Abstract
We conducted an experimental landscape study to test the hypotheses that: (1) habitat removal results in short-term increases in population density in the remaining habitat patches (the crowding effect); (2) following habitat removal, density is higher in landscapes with more, smaller patches and more habitat edge (i.e., a higher level of habitat fragmentation per se) than in less fragmented landscapes, for the same total amount of habitat on the landscapes; (3) this positive effect of fragmentation per se on density is larger in landscapes with smaller inter-patch distances; and (4) these last two effects should be reduced or disappear over time following habitat removal. Our results did not support the first hypothesis, but they provided some support for the other three hypotheses, for two of the four Coccinellid species studied. As in other empirical studies of fragmentation per se on population density, the effects of fragmentation per se were weak and positive (when they did occur). This is the first study to document a transient effect of fragmentation per se on population density, and to show that this effect depends on inter-patch distances. We suggest that fragmentation per se increased the rate of immigration to patches, resulting in higher population densities in more fragmented landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Despite the huge literature on effects of habitat fragmentation on population density (reviewed in Fahrig 2003), only a handful of empirical studies have evaluated the effects of habitat fragmentation per se, i.e., the breaking apart of habitat independent of the loss of habitat, on population density (McGarigal and McComb 1995; Collins and Barrett 1997; Wolff et al. 1997; Collinge and Forman 1998; Flather et al. 1999; Hovel and Lipcius 2001; Caley et al. 2001). Evidence from these studies suggests that the effects of fragmentation per se on population density are not ubiquitous; effects have been found in fewer than half of the cases studied. In addition, when effects of fragmentation per se have been found, they were usually weak, typically accounting for <20% of the variation in population density.
These empirical studies also suggest that when there is an effect of fragmentation per se on population density it is usually positive; population density increases with increasing degree of habitat fragmentation, i.e., when a constant amount of habitat is broken into more, smaller patches. Authors of three of the studies attribute this positive effect of fragmentation per se to altered social interactions, leading to reduced competition for space in more fragmented landscapes. Collins and Barrett (1997) argue that because female meadow voles use patch edges to define territory boundaries, they are better able to defend territories and consequently have higher reproduction in more fragmented landscapes. Wolff et al. (1997) suggest that gray-tailed voles obtained higher densities in more fragmented landscapes because individuals in smaller patches are more likely to encounter familiar than unfamiliar animals, which they are more likely to tolerate nearby. In both these studies the experimental landscapes were enclosed, so movement was not possible into or out of the landscapes. Caley et al. (2001) argue that experimentally dividing a coral colony increased the total number of territories available to trapezoid crabs, because a single coral colony usually contains no more than two crab territories, irrespective of its size. Other proposed explanations for observed positive effects of fragmentation per se on population density include reduced predation on juvenile blue crabs in more fragmented landscapes (Hovel and Lipcius 2001), and increased habitat heterogeneity leading to higher densities of late-seral forest birds in more fragmented landscapes (McGarigal and McComb 1995).
Perhaps the simplest explanation was proposed by Collinge and Forman (1998), who suggest that their observed positive effect of fragmentation per se on insect population density could be due to an extension of the “crowding effect” (Debinski and Holt 2000). When habitat is removed from a landscape, surviving individuals in the portion of the landscape that is converted to matrix (non-habitat) will move into the remaining habitat fragments. This will result in an increase in population density relative to the density before habitat removal. As noted by Collinge and Forman (1998), the increase in density should depend not only on the amount of habitat removed from the landscape, but also on the pattern of habitat removal. Landscapes with a larger number of smaller patches, i.e., more fragmented landscapes, will have more habitat edge and should therefore “collect” more of the stranded individuals, which should produce higher densities in the habitat in more fragmented landscapes than in less fragmented landscapes containing the same total amount of habitat. This argument assumes that the stranded individuals move at or near ground level and are not disproportionately attracted to large patches (Bowman et al. 2002).
The crowding effect following habitat removal is typically transient, as the inflated densities settle down to levels that can actually be supported by the remaining habitat (Debinski and Holt 2000). Collinge and Forman (1998) argue that the crowding effect due to habitat fragmentation per se (increasing the number of patches, while removing the same total amount of habitat) should also be transient. Although in their study of grassland insects the positive effect of habitat fragmentation per se on population density did not disappear over time, they suggest that this is because they did not sample for a long enough time period following the establishment of their experimental grassland landscapes.
On the other hand, Bowman et al. (2002) suggest that inflated densities in more fragmented landscapes might be maintained in situations where there is a high rate of immigration from outside the landscape. Immigrating individuals that “land” in the matrix will be more likely to encounter habitat in more fragmented landscapes because, again, these landscapes contain more habitat edge per area of habitat. If the rate of immigration to the landscape declines over time, the inflated densities in the more fragmented landscapes should decline leading, again, to a transient effect of fragmentation per se on population density.
However, an increase in the amount of habitat edge in the landscape could also result in an increase in the rate of emigration from patches (Bevers and Flather 1999; Grez and Prado 2000). In the absence of immigration from outside the landscape, this could result in a negative effect of habitat fragmentation per se on population density. Therefore, in the absence of other possible fragmentation effects (above), the effect of habitat fragmentation per se on population density should depend on the net result of its positive effect through increased immigration and its negative effect through increased emigration.
This net effect should depend on the distances among the habitat fragments, relative to the movement range of the organism (Fahrig and Paloheimo 1988). If the remaining patches are close together, individuals that emigrate from patches are likely to be “captured” by another patch in the same landscape, so emigration from the whole landscape should be lower than when patches are farther apart. Therefore, the positive effects of fragmentation per se on population density should be larger when patches are closer together than when they are farther apart. The distance will depend on the organism’s movement speed in the matrix and on its giving up time, i.e., the amount of time it spends looking for habitat, before leaving the landscape altogether. Note this assumes that the organisms within a patch cannot detect the distance to the next patch, and do not “decide” to remain in a patch, if that distance is large (e.g., Jonsen and Taylor 2000) .
We tested for a transient positive effect of fragmentation per se in a landscape-scale experiment (sensu Brennan et al. 2002) of the population responses of Coccinellid beetles to removal of a constant amount of alfalfa habitat to create landscapes with different habitat spatial patterns, i.e., different levels of fragmentation per se, and different inter-patch distances. Our experiment was an open system situated in a larger region containing other alfalfa fields. This reduces the likelihood of fragmentation effects through mechanisms such as intra-specific and inter-specific interactions (above). Our intention was to test predictions resulting from the hypothesized effects of fragmentation per se on inter-patch movement described above. The treatment landscapes represented all four combinations of low and high levels of fragmentation per se (four fragments vs. 16 fragments), with small and large inter-patch distances (2 m vs. 6 m; Fig. 1), but with the same amount of habitat removal. We estimated densities of four Coccinellid species: (1) before the treatment landscapes were created through alfalfa removal (week −1), (2) during the first few weeks following creation of the treatment landscapes (weeks 1–7), and (3) several months later (weeks 13 and 15). We tested the following predictions: (1) Coccinellid densities should increase more in all treatment landscapes immediately following alfalfa removal (from week −1 to week 1) than in landscapes with no habitat removal, due to the crowding effect; (2) landscapes with a higher degree of fragmentation per se (16 smaller fragments) should have higher Coccinellid densities than the less fragmented landscapes (four larger patches) during the first few weeks following alfalfa removal (weeks 1–7); (3) this effect of fragmentation should depend on inter-patch distances such that the positive effects of fragmentation on Coccinellid density should be larger when inter-patch distances are small (2 m) than when inter-patch distances are large (6 m); and (4) these last two effects should be reduced or disappear during the later sampling period (weeks 13 and 15), when population processes other than immigration, such as reproduction and mortality, will be the main determinants of population density.
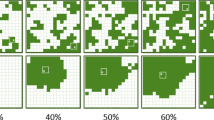
A–E Illustration of the five types of experimental landscapes. Each 30×30-m plot represents a landscape with alfalfa (black areas) and bare ground (white areas; the matrix). B–E Landscapes contain 16% alfalfa at two fragmentation levels [16 or four fragments (fr)] and two inter-patch distances (6 m or 2 m). The five types of landscapes were replicated 3 times in the field, for a total of 15 landscapes, with a 20-m buffer zone between landscapes
Materials and methods
Experimental landscapes
The study was conducted at Antumapu Experimental Research Station, University of Chile, Santiago, Chile (33°34′S, 70°37′W), during the 2002–2003 growing season. On 4 September 2002, we ploughed the entire study area and then sowed alfalfa (Pioneer 5683) in each of fifteen 30×30-m plots, which became our experimental landscapes. The landscape size for our study was selected based on other studies dealing with similar questions using similar organisms (Kareiva 1987; Ives et al. 1993; Banks 1999; With et al. 2002). The landscapes were separated from each other by at least 20 m. Alfalfa was irrigated every 2 weeks and no insecticides were used. On 25 November, the alfalfa was cut to a height of 20 cm. On 12 December, we created our treatment landscapes by ploughing up 84% of the alfalfa in each of 12 of the experimental landscapes. We created three replicates of each of four combinations of two fragmentation levels (four 6×6-m fragments, or sixteen 3×3-m fragments) and two inter-patch distances (2 m or 6 m) (Fig. 1). No alfalfa was removed from the remaining three control landscapes. The alfalfa was cut again on 21 January and 20 February to 20 cm, to encourage growth of fresh leaves and maintain habitat quality. The matrix (non-alfalfa) areas within the treatment landscapes were maintained free of vegetation throughout the experiment by herbicide application and ploughing as needed.
The two inter-patch distance levels used (2 m and 6 m) were based on movement and dispersal experiments with three of the Coccinellid beetle species in our study (Grez et al., in press). These experiments suggested that 15–20% of Coccinellid beetles placed on bare ground will walk at least 2 m without taking flight, whereas almost all individuals took flight before reaching 6 m. Therefore, between-patch movements should be much more common in landscapes in which patches are separated by 2 m than in landscapes with patches separated by 6 m. This was confirmed in mark-recapture experiments using Eriopis connexa (Germ.).
Insect sampling
Adults of four species of Coccinellid beetles were sampled in the landscapes, E. connexa, Hippodamia variegata (Goeze), Hippodamia convergens (Guerin), and Hyperaspis sphaeridioides (Mulsant). These insects eat mainly aphids, but also other small insects, Lepidopteran eggs, and pollen (Ferran and Dixon 1993). Adults lay eggs in early to mid summer. Development to adulthood takes about 4 weeks for E. connexa and about 6 weeks for H. convergens (Montes 1970; Etchégaray and Barrios 1979; Ferran and Dixon 1993). Development times for the other two species are likely similar but they have not been documented. Adults can survive for several months. Previous studies close to our study site indicate that E. connexa appears earlier in the field than H. variegata and H. convergens, with the former species having its peak abundance in December while the latter two species peak in January and March, respectively (Grez 1997).
Adult Coccinellids were sampled by sweep-netting at 16 points in each landscape. In the landscapes with four fragments, four samples were taken in each fragment and in the landscapes with 16 fragments one sample was taken in each fragment. For each sample, we passed the sweep net 4 times, covering a total area of approximately 2 m2. To reduce spatial autocorrelation of the data in the 6×6-m fragments, samples were taken at least 3 m apart (A. A. Grez et al., unpublished data). All samples were taken between 0900 and 1300 hours on warm, sunny days. Insects were counted and released back into the location where they were collected. Sampling was conducted once before alfalfa removal, on 5 December (week −1). Sampling was also conducted 4 times during the 7 weeks immediately following alfalfa removal, on 19 December, 2 January, 16 January and 30 January (weeks 1, 3, 5, and 7 respectively). The Coccinellids collected during this time period represented mainly insects that immigrated into our study area after having survived the winter elsewhere, although since E. connexa colonizes earlier than the other species, it was already reproducing at the time of alfalfa removal, as confirmed by the presence of their larvae at that time (unpublished data). We refer to this first period of sampling (weeks 1–7) as the “early sampling period”. Sampling was also conducted twice during a second period of sampling over 3 months following alfalfa removal, on 12 March and 25 March (weeks 13 and 15), i.e., at the beginning of fall in the southern hemisphere. The Coccinellids collected during this time period represented mainly insects arising from eggs laid during the summer of our experiment. We refer to this period of sampling as the “late sampling period”. For each sample date, we calculated the mean density per 2 m2 of each of the four species in each landscape, i.e., three mean values from three replicate landscapes per species per treatment. We used these mean values as the data points in all statistical analyses (below).
Statistical analyses
To test our first prediction we compared, for each species, the density in the sample immediately preceding alfalfa removal (week −1) to the density in the sample immediately following alfalfa removal (week 1) in the treatment landscapes. To account for the possibility that any increase in density was simply due to area-wide colonization, we included the control landscapes in the analysis. We conducted repeated measures ANOVAs of beetle density on week (week −1 vs. week 1), habitat amount [0% habitat removal (controls) vs. 84% habitat removal (treatments)] and their interaction. Area-wide colonization of the alfalfa plots between 5 and 19 December would result in an effect of week on population density. Our prediction of increased density due to habitat removal (crowding effect) would be confirmed by a significant interaction effect between week and habitat amount, i.e., the increase in density between weeks −1 and 1 should be greater for landscapes with habitat removal than for landscapes with no habitat removal.
To test our second and third predictions we conducted four analyses, one for each of the four species, of the density data during the early sampling period following alfalfa removal (weeks 1–7). We used the 12 treatment landscapes only, because we were evaluating the effect of habitat fragmentation per se, given a constant amount of habitat (16% in our case). For each species, we conducted a repeated measures ANOVA of density on fragmentation level (four or 16 fragments), inter-patch distance level (2 m or 6 m), and their interaction, where the density measures were repeated 4 times (weeks 1, 3, 5, and 7). Our predictions would be confirmed by: (1) a significant positive effect of fragmentation level on density, and (2) a significant interaction effect between fragmentation level and inter-patch distance, such that fragmentation level should have a stronger positive effect on density when inter-patch distance is 2 m than when inter-patch distance is 6 m.
To test our fourth prediction, we conducted a similar set of four repeated measures ANOVAs of density during the late sampling period on fragmentation level, inter-patch distance, and their interaction, where the density measures were repeated twice (weeks 13 and 15). We predicted that any significant effects of fragmentation level or interactions between fragmentation level and inter-patch distance that we had found in the analyses of the early sampling period (described above; weeks 1–7) would be much smaller or absent in the late sampling period.
Results
In all, we sampled 6,209 adult Coccinellids: 2,112 E. connexa, 3,443 H. variegata, 376 H. convergens, and 278 H. sphaeridioides. The time trends for the four species are shown in Fig. 2.
Density (per 2 m2) of four Coccinellid species, aEriopis connexa, bHippodamia convergens, cH. variegata, dHyperaspis sphaeridioides, over time in alfalfa within five experimental landscapes (see Fig. 1). Treatment landscapes were created at week 0 by removal of alfalfa. Densities in the control landscapes (no alfalfa removal) were compared to those in the treatment landscapes in weeks −1 and 1 to test for the crowding effect. Densities in the treatment landscapes were used to test for fragmentation and inter-patch distance effects during the early sampling period (weeks 1, 3, 5, and 7) and the late sampling period (weeks 13 and 15). SE bars are based on three data points, i.e., three replicate landscapes for each landscape type
Three of the four species showed a significant increase in density in the week immediately following alfalfa removal (Table 1; compare week −1 to week 1 in Fig. 2). However, contrary to our first prediction, there were no significant interaction effects between week (week −1 vs. week 1) and level of habitat loss (0 vs. 84%) for any of the four species (Table 1).
The repeated measures ANOVAs from the early sampling period, i.e., the first 7 weeks following alfalfa removal, comparing only the treatment landscapes, revealed a marginally significant effect of fragmentation level, and a marginally significant effect of the interaction between fragmentation level and inter-patch distance for H. variegata, and a marginally significant effect of the interaction between fragmentation level and inter-patch distance for H. convergens (Table 2). These species showed the predicted higher density in the more fragmented landscapes (16 patches) than in the less fragmented landscapes (four patches), when the inter-patch distance was small (2 m; Fig. 3). This effect of fragmentation per se can also be seen by comparing the early part (weeks 1–7) of the curves in Fig. 3. The 16fr-2 m curves for H. variegata and H. convergens rise above the other curves during the early sampling period for these two species, indicating positive effects of fragmentation per se.
Interaction effect between the level of habitat fragmentation and inter-patch distance on density (individuals per 2 m2±1 SE) of two Coccinellid species, H. variegata (a) and H. convergens (b) during the early sampling period, i.e., the first 7 weeks following alfalfa removal. Within landscapes the values were averaged across the four weeks (1, 3, 5, and 7); the SE bars are based on the three replicate temporal means for each treatment
As predicted, the effects of fragmentation per se and its interaction with inter-patch distance that we found in the early sampling period (weeks 1–7) had disappeared by the late sampling period (weeks 13 and 15). There were no effects of fragmentation level or inter-patch distance or their interaction on Coccinellid densities in the late sampling period (Table 2).
Discussion
In some respects, our results are consistent with previous empirical studies of the effect of habitat fragmentation per se on population density (McGarigal and McComb 1995; Collins and Barrett 1997; Wolff et al. 1997; Collinge and Forman 1998; Flather et al. 1999; Hovel and Lipcius 2001; Caley et al. 2001). First, as in previous empirical studies, the effect of habitat fragmentation per se was not ubiquitous; it was only detected in two species of four. When an effect of fragmentation per se did occur, the effect was weak, explaining only 5 and 8% of the variation in population density, or 12 and 16% of the variation in density for the sum of the main effect of fragmentation level and the interaction effect between fragmentation level and inter-patch distance (for H. convergens and H. variegata, respectively).
In our analyses of the effects of fragmentation per se, i.e., including only the 12 landscapes from which alfalfa had been removed, habitat amount was held constant among treatments at 16% of the landscape. This habitat amount is under the threshold level of 20–30%, below which theoretical studies predict that habitat fragmentation per se should have its largest effect on population density (Fahrig 1998; Flather and Bevers 2002). Note that three other experimental studies of the effect of fragmentation per se on density used habitat amount levels of 16, 30 and 25% (Collins and Barrett 1997; Wolff et al. 1997; Collinge and Forman 1998, respectively). Our results and these other studies suggest that the effect of fragmentation per se on population density is not strong, even in the range of habitat amount values where it is predicted to be strongest.
The second major way in which our results are consistent with previous empirical studies of fragmentation per se on population density is that the effect of fragmentation, when it did occur, was positive, i.e., population density was higher in the landscapes containing 16 small patches than in the landscapes containing four larger patches. All other experimental studies of the effects of fragmentation per se on population density have similarly found positive effects (Collins and Barrett 1997; Wolff et al. 1997; Collinge and Forman 1998; Caley et al. 2001; Bancroft and Turchin 2003). Note, however, that this result is not consistent with most current theory, which predicts negative effects of fragmentation per se on population density (Fahrig 1998, Burkey 1999; With and King 1999; Hill and Caswell 1999; Urban and Keitt 2001).
We suggest the following interpretation for our results. First, immediately following habitat removal, insect densities were not higher in landscapes where habitat had been removed than in the control landscapes. This suggests that many or most of the insects that had been in alfalfa before it was converted to bare ground were either killed during plowing, or engaged in immediate long-distance movements and therefore did not enter the remaining fragments of alfalfa in significant numbers. However, we did observe a weak positive effect of the level of fragmentation on density of H. variegata, i.e., higher density in the landscapes with 16 patches than in the landscapes with four patches, during the early sampling period following alfalfa removal (Table 2). We suggest that increased fragmentation per se increased the ability of migrating adults arriving at our experimental area to find alfalfa habitat. These insects were more likely to find alfalfa in landscapes containing 16 small fragments than in those containing four large fragments (with the same total amount of habitat), because the more fragmented landscapes had more habitat edge. This is consistent with the hypothesis of Bowman et al. (2002). Some animals, including many insects, make long-distance movements aerially, but when they alight on the ground they move in a random pattern until they intercept habitat (e.g., Fahrig and Paloheimo 1987; Gillespie 1987). Such animals are more likely to intercept habitat in landscapes containing more habitat edge, i.e., more fragmented landscapes. This is also true for animals with directed walking, unless large patches disproportionately attract moving individuals. We suggest that this sort of behavior led to the small positive effect of fragmentation per se on the density of H. variegata (higher density in landscapes with 16 patches than in landscapes with four patches) during the early sampling period.
Our results support our hypothesis that the effect of habitat fragmentation should depend on inter-patch distances. The positive effect of fragmentation per se was stronger in landscapes where the inter-patch distances were smaller (2 m) than where inter-patch distances were larger, for both H. variegata and H. convergens (6 m; Fig. 3). To our knowledge, this study is the first demonstration of this interaction effect between fragmentation per se and inter-patch distance. Coccinellids are more likely to emigrate from patches with a higher perimeter to area ratio (Grez and Prado 2000); therefore, in our experimental landscapes the emigration rate from patches should be higher in the more fragmented landscapes (16 patches) than in the less fragmented landscapes (four patches). However, when the distance between fragments is small, emigrants from fragments are more likely to find new fragments within the landscape. Coccinellids are more likely to fly when they are on bare ground than when they are in alfalfa (A.A. Grez et al., unpublished data). We hypothesize that beetles leaving habitat patches by walking were more likely to find nearby patches than more distant patches, before taking flight. Therefore, when small patches were close together, the positive effect of fragmentation per se on immigration was not countered by an increase in emigration from the landscape.
As we predicted, the effects of fragmentation per se on population density, i.e., the higher densities in the landscapes with 16 fragments than in the landscapes with four fragments, were only apparent during the early sampling period. This is the first demonstration of which we are aware, of the predicted transient effect of fragmentation per se on population density. Most of the adult Coccinellids captured during the late sampling period were from a second generation and likely originated largely from reproduction within our plots, and not from a continuing immigration process. Most species of Coccinellids in temperate regions hibernate in different places than where they feed and reproduce (Dixon 2000). Therefore, every year they colonize new habitats during spring and early summer, feed, reproduce, and in fall the adults from the summer generation seek overwintering habitats. Our results, along with these life history traits, suggest that, at longer time scales, when immigration had decreased, factors affecting reproduction and mortality had a much larger effect on population density than any possible effect of habitat fragmentation per se on density. This is particularly likely for species with high fecundities such as H. variegata and H. convergens (With and King 1999; Fahrig 2001). By 3 months following alfalfa removal, the effect of the level of fragmentation (four patches vs. 16 patches) on immigration was probably so small relative to these demographic effects, that it was not detectable.
If, as we propose, immigration was the main cause of the observed positive effect of fragmentation per se on Coccinellid density, then the timing of alfalfa removal relative to the immigration period of each species likely determined whether or not such an effect was detected for that species. Based on the temporal dynamics of the Coccinellid species reported here and in previous studies (Grez 1997), it seems likely that in our experiment alfalfa removal occurred before the bulk of immigration for Hippodamia species, and after the bulk of immigration for E. connexa. We hypothesize that this difference in timing resulted in our observation of a positive effect of fragmentation for the Hippodamia species, but no effect for E. connexa. We do not have a clear idea of when immigration occurs for Hyperaspis.
There are other possible mechanisms that could produce positive relationships between fragmentation per se and population density (Fahrig 2003; L. Tischendorf et al., unpublished data). These include release from competitors or predators, improved access to other resources on the landscape (“landscape complementation”), and higher reproduction or lower mortality at habitat edges. If these mechanisms were operating in our system we would have expected the positive effect of fragmentation per se to persist over time. Since it did not, we believe a temporary increase in immigration rate caused by the larger amount of habitat edge in the more fragmented landscapes, as discussed above, is the most likely explanation for our results, although we cannot definitively reject the possibility that other mechanisms were also operating.
In summary, we found that habitat fragmentation per se (controlling for habitat amount) had a weak positive effect on the population density of two of four Coccinellid species, in the short term following habitat removal. There was no effect of fragmentation per se on the other two species. The positive effect of fragmentation level depended on inter-patch distances; it occurred only where fragments were close together. The effect was also transient; it had disappeared 3 months following habitat removal. This is the first study to document a transient positive effect of fragmentation per se on population density, and to show that this positive effect depends on inter-patch distances. Our results are consistent with the hypothesis that fragmentation per se can increase population density through an increase in immigration rate. Theoretical work is needed to determine the conditions under which this mechanism is most likely to cause positive effects of fragmentation per se on population density. Also, more empirical studies of the effect of fragmentation per se are needed, to determine whether this is an important mechanism in nature.
References
Bancroft JS, Turchin P (2003) An experimental test of fragmentation and loss of habitat with Oryzaephilus surinamensis. Ecology 84:1756–1767
Banks JE (1999) Differential response of two agroecosystem predators Pterostichus melanarius (Coleoptera: Carabidae) and Coccinella septempunctata (Coleoptera: Coccinellidae) to habitat-composition and fragmentation-scale manipulations. Can Entomol 131:645–657
Bevers M, Flather CH (1999) Numerically exploring habitat fragmentation effects on population using cell-based coupled map lattices. Theor Popul Biol 55:61–76
Bowman J, Cappuccino N, Fahrig L (2002) Patch size and population density: the effect of immigration behavior. Conservation Ecol 6:9. http://www.consecol.org/vol6/iss1/art9
Brennan JM, Bender DJ, Contreras TA, Fahrig L (2002) Focal patch landscape studies for wildlife management: optimizing sampling effort across scales. In: Liu J, Taylor WW (eds) Integrating landscape ecology into natural resource management. Cambridge University Press, Cambridge, pp 68–91
Burkey TV (1999) Extinction in fragmented habitats predicted from stochastic birth–death processes with density dependence. J Theor Biol 199:395–406
Caley MJ, Buckley KA, Jones GP (2001) Separating ecological effects of habitat fragmentation, degradation, and loss on coral commensals. Ecology 82:3435–3448
Collinge SK, Forman RTT (1998) A conceptual model of land conversion processes: predictions and evidence from a microlandscape experiment with grassland insects. Oikos 82:66–84
Collins RJ, Barrett GW (1997) Effects of habitat fragmentation on meadow vole (Microtus pennsylvanicus) population dynamics in experiment landscape patches. Landscape Ecol 12:63–76
Debinski DM, Holt RD (2000) A survey and overview of habitat fragmentation experiments. Conserv Biol 14:342–355
Dixon AFG (2000) Insect predator-prey dynamics: ladybird beetles and biological control. Cambridge University Press, Cambridge
Etchégaray J, Barrios S (1979) Ciclo de vida de E. connexa (Germar, 1824) (Coleoptera: Coccinellidae). Anal Mus Hist Nat Valparaíso 12:185–190
Fahrig L (1998) When does fragmentation of breeding habitat affect population survival? Ecol Model 105:273–292
Fahrig L (2001) How much habitat is enough? Biol Conserv 100:65–74
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Syst 34:487–515
Fahrig L, Paloheimo JE (1987) Interpatch dispersal of the cabbage butterfly. Can J Zool 65:616–622
Fahrig L, Paloheimo JE (1988) Determinants of local population size in patchy habitats. Theor Popul Biol 34:194–213
Ferran A, Dixon FG (1993) Foraging behaviour of ladybird larvae (Coleoptera: Coccinellid). Eur J Entomol 90:383–402
Flather CH, Bevers M (2002) Patchy reaction-diffusion and population abundance: the relative importance of habitat amount and arrangement. Am Nat 159:40–56
Flather CH, Bevers M, Cam E, Nichols J, Sauer J (1999) Habitat arrangement and extinction thresholds: do forest birds conform to model predictions? Landscape ecology: the science and the action. In: Proceedings of 5th World Congress of the International Association for Landscape Ecology , vol 1. Snowmass, Colo., pp 44–45
Gillespie RG (1987) The mechanism of habitat selection in the long-jawed orb-weaving spider Tetragnatha elongata (Araneae, Tetragnathidae). J Arachnol 15:81–90
Grez AA (1997) Effect of habitat subdivision on the population dynamics of herbivorous and predatory insects in central Chile. Rev Chil Hist Nat 70:481–490
Grez AA, Prado E (2000) Effect of plant patch shape and surrounding vegetation on the dynamics of predatory coccinellids and their prey Brevicoryne brassicae (Hemiptera: Aphididae). Environ Entomol 29:1244–1250
Grez AA, Zaviezo T, Ríos M Ladybird (in press) (Coleoptera: Coccinellid) dispersal in fragmented alfalfa landscapes. Eur J Entomol
Hill MF, Caswell H (1999) Habitat fragmentation and extinction thresholds on fractal landscapes. Ecol Lett 2:121–127
Hovel KA, Lipcius RN (2001) Habitat fragmentation in a seagrass landscape: patch size and complexity control blue crab survival. Ecology 82:1814–1829
Ives AR, Kareiva P, Perry R (1993) Response of a predator to variation in prey density at three hierarchical scales: lady beetles feeding on aphids. Ecology 74:1929–1938
Jonsen I, Taylor PD (2000) Fine-scale movement behaviors of Calopterygid damselflies are influenced by landscape structure: an experimental manipulation. Oikos 88:553–562
Kareiva P (1987) Habitat fragmentation and the stability of predator–prey interactions. Nature 326:388–390
McGarigal K, McComb WC (1995) Relationships between landscape structure and breeding birds in the Oregon Coast Range. Ecol Monogr 65:235–260
Montes F (1970) Biología y morfología de Eriopis connexa Germar 1824 y de Adalia bipunctata Linnaeus 1758 (Coleoptera). Publicaciones del Centro de Estudios Entomológicos, Universidad de Chile, vol 10. Centro de Estudios Entomológicos, Universidad de Chile, pp 43–56
Urban D, Keitt T (2001) Landscape connectivity: a graph-theoretic perspective. Ecology 82:1205–1218
With KA, King AW (1999) Dispersal success on fractal landscapes: a consequence of lacunarity thresholds. Landscape Ecol 14:73–82
With KA, Pavuk DM, Worchuck JL, Oates RK, Fisher JL (2002) Threshold effects of landscape structure on biological control in agroecoystems. Ecol Appl 12:52–65
Wolff JO, Schauber EM, Edge WD (1997) Effects of habitat loss and fragmentation on the behavior and demography of gray-tailed voles. Conserv Biol 11:945–956
Acknowledgements
We thank Astrid Pérez, Denise Donoso, Marta Ríos, Sandra Díaz, Bernardino Camousseigt and Susana Reyes for their field assistance. We particularly thank Manuel Bustos for maintaining the experimental plots. We thank members of the Landscape Ecology Laboratory at Carleton University for helpful comments on the manuscript. This work was funded by the Chilean government through FONDECYT 1011041 and 1011071.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grez, A., Zaviezo, T., Tischendorf, L. et al. A transient, positive effect of habitat fragmentation on insect population densities. Oecologia 141, 444–451 (2004). https://doi.org/10.1007/s00442-004-1670-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-004-1670-8