Abstract
Cartilage generation and degradation are controlled by miRNAs. Our previous study showed miR-23a-3p was downregulated during chondrogenic differentiation in chondrogenic human adipose-derived mesenchymal stem cells (hADSCs). In the present study, we explored the function of miR-23a-3p in chondrogenesis differentiation. The role of miR-23a-3p in chondrogenic differentiation potential of hADSCs was assessed by Alcian blue staining, quantitative real-time polymerase chain reaction (qRT-PCR), and Western blot. We show that miR-23a-3p suppressed the chondrogenic differentiation of hADSCs. LncRNA SNHG5 interacted with miR-23a-3p, and suppression or overexpression of SNHG5 correlates with inhibition and promotion of hADSC chondrogenic differentiation, respectively. We have determined that SNHG5 can sponge miR-23a-3p to regulate the expression of SOX6/SOX5, transcription factors that play essential roles in chondrocyte differentiation. Furthermore, the overexpression of SNHG5 activates the JNK/MAPK/ERK pathway. In conclusion, miR-23a-3p regulated by lncRNA SNHG5 suppresses the chondrogenic differentiation of human adipose-derived stem cells via targeting SOX6/SOX5.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoarthritis (OA) is a progressive joint disease involving mainly degeneration of articular cartilage and inflammation of the synovium, causing joint pain and significantly reducing motor functions (Andarwulan et al. 2010). The prevalence rate of OA in people over 60 years old is higher than that of lumbago, neck and shoulder pain, inflammatory arthritis, osteoporosis, and spinal deformity, and OA has been one of the main diseases leading to the decline of activity dysfunction and quality of life in the middle-aged and older people (Pereira et al. 2011). Autogenous chondrocyte implantation is widely used in cartilage defect repair, and its clinical effect has been confirmed (Komarek et al. 2010; Niethammer et al. 2016). However, the sources of autologous chondrocytes are limited, and the materials are inconvenient (arthroscopy or surgical materials are required); the ability of traditional culture to pass through and proliferate is limited, and in vitro culture is prone to dedifferentiation. Its activity decreases significantly with age (Schulze-Tanzil 2009). The clinical application of autologous chondrocyte repair is limited. Developing an alternative treatment strategy in the repair of damaging cartilage is quite necessary.
Recently, stem cell-based tissue engineering has been regarded as a potential ideal strategy to address this problem (Lai et al. 2015). According to previous reports, mesenchymal stem cells (MSCs), particularly adipose-derived stem cells (ADSCs) (Im 2017; Polymeri et al. 2016), are commonly used in clinical applications due to their ease of isolation, strong proliferation ability, and multi-differentiation potential (Miura 2016). Furthermore, compared with bone marrow-derived mesenchymal stem cells (BMMSCs), ADSCs do have an equal potential to differentiate into cells and tissues of mesodermal origin, such as adipocytes, cartilage, bone, and skeletal muscle. On the other hand, the easy and repeatable access to subcutaneous adipose tissue and the simple isolation procedures provide a clear advantage (Schäffler and Büchler 2007, Zhu et al. 2008). Different studies have shown that the application of ADSCs as therapy in the treatment of OA has improved the symptoms suffered by patients, particularly after more than 6 months of follow-up (Lee et al. 2019, Pers et al. 2018).
MicroRNAs (miRNAs) are a group of small, noncoding single-stranded RNA with a tissue or developmental stage specificity, which plays a regulatory role through post-transcriptional binding to specific sequences of target genes. Several miRNAs were verified to take part in the processes of cartilage differentiation and degradation (Dong et al. 2012; Kobayashi et al. 2008). Our previous study revealed that miR-23a-3p was downregulated during chondrogenic differentiation. We likewise anticipated possible target genes of miR-23a-3p predicted the potential target genes of miR-23a-3p using prediction software and the literature (Yang et al. 2015). In this work, we continue to explore certain biological functions as well as mechanisms of miR-23a-3p based on our previous study.
Materials and methods
Isolation and culture of human ADSCs
hADSCs were harvested and cultured from donors who underwent elective liposuction or other abdominal surgery at the Tianjin Haihe Hospital (Tianjin, China). All the donors were obtained after written informed consent was provided and ethical approval. hADSCs were cultured and induced as our previous study described (Yang et al. 2018).
Alcian blue staining
The cells were fixed with 4% paraformaldehyde for 20 min and then stained with Alcian blue for 30 min. After culture, the cells were washed three times with PBS, and the samples were observed with a phase-contrast microscope.
Cell transfection
The miR-23a-3p mimic, miR-23a-3p inhibitor, pcDNA3.1-SNHG5, and short hairpin RNA (sh-SNHG5) were designed and synthesized by GenePharma Company (Shanghai, China). Cell transfections were performed using Lipofectamine 2000 reagent (Life Technologies Corporation, USA) following the manufacturer’s protocol.
Dual-luciferase reporter assays
Dual-luciferase reporter assay was performed to confirm whether SNHG5 directly targets miR-23a-3p, and miR-23a-3p directly targets SOX6/SOX5. Cells were co-transfected with SNHG5-WT (or -MUT) or SOX6/SOX5 3′-untranslated region (UTR) -WT (or -MUT) reporter plasmids and miR-23a-3p mimics (or mimic control). The luciferase activities were measured by a dual-luciferase reporter assay kit (Promega, USA) after transfection.
Real-time quantified PCR
Total RNA was isolated with Trizol reagent (Takara Bio Inc., Japan) and also was reverse recorded. The GAPDH was made use of as an internal control for determining the mRNA material. After drawing the standard curve of the PCR reaction, the expression level of mRNA is computed. The outcomes were analyzed by using the organic system 7300 rapid real-time PCR system.
Western blot assay
Total protein separated from hADSC cells using a protein extraction kit (Bio-Rad, USA). Then, protein samples (50 μg) were resolved using SDS-PAGE and then moved to polyvinylidene fluoride membrane (PVDF) (Millipore, USA) with being probed with GAPDH (Proteintech), Collagen II, SOX9, Aggrecan (Abcam), and SOX6 and SOX5 (cell signaling) antibodies followed by appropriate secondary antibody. All Western blots were repeated at least three times.
Statistical analysis
The data in this research exist as the mean ± standard error indicates (SEM) from at least three independent experiments. The differences among the groups’ one-way variation analysis and Duncan multiple range examinations were used to determine considerably different groups. P values less than 0.05 were taken into consideration as considerable distinctions for all statistical estimations.
Results
miR-23a-3p suppressed chondrogenic differentiation of hADSCs
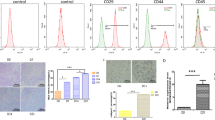
Following induction for 21 days, hADSCs were positive for Alcian blue staining (Fig. 1a). Expression of chondrocyte-specific markers and miR-23a-3p was detected by RT-qPCR in chondrogenic hADSCs. Our results showed that expression of COL2A1, SOX9, and Aggrecan increased, whereas miR-23a-3p level decreased during the chondrogenesis process (Fig. 1b, c). To test whether miR-23a-3p inhibits chondrogenic differentiation, we suppressed or overexpressed miR-23a-3p in hADSCs. The number of GFP-positive cells was examined by a microscope to evaluate the infection efficiency. As shown in Fig. 1d, e, the percentage of GFP-positive cells in all groups was approximately 80–90%. The results of qRT-PCR showed that the transfection was successful (Fig. 1f). The Alcian blue staining showed that proteoglycan secretion of chondrocytes increased in the groups with less miR-23a-3p (Fig. 1g). Moreover, results from qRT-PCR and Western blot indicated that COL2A1, SOX9, and Aggrecan mRNA and protein expression were significantly decreased in miR-23a-3p mimics group, whereas they were increased in the miR-23a-3p inhibitor group (Fig. 1h, i). These data suggested that miR-23a-3p inhibited chondrogenic differentiation in hADSC cells.
miR-23a-3p suppressed cell chondrogenic differentiation of hADSCs. a Alcian blue staining of hADSCs (scale bar = 50 μm). b The expression of levels of SOX9, COL2A1, and Aggrecan was determined using RT-qPCR. c The expression of miR-1307-3p was determined by qRT-PCR. d, e Infection efficiency was determined by microscopic examination of the GFP-expressing cells (scale bar = 100 μm). f Expression of miR-23a-3p after mimics or inhibitor transfection. g The deposition of cartilage matrix proteoglycans was determined by Alcian blue staining (scale bar = 50 μm). h The mRNA and i protein expression levels of SOX9, COL2A1, and Aggrecan were determined using RT-qPCR and Western blotting, respectively. Data are expressed as mean ± SEM and *P < 0.05, **P < 0.01, and ***P < 0.001
The lncRNA SNHG5 interacts with miR-23a-3p
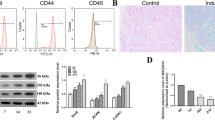
Previous studies have suggested that lncRNAs play an essential role in gene expression regulation by acting as ceRNAs (Salmena et al. 2011). The potential lncRNA–miRNA mutual binding sites were predicted by the bioinformatics tool (Starbase). According to the results of the prediction algorithm and the previous study about long noncoding RNAs involved in bone generation from mesenchymal stem cells (Peng et al. 2018), we found MEG3, MALAT1, SNHG5, GAS5, XIST, SHNG16, and NEAT1 may interact with miR-23a-3p. Next, to evaluate the expression of each lncRNA in hADSCs, RT-PCR was performed. As shown in Fig. 2a, hADSCs expressed very high SNHG5 lncRNA levels, compared with other groups. As Fig. 2b indicated, there are potential binding sites at the 3′- and 5′-UTR among the SNHG5 and miR-23a-3p. To validate this bioinformatics prediction, we used a dual-luciferase reporter gene assay. The results indicated that SNHG5-WT presented lower luciferase activity than the corresponding MUT group, confirming the binding relationship of SNHG5 and miR-23a-3p (Fig. 2c). Overall, these findings suggested that lncRNA SNHG5 interacted with miR-23a-3p.
The lncRNA SNHG5 targeted miR-23a-3p in hADSCs. a RT-PCR for lncRNAs (MEG3, MALAT1, SNHG5, GAS5, XIST, SHNG16, and NEAT1 targeted miR-23a-3p) level in hADSCs. b Potential binding sites of lncRNA-SNHG5 and miR-23a-3p. c Relative luciferase activity in hADSCs co-transfected with luciferase reporter containing SNHG5-WT or SNHG5-MUT with either miR-23a-3p mimics or miR-NC and fluorescence was quantified. Data are expressed as mean ± SEM and *P < 0.05, **P < 0.01, and ***P < 0.001
SNHG5 promoted cell chondrogenic differentiation of hADSCs
To further elucidate the role of SNHG5 in the regulation of osteogenic differentiation of hADSCs, pcDNA3.1-SNHG5 or sh-SNHG5 was transfected into hADSCs. The SNHG5 level was markedly upregulated by pcDNA3.1-SNHG5 and substantially downregulated by sh-SNHG5 (Fig. 3a). Furthermore, osteogenic differentiation was significantly promoted after overexpression of SNHG5 and inhibited after reduction of SNHG5 (Fig. 3b), as indicated by the Alcian blue staining, as well as the expression change of the chondrogenic differentiation markers, Aggrecan, SOX9, and COL2A1 (Fig. 3c, d).
SNHG5 promoted the chondrogenic differentiation of hADSCs. a RT-PCR analyzed the expression of SNHG5 among the four groups. b The Alcian blue staining at day 14 showed the deposition of cartilage matrix proteoglycans after SNHG5 overexpression and suppression (scale bar = 50 μm). c RT-PCR and Western blot d analysis of chondrogenic differentiation markers. Data are expressed as mean ± SEM and *P < 0.05, **P < 0.01, and ***P < 0.001
SNHG5 can reverse the effect of miR-23a-3p on cell chondrogenic differentiation of hADSCs
To verify the regulatory relationship between SNHG5 and miR-23a-3p was also determined in our study. As shown in Fig. 4a, miR-23a-3p expression was remarkably upregulated in the miR-23a-3p-mimics+pcDNA3.1-SNHG5 group compared with that in miR-23a-3p-mimics group+pcDNA3.1 group and downregulated in the miR-23a-3p inhibitor+sh-SNHG5 group relative to that in miR-23a-3p inhibitor+sh-NC group, indicating that SNHG5 negatively regulated the expression of miR-23a-3p. Moreover, we co-transfected miR-23a-3p mimics and SNHG5 overexpression plasmids into hADSCs to determine whether SNHG5 can inhibit miR-23a-3p-induced cell chondrogenic differentiation of hADSCs. The results showed that the mRNA and protein levels of the chondrogenic differentiation markers were remarkably elevated in the miR-23a-3p mimics+pcDNA3.1-SNHG5 group when compared with the miR-23a-3p overexpression group alone (Fig. 4b, c). These results demonstrated that the upregulation of SNHG5 could reverse the inhibitory effect of overexpressed miR-23a-3p on proteoglycan deposition as well as Aggrecan, SOX9, and COL2A1 expression.
Overexpression of SNHG5 could reverse the inhibitory effect caused by miR-23a-3p upregulation. a RT-PCR analysis of miR-23a-3p expression level. b, c Overexpression of SNHG5 could reverse the decrease of Aggrecan, SOX9, and COL2A1 expression levels caused by miR-23a-3p upregulation. Data are expressed as mean ± SEM and *P < 0.05, **P < 0.01, and ***P < 0.001
SOX6/SOX5 can reverse the effect of SNHG5 on cell chondrogenic differentiation of hADSCs
To identify potential target genes of miR-23a-3p, the TargetScan, miRanda, and Starbase were used. SOX6/SOX5 was the potential target of mir-23a-3p, according to TargetScan, and the binding sequence was shown in Fig. 5a. Also, the luciferase reporter gene experiment results confirmed that miR-23a-3p could target SOX6/SOX5 3′UTR (Fig. 5b). Besides, miR-23a-3p mimic significantly inhibited the expression of SOX6/SOX5 at both mRNA and protein levels, while miR-23a-3p inhibitor markedly increased the expression levels of SOX6/SOX5 (Fig. 5c, d). These results indicated the negative regulatory relationship between miR-23a-3p and SOX6/SOX5. To further explore the effect of SNHG5 on SOX6/SOX5, hADSCs were transfected with pcDNA3.1-SNHG5, sh-SNHG5, and sh-SOX6/SOX5 which was detected by Western blot. As shown in Fig. 5e, SNHG5 overexpression increased SOX6/SOX5 expression level. Consistently, the downregulation of SNHG5 reduced the abundance of SOX6/SOX5. Besides, we also detected the expression of cartilage differentiation markers Aggrecan, Sox9, and COL2A1 by downregulating SOX6/SOX5. As shown in Fig. 5f, inhibition of SOX6/SOX5 can reduce the expression level of Aggrecan, SOX9, and COL2A1 that was induced by SNHG5. These data suggested that SOX6/SOX5 is essential for the function of SNHG5 in the chondrogenic differentiation of hADSCs.
SOX6/SOX5 can reverse the effect of SNHG5 on cell chondrogenic differentiation of hADSCs. a The binding sequence of miR-23a-3p and SOX6/SOX5. b Luciferase reporter assay confirmed the target relationship between miR-23a-3p and SOX6/SOX5. c, d The expression levels of SOX6/SOX5 mRNA and protein in hADSCs transfected with miR-23a-3p mimic, miR-23a-3p inhibitor, and their negative controls. e hADSCs were transfected with pcDNA3.1, pcDNA3.1-SNHG5, pcDNA3.1-SNHG5+sh-NC, pcDNA3.1-SNHG5+sh-SOX6, and pcDNA3.1-SNHG5+sh-SOX5. The protein level of SOX6/SOX5 was detected with an anti-SOX6/SOX5 antibody. f The relative expression level of the Aggrecan, SOX9, and COL2A1 of hADSCs harboring the indicated expressing vector was detected. Data are expressed as mean ± SEM and *P < 0.05, **P < 0.01, and ***P < 0.001
Overexpression of SNHG5 enhances chondrogenic differentiation via activation of JNK/MAPK/ERK pathway
We further investigated the possible signal pathways regulated by SNHG5/miR-23a-3p/SOX6/SOX5. It is found that the JNK/MAPK/ERK pathway plays a crucial role in the process of cartilage differentiation and activation. As shown in Fig. 6a, pcDNA3.1-SNHG5+miR-23a-3p mimics+pcDNA3.1-SOX5/SOX6 groups increased the expression levels of p-JNK, p-ERK1/2, and p-P38 MAPK compared with pcDNA3.1-SNHG5+miR-23a-3p mimics group, confirming that overexpression of SNHG5 can relieve the inhibition of miR-23a-3p on SOX6/SOX5 expression and activate JNK/MAPK/ERK pathway. To better confirm these findings, hADSCs were treated with 1 μM Cl-1040 (MEK inhibitor) before transfection with pcDNA-SNHG or pcDNA3.1. The WB results showed that Cl-1040 therapy considerably reversed the impacts of overexpression of SNHG5 on cell differentiation by reducing the expression of Aggrecan, SOX9, and COL2A1 (Fig. 6b), suggesting that JNK/MAPK/ERK pathway was an essential mechanism mediating the function of SNHG5 in OA development. All these data demonstrated that SNHG5 modulates SOX6/SOX5 to activate the JNK/MAPK/ERK pathway, affecting hADSC differentiation by sponge miR-23a-3p.
Overexpression of SNHG5 activates the JNK/MAPK/ERK pathway by abrogating the inhibitory effect of miR-23a-3p on SOX6/SOX5 expression. a The protein expression of JNK, p38 MAPK, ERK, p-JNK, p-p38 MAPK, and p-ERK in hADSCs transfected with pcDNA-SNHG5, miR-23a-3p mimic, pcDNA-SOX6/SOX5, or their controls. b hADSCs were treated with 1 μM of MEK inhibitor CI-1040 before transfection with pcDNA-SNHG or pcDNA. The expression of differentiation markers (Aggrecan, SOX9, and COL2A1) in different transfected groups. All experiments were independently repeated three times. Data are expressed as mean ± SEM and *P < 0.05, **P < 0.01, and ***P < 0.001
Discussion
Cartilage differentiation of stem cells is under precise gene regulation at different levels of expression (Almalki and Agrawal 2016; Lin and Hankenson 2011), including a variety of cytokines (BMPs, FGFs, TGF-β), signaling pathways (Smad, MAPK, Notch, Wnt), and transcription factors (Runx2, Osterix, ATF4, SOCS1, SATB2). In recent years, studies have proved that cartilage differentiation of stem cells is strictly regulated by miRNA, and the main signal pathway and related transcription factors of cartilage differentiation of stem cells are regulated by miRNA (Gangaraju and Lin 2009; Gao et al. 2011). In our previous study, we demonstrated that the expression level of miR-23a-3p was significantly decreased in hADSCs (Yang et al. 2015). However, the regulatory function of miR-23a-3p in hADSCs is still unknown. In this study, we found that miR-23a-3p regulated by lncRNA SNHG5 suppresses the chondrogenic differentiation of hADSCs via targeting SOX6/SOX5.
MiRNA is paired with the 3′UTR base of the target mRNA and silences the gene at the post-transcriptional level by inhibiting the translation of the mRNA or directly degrading the mRNA (Liao et al. 2010). More and more evidence shows that the gene regulatory mechanism regulated by miRNA is a vital way to regulate gene expression levels during chondrogenic differentiation of various types of human cells (Hodges et al. 2017). Kang et al. (2016a) demonstrated that miR-23a-3p could induce the decrease of vascular endothelial growth factor level and chondrocyte apoptosis by targeting the inhibition of Smad3 in cartilage, thereby promoting the pathogenesis of OA. Besides, our study demonstrated that miR-23a-3p suppressed hADSC differentiation; this proved that miR-23a-3p was a negative regulator of cartilage differentiation in hADSCs.
Numerous studies have revealed that lncRNAs can function as a competing endogenous RNA (ceRNA) to bind miRNA and inhibit miRNA binding to their targets, thus regulating gene expression (Li et al. 2019). In a previous study, the lncRNA SNHG5 acts as a ceRNA of miR-205-5p to promote Imatinib resistance in the K562 cell lines (Wu et al. 2018). In our study, we found that SNHG5 promoted cell chondrogenic differentiation and reverse the effect of miR-23a-3p on cell chondrogenic differentiation of hADSCs, suggesting that SNHG5 was also confirmed to be a ceRNA of miR-23a-3p. Besides, it is well-known that miRNA regulated gene expression via targeting the 3′UTR. By using bioinformatics analysis, SOX6/SOX5 was shown to be the targets of miR-23a-3p. Chondrogenic transcription factors Sox5 and Sox6 are needed for the establishment of multilayered growth plates and thereby for the proper and timely development of endochondral bones (Smits et al. 2004). We found that SOX6/SOX5 can reverse the effect of SNHG5 on cell chondrogenic differentiation of hADSCs. A recent study showed that Sox5 and Sox6 are necessary cells autonomously for articular and meniscal chondrocyte differentiation and proper joint maturation (Dy et al. 2010).
Furthermore, the association between SNHG5 and JNK/MAPK/ERK pathway was examined, and the results showed that overexpression of SNHG5 enhances chondrogenic differentiation via activation of JNK/MAPK/ERK pathway through abrogating the inhibitory effect of miR-23a-3p on SOX6/SOX5 expression. MAPK signaling pathways play an essential regulatory role in normal chondrocyte physiology and osteoarthritis pathology (Kang et al. 2016b). Previous evidence has pointed out that IL-1β can reduce the expression of miR-558 and induce the expression of COX-2 in chondrocytes by activating the MAPK pathway (Park et al. 2013). Moreover, histone deacetylase-4 and histone deacetylase-8 regulate interleukin-1β-induced cartilage catabolic degradation via MAPK/JNK/ERK pathways (Wang et al. 2018). In this study, we identified that SNHG5 could activate JNK/MAPK/ERK pathway by acting as a sponge for miR-23a-3p to relieve SOX6/SOX5 expression. Overexpression of SNHG5 promotes the expression of Aggrecan, SOX9, and COL2AL1 through activation of the JNK/MAPK/ERK pathway by abrogating the inhibitory effect of miR-23a-3p on SOX6/SOX5 expression.
In conclusion, this study illustrated the molecular interaction between miR-23a-3p, SNHG5, and SOX6/SOX5 in hADSCs. The results from our study suggested that SNHG5 modulates SOX6/SOX5 to activate the JNK/MAPK/ERK pathway, affecting hADSC differentiation by sponge miR-23a-3p. Therefore, SNHG5 may have potential utility in enhancing stem cell chondrogenesis for therapeutic applications such as cartilage tissue engineering or the treatment of OA.
References
Almalki SG, Agrawal DK (2016) Key transcription factors in the differentiation of mesenchymal stem cells. Differentiation 92:41–51
Andarwulan N, Batari R, Sandrasari DA, Bolling B, Wijaya H (2010) Flavonoid content and antioxidant activity of vegetables from Indonesia. Food Chem 121:1231–1235
Dong S, Yang B, Guo H, Kang F (2012) MicroRNAs regulate osteogenesis and chondrogenesis. Biochem Biophys Res Commun 418:587–591
Dy P, Smits P, Silvester A, Penzo-Mendez A, Dumitriu B, Han Y, de la Motte CA, Kingsley DM, Lefebvre V (2010) Synovial joint morphogenesis requires the chondrogenic action of Sox5 and Sox6 in growth plate and articular cartilage. Dev Biol 341:346–359
Gangaraju VK, Lin H (2009) MicroRNAs: key regulators of stem cells. Nat Rev Mol Cell Biol 10:116–125
Gao J, Yang T, Han J, Yan K, Qiu X, Zhou Y, Fan Q, Ma B (2011) MicroRNA expression during osteogenic differentiation of human multipotent mesenchymal stromal cells from bone marrow. J Cell Biochem 112:1844–1856
Hodges WM, O’Brien F, Fulzele S, Hamrick MW (2017) Function of microRNAs in the osteogenic differentiation and therapeutic application of adipose-derived stem cells (ASCs). Int J Mol Sci 18:2597
Im GI (2017) Bone marrow-derived stem/stromal cells and adipose tissue-derived stem/stromal cells: their comparative efficacies and synergistic effects. J Biomed Mater Res A 105:2640–2648
Kang L, Yang C, Song Y, Liu W, Wang K, Li S, Zhang Y (2016a) MicroRNA-23a-3p promotes the development of osteoarthritis by directly targeting SMAD3 in chondrocytes. Biochem Biophys Res Commun 478:467–473
Kang SW, Kim J, Shin DY (2016b) Inhibition of senescence and promotion of the proliferation of chondrocytes from articular cartilage by CsA and FK506 involves inhibition of p38MAPK. Mech Ageing Dev 153:7–13
Kobayashi T, Lu J, Cobb BS, Rodda SJ, McMahon AP, Schipani E, Merkenschlager M, Kronenberg HM (2008) Dicer-dependent pathways regulate chondrocyte proliferation and differentiation. Proc Natl Acad Sci 105:1949–1954
Komarek J, Valis P, Repko M, Chaloupka R, Krbec M (2010) Treatment of deep cartilage defects of the knee with autologous chondrocyte transplantation: long-term results. Acta Chir Orthop Traumatol Cechoslov 77:291–295
Lai RC, Yeo RW, Lim SK (2015) Mesenchymal stem cell exosomes. Semin Cell Dev Biol 40:82–88
Lee W-S, Kim HJ, Kim K-I, Kim GB, Jin W (2019) Intra-articular injection of autologous adipose tissue-derived mesenchymal stem cells for the treatment of knee osteoarthritis: a phase IIb, randomized, placebo-controlled clinical trial. Stem Cells Transl Med 8:504–511
Li X, Liu L, Luo Y, Cui S, Chen W, Zeng A, Shi Y, Luo L (2019) Long noncoding RNA SNHG5 promotes glioma progression via miR-205/E2F3 axis. Biosci Rep 39:1-11
Liao JY, Ma LM, Guo YH, Zhang YC, Zhou H, Shao P, Chen YQ, Qu LH (2010) Deep sequencing of human nuclear and cytoplasmic small RNAs reveals an unexpectedly complex subcellular distribution of miRNAs and tRNA 3′ trailers. PLoS One 5:e10563
Lin GL, Hankenson KD (2011) Integration of BMP, Wnt, and notch signaling pathways in osteoblast differentiation. J Cell Biochem 112:3491–3501
Miura Y (2016) Human bone marrow mesenchymal stromal/stem cells: current clinical applications and potential for hematology. Int J Hematol 103:122–128
Niethammer TR, Pietschmann MF, Ficklscherer A, Gulecyuz MF, Hammerschmid F, Muller PE (2016) Incomplete defect filling after third generation autologous chondrocyte implantation. Arch Med Sci 12:785–792
Park SJ, Cheon EJ, Kim HA (2013) MicroRNA-558 regulates the expression of cyclooxygenase-2 and IL-1β-induced catabolic effects in human articular chondrocytes. Osteoarthr Cartil 21:981–989
Peng S, Cao L, He S, Zhong Y, Ma H, Zhang Y, Shuai C (2018) An overview of long noncoding RNAs involved in bone regeneration from mesenchymal stem cells. Stem Cells Int 2018:8273648
Pereira D, Peleteiro B, Araujo J, Branco J, Santos RA, Ramos E (2011) The effect of osteoarthritis definition on prevalence and incidence estimates: a systematic review. Osteoarthr Cartil 19:1270–1285
Pers Y-M, Quentin J, Feirreira R, Espinoza F, Abdellaoui N, Erkilic N, Cren M, Dufourcq-Lopez E, Pullig O, Nöth U, Jorgensen C, Louis-Plence P (2018) Injection of adipose-derived stromal cells in the knee of patients with severe osteoarthritis has a systemic effect and promotes an anti-inflammatory phenotype of circulating immune cells. Theranostics 8:5519–5528
Polymeri A, Giannobile WV, Kaigler D (2016) Bone marrow stromal stem cells in tissue engineering and regenerative medicine. Horm Metab Res 48:700–713
Salmena L, Poliseno L, Tay Y, Kats L, Pandolfi PP (2011) A ceRNA hypothesis: the Rosetta Stone of a hidden RNA language? Cell 146:353–358
Schäffler A, Büchler C (2007) Concise review: adipose tissue-derived stromal cells-basic and clinical implications for novel cell-based therapies. Stem Cells 25:818–827
Schulze-Tanzil G (2009) Activation and dedifferentiation of chondrocytes: implications in cartilage injury and repair. Ann Anat 191:325–338
Smits P, Dy P, Mitra S, Lefebvre V (2004) Sox5 and Sox6 are needed to develop and maintain source, columnar, and hypertrophic chondrocytes in the cartilage growth plate. J Cell Biol 164:747–758
Wang P, Mao Z, Pan Q, Lu R, Huang X, Shang X, Zhang R, You H (2018) Histone deacetylase-4 and histone deacetylase-8 regulate interleukin-1beta-induced cartilage catabolic degradation through MAPK/JNK and ERK pathways. Int J Mol Med 41:2117–2127
Wu DM, Wang S, Wen X, Han XR, Wang YJ, Shen M, Fan SH, Zhang ZF, Shan Q, Li MQ, Hu B, Lu J, Chen GQ, Zheng YL (2018) LncRNA SNHG15 acts as a ceRNA to regulate YAP1-Hippo signaling pathway by sponging miR-200a-3p in papillary thyroid carcinoma. Cell Death Dis 9:947
Yang Z, Hao J, Hu ZM (2015) MicroRNA expression profiles in human adipose-derived stem cells during chondrogenic differentiation. Int J Mol Med 35:579–586
Yang Z, Li R, Ao J, Wa QD, Zhang Y, Chen L, Wen J, Chen B, Pan W, Li B, Tian XB (2018) miR-1307-3p suppresses the chondrogenic differentiation of human adipose-derived stem cells by targeting BMPR2. Int J Mol Med 42:3115–3124
Zhu Y, Liu T, Song K, Fan X, Ma X, Cui Z (2008) Adipose-derived stem cell: a better stem cell than BMSC. Cell Biochem Funct 26:664–675
Funding
This work was supported by National Natural Science Foundation of China (31660265, 31960208, 81060145, and 81560356), Youth Fund of Guizhou Provincial People’s Hospital (GZSYQN(2015)06), Subsidy Foundation of National Natural Science Foundation of Guizhou Provincial People’s Hospital (Guizhou Science and Technology Platform (2017)5724), and Science and Technology Foundation of Guizhou Province (Guizhou Science and Technology J Word (2015)2096).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were following the ethical standards of the Ethics Committee of Tianjin Haihe Hospital and written informed consent was obtained from all the patients.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, Z., Ren, Z., She, R. et al. miR-23a-3p regulated by LncRNA SNHG5 suppresses the chondrogenic differentiation of human adipose-derived stem cells via targeting SOX6/SOX5. Cell Tissue Res 383, 723–733 (2021). https://doi.org/10.1007/s00441-020-03289-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-020-03289-4