Abstract
In order to study the mechanism by which an omental pedicle promotes healing when applied to an injured site, we injected a foreign body into the abdominal cavity to activate the omentum. One week after the injection, we isolated the omentum and measured blood vessel density, blood content, growth and angiogenesis factors (VEGF and others), chemotactic factors (SDF-1α), and progenitor cells (CXCR-4, WT-1). We found that the native omentum, which consisted mostly of adipose tissue, expanded the mass of its non-adipose part (milky spots) 15– to 20-fold. VEGF and other growth factors increased by two– to four-fold, blood vessel density by three-fold, and blood content by two-fold. The activated omentum also showed increases in SDF-1α, CXCR-4, and WT-1 cells (factors and cells positively associated with tissue regeneration). Thus, we propose that an omentum activated by a foreign body (or by injury) greatly expands its milky-spot tissue and becomes rich in growth factors and progenitor cells that facilitate the healing and regeneration of injured tissue.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The omentum, a sheet-like tissue attached to the greater curvature of the stomach, has been referred to as an adipose tissue with islands of compact tissue containing immune cells called “milky spots” (Liebermann-Meffert 2000). Although its natural role is not clear, transposing the omental pedicle to injured organs (omental transposition) has been used for over 100 years to promote healing (Cannaday 1948; Vineberg et al. 1966). The omentum also provides a “safe haven” for sustaining transplanted pancreatic islets, expanding cultured hepatocytes, and growing embryonic kidney and pancreas anlagen into adult organs (Kin et al. 2003; Sigrist et al. 2003; Lee et al. 2003; Hammerman 2004; Rogers et al. 2003; Takashi et al. 2006).
More recently, Goldsmith’s group has demonstrated that omental transposition promotes healing and even regeneration of neurons across a transected spinal cord in experiments in cats and also in one patient (Goldsmith and de la Torre 1992; de la Torre and Goldsmith 1994; Goldsmith et al. 2000, reviewed in Goldsmith 2004) resulting in the unexpected recovery of limb function. Despite reports of the benefits of omental transposition in acute injury, studies addressing the mechanism by which the omentum exerts such effects are lacking. However, in contrast to the functional recovery observed in cats in which omental transposition is performed hours after injury, the benefits of omental transposition in patients with long standing spinal injury remain questionable (Clifton et al. 1996; Duffill et al. 2001).
The omentum has the innate property of sensing injured sites in the abdominal cavity and of firmly adhering to them. It likewise reacts to foreign bodies present in the abdomen (Halverson et al. 1994; Luijendijk et al. 1996). On sensing the foreign body, the omentum rapidly extends and expands to encapsulate the object as if to protect the adjoining internal organs from contact with it. A study of this reaction to a foreign body and of the factors produced in the process might thus be relevant to understanding the way that omental fusion brings about healing and regeneration. We have therefore activated the omentum with a slurry of inert polydextran particles or with a polyvinyl chloride (PVC) solid rod attached to the stomach wall and have measured the density of the blood vessels, possible changes in growth, angiogenesis, and chemotactic factors, and the presence of progenitor cells in order to gain insights into the healing and regenerative properties of the omentum.
Materials and methods
Activation of omentum by foreign bodies
A group of rats (Sprague Dawley, 200–250 g) were injected with 5 ml polydextran particle slurry (Biogel P-60, 120 μM; Biorad Laboratories, Richmond, Calif.; 1:1 in normal saline) intraperitoneally. After 1–3 weeks, the animals were laparotomized, and the expanded omenta were harvested and processed as described below. Native omenta (control) were harvested and processed from age-matched normal rats injected with normal saline.
Another group of rats were laparotomized, and a piece of PVC tubing (length: 25 mm, internal diameter: 3 mm; PVC 180, Nalge Nunc International, Rochester, N.Y.) that had been heat-sealed at both open ends was stitched to the stomach wall via one end, with the other end lying free in the abdominal cavity.
The polydextran slurry was stored in 70% alcohol for disinfection and washed vigorously with sterile saline before injection. The PVC rod was also kept in 70% alcohol for sterilization and air-dried before implantation. The omentum was allowed to expand around the tube for 1–6 weeks before being harvested.
The omentum from rats that were not injected/implanted with a foreign body served as the control omentum (henceforth called native omentum).
Histological processing of omental tissue and quantification of areas of milky spots and adipose tissue
Pieces of native and activated omenta were fixed for histology and immunochemistry by immersion in Histochoice (Amersco, Solon, Ohio). Following dehydration and paraffin embedment, tissues were sectioned and stained by hematoxylin-eosin (H&E). Areas (square pixels) of milky spots and adipose tissue were measured from random digital micrographs of the omenta for calculation of the percentage area occupied by milky spots.
Of note, the milky spots in rat omentum, unlike those of human omentum (and omenta from several other species), are not visible by the naked eye. However, these compact islands of stromal cells, which are essentially similar in function across species, can be easily distinguished histologically in the rat omentum. We will therefore continue to term such areas as milky spots in the work described below.
Immunostaining for type IV collagen, stromal cell-derived factor 1α, chemokine receptor-4, and Wilms’ tumor-1
Immunostaining of the sections was carried out by first incubating them with goat anti-type IV collagen (Southern Biotechnology Associates, Birmingham, Ala.), anti-chemokine receptor-4 (CXCR-4), anti-stromal cell-derived factor 1α (SDF-1α; Chemicon International, Temecula, Calif.), or mouse anti-Wilm’s tumor-1 (WT-1; Dako, Glostrup Denmark), followed by washes and incubation with a secondary anti-goat IgG-peroxidase conjugate or other appropriate peroxidase-conjugated secondary antibodies or a fluorescein-isothiocyanate-labeled secondary antibody (Sigma, St Louis, Mo.). The slides were then washed, and those sections incubated with peroxidase-conjugated secondary antibodies were developed with diaminobenzidine- H2O2 (brown color) and viewed by light microscopy, whereas fluorescein-labeled sections were wet-mounted in glycerol in phosphate-buffered saline (PBS) and viewed by epifluorescence. The sections were digitally photographed (Nikon, New York).
Visualization of blood vessels
Blood vessels and lymphatic vessels were visualized by type IV collagen immunostaining as described above. From previous studies, we had established that new and existing blood vessels and microvessels were better visualized by type IV collagen immunostaining than by von Willebrand factor (also known as Factor VIII) immunostaining (Gudehithlu et al. 2005).
Quantification of blood content in omental extracts by hemoglobin determination
For the determination of blood content in the activated omentum, we used omenta activated by a PVC rod, because the omenta activated by polydextran particles contained embedded polydextran particles that made the determination of wet weights difficult. Freshly harvested omenta were wet-weighed and placed in ice-cold water containing heparin to promote red cell lysis and avoid blood clotting. The tissue was homogenized in a Polytron homogenizer. The homogenate was centrifuged at 15,000g for 10 min. The supernatant was filtered through 0.45-μm filters to obtain a clear filtrate, which was scanned in a spectrophotometer in the 400–800 nm range to measure the two typical absorption peaks of hemoglobin at 540 and 575 nm (Gudehithlu et al. 2005). The hemoglobin concentration was determined from the absorption at 575 nm by using a standard curve constructed from freshly diluted human blood with known hemoglobin concentrations, also read at 575 nm. On the assumption that the hemoglobin content in normal blood was 15 g/dl, the hemoglobin concentration was converted to microliters of blood per gram wet weight of tissue.
Preparation of cell-free omental extracts for measurement of vascular endothelial factor and other growth factors
Because assay kits for specific growth factors of rat specificity (except for vascular endothelial factor [VEGF], see below) were unavailable, we assayed the mixture of growth factors present in the activated omentum by comparing the efficacy of cell-free omental extract with that of fetal bovine serum (FBS; a fluid rich in growth factors and commonly used as an additive in tissue culture media) in supporting the growth of four cell lines in tissue culture. Cell-free omental extract was prepared by dispersing the activated omenta or native omenta in a 1:1 volume of PBS by using a Potter-Elvehjm tissue homogenizer (mechanized glass tube and Teflon pestle with a clearance of 125 μm), which lacerated the tissue but prevented cell breakage. The extract was centrifuged at 5,000g to remove cells and polydextran particles. The supernatant was filtered through a sterile 0.22-μm filter before being tested on cultured cells. The protein concentration in the native and activated omental extracts was determined by the Biorad assay, and the extracts were diluted with basal RPMI medium (or EBM medium for endothelial cells; see below) to a final concentration of 5 mg/ml to allow comparisons with standard tissue-culture medium (RPMI containing 10% FBS) Four mammalian cell lines were tested for growth properties after the cells had been seeded in equal numbers in (1) basal RPMI medium (basal medium, BM), (2) RPMI medium containing the cell-free native omental extract (Native-O), (3) RPMI medium containing cell-free activated omental extract (Active-O), or (4) RPMI containing 10% FBS (FBS). The four cell lines tested were: a glomerular epithelial cell line (GEC; obtained originally from Kreisberg et al. 1978), human umbilical vein endothelial cells (HUVEC, a prototype endothelial cell used in culture studies and obtained from Cambrex Bio Science Rockland, East Rutherford, N.J.), primary rat glomerular mesangial cells grown by seeding rat glomeruli as described previously (Singh et al. 2003), and a human proximal tubular cell line (HK-2) obtained from the American Type Culture Collection (Washington DC, USA). For HUVEC, the basal medium used was EBM, and the standard growth medium containing growth factors was EGM (instead of FBS), both supplied by Cambrex Bio Science Rockland. Following incubation for 2 days in the various media, the cells were detached from their dishes by trypsinization, and growth was measured by cell counting with a hemocytometer.
VEGF was measured in the cell-free omental extracts by a sandwich enzyme-lnked immunosorbent assay by using a commercial murine VEGF assay kit (R and D Systems, Minneapolis, Minn.).
Histological quantification and statistical methods
The data are presented as means ± SEM with the number of observations (n) being indicated in the figure legends or Table. Statistical comparisons were made by using the Student t-test, with P < 0.05 being considered as significant. Tissue areas in square pixels were measured from digital micrographs of H&E-stained or immunostained sections by using Image J software (JAVA imaging software inspired by the National Institute of Health and available free at http://rsb.info.nih.gov). Blood vessels were manually counted from digital micrographs of sections immunostained for type IV collagen, which highlighted small and large vessels. Blood vessel density was determined as: number of blood vessels/10,000 square pixels.
Results
Expansion of omentum upon contact with foreign bodies
Polydextran particles (100 μm in diameter) injected intraperitoneally caused the omentum to spread rapidly and to enclose each bead separately (Fig. 1a). By 1 week, all the injected particles were included in the omental tissue and peritoneal washes at this time rarely showed any free particles. The histological image of the omentum at 2 or 3 weeks after polydextran injection remained the same as that after 1 week (Fig. 1a). The shape of the omentum was variable and differed from animal to animal depending on the location of the beads. Usually, the omentum extended from its origin at the stomach wall to the lobes of the liver and to large portions of the upper gastrointestinal tract, also resulting in adhesions to these organs. The extended omentum appeared reddish in color because of the extensive blood vessel growth in the tissue.
Histology of native and activated omentum. a Native omentum mostly consisted of adipose cells with small islands of lymphoid cells (arrows; also termed milky spots). b Higher magnification of milky spot boxed in a. c Omentum activated by polydextran particles showing the greatly extended non-adipose tissue surrounding the individual polydextran particles. Several polydextran particles (intense purple) are visible. H&E staining. Bars 100 μm
Native omentum was composed of mostly adipose tissue (93% of omental area) and small islands of non-adipose lymphoid tissue called “milky spots”. After activation, the non-adipose component of the omentum, probably the milky spots, had extended, whereas the adipose tissue appeared not to change in size (Fig. 1a–c). Area measurements of the adipose and non-adipose portions of the activated omentum showed that the milky spots (shown magnified in Fig. 1b), which occupied 7% of the omental area in the native state, extended to occupy 76% of the omentum in its activated state (Fig. 1c, Table 1).
Native omentum in the control rats, which weighed approx 0.1 g (wet weight), grew to approximately 2.0 g wet weight (after subtraction of the weight of the injected particles) by 1 week following polydextran injection and remained at the same wet weight for up to 3 weeks (Table 1).
When stimulated by a PVC rod, the omentum grew to form a pedicle along the length of the rod and tended to encapsulate it. After 1 week, the omentum grew to a wet weight of approximately 1.5 g, but unlike the polydextran-activated omentum, it continued to grow for several weeks until the rod was completely encapsulated (6 weeks; ~2 g after two weeks, ~3 g after three weeks, ~7 g after 6 weeks). The tissue growth was paralleled by the growth of major blood vessels along the length of the pedicle. Unlike the omentum activated by polydextran seen in Fig. 1c, the omentum activated by a PVC rod was more compact in several regions. In these compact regions, it was highly vascular, and the blood vessels were larger (see below).
Increased blood vessel density and blood content in activated omentum
Native omentum, which consists of adipose tissue and milky spots, is richly supplied by blood and lymphatic vessels (Fig. 2a). However, the blood vessel densities in the adipose part and in the milky spots of the native omentum are highly disparate and, the blood vessel density of the milky spots is four times greater than in the adipose part (Fig. 2a,b, Table 1).
When the omentum was activated by polydextran, the major and minor blood vessels extended into the new non-adipose tissue, so that the blood vessel density became three times as great as that of the adipose part and was similar to that of the milky spots (Fig. 2c, Table 1). At the edge of the activated omentum, we noted a pronounced increase in fine vessels that were suggestive of lymphatic channels (Fig. 2d).
Blood vessels in the native and activated omentum revealed by Type IV collagen immunostaining. a Blood vessels in the adipose part of native omentum. b Milky spot of native omentum showing fine blood vessels at a higher density than those in the adipose part of native omentum. c Blood vessel density in the omentum activated by polydextran particles was higher than that in the adipose part of native omentum and resembled that seen in the milky spots of native omentum. d Edge of activated omentum showing fine vessels, probably lymphatic channels. Bars 100 μm
In the omentum activated by a PVC rod, the overall density of blood vessels is also three-fold higher than that in the adipose part and as high as in the milky spots of the native omentum (Table 1). Figure 3b shows blood vessels stained for Type IV collagen in a compact region of PVC-activated omentum. In the few compact regions, the vascularity appears to be higher than that in polydextran-activated omentum (cf. Fig. 2b), but the overall vascularity in the tissue is similar to that seen in polydextran-activated omentum (Table 1).
Omentum activated by a PVC rod attached to the stomach wall (rod not visible). a The omentum is greatly expanded and more compact in a few regions than an omentum activated by polydextran particles. In the compact regions, the omentum appears to be highly vascularized with large blood vessels. H&E staining. b Blood vessels (higher power) revealed by Type IV collagen immunostaining in a compact region of PVC-activated omentum. Although the vascularity appears to be higher than in polydextran-activated omentum (cf. Fig. 2b), the overall vascularity is similar to that seen in polydextran-activated omentum and milky spots of native omentum (see Table 1). Bars 100 μm
To substantiate the increase in blood vessel density in the activated omentum, blood content was compared in native and PVC-rod-activated omentum and was found to be increased by two-fold (Table 1). Although the increase in hemoglobin content might have been caused by vessel congestion, no a priori reason led us to believe that such was the case. Indeed, the increase in hemoglobin was consistent with the increase in blood vessel density, thereby supporting our hypothesis of increased angiogenesis.
Increase of VEGF and other growth factors in activated omentum
VEGF is one of the most powerful mitogens for endothelial cells and is responsible for new blood vessel formation. Of note, the concentration of VEGF found in the native omentum is itself 10– to 100-fold higher than levels usually found in other adult rat tissues (Singh et al. 2004), as also reported by others (Zhang et al. 1997).
Cell-free extracts from omentum activated by polydextran particles exhibited a VEGF concentration 3.5 times higher than that in native omentum (Fig. 4). Since the omentum activated by polydextran expanded by 20-fold in mass within 1 week, the total content of VEGF must have increased by 70-fold in the activated omentum compared with the native omentum.
VEGF levels in cell-free extracts of native and polydextran-activated omenta. Extracts of omentum activated by polydextran particles conatined 3.5 times more VEGF than extracts from native omentum. For comparison, the level of VEGF in adult rat kidney was 10-fold less (approximately 0.2 ng/mg protein; Luijendijk et al. 1996). *Statistically significant difference from the native omentum at P < 0.05
Growth factors other than VEGF could not be individually assayed in the cell-free omental extracts because assay kits of rat specificity were unavailable. Therefore, the mixture of other growth factors in the activated omentum was tested by the ability of the cell-free omental extract to support the growth of four different types of mammalian cells in culture. We deliberately tested four cell lines for comparison as they represented different cell types of human and rat origin. These were tubular epithelial cells (HK-2, human; GEC, rat), HUVEC (human endothelial), and primary cells of mesodermal origin (primary rat glomerular mesangial cells). Growth in cell-free omental extract was compared with growth in the “gold-standard” FBS-containing RPMI medium (and also with basal medium and medium containing native omental extract). The basal medium by itself was poor in supporting the growth of all four cell types as expected. Medium containing native omental extract gave slightly better results than the basal medium. The medium containing activated omental extract or FBS was two to four times more potent in supporting the growth of the four cell types than the medium containing native omental extract. In the cases of HK-2 cells and primary mesangial cells, the medium containing activated omental extract was superior to the gold-standard FBS-containing medium (Figs. 5, 6).
The high growth factor content of activated omentum as demonstrated by the excellent growth of glomerular epithelial cells (GEC) and primary mesangial cells (Pr MC) in medium containing activated cell-free omental extract. Cell growth was comparable with that attained in the gold-standard medium containing FBS (see Fig. 6 for quantitative data on the four cultured cell types). Bars 100 μm
Comparison of growth-factor content of activated and native omenta by using the growth of four different mammalian cell types cultured in medium containing either native or activated omental extract (Native-O or Active-O, respectively). Cell growth in basal medium (BM, negative control) and in medium containing FBS (FBS, positive control) is also shown. Medium containing activated omental extract is clearly more potent in supporting cell growth than medium containing native omental extract, suggesting a three to four times higher growth factor content in activated omentum than in native omentum. Growth-promoting activity of medium containing activated omental extract equals or exceeds the activity of medium containing FBS (GEC glomerular epithelial cells, HUVEC human umbilical vein endothelial cells, HK-2 human proximal tubular cell line, pr MC primary mesangial cells). In the case of HUVEC, the purchased medium EBM was better than the activated omentum. *Statistically significant difference from native omental extract at P < 0.05. #Statistically significant difference from FBS (or EBM for HUVEC) at P < 0.05
Abundance of progenitor cells (positive for SDF-1α, CXCR-4, and WT-1) in activated omentum
Activated and native omenta were immunostained for several markers of progenitor cell activity. The presence of cells positive for SDF-1α (a chemokine), CXCR-4 (chemokine receptor for SDF-1α present on progenitor cells of bone marrow and local tissue), or WT-1 (marker of mesenchymal cells preceding differentiation to epithelial cells during development) is the hallmark of a regenerating (or developing) tissue.
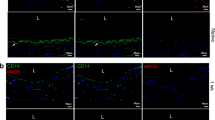
Native omentum generally showed patchy SDF-1α and CXCR-4 immunoreactivity in areas limited to around blood vessels. More concentrated reactivity was seen in cells of the milky spots (Figs. 7a, 8a). Consistent with the expansion of milky spots by polydextran particles, activated omentum showed larger areas positive for SDF-1α and CXCR-4, with more intense staining (similar to that seen in milky spots) in cells surrounding the polydextran particles (Figs. 7b, 8b). Upon careful examination of the micrographs, we observed that, whereas CXCR-4 immunoreactivity was more concentrated in cells immediately surrounding the particles, SDF-1α reactivity was present diffusely at greater distances from the polydextran particles.
Immunostaining of native and activated omentum for stromal-cell-derived factor-1α (SDF-1α). a Native omentum showing concentrated SDF-1α immunoreactivity in milky spots (arrow) and patchy staining in areas around blood vessels. b Activated omentum showing larger SDF-1α-positive areas with more intense staining (similar to reactivity in milky spots) in cells surrounding the polydextran particles. Bars 100 μm
Immunostaining of native and activated omentum for the chemokine receptor CXCR-4. a Native omentum showing concentrated CXCR-4 immunoreactivity in milky spots (arrow) and patchy staining in areas around blood vessels. b Activated omentum showing larger CXCR-4-positive areas with more intense staining (similar to reactivity in milky spots) in cells surrounding the polydextran particles. The staining pattern is similar to that seen with SDF-1α (Fig. 7). Bars 100 μm
Similarly, WT-1-positive cells were concentrated in the milky spots of the native omentum but were present only in small numbers in the adipose part of the native omentum. In the activated omentum, WT-1-positive areas were greatly expanded and, with respect to density, appeared to be similar to that seen in the milky spots of the native omentum (Fig. 9a,b).
WT-1-positive cells in native and activated omentum revealed by immunostaining. a Native omentum showing WT-1-positive cells concentrated in the milky spots but present only in low numbers in the adipose part of the omentum (arrow). b Activated omentum showing that the WT-1 cells have greatly increased and have a density similar to that seen in the milky spots of native omentum. Bars 100 μm
Adult rat kidney was used as a control for the above markers (not shown). Normal adult rat kidney structures were found to be completely negative for CXCR-4, mildly positive for SDF-1α (only in distal tubules), and strongly nuclear-positive for WT-1, but only in glomerular podocytes (Pritchard-Jones et al. 1990; Kreidberg et al. 1993).
Discussion
We have observed that, on contact with a foreign body, the omentum expands rapidly in size and mass, thereby completely encapsulating it. Remarkably, the non-adipose portion of the omentum (milky spots, which are islands of lymphoid and phagocytic cells in the native state) expands, rather than the adipose part of the organ. Accordingly the omentum, which in its native state in the rat is a flimsy structure, now assumes the appearance of a solid mass of tissue that resembles an expanded milky spot. In this expanded state, we have found that the omentum exhibits large increases in the densities of blood vessels and lymphatic vessels, in blood content, in the angiogenesis factor VEGF, in other growth and chemotactic factors (which attract progenitor cells), and in progenitor cells. These changes, originally present in the milky spots, are now manifested in the entire organ, almost as though the entire activated omentum has become transformed into a large expanded milky spot.
In our experiments, the omentum appears to be a pliable tissue, the size and shape of which can be dictated by the form of the foreign body. When stimulated by polydextran particles, the omentum takes an unpredictable shape depending on the location of the particles in the peritoneal cavity. However, when stimulated by a rigid body such as a PVC rod attached to the stomach wall, the omentum assumes a cylindrical form that follows the contours of the rigid foreign body. This property might be useful in experimentally creating an omental pedicle tailored to the length and form required for omental transposition.
We have also found that an omentum activated by foreign bodies induces a large increase in the concentration and content of VEGF. However, since VEGF has been measured in the cell-free extracts of the total omentum and not specifically in milky spots, we have been unable to determine whether the milky spots are richer in VEGF than the rest of the omental tissue, such is the case with other tested markers (SDF-1α, CXCR-4, and WT-1). As VEGF is a diffusible factor that is unpredictably lost during the fixation and processing of tissue, it cannot be identified reliably by immunostaining in the milky spots; all other factors have been identified in the milky spots and occur at higher levels than those in the adipose tissue of native omentum.
At present, the mechanism whereby the omentum prevents infections and promotes healing of injured organs remains poorly understood. By analogy with its response to foreign bodies, the omentum in vivo might similarly be activated by sensing that blood clots, tissue debris, and infectious agents are “foreign” and thus being attracted to the injured area. The release of high amounts of angiogenic (VEGF) and other growth factors facilitates the growth of new blood vessels and lymphatic vessels that anastamose with those of the injured organ, helping to clear tissue debris and clotted blood, assuring a fresh blood supply, and thus preventing tissue death by ischemia. Moreover, the revascularized injured tissue is thus supplied with a potent mixture of growth factors (VEGF and others), progenitor cells (such as cells positive for CXCR-4 and WT-1), and chemotactic factors (such as SDF-1α), which further helps in the recruitment of progenitor cells from the bone marrow and local tissue to accelerate tissue repair.
The ability of the activated omentum to form new blood vessels that anastamose with organ vessels has been utilized in regenerating embryonic tissues, thereby sustaining transplanted pancreatic islets and expanding cultured liver cells implanted in the omentum (Kin et al. 2003; Sigrist et al. 2003; Lee et al. 2003; Hammerman 2004; Rogers et al. 2003; Takashi et al. 2006). Thus, blood vessels vascularizing implanted xenogenic embryonic tissue have been shown to be derived from the host omentum, suggesting that these omental vessels can differentiate with respect to the specificity of the vascularized organ (Hammerman 2004; Takashi et al. 2006). For example, mouse kidney anlagen implanted in the rat omentum gives rise to a fully grown mouse kidney with rat blood vessels. The glomerular vessels of this kidney, obviously derived from the omentum, are even capable of ultrafiltration. These findings demonstrate the exceptional adaptability of omental blood vessels (Hammerman 2004; Takashi et al. 2006).
Large amounts of ambient growth factors and chemokines are increasingly recognized as being required to stimulate the differentiation of adult and embryonic stem cells (Humes et al. 1989; Nigam and Lieberthal 2000). The chemokines/chemokine receptor axis SDF-1α/CXCR-4 is particularly important in embryogenesis and tissue repair (Hatch et al. 2002; Kayali et al. 2003; Sordi et al. 2005), and their gene deletion or mutation results in embryonic death. We have found that SDF-1α, which usually occurs in developing and injured organs but not in adult tissues, is present in the milky spots of native omentum and, more abundantly, in activated omentum. The presence of SDF-1α might create a milieu in which progenitor cells from injured tissue or bone marrow are attracted to the injured site. Consistent with this, we have detected an abundance of cells positive for CXCR-4 and WT-1 in the activated omentum.
WT-1 is a nuclear regulatory protein expressed during the differentiation of mesenchymal cells into epithelial cells at the time of the development of genitourinary and other mesodermally derived tissues (Pritchard-Jones et al. 1990; Kreidberg et al. 1993; for a review, see Hastie 2001). As the activated omentum is a newly formed tissue, we have hypothesized that it must be a differentiating tissue and have thus chosen WT-1 to mark such differentiating cells. The finding of high WT-1 expression in the activated omentum suggests that active epithelial differentiation (or mesenchymal-epithelial transformation) takes place in the tissue as it expands to form blood vessels and interstitial tissue. On contact with the high levels of growth factors generated in the omentum, such progenitor WT-1 cells can begin to differentiate, and with added cues from the injured organ, to initiate tissue regeneration with the specificity of the injured tissue. Since the natural regenerative process is highly orchestrated and depends on specific growth factors at different times, prediction of the outcome of the regenerative process brought about by omental contact is difficult. Nevertheless, the fusion of injured tissue with activated omentum might set the stage in which tissue regeneration can take place, as reported by the Goldsmith group (Goldsmith and de la Torre 1992; de la Torre and Goldsmith 1994; Goldsmith et al. 2000; Goldsmith 2004) following omental transposition in association with neurons of the transected spinal cord. Whether omental transposition can induce tissue regeneration in other organs needs to be re-investigated, especially if this can be aided by bridging the injured site and the omentum with artificial collagen, as shown by Goldsmith et al. (2000) and Goldsmith (2004).
In conclusion, our study shows that the activation of the omentum by contact with injured tissue leads to an expansion of its milky spot area. At the same time, its content of “healing factors” (growth, angiogenesis, and chemotactic factors and progenitor cells) increases, all of which promote the healing and regeneration of the injured tissue.
References
Cannaday JE (1948) Some uses of undetached omentum in surgery. Am J Surg 76:502–505
Clifton GL, Donovan WH, Dimitrijevic MM, Allen SJ, Ku A, Potts JR III, Moody FG, Boake C, Sherwood AM, Edwards JV (1996) Omental transposition in chronic spinal cord injury. Spinal Cord 34:193–203
Duffill J, Buckley J, Lang D, Neil-Dwyer G, McGinn F, Wade D (2001) Prospective study of omental transposition in patients with chronic spinal injury. J Neurol Neurosurg Psychiatry 71:73–80
Goldsmith HS (2004) The evolution of omentum transposition: from lymphedema to spinal cord, stroke and Alzheimer’s disease. Neurol Res 26:586–593
Goldsmith HS, de la Torre JC (1992) Axonal regeneration after spinal cord transaction and reconstruction. Brain Res 589:217–224
Goldsmith HS, Brandt M, Waltz T (2000) Near total transaction of human spinal cord: a functional return following omentum-collagen reconstruction. In: Goldsmith HS (ed) The omentum: application to brain and spinal cord Forefront, Wilton, pp 76–92
Gudehithlu KP, Ahmed N, Yu H, Litbarg, NO, Garber SL, Arruda, JAL, Dunea G, Singh AK (2005) Antagonism of VEGF results in microvessel attrition and disorganization of wound tissue. J Lab Clin Med 145:194–203
Halverson JM, Butterman MK, Legeir JF, Mann WJ Jr, Hoefer RA Jr (1994) Perforation of Meckel’s diverticulum caused by injestion of a coin. South Med J 87:823–824
Hammerman MR (2004) Renal organogenesis from transplanted metanephric primordia. J Am Soc Nephrol 15:1126–1132
Hastie ND (2001) Life, sex, and WT-1 isoforms—three amino acids can make all the difference. Cell 106:391–394
Hatch HM, Zheng D, Jorgensen ML, Petersen BE (2002) SDF-1α/CXCR-4; a mechanism for hepatic oval cell activation and bone marrow stem cell recruitment to the injured livers in rats. Cloning Stem Cells 4:339–351
Humes HD, Cielinski DA, Coimbra TM, Messana JM, Galvao C (1989) Epidermal growth factor enhances renal tubule cell regeneration and repair and accelerates the recovery of renal function in postischemic acute renal failure. J Clin Invest 84:1757–1761
Kayali AG, Gunst KV, Campbell IL, Stotland A, Kritzik M, Guoxun L, Flodstrom-Tullberg M, Zhang Y-Q, Sarvetnick N (2003) The stromal cell-derived factor-1α (SDF-1α)/CXCR-4 ligand-receptor axis is critical for progenitor survival and migration in the pancreas. J Cell Biol 163:859–869
Kin T, Korbutt GS, Rajotte RV (2003) Survival of islets in omental pouch. Am J Tranplant 3:281–285
Kreidberg JA, Sariola H, Loring JM, Maeda M, Pelletier J, Housman D, Jaenisch R (1993) WT-1 is required for early kidney development. Cell 74:679–691
Kreisberg JL, Hoover RL, Karnowsky MJ (1978) Isolation and characterization of rat glomerular epithelial cells in vitro. Kidney Int 14:21–30
Lee H, Cusick RA, Utsunomiya H, Ma PX, Langer R, Vacanti JP (2003) Effect of implantation site on hepatocytes heterotopically transplanted on biodegradable polymer scaffolds. Tissue Eng 9:1227–1232
Liebermann-Meffert D (2000) The greater omentum: anatomy, embryology, and surgical applications. Surg Clin North Am 80:275–293
Luijendijk RW, Lange DCD de, Wauters CCAP, Hop WCJ, Duron JJ, Pailler JL, Camprodon BR, Gelsdorp HJ van, Jeekel J (1996) Foreign material in postoperative adhesions. Ann Surg 223:242–248
Nigam S, Lieberthal W (2000) Acute renal failure. III. The role of growth factors in the process of renal regeneration and repair. Am J Physiol Renal Physiol 279:F3–F11
Pritchard-Jones K, Fleming S, Davidson D, Bickmore W, Porteous D, Gosden C, Bard J, Buckler A, Pelletier J, Housman D (1990) The candidate Wilms’ tumor gene is involved in genitourinary development. Nature 346:194–197
Rogers SA, Chen F, Talcott M, Hammerman MR (2003) Islet cell engraftment and control of diabetes after transplantation of pig pancreatic anlagen. Am J Physiol Endocrinol Metab 286:E502–E509
Sigrist S, Machine-Neuville A, Mandes K, Calenda V, Legeay G, Bellocq J-P, Pinget M, Kessler L (2003) Induction of angiogenesis in omentum with vascular endothelial growth factor: influence of encapsulated rat pancreatic islets during transplantation. J Vasc Res 40:359–367
Singh R, Singh AK, Alavi N, Leehey DJ (2003) Mechanism of increased angiotensin II levels in glomerular mesangial cells cultured in high glucose. J Am Soc Nephrol 14:873–880
Singh AK, Gudehithlu KP, Pegoraro AA, Singh GK, Bashiruddin K, Robey RB, Arruda Jose AL, Dunea G (2004) Vascular factors altered in glucose-treated mesangial cells and diabetic glomeruli. Changes in vascular factors impair endothelial cell growth and matrix. Lab Invest 84:597–606
Sordi V, Malosio ML, Marchesi F, Marcalli A, Melzi R, Giordano T, Belmonte L, Ferrari G, Leone BE, Bertuzzi F, Zerbini G, Allavena P, Bonifacio E, Piemonti L (2005) Bone marrow mesenchymal stem cells express a restricted set of functionally active chemokine receptors capable of promoting migration to pancreatic islets. Blood 106:419–427
Takashi Y, Fukui A, Ohashi T, Miyazaki Y, Utsunomiya Y, Kawamura T, Hosoya T, Okabe M, Kobayashi E (2006) Xenobiotic kidney organogenesis from human mesenchymal stem cells using a growing rodent embryo. J Am Soc Nephrol 17:1026–1034
Torre JC de la, Goldsmith HS (1994) Coerulospinal fiber regeneration in transected feline spinal cord. Brain Res Bull 35:413–417
Vineberg AM, Kato Y, Pirozynski WJ (1966) Experimental revascularization of the entire heart; evaluation of epicardiectomy, omental graft, and/or implantation of the internal mammary artery in preventing myocardial necrosis and death of the animal. Am Heart J 72:79–93
Zhang Q-X, Magovern CJ, Mack CA, Budenbender KT, Wilson K, Rosengart TK (1997) Vascular endothelial growth factor is the major angiogenic factor in omentum: mechanism of the omentum-mediated angiogenesis. J Surg Res 67:147–154
Acknowledgements
We thank Ms. Valentina Svoren and Dr. Lev Rappoport for help with the histological processing and immunostaining of tissues.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was partly supported by a grant (no. 2000–241 to A.K.S.) from the Juvenile Diabetes Foundation International.
Rights and permissions
About this article
Cite this article
Litbarg, N.O., Gudehithlu, K.P., Sethupathi, P. et al. Activated omentum becomes rich in factors that promote healing and tissue regeneration. Cell Tissue Res 328, 487–497 (2007). https://doi.org/10.1007/s00441-006-0356-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-006-0356-4