals (Williams et al. 1999; Seaman et al. 2005; Cerasale and Guglielmo 2006a).
Certain plasma metabolites indicate changes in body mass of birds (Jenni-Eiermann and Jenni 1994; Williams et al. 1999). Triglycerides are the major form of stored fuel in birds, and plasma concentrations of triglycerides have been positively correlated with fat deposition in captive sandpipers (Stevens 1996; Williams et al. 1999; Seaman et al. 2005). During periods of fat deposition and increasing body mass, triglycerides of dietary and hepatic origin are transported to adipose tissue as lipoproteins (Ramenofsky 1990; Stevens 1996). Lipoproteins also contain endogenous and dietary phospholipids, important components of cell membranes that have been suggested to play an energy-delivery role in refueling birds (Guglielmo et al. 2002b; Cerasale and Guglielmo 2006a; Cerasale and Guglielmo 2006b). Plasma concentrations of triglycerides, and in some cases phospholipids, are elevated when energy intake exceeds energy demand (e.g., during refueling bouts). Conversely, when energy demand exceeds intake (e.g., restricted feeding, fasting, or endurance flight) stored lipids are mobilized from adipose tissue resulting in elevated concentrations of plasma glycerol (Ramenofsky 1990). When fat catabolism is elevated, the liver produces β-OH-butyrate as an alternative metabolic fuel for nervous and other tissues (Stevens 1996). Elevated plasma glycerol and β-OH-butyrate generally indicate declining body mass in birds (Williams et al. 1999). However, recent work suggests that glycerol may also increase at very high rates of fat deposition (Guglielmo et al. 2005).
In this study we test for spatial and temporal variation in four plasma lipid metabolites in free-living semipalmated sandpipers (Calidris pusilla) before and during vernal migration. To assess the spatial variation in metabolites, we collected plasma samples at one wintering area in the Dominican Republic and at stopover sites in the South Atlantic (three sites) and Mid-Atlantic (one site) Coastal Plain regions of North America. We describe refueling performance along a latitudinal gradient and place the stopover sites in the context of the entire migration system to increase our understanding of vernal migration strategies of semipalmated sandpipers. Specifically, we test a regional hypothesis describing how physiographic regions may function to support migrants and contribute to successful migration. To assess temporal variation, we also collected samples throughout the migration season at one stopover site in the South Atlantic Coastal Plain. Finally, based on both spatial and temporal variation of plasma lipid metabolites, we determine the best indicator(s) of refueling rate and stopover habitat quality, and describe their usefulness to future research on migration physiology.
Materials and methods
Study sites and field methods
This study was conducted at five latitudes encompassing three geographic regions: Dominican Republic, South Atlantic, and Mid-Atlantic Coastal Plain. Regions were defined by dominant wetland landscapes, available prey resources, and position in the migratory route (pre-migration, stopover, and last staging area before breeding). The study area thus included the northern part of the winter range and a substantial portion of the northward migratory route of semipalmated sandpipers in North America (Gratto-Trevor 1992; Rice et al. 2007). We collected blood samples during northward migration in 2001 at Yawkey Wildlife Center (YWC), Georgetown, South Carolina (33°10′N, 79°13′W) to assess intraseasonal temporal variation, and during northward migration in 2002 at five sites described below, including YWC, to assess spatial variation in metabolite concentrations along a latitudinal gradient.
YWC comprises 5,425 ha of tidal marsh, barrier beach, maritime forest, and managed wetlands. Field work for this study was conducted entirely within the 12 managed wetlands of the center, which are non-tidal, brackish water (9–35 ppt) impoundments ranging in size from 4 to 98 ha (Weber and Haig 1997). The invertebrate prey base of YWC wetlands is dominated by chironomid larvae, oligochaetes, and polychaetes, especially Leonareis culveri (Wenner 1987; Weber and Haig 1997). In 2001, we collected blood samples from 70 birds captured with mist nets placed over foraging areas. Nets were operated in the morning on 2–3 consecutive days each week in May; 90% of all birds were sampled between 0800 and 1200 hours. Birds were held in boxes before banding and blood sampling. Birds were weighed to the nearest 0.1 g using a digital balance, measured (wing chord, tarsus length, exposed culmen), and banded with a U.S. Fish and Wildlife Service (USFWS) numbered, metal band and a unique combination of color bands (as part of ongoing research). Each bird was classified as adult or immature based on plumage characteristics (Prater et al. 1977).
In 2002, we collected blood samples at one wintering area in the Caribbean, three stopovers in the South Atlantic Coastal Plain, and one in the Mid-Atlantic Coastal Plain. Lago Enriquillo, Dominican Republic (18°27′N, 71°41′W) is a hypersaline lake of about 20,000 ha near the northern limit of the winter range of C. pusilla (Gratto-Trevor 1992; Rice et al. 2007). Salinity ranges from 35 to 80 p.p.t. and invertebrate fauna include crustaceans (especially Artemia sp.), Foraminifera, Ostracoda, and Gastropoda (Buck et al. 2005, personal observation). We collected blood samples from 25 birds at Lago Enriquillo during 29 March–1 April (0730–1220 hours). During 6–9 May (0650–0950 hours), we collected blood samples from 20 birds at Merritt Island National Wildlife Refuge (NWR), Titusville, Florida (28°39′N, 80°46′W). Merritt Island NWR is a 55,000-ha barrier beach complex that includes 76 brackish water impoundments, with salinity ranging from 10 to 42 p.p.t. (Provancha and Sheidt 2000; Herring and Collazo 2005); the dominant prey species are chironomid larvae, oligochaetes, and polychaetes (Herring and Collazo 2001). We returned to YWC during 14–16 May 2002 (0715–1100 hours) and sampled 43 birds in some of the same managed wetlands as 2001. Pea Island NWR (35°44′N, 75°30′W), located on the Outer Banks of North Carolina, includes three brackish water impoundments (salinity <30 p.p.t.) with a total area of 380 ha; dominant prey species include chironomid larvae, oligochaetes, polychaetes, amphipods, and coleopteran larvae (Collazo et al. 2002). We sampled 49 birds during 21–24 May (0600–1050 hours) at Pea Island NWR. Our fourth stopover area was Fortescue Beach, Fortescue, New Jersey (39°13′N, 75°10′W), on the north shore of Delaware Bay in the Mid-Atlantic Coastal Plain, where we sampled 50 birds during 28–31 May (0650–1700 hours). The dominant prey item of birds on beaches of Delaware Bay is eggs of the horseshoe crab (Limulus polyphemus; Castro and Myers 1993; Tsipoura and Burger 1999) Twenty-seven birds (54%) at Fortescue Beach were captured at a high tide roost site with a cannon net. The remaining birds at Fortescue Beach, and all birds at other sites, were captured over foraging areas using mist nets. We handled birds and collected blood samples using methods similar to those described above for YWC in 2001. In 2002, we recorded the number of minutes between capture and blood-sampling for each bird to use as a covariate (hereafter “handling time”); we did not collect data on handling time in 2001, but we assume that variation in handling time was minimal because we were only at one site in 2001 and procedures were similar on each day that we captured birds.
Blood sampling and metabolite assays
A 200- to 300-μl blood sample was collected from the brachial vein using a 26-gauge needle and heparinized capillary tubes. Blood samples were stored on ice in a small cooler in the field and centrifuged in capillary tubes at 6,000 r.p.m. for 10 min; plasma was extracted using a 100-μl syringe. Plasma samples were stored in a conventional freezer 1–4 weeks and then stored at −80° C prior to analysis.
Lipid metabolites were assayed in a microplate spectrophotometer. Total triglyceride and free glycerol were measured sequentially by endpoint assay (Sigma Diagnostics Trinder reagents A and B; Williams et al. 1999). True triglyceride concentration was calculated by subtracting glycerol from total triglyceride. β-OH-butyrate was measured by kinetic assay (Sigma; Williams et al. 1999) and phospholipids were measured by endpoint assay using a calorimetric kit (Wako Diagnostics; Guglielmo et al. 2005). Birds were captured, marked and sampled under a USFWS permit, and the capture, handling, and blood-sampling protocols were approved by the North Carolina State University Institutional Animal Care and Use Committee permit 01-050-0.
Statistical analyses
We used model-based data analyses to evaluate the support in the data for various hypotheses describing plasma metabolite concentrations during migration. We used Akaike’s information criterion adjusted for small sample sizes (AIC c ; Akaike 1973; Hurvich and Tsai 1989) to compare general linear models which we fit using the GLM procedure of SAS/STAT software (SAS Institute 2004). AIC c values were calculated using residual sums-of-squares according to Burnham and Anderson (2002, pp 63–66). Models were ranked by AIC c , where the model with the minimum AIC c was the model with the most support in the data. The difference in AIC c units between the best supported model and any other model (ΔAIC c ) was used to calculate AIC c model weights, which indicate the relative likelihood of the model given the data (Burnham and Anderson 2002). AIC c model weights are normalized across the set of candidate models to sum to one. Therefore, a model with AIC c weight approaching one is unambiguously supported by the data and models with equal weights have a similar level of support in the data.
We created two a priori candidate model sets that would allow us to test hypotheses about spatial and temporal variation in plasma metabolites. Our first candidate model set focused on temporal variation in metabolites during 2001 at YWC. We used Julian date of capture (as a continuous variable) to model temporal changes in plasma metabolites as the migration season progressed. The candidate model set included 14 models: a null model (intercept only); all one, two, and three factor combinations of date, age (adult vs. immature) and body mass; and a subset of interaction models. We included the following six interaction models based on biological reasoning and interest (Guglielmo et al. 2002a; Guglielmo et al. 2005): (1) [Date, Age, Date × Age]; (2) [Date, Age, Mass, Date × Age]; (3) [Date, Age, Mass, Age × Mass]; (4) [Date, Age, Mass, Date × Mass, Age × Mass]; (5) [Age, Mass, Age × Mass]; and (6) [Date, Date2]. We included both a linear and quadratic model on date because the model set focused on temporal variation and we had no a priori basis to predict the functional form of this relationship. An interaction between age and body mass accounted for possible age-specific body composition and migration physiology. An interaction between date and age accounted for possible age-specific migration schedules; adults may precede juveniles in the spring (Gratto-Trevor 1992).
In our second candidate model set, we modeled metabolite concentrations at two alternative spatial scales: regional and site-specific. The regional hypothesis predicts that refueling performance for the three South Atlantic Coastal Plain stopovers (Merritt Island NWR, YWC, and Pea Island NWR) will be equal to each other, yet unequal to either the Dominican Republic region (i.e., Lago Enriquillo) or the Mid-Atlantic Region (i.e., Fortescue Beach). We predicted that refueling rate at the South Atlantic Coastal Plain stopovers would be equal because the prey base and foraging habitats are similar across these sites. We predicted that metabolic profiles would indicate greatest refueling performance in the Mid-Atlantic Coastal Plain based on the empirical evidence for substantial body mass increase of shorebirds feeding on horseshoe crab eggs in this region (Castro and Myers 1993; Tsipoura and Burger 1999). Although we expected that birds in the Dominican Republic might be engaged in pre-migratory fueling before a non-stop flight to North America (Rice et al. 2007), we did not make specific predictions about refueling performance and metabolic profiles at Lago Enriquillo because there was little available information on which to base quantitative predictions. We compared the likelihood of models based on the regional hypothesis with models where metabolite concentrations varied on a site-by-site basis. If the mean metabolite concentration at any South Atlantic Coastal Plain site was not equal to the other sites in the region, the regional hypothesis would not be supported. Finally, our null hypothesis is that all sites are equal (i.e., no spatial variation). The candidate model set included 15 models. Region and site effects occurred in five models each, yet never in the same model. The five models included all one, two, and three factor combinations between region (or site), body mass, and handling time, and the interaction model [Region/Site, Mass, Handling time, Handling time × Mass]. We also included a null model (intercept only) and two additional models based on biological reasoning and interest: [Handling time, Mass], [Handling time, Mass, Handling time × Mass]. We included body mass and handling time as covariates because these factors may influence metabolite concentration (Guglielmo et al. 2002a; Guglielmo et al. 2005); it was not possible to include age as a covariate due to insufficient captures of immature birds in 2002. To assess the cumulative support in the data for Region versus site effects, we calculated relative importance values for these two factors according to Burnham and Anderson (2002); we did not calculate relative importance values for the covariates because some covariates occurred in more models than others. We present mean metabolite concentrations for regions and sites using least-squares means; least-squares means represent the expected mean metabolite concentration at the mean of all predictor variables in the model with the most support (SAS Institute 2004).
Results
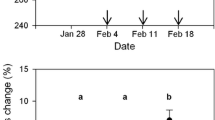
Plasma triglyceride concentrations decreased as the 2001 migratory season progressed at YWC, South Carolina. The earliest migrants clearly had the highest levels of triglyceride; concentrations declined and leveled off later in the season (Fig. 1a). There was substantial evidence that the decline was not linear: a quadratic model on date received 88% of the AIC c model weight, with the high weight indicating little uncertainty in model selection (Table 1). The daily mean phospholipid concentration was nearly constant across all sample dates, with substantial variation within each sample date (Fig. 1c). There was considerable model uncertainty in the phospholipid models and the null model received the most support (31% of the AIC c model weight; Table 1). Nevertheless, the second-ranked model suggested that a linear model on date might account for some of the variation in phospholipid (ΔAIC c = 1.169 and 18% of the AIC c model weight). β-OH-butyrate concentration increased linearly with date and decreased with body mass (Fig. 1b, Table 1). The effect of body mass on plasma β-OH-butyrate, however, was marginal: the 95% confidence intervals (CI) for the body mass coefficient in the general linear model included zero (−0.037, 0.001), and a model with only date received substantial support (ΔAIC c = 1.36 and 20% of the AIC c model weight). There was a great deal of model selection uncertainty for the 2001 glycerol assay data. There were four models with relatively high AIC c model weights, ranging from 0.11 to 0.28 (Table 1). Although no clear effects of date, age, or body mass were supported, age and body mass dominated the top four glycerol models (Table 1, Fig. 1d). Birds captured early and late in the season had relatively low and high body mass, respectively (Table 2). Adult birds had greater concentrations of plasma glycerol [95% CI: (0.42, 0.51)] than immature birds [95% CI: (0.31, 0.42)].
Distribution of plasma metabolite concentrations of semipalmated sandpipers (Calidris pusilla) captured during spring migration at Yawkey Wildlife Center, South Carolina, USA in May 2001. Each week comprised 2–3 consecutive days of capture beginning on the date shown. Heavy lines indicate the median; boxes the middle 50% of the data; whiskers the range of the data excluding outliers, which are indicated with open circles. See Table 2 for sample sizes
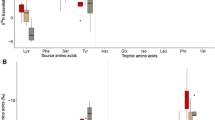
In 2002, we collected blood samples from 187 birds at five sites in three geographic sampling regions from 29 March to 31 May. Triglyceride varied among regions (Table 3) and was substantially higher at Fortescue Beach in the Mid-Atlantic Coastal Plain than any other region (Fig. 2a). Model [Region, Handling time, Mass, Handling time × Mass] was selected as the top model, indicating that triglyceride concentrations were similar at Merritt Island NWR, YWC, and Pea Island NWR in the South Atlantic Coastal Plain (Table 3). Triglyceride concentrations in the Dominican Republic were similar to sites in the South Atlantic Coastal Plain, i.e., lower than at Fortescue Beach (Fig. 2a). In the triglyceride analysis, relative importance values for Region and Site were 0.63 and 0.12, respectively. The best supported model for 2002 phospholipid assay data, [Region, Handling time, Mass, Handling time × Mass], was identical to the 2002 triglyceride model (Table 3) and received 76% of the AIC c model weight. Phospholipid was lower in the Dominican Republic than either the South Atlantic or Mid-Atlantic Coastal Plain, where phospholipid concentrations were similar (Fig. 2c). In the phospholipid analysis, relative importance values for Region and Site were 0.78 and 0.21, respectively. β-OH-butyrate plasma concentrations, unlike triglyceride and phospholipid, varied among sites, rather than just among regions. While β-OH-butyrate concentrations were greatest at Lago Enriquillo and lowest at Fortescue Beach, β-OH-butyrate at YWC was higher relative to the other South Atlantic sites than predicted by the regional hypothesis (Fig. 2b). The model selection results indicated little uncertainty about the best model for the β-OH-butyrate assay data. The top model, which received 65% of the AIC c model weight, included effects of site, handling time, and body mass (Table 3); the relative importance values for Region and Site were 0.01 and 0.99, respectively. β-OH-butyrate increased with handling time [95% CI for β-OH-butyrate coefficient in general linear model, (0.009, 0.025)] and decreased with body mass [95% CI for body mass coefficient in general linear model, (−0.191, −0.016)]. Glycerol concentrations in 2002 also varied among sites rather than regions (Fig. 2d). In contrast to our predictions, glycerol was high at Fortescue Beach (where body mass was highest) as well as Lago Enriquillo [where body mass was low (Table 4)]. There was essentially no model selection uncertainty, with [Site, Handling time, Mass, Handling time × Mass] receiving 99% of AIC c model weight (Table 3); the relative importance of Region and Site were 0.01 and 0.99, respectively.
Estimated plasma metabolite concentrations (least squares means and 95% confidence interval) for semipalmated sandpipers along a latitudinal gradient. Least-squares means are presented by a, c region or b, d site based on all predictor variables in the best supported model identified in Tables 1 and 3 for each metabolite. Regions and sites are arranged from south to north. Regions are Dominican Republic, South Atlantic Coastal Plain (South ACP), and Mid-Atlantic Coastal Plain (Mid-ACP); see text for description of regions. Sites are Lago Enriquillo, Dominican Republic (DR1); Merritt Island National Wildlife Refuge (NWR), Florida (SA1); Yawkey Wildlife Center, South Carolina (SA2); Pea Island NWR, North Carolina (SA3); and Fortescue Beach, New Jersey (MA1). See Table 4 for sample sizes
Discussion
Metabolite profiles in this study provided information about refueling performance at specific stopover sites in relation to each other, and in the context of vernal migration of semipalmated sandpipers in eastern North America. Elevated triglyceride concentrations indicated rapid refueling, whereas elevated β-OH-butyrate indicated slower weight gain. Birds at Lago Enriquillo, Dominican Republic had low body mass, intermediate concentrations of triglyceride, and high β-OH-butyrate concentrations. Many of these birds were immature and probably were not preparing to migrate north. Approximately two-thirds of all immature semipalmated sandpipers do not migrate to the breeding grounds as yearlings, but spend the boreal summer on wintering areas (Gratto-Trevor 1992). Birds captured at stopovers on the South Atlantic Coastal Plain (i.e., Florida, South Carolina, and North Carolina) showed similar metabolic profiles across sites: intermediate concentrations of both triglyceride and β-OH-butyrate. Given the energy reserves and metabolite concentrations of migrants at sites in this region, it is unlikely that these birds are making non-stop flights to the breeding grounds (Harrington and Morrison 1979). Birds captured at Fortescue Beach on Delaware Bay, however, were in a different refueling class. At Fortescue, birds had highest triglyceride and lowest β-OH-butyrate concentrations, suggesting that once birds reach this site, the rate of mass increase and lipid storage is substantially elevated. Results of this study suggest that birds stopping on the South Atlantic Coastal Plain likely move north along the coast until reaching Delaware Bay, then stage for longer periods and gain substantial lipid reserves before making a direct flight to the breeding grounds (Morrison 1984; Mizrahi and Peters 2006). This does not suggest, however, that the South Atlantic Coastal Plain is of little consequence to successful migration. On the contrary, the stopover sites in this region probably host a large proportion of breeding populations of semipalmated sandpipers from the central and eastern Canadian Arctic each year (Harrington and Morrison 1979; Morrison 1984). These stopovers provide habitats where birds can maintain energy reserves, and act as stepping stones to the major staging area at Delaware Bay.
Two previous studies have examined refueling rate at stopover areas over a large geographic area. Schaub and Jenni (2001) estimated fuel deposition rates using triglycerides and β-OH-butyrate concentrations of four passerine species captured at 14 stopover sites from northern Europe to sub-Saharan Africa. Refueling rate varied among sites but was not correlated with latitude; furthermore, in only one of four species was refueling rate impacted by impending ecological barriers, e.g., sea, desert, mountain range (Schaub and Jenni 2001). The results of Schaub and Jenni (2001) suggest that it will be difficult for migrants to predict the refueling rate at the next stopover site, whereas our results, at least for triglycerides, indicate that refueling rate may be predictable within certain physiographic regions. Williams et al. (2007) found that triglyceride concentrations of western sandpipers (Calidris mauri) not only varied among sites from Mexico to Alaska, but also increased linearly with latitude. Williams et al. (2007) also measured invertebrate biomass along the migration route and found that prey resources did not increase with latitude, suggesting that migrants increased refueling rate at more northerly stopovers via physiological or behavioral mechanisms (Williams et al. 2007). We did not find a linear increase in refueling rate with latitude, and the contrasting results emphasize the importance of spatial variation in resources at multiple scales, including site, region, and flyway levels. The spatial arrangement of resources, and the timing of resource availability, within each flyway present unique opportunities and constraints to migrants, and may explain the different latitudinal patterns of Williams’ et al. (2007) and our study.
Plasma metabolites also suggest different strategies for early and late migrants based on our season-long sampling at YWC. Early migrants had the highest levels of triglycerides, indicating substantial weight gains, whereas late migrants had lower levels of triglyceride (Fig. 1). β-OH-butyrate, an indication of fasting, showed the opposite temporal pattern: β-OH-butyrate levels were low among early migrants and highest among the late arrivals. Taken together, these two metabolites indicate that early and late migrants may use two different refueling strategies on the South Atlantic Coastal Plain. Early migrants appear to be gaining weight at stopover sites in the region as they move northward. Late migrants, on the other hand, appear to travel with higher fuel loads and may use the region primarily for resting and to maintain fuel levels, rather than for additional weight gain. Furthermore, male semipalmated sandpipers precede females in the southeast during spring passage (Lyons 1994), and these results suggest that males and females have different spring migration strategies, which may affect their body condition and timing of arrival on the breeding grounds. Schaub and Jenni (2001) did not find a temporal trend in refueling rate within a season, but they sampled only near the peak of migration, rather than across the season, and therefore did not include early and late-migrating individuals.
Our results are consistent with previous studies for some metabolites and inconsistent for others. Among the metabolites we examined, triglyceride and β-OH butyrate appear to be the most useful for studies of migration physiology (Guglielmo et al. 2005; Seaman et al. 2006). Triglyceride concentrations were consistent with prior independent information about habitat quality and refueling performance at our study sites. Guglielmo et al. (2005) also found triglyceride to be a useful indicator of refueling performance of wild birds, and of habitat quality for sites of known quality. Phospholipids may be less useful in studies of migration physiology (but see Cerasale and Guglielmo 2006b). Phospholipids were low at Lago Enriquillo and provided corroborating evidence that birds were not refueling rapidly, if at all, at this site, but similar information was provided by triglyceride and β-OH-butyrate. Phospholipids were not useful as a measure of refueling rate over time because mean phospholipid concentration did not change substantially over time in South Carolina, whereas triglycerides and β-OH-butyrate indicated substantial variation in refueling performance. Furthermore, variation about the mean phospholipid concentration was substantial, reducing power to detect changes over time or among habitats. Guglielmo et al. (2005) found that phospholipid concentrations, when examined alone, did not vary among habitats of different quality, yet when used in a principle components analysis with other metabolites, phospholipids helped distinguish habitats of low and high quality.
Glycerol may be particularly unsuited for use in investigations of migration physiology. Glycerol has been correlated with mass loss and poor body condition in captive studies (Williams et al. 1999), and we initially predicted it might decrease along the migration route as birds moved to the major staging area at Fortescue Beach. Contrary to predictions, however, glycerol levels were high at Fortescue Beach as well as Lago Enriquillo in the Dominican Republic (Fig. 2). This pattern could result from two mechanisms. First, glycerol is produced by lipolysis of triglycerides in adipose tissue and muscle during periods of negative energy balance and exercise (Stevens 1996). Elevated glycerol levels at Laqo Enriquillo and Delaware Bay may have been the result of strenuous exercise at each site, although for different reasons. We observed several American kestrels (Falco sparverius) and merlins (Falco columbarius) hunting shorebirds at Lago Enriquillo; these predators made multiple attacks on shorebirds in the area. Migrants at Fortescue Beach were likely preparing for non-stop flight to the breeding grounds, and were observed to repeatedly make pre-migratory flights, circling overhead and returning to the beach again and again. Furthermore, these birds may have exercised as we moved them to the target area for the cannon net. A second mechanism for the U-shaped pattern of glycerol we observed was suggested by Guglielmo et al. (2005). They reported that glycerol concentrations increased at very high plasma triglyceride concentrations, such as we measured at Fortescue Beach, possibly because of very high rates of hydrolysis of plasma triglycerides during fatty acid uptake by adipocytes. Our findings reinforce the assertion that plasma glycerol on its own is not suitable for evaluating refueling performance in field studies.
Future field studies should strive to minimize handling time. In this study, triglyceride and phospholipids tended to decrease with handling time, whereas β-OH-butyrate and glycerol tended to increase. We were able to account for effects of handling time in our study of spatial variation in metabolites along the latitudinal gradient because we collected data on this effect and included it as a covariate in the candidate model set. We were not able to include handling time in the candidate model set for temporal variation in metabolites (season-long sampling) because we did not collect data on time between capture and blood sampling in 2001. While we believe that we were able to minimize variation in handling time among days because all sampling was conducted at the same site with identical field procedures on each sampling day in 2001, our results for temporal variation in metabolites should be interpreted with caution. Future observational studies of refueling performance should strive to collect blood samples immediately after capture if possible to ensure accurate estimates of plasma metabolite concentrations.
This study provides further evidence that plasma metabolites, especially triglycerides and β-OH-butyrate, provide information on refueling performance useful for conservation and management of migratory shorebirds (Guglielmo et al. 2002a; Seaman et al. 2006). In fact, plasma metabolite analysis may be the only viable option for studying refueling in many shorebird species. Analysis of recapture data is unlikely to be fruitful because daily recaptures of shorebirds are extremely rare at most sites. Regression of mass against time of day is not suitable because many shorebirds feed in response to tidal cycle rather than time of day, they may not be accessible for capture at all times, or may even feed day and night.
Shorebird populations around the world are in decline, and long-distance migratory populations are more likely to decline than short-distance migratory or resident populations (Wetlands International 2002; Zöckler et al. 2003; Thomas et al. 2006). The potential causes of population declines include habitat loss and degradation along the migration route, which may impact the ability of migrants to acquire sufficient energy reserves for successful migration (Baker et al. 2004; Skagen 2006). Given the association between long-distance migration and population status, a better understanding of migration physiology and stopover behavior may help identify causes and effects of change in migratory shorebird populations. Plasma lipid metabolites provide a model system for testing hypotheses about population dynamics and monitoring stopover habitat quality to help maintain viable populations and migration systems.
References
Akaike H (1973) Information theory as an extension of the maximum likelihood principle. In: Petrov BN, Csaki F (eds) Second international symposium on information theory. Akademiai Kiado, Budapest, pp 267–281
Alerstam T, Lindström Å (1990) Optimal bird migration: the relative importance of time, energy, and safety. In: Gwinner E (ed) Bird migration: physiology and ecophysiology. Springer, Berlin, pp 331–351
Baker AJ, González PM, Piersma T, Niles LJ, de Lima Serrano do Nascimento I, Atkinson PW, Clark NA, Minton CDT, Peck MK, Aarts G (2004) Rapid population decline in red knots: fitness consequences of decreased refuelling rates and late arrival in Delaware Bay. Proc R Soc Lond B 271:875–882
Buck DG, Brenner M, Hodell DA, Curtis JH, Martin JB, Pagani M (2005) Physical and chemical properties of hypersaline Lago Enriquillo, Dominican Republic. Verh Int Verein Limnol 29:725–731
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, New York
Castro G, Myers J (1993) Shorebird predation on eggs of horseshoe crabs during spring stopover on Delaware Bay. Auk 110:927–930
Cerasale DJ, Guglielmo CG (2006a) Dietary effects on prediction of body mass changes in birds by plasma metabolites. Auk 123:836–846
Cerasale DJ, Guglielmo CG (2006b) Plasma metabolite profiles: effects of dietary phospholipids in a migratory passerine (Zonotrichia leucophrys gambelii). Physiol Biochem Zool 79:754–762
Collazo JA, O’Harra DA, Kelly CA (2002) Accessible habitat for shorebirds: Factors influencing its availability and conservation implications. Waterbirds 25(Special Publication 2):13–24
Davidson NC (1983) Formulae for estimating the lean weight and fat reserves of live shorebirds. Ring Migr 4:159–166
Gratto-Trevor CL (1992) Semipalmated sandpiper. In: Poole A, Stettenheim P, Gill F (eds) The birds of North America, no. 6. Academy of Natural Sciences, Philadelphia
Guglielmo CG, O’Hara PD, Williams TD (2002a) Extrinsic and intrinsic sources of variation in plasma lipid metabolites of free-living western sandpipers (Calidris mauri). Auk 119:437–445
Guglielmo CG, Williams TD, Zwingelstein G, Brichon G, Weber J (2002b) Plasma and muscle phospholipids are involved in the metabolic response to long-distance migration in a shorebird. J Comp Physiol B 172:409–417
Guglielmo CG, Cerasale DJ, Eldermire C (2005) A field validation of plasma metabolite profiling to assess refueling performance of migratory birds. Physiol Biochem Zool 78:116–125
Harrington B, Morrison R (1979) Semipalmated sandpiper migration in North America. In: Pitelka F (eds) Studies in avian biology, vol 2. Cooper Ornithological Society, Camarillo, pp 83–100
Herring G, Collazo JA (2001) Habitat quality and use by waterfowl in impounded wetlands under various hydrological treatments and adjacent estuarine areas at Merritt Island National Wildlife Refuge. North Carolina State University, Raleigh
Herring G, Collazo JA (2005) Habitat use, movements, and home range of wintering lesser scaup in Florida. Waterbirds 28:71–78
Hurvich C, Tsai C-L (1989) Regression and time series model selection in small samples. Biometrika 76:297–307
Jenni-Eiermann S, Jenni L (1994) Plasma metabolite levels predict individual body-mass changes in a small long-distance migrant, the garden warbler. Auk 11:888–899
Lyons JE (1994) Eco-physiology of shorebirds during winter and migration at South Island, South Carolina. M.S. thesis, Clemson University
Lyons JE, Haig SM (1995) Fat content and stopover ecology of spring migrant semipalmated sandpipers in South Carolina. Condor 97:427–437
Mizrahi D, Peters K (2006) Semipalmated and least sandpiper stopover dynamics on Delaware Bay during spring migration. New Jersey Audubon, Cape May Court House
Morrison R (1984) Migration systems of some New World shorebirds. In: Burger J, Olla B (eds) Behavior of marine animals, vol 6. Plenum, New York, pp 125–202
Prater T, Marchant J, Vuorinen J (1977) Guide to the identification and ageing of Holarctic waders. British Trust for Ornithology, Tring
Provancha JA, Sheidt DM (2000) Long-term trends in seagrass beds in the Mosquito Lagoon and northern Banana River, Florida. In: Dennish MJ (ed) Seagrasses: monitoring, ecology, physiology, and management. CRC, Boca Raton, pp 177–193
Ramenofsky M (1990) Fat storage and fat metabolism in relation to migration. In: Gwinner E (ed) Bird migration: physiology and ecophysiology. Springer, New York, pp 214–231
Rice S, Collazo JA, Alldredge M, Harrington B, Lewis A (2007) Local annual survival and seasonal residency rates of semipalmated sandpipers in Puerto Rico. Auk 124:1397–1406
SAS Institute (2004) SAS OnlineDoc 9.1.3. SAS Institute, Cary
Schaub M, Jenni L (2001) Variation in fuelling rates among sites, days and individuals in migrating passerine birds. Funct Ecol 15:584–594
Seaman DA, Guglielmo CG, Williams TD (2005) Effects of physiological state, mass change and diet on plasma metabolite profiles in western sandpiper Calidris mauri. J Exp Biol 208:761–769
Seaman DAA, Guglielmo CG, Elner RW, Williams TD (2006) Landscape-scale physiology: site differences in refueling rates indicated by plasma metabolite analysis in free-living migratory sandpipers. Auk 123:563–574
Skagen SK (2006) Migration stopovers and the conservation of arctic-breeding calidridine sandpipers. Auk 123:313–322
Stevens L (1996) Avian biochemistry and molecular biology. Cambridge University Press, Cambridge
Summers RW (1988) The use of linear measurements when comparing masses. Bird Study 36:77–79
Thomas GH, Lanctot RB, Szekely T (2006) Can intrinsic factors explain population declines in North American breeding shorebirds? A comparative analysis. Anim Conserv 9:252–258
Tsipoura N, Burger J (1999) Shorebird diet during spring migration stopover on Delaware Bay. Condor 101:635–644
Weber LM, Haig SM (1997) Shorebird–prey interactions in South Carolina coastal soft sediments. Can J Zool 75:245–252
Wenner EL (1987) Benthic macrofauna. In: DeVoe MR, Baughman DS (eds) South Carolina coastal wetland impoundments, vol II. South Carolina Sea Grant Consortium, Charleston, pp 255–293
Wetlands International (2002) Waterbird population estimates, 3rd edn. Wetlands International, Wageningen
Williams TD, Guglielmo CG, Egeler O, Martyniuk CJ (1999) Plasma lipid metabolites provide information on mass change over several days in captive western sandpipers. Auk 116:994–1000
Williams TD, Warnock N, Takekawa JY, Bishop MA (2007) Flyway scale variation in plasma triglyceride levels as an index of refueling rate in spring migrating western sandpipers. Auk 124:886–897
Winker K, Warner DW, Weisbrod AR (1992) Daily mass gains among woodland migrants at an inland stopover site. Auk 109:853–862
Zöckler C, Delany S, Hagemeijer W (2003) Wader populations are declining—how will we elucidate the reasons? Wader Study Group Bull 100:202–211
Acknowledgements
We thank Aaron Brees, Francisco Collazo, Garth Herring, Jeff Hostetler, and Jordan Perkins for excellent assistance in the field. Sarah Converse and two anonymous reviewers provide valuable comments on the manuscript. Several individuals and institutions provided logistical support and we would like to specifically thank Robert Joyner, YWC; Marc Epstein, Merritt Island NWR; Bob Noffsinger, Pea Island NWR; and David Mizrahi, New Jersey Audubon Society. This work was funded by the Species at Risk Program of the U.S. Geological Survey and approved by the North Carolina State University Institutional Animal Care and Use Committee (permit 01-050-0).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Carol Vleck.
Rights and permissions
About this article
Cite this article
Lyons, J.E., Collazo, J.A. & Guglielmo, C.G. Plasma metabolites and migration physiology of semipalmated sandpipers: refueling performance at five latitudes. Oecologia 155, 417–427 (2008). https://doi.org/10.1007/s00442-007-0921-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-007-0921-x