Abstract
Vector-borne diseases are among the greatest causes of human suffering globally. Several studies have linked climate change and increasing temperature with rises in vector abundance, and in the incidence and geographical distribution of diseases. The microbiome of vectors can have profound effects on how efficiently a vector sustains pathogen development and transmission. Growing evidence indicates that the composition of vectors’ gut microbiome might change with shifts in temperature. Nonetheless, due to a lack of studies on vector microbiome turnover under a changing climate, the consequences for vector-borne disease incidence are still unknown. Here, we argue that climate change effects on vector competence are still poorly understood and the expected increase in vector-borne disease transmission might not follow a relationship as simple and straightforward as past research has suggested. Furthermore, we pose questions that are yet to be answered to enhance our current understanding of the effect of climate change on vector microbiomes, competence, and, ultimately, vector-borne diseases transmission.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Vector-borne pathogens cause some of the most prevalent and deadly human (World Health Organization, WHO 2014, 2020) and wildlife diseases (Perkins 2001; Marzal 2012; Tomé et al. 2019). For instance, malaria alone is responsible for up to 400 thousand human deaths yearly (WHO 2020). Due to the high human and conservation burden imposed by vector-borne pathogens, those pathogens have been the focus of studies in several distinct areas of science (e.g., immunology, pathology, and ecology) for decades. In the twenty-first century, research has aimed to predict how global change (i.e., contemporary environmental/climatic modification due to anthropogenic effects), focusing mostly on increasing temperature, will affect transmission dynamics of pathogens borne by vectors (Atkinson and LaPointe 2009; Garamszegi 2011; Loiseau et al. 2013; Rocklöv and Dubrow 2020). Indeed, the human and wildlife burden associated with these pathogens is forecasted to increase in intensity and distribution due to the effects of global warming on the abundance and distribution of arthropod vectors (e.g., mosquitoes and ticks) (Kelly-Hope et al. 2009; Garamszegi 2011; Pérez-Rodríguez et al. 2014; Li et al. 2021; Gray and Ogden 2021). However, researchers have often ignored important biological traits associated with infection, such as the optimum temperature for parasite development and, more recently, the emerging recognition that ambient temperature affects the composition of vector microbiome (the microbial community living within vectors) (Mouton et al. 2006; Moghadam et al. 2018; Onyango et al. 2020). Here, we assess how such neglected traits might shape vector-borne disease transmission in a world of changing climate, focusing mostly on mosquito-borne pathogens.
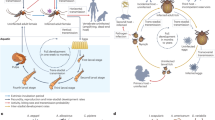
The microbiome (including bacteria, archaea, viruses, and protozoa) can deeply modify an organism’ health and, hence, the outcomes or odds of infection in both hosts and vectors (Grice and Segre 2012; Johnson 2015; Ippolito et al. 2018; McLoughlin et al. 2021; Pimentel et al. 2021; Videvall et al. 2021). Many studies have evaluated the role of the gut microbiome in modulating general health and fitness, behavior, and immune response to pathogens (Ezenwa et al. 2012; Salem et al. 2018; Zeevi et al. 2019; Durack and Lynch 2019; Manor et al. 2020). The presence of certain microbes (e.g., Wolbachia) seems to have profound effects on a vector’s ability to sustain the development of pathogens and their transmission (i.e., vector competence) (Weiss and Aksoy 2011; Johnson 2015; Pimentel et al. 2021; Cansado-Utrilla et al. 2021). This provides evidence that at least some components of a vector’s microbiome can influence its ability to transmit diseases. Since vector-borne diseases represent one of the main human burdens worldwide, several researchers have attempted to develop vector strains resistant to pathogen development as a tool to prevent human infection (Iturbe‐Ormaetxe et al. 2011; Caragata et al. 2021). Nonetheless, since vectors are ectothermic organisms and, as a result, their microbiome is subject to climatic/environmental changes, here we speculate on whether the environmental disruptions associated with global change could (i) affect the microbiome of vectors (Fig. 1A), and, mostly importantly, (ii) whether this altered microbiome can modulate the future dynamics of vector-borne pathogen transmission (Fig. 1B).
Forecasted scenarios for microbiome turnover and vector competence associated with global change. A Representation of presumed microbiome turnover resulting from modification from current to future environmental conditions. B Illustration of principal coordinates analysis comparing the composition of vector gut microbiome between present-day vectors and vectors subject to future environmental conditions. C Two possible scenarios of changes in vector competence (e.g., increase or decrease) associated with variation in microbiome composition linked to environmental conditions explained by the first axis of the principal coordinate analyses (i.e., the axis that explains most of the variation observed in the data)
Can global change affect the microbiome of vectors?
The increasing emissions of carbon dioxide in the atmosphere shall raise global temperature by 2 to 4° by 2100 (IPCC 2021). This rise in temperature will not be uniform throughout the globe and is expected to be particularly high in the continental areas of the North hemisphere (IPCC 2021). Previous studies have indicated that temperature can affect the microbiome composition of various organisms (Lokmer and Mathias Wegner 2015; Moghadam et al. 2018; Huus and Ley 2021), while other anthropogenic changes can also alter microbiome communities. For instance, urbanization has recently been associated with an increase in infectious air microbes (Li et al. 2019) whereas pesticides are known to modify soil microbial composition (Bond-Lamberty et al. 2016). Pesticide use could modify insect gut microbiomes by increasing selection pressure for insects that harbor microbes that provide resistance to chemicals (e.g., phenolic glycoside oleuropein and pinene) (Gressel 2018). Therefore, mosquito microbiomes might be subject to multiple environmental factors associated with global change.
Anthropogenic landscape changes that lead to shifts in environmental microbiome could directly affect mosquito microbiome composition as mosquito microbiome often comes from their aquatic larval habitats (Guégan et al. 2018b). Water pollution can alter the environmental microbiome (Chen et al. 2019), which could, as a result, modulate insect microbiomes. At the same time, quantitative differences in environmental resources and exposure to insecticides result in distinct bacterial microbiome composition in mosquitoes (Guégan et al. 2018b; Oke et al. 2022). Another major environmental change that is predicted to impact wildlife and vector-borne infectious diseases in the next decades is habitat loss (Sehgal 2010; Betts et al. 2019; Bernardo-Cravo et al. 2020). This stressor also induces significant reductions in microbiome biodiversity and modifies general microbial composition (Kiesewetter and Afkhami 2021). Global warming will increase drought in several regions of the world and, as a result, will constrain the suitable habitat for all vector life stages (i.e., larvae, pupae, and adults) (Caminade et al. 2019). Changes in landscape associated with anthropogenic global change may also disrupt mosquito microbiomes, and consequently the putative outcomes of microbial compositional changes on mosquito development, life history, and vectorial competence.
The most notorious environmental change associated with global change is increasing temperature. Vectors, their symbiotic microbes, and pathogen development all have their particular temperature optimum (Beier 1998; Mestre et al. 2020; Bellone and Failloux 2020). As a result, since vectors are ectothermic organisms and their microbes experience external temperature conditions, the forecasted environmental warming may impact vector microbiome composition due to differences in the tolerance or ability of distinct microbes to develop in higher temperatures. Indeed, remarkable differences in microbiome composition were found among oysters subject to distinct temperature conditions, demonstrating that changing temperature can drive symbiont composition in ectothermic organisms (Lokmer and Mathias Wegner 2015; Scanes et al. 2021). Moghadam et al. (2018) observed that flies subject to low temperature during their development were most cold-tolerant and harbored the highest abundance of Wolbachia, whereas flies that developed at high temperatures harbored the greatest abundance of Acetobacter and were the most resistant to heat. These results indicate that different microbiome composition in flies might confer tolerance to the distinct temperature range to which the insects were exposed (Moghadam et al. 2018), suggesting shifts in insect microbiomes could help them tolerate distinct environmental conditions. However, comparing the microbiomes of flies naturally found at different temperatures is only correlative evidence, and more direct experimental tests would help establish the link between the heat tolerance of vectors and shifts in their microbial communities. Increasing temperature also changes the environmental microbial composition (Bond-Lamberty et al. 2016; Onyango et al. 2020), modifying the microbial community to which vectors are exposed during their development and, ultimately, potentially driving the composition of vector microbiomes. Overall, global change has a clear potential to modify the vector’s microbiome. In the next section, we discuss the potential outcomes of changes in vector microbiomes for vector-borne pathogen transmission.
Microbiome effects on vector competence
Gut microbiome of mosquitoes can modulate their vector competence for several distinct pathogens (Weiss and Aksoy 2011; van den Hurk et al. 2012; Dennison et al. 2014; Johnson 2015; Feng et al. 2022). Distinct taxa of microbes can suppress or facilitate pathogen development. For instance, while most strains of Wolbachia prevent pathogenic human viruses from successfully replicating within their mosquito vectors, some strains enhance viral transmission (Johnson 2015). At the same time, the extent of pathogen suppression is neither constant nor predictable, depending on the particular strain of the symbiotic Wolbachia, the mode and titer of infection, and the particular strains of chikungunya and yellow fever viruses (van den Hurk et al. 2012). For this reason, many researchers have investigated the outcomes of multiple pathogen–Wolbachia strain combinations (van den Hurk et al. 2012; Johnson 2015; Pimentel et al. 2021; Caragata et al. 2021). Symbiotic microbes within the vector can suppress pathogens by competition or by remodeling the vector environment. For example, Wolbachia strains can compete with pathogens for host resources and/or increase reactive oxygen concentrations within their vectors (Johnson 2015; Manokaran et al. 2020). On the other hand, certain microbes (e.g., Enterobacteriaceae) can enhance pathogen transmission by weakening the vector’s immune defenses or disrupting transmission barriers (e.g., salivary glands) (Vaughan and Turell 1996; Gao et al. 2020).
Currently, most research focuses on microbes that have the potential to reduce transmission of pathogens to human populations. In this context, the best-studied group are Wolbachia bacteria not only due to their known effect of reducing viral replication in Aedes aegypti but also due to the bacteria’s ability to be vertically transmitted (Johnson 2015; Caragata et al. 2021). This ability induces not only a decrease in pathogen transmission within an infected mosquito, but it can also reduce vector abundance in nature due to reduced production of viable offspring through cytoplasmic incompatibility (i.e., incompatibility between infected and non-infected gametes) (Lambrechts et al. 2015). This happens because infected mosquitoes can only successfully generate offspring if they mate with other infected individuals carrying a similar or compatible Wolbachia strain (Caragata et al. 2021). Nonetheless, other microbes can also constrain pathogen transmission. For instance, Anopheles mosquitoes infected with a specific microsporidian (microsporidia MB) present significantly lower Plasmodium falciparum loads and reproductive success than mosquitoes not harboring the microsporidian (Herren et al. 2020). Additionally, the gut bacteria Pseudomonas alcaligenes seem to prevent Plasmodium infections in Anopheles by catabolizing tryptophan (Feng et al. 2022). However, distinct pathogens are unevenly affected by the same microbial taxa: for example, the bacterium Serratia marcescens can inhibit Plasmodium berghei development in Anopheles, but this same bacterium intensifies dengue virus transmission in A. aegypti Rockefeller strain (Wu et al. 2019; Gao et al. 2020). This fact illustrates how the effect of microbes on vector-borne pathogen control depends primarily on individual pathogen characteristics; consequently, different pathogen strains should react differently to microbiome shifts associated with climate change.
There are a few known examples of pathogen facilitation mediated by other microbes. For example, Trypanosoma-infected triatomine bugs carry higher loads of Enterobacterales and Petrimona sp. than non-infected individuals (Mann et al. 2020), indicating either that the presence of these bacteria facilitates infection or that Trypanosoma infection promotes increases in both bacterial populations. On the other hand, Eberhard et al. (2022) demonstrated that Trypanosoma parasites diminished the abundance of triatomine gut microbes (i.e., bacteria, fungi, and viruses) post-infection. These studies suggest that the composition of the triatomine gut microbiome can shape the transmission of Chagas disease. Furthermore, a prior Plasmodium infection seems to improve the development of a new malaria parasite in mosquitoes without constraining the first infection or mosquito survival (Pollitt et al. 2015). Therefore, Plasmodium development could have the potential to facilitate secondary malaria infections among mosquitoes. Similarly, Wang et al. (2021) showed that the commensal bacteria Asaia facilitate Plasmodium development by increasing the vector’s midgut pH. As discussed previously, global change has the potential to change vector microbiomes, indirectly modulating pathogen development and subsequent transmission. Moreover, the microbiome could also drive pathogen transmission through effects on the vectors’ reproductive success. For instance, Amblyomma ticks exposed to treatment against their microbiome presented reduced larval survival and fewer hatched eggs (Narasimhan and Fikrig 2015). However, our current understanding of symbiotic microbial tolerance to warming, their interaction with pathogens, and the possible outcome for vectorial competence is still very limited. For this reason, the effects of global change on vector microbiomes and microbiome-mediated pathogen suppression/facilitation must become an urgent topic of research.
Will global change affect vector competence?
Environmental modifications associated with global change could shape the composition and diversity of vector microbiomes, and also the latter’s effects in blocking the development of pathogens (Weiss and Aksoy 2011; Murdock et al. 2015). High temperatures (28 °C and above) improve the ability of Wolbachia to reduce Plasmodium oocyst burden, whereas at medium temperatures (24 °C), the bacteria enhanced the reproduction of Plasmodium oocyst within mosquitoes (Murdock et al. 2015). This fact suggests that human malaria prevention strategies using Wolbachia-infected mosquitoes might have negative or positive effects on human burden around the globe depending on local temperature conditions, by either increasing or preventing Plasmodium transmission. Nonetheless, global warming might expand the range of localities where this disease control strategy could be applied. Global warming can also affect the abundance and diversity of mosquito gut microbes (Mouton et al. 2006; Guégan et al. 2018a). Mosquitoes carrying higher abundance and diversity of microbes in their guts might be more resistant to pathogen infections and less competent vectors due to the immune modulation elicited by their symbiotic bacterial fauna (Francino 2016; Strand 2017; Raymann and Moran 2018). Therefore, global warming might lead to reduced vector competence in regions subject to high average temperatures (> 26–28 °C) (Murdock et al. 2012).
Interestingly, temperature directly modulates the mosquito immune system (Murdock et al. 2012). This immune modulation alters the mosquito’s immune response in different ways; lower temperatures (18 °C) were associated with the highest rates of melanization, phagocytosis, and expression of antimicrobial peptide defenses, suggesting that mosquitoes might become more susceptible to pathogens in the future (Murdock et al. 2012). Most importantly, vector competence might not correlate indefinitely with environmental temperature due to temperature constraints for the pathogen itself. Paaijmans et al. (2012) have shown that the incidence of malaria parasites in the salivary glands of mosquitoes is highest at 22 °C (50% of mosquitoes) while at 26 °C, only 10% of mosquitoes carry parasites in their salivary glands. This fact reinforces the idea that vector-borne pathogen transmission should not rise linearly following increases in global temperature. Global change ought to select for a heat tolerant microbiome in vectors and other ectothermic organisms (Onyango et al. 2020), which may (or not) result in a higher pathogen abundance. To confirm this prediction, new studies must focus on how important vector-borne pathogens (e.g., Plasmodium, Leishmania, and dengue and yellow fever viruses) react to increasing heat in order to predict which pathogens could emerge as future threats to human populations.
In summary, we argue that there are still several gaps in our current knowledge that limit any attempt to forecast the effects of global change on vector competence and, as a result, vector-borne disease transmission. Here, we argue the outcomes of global change for pathogen transmission must be treated individually since each specific pathogen strain will be distinctively impacted by global warming and the associated changes to its vector’s microbiomes. This is the outcome of the uneven interactions between temperature, pathogens, and particular microbes. For example, Serratia marcescens facilitates Plasmodium development while inhibiting dengue virus transmission. Likewise, Wolbachia can either enhance or suppress the establishment of malaria parasites within Anopheles mosquitoes depending on the external temperature. For this reason, experimental research should be conducted to clarify the effects of temperature and anthropogenic landscape changes on vector microbiome composition and its implication for multiple human and wildlife pathogens. Future research should also aim to uncover the mechanisms that confer to some microbes the ability of blocking or enhancing vector competence. This information should further be embodied by new eco-epidemiological models to enhance the predictive quality models of disease spread and outbreaks (Wells and Flynn 2022). Lastly, we conclude that our current understanding of the dynamics of global change and vector microbiome and competence is too deficient to enable predictions of how environmental and climate changes will impact the incidence and dynamics of vector-borne diseases.
Data availability
Not applicable.
References
Atkinson CT, LaPointe DA (2009) Introduced avian diseases, climate change, and the future of Hawaiian honeycreepers. J Avian Med Surg 23:53–63. https://doi.org/10.1647/2008-059.1
Beier JC (1998) Malaria parasite development in mosquitoes. Annu Rev Entomol 43:519–543. https://doi.org/10.1146/annurev.ento.43.1.519
Bellone R, Failloux A-B (2020) The role of temperature in shaping mosquito-borne viruses transmission. Front Microbiol 11:584846. https://doi.org/10.3389/fmicb.2020.584846
Bernardo-Cravo AP, Schmeller DS, Chatzinotas A et al (2020) Environmental factors and host microbiomes shape host–pathogen dynamics. Trends Parasitol 36:616–633. https://doi.org/10.1016/J.PT.2020.04.010
Betts MG, Wolf C, Pfeifer M et al (2019) Extinction filters mediate the global effects of habitat fragmentation on animals (1979). Science 366:1236–1239. https://doi.org/10.1126/science.aax9387
Bond-Lamberty B, Bolton H, Fansler S et al (2016) Soil respiration and bacterial structure and function after 17 years of a reciprocal soil transplant experiment. PLoS ONE 11:e0150599. https://doi.org/10.1371/journal.pone.0150599
Caminade C, McIntyre KM, Jones AE (2019) Impact of recent and future climate change on vector-borne diseases. Ann N Y Acad Sci 1436:157–173. https://doi.org/10.1111/nyas.13950
Cansado-Utrilla C, Zhao SY, McCall PJ et al (2021) The microbiome and mosquito vectorial capacity: rich potential for discovery and translation. Microbiome 9:111. https://doi.org/10.1186/s40168-021-01073-2
Caragata EP, Dutra HLC, Sucupira PHF et al (2021) Wolbachia as translational science: controlling mosquito-borne pathogens. Trends Parasitol 37:1050–1067. https://doi.org/10.1016/j.pt.2021.06.007
Chen J, McIlroy SE, Archana A et al (2019) A pollution gradient contributes to the taxonomic, functional, and resistome diversity of microbial communities in marine sediments. Microbiome 7:104. https://doi.org/10.1186/s40168-019-0714-6
Dennison NJ, Jupatanakul N, Dimopoulos G (2014) The mosquito microbiota influences vector competence for human pathogens. Curr Opin Insect Sci 3:6–13. https://doi.org/10.1016/j.cois.2014.07.004
Durack J, Lynch SV (2019) The gut microbiome: relationships with disease and opportunities for therapy. J Exp Med 216:20–40. https://doi.org/10.1084/jem.20180448
Eberhard FE, Klimpel S, Guarneri AA, Tobias NJ (2022) Exposure to Trypanosoma parasites induces changes in the microbiome of the Chagas disease vector Rhodnius prolixus. Microbiome 10(1):45. https://doi.org/10.1186/s40168-022-01240-z
EzSenwa VO, Gerardo NM, Inouye DW et al (2012) Animal behavior and the microbiome (1979). Science 338:198–199. https://doi.org/10.1126/science.1227412
Feng Y, Peng Y, Song X et al (2022) Anopheline mosquitoes are protected against parasite infection by tryptophan catabolism in gut microbiota. Nat Microbiol 7:707–715. https://doi.org/10.1038/s41564-022-01099-8
Francino MP (2016) Antibiotics and the human gut microbiome: dysbioses and accumulation of resistances. Front Microbiol 6. https://doi.org/10.3389/fmicb.2015.01543
Gao H, Cui C, Wang L et al (2020) Mosquito microbiota and implications for disease control. Trends Parasitol 36:98–111
Garamszegi LZ (2011) Climate change increases the risk of malaria in birds. Glob Chang Biol 17:1751–1759. https://doi.org/10.1111/j.1365-2486.2010.02346.x
Gray JS, Ogden NH (2021) Ticks, human Babesiosis and climate change. Pathogens 10:1430. https://doi.org/10.3390/pathogens10111430
Gressel J (2018) Microbiome facilitated pest resistance: potential problems and uses. Pest Manag Sci 74:511–515. https://doi.org/10.1002/ps.4777
Grice EA, Segre JA (2012) The human microbiome: our second genome. Annu Rev Genomics Hum Genet 13:151–170. https://doi.org/10.1146/annurev-genom-090711-163814
Guégan M, Minard G, Tran FH et al (2018a) Short-term impacts of anthropogenic stressors on Aedes albopictus mosquito vector microbiota. FEMS Microbiol Ecol 94:fiy188. https://doi.org/10.1093/femsec/fiy188
Guégan M, Zouache K, Démichel C et al (2018) The mosquito holobiont: fresh insight into mosquito-microbiota interactions. Microbiome 6:49
Herren JK, Mbaisi L, Mararo E et al (2020) A microsporidian impairs Plasmodium falciparum transmission in Anopheles arabiensis mosquitoes. Nat Commun 11:2187. https://doi.org/10.1038/s41467-020-16121-y
Huus KE, Ley RE (2021) Blowing hot and cold: body temperature and the microbiome. mSystems 6:e00707–21 https://doi.org/10.1128/msystems.00707-21
IPCC 2021 (2021) Climate change 2021: the physical science basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. United Kingdom and New York, NY, USA
Ippolito MM, Denny JE, Langelier C et al (2018) Malaria and the microbiome: a systematic review. Clin Infect Dis 67:1831–1839. https://doi.org/10.1093/cid/ciy374
Iturbe-Ormaetxe I, Walker T, O’Neill SL (2011) Wolbachia and the biological control of mosquito-borne disease. EMBO Rep 12:508–518. https://doi.org/10.1038/embor.2011.84
Johnson K (2015) The impact of Wolbachia on virus infection in mosquitoes. Viruses 7:5705–5717. https://doi.org/10.3390/v7112903
Kelly-Hope LA, Hemingway J, McKenzie FE (2009) Environmental factors associated with the malaria vectors Anopheles gambiae and Anopheles funestus in Kenya. Malar J 8:1–8. https://doi.org/10.1186/1475-2875-8-268
Kiesewetter KN, Afkhami ME (2021) Microbiome-mediated effects of habitat fragmentation on native plant performance. New Phytol 232:1823–1838. https://doi.org/10.1111/nph.17595
Lambrechts L, Ferguson NM, Harris E et al (2015) Assessing the epidemiological effect of Wolbachia for dengue control. Lancet Infect Dis 15:862–866. https://doi.org/10.1016/S1473-3099(15)00091-2
Li C, Gao Y, Chang N et al (2021) Risk assessment of Anopheles philippinensis and Anopheles nivipes (Diptera: Culicidae) invading China under climate change. Biology (basel) 10:998. https://doi.org/10.3390/biology10100998
Li H, Zhou X-Y, Yang X-R et al (2019) Spatial and seasonal variation of the airborne microbiome in a rapidly developing city of China. Sci Total Environ 665:61–68. https://doi.org/10.1016/j.scitotenv.2019.01.367
Loiseau C, Harrigan RJ, Bichet C et al (2013) Predictions of avian Plasmodium expansion under climate change. Sci Rep 3:1–6. https://doi.org/10.1038/srep01126
Lokmer A, Mathias Wegner K (2015) Hemolymph microbiome of Pacific oysters in response to temperature, temperature stress and infection. ISME J 9:670–682. https://doi.org/10.1038/ismej.2014.160
Mann AE, Mitchell EA, Zhang Y et al (2020) Comparison of the bacterial gut microbiome of North American Triatoma spp. with and without Trypanosoma cruzi. Front Microbiol 11:364. https://doi.org/10.3389/fmicb.2020.00364
Manokaran G, Flores HA, Dickson CT et al (2020) Modulation of acyl-carnitines, the broad mechanism behind Wolbachia-mediated inhibition of medically important flaviviruses in Aedes aegypti. Proc Natl Acad Sci 117:24475–24483. https://doi.org/10.1073/pnas.1914814117
Manor O, Dai CL, Kornilov SA et al (2020) Health and disease markers correlate with gut microbiome composition across thousands of people. Nat Commun 11:5206. https://doi.org/10.1038/s41467-020-18871-1
Marzal A (2012) Recent advances in studies on avian malarial parasites. In: Reference Info Is in Pdf. pp 135–158
McLoughlin IJ, Wright EM, Tagg JR et al (2021) Skin microbiome—the next frontier for probiotic intervention. Probiotics Antimicrob Proteins. https://doi.org/10.1007/s12602-021-09824-1
Mestre A, Poulin R, Hortal J (2020) A niche perspective on the range expansion of symbionts. Biol Rev 95:491–516. https://doi.org/10.1111/brv.12574
Moghadam NN, Thorshauge PM, Kristensen TN et al (2018) Strong responses of Drosophila melanogaster microbiota to developmental temperature. Fly (austin) 12:1–12. https://doi.org/10.1080/19336934.2017.1394558
Mouton L, Henri H, Bouletreau M, Vavre F (2006) Effect of temperature on Wolbachia density and impact on cytoplasmic incompatibility. Parasitology 132:49–56. https://doi.org/10.1017/S0031182005008723
Murdock CC, Blanford S, Hughes GL et al (2015) Temperature alters Plasmodium blocking by Wolbachia. Sci Rep 4:3932. https://doi.org/10.1038/srep03932
Murdock CC, Paaijmans KP, Cox-Foster D et al (2012) Rethinking vector immunology: the role of environmental temperature in shaping resistance. Nat Rev Microbiol 10:869–876
Narasimhan S, Fikrig E (2015) Tick microbiome: the force within. Trends Parasitol 31:315–323. https://doi.org/10.1016/j.pt.2015.03.010
Oke CE, Ingham VA, Walling CA, Reece SE (2022) Vector control: agents of selection on malaria parasites? Trends Parasitol. https://doi.org/10.1016/j.pt.2022.07.006
Onyango GM, Bialosuknia MS, Payne FA et al (2020) Increase in temperature enriches heat tolerant taxa in Aedes aegypti midguts. Sci Rep 10:19135. https://doi.org/10.1038/s41598-020-76188-x
Paaijmans KP, Blanford S, Chan BHK, Thomas MB (2012) Warmer temperatures reduce the vectorial capacity of malaria mosquitoes. Biol Lett 8:465–468. https://doi.org/10.1098/rsbl.2011.1075
Pérez-Rodríguez A, de la Hera I, Fernández-González S, Pérez-Tris J (2014) Global warming will reshuffle the areas of high prevalence and richness of three genera of avian blood parasites. Glob Chang Biol 20:2406–2416. https://doi.org/10.1111/gcb.12542
Perkins SL (2001) Phylogeography of Caribbean lizard malaria: tracing the history of vector-borne parasites. J Evol Biol 14:34–45. https://doi.org/10.1046/j.1420-9101.2001.00261.x
Pimentel AC, Cesar CS, Martins M, Cogni R (2021) The antiviral effects of the symbiont bacteria Wolbachia in insects. Front Immunol 11:626329. https://doi.org/10.3389/fimmu.2020.626329
Pollitt LC, Bram JT, Blanford S et al (2015) Existing infection facilitates establishment and density of malaria parasites in their mosquito vector. PLoS Pathog 11:e1005003. https://doi.org/10.1371/journal.ppat.1005003
Raymann K, Moran NA (2018) The role of the gut microbiome in health and disease of adult honey bee workers. Curr Opin Insect Sci 26:97–104. https://doi.org/10.1016/J.COIS.2018.02.012
Rocklöv J, Dubrow R (2020) Climate change: an enduring challenge for vector-borne disease prevention and control. Nat Immunol 21:479–483. https://doi.org/10.1038/s41590-020-0648-y
Salem I, Ramser A, Isham N, Ghannoum MA (2018) The gut microbiome as a major regulator of the gut-skin axis. Front Microbiol 9:459. https://doi.org/10.3389/fmicb.2018.01459
Scanes E, Parker LM, Seymour JR et al (2021) Climate change alters the haemolymph microbiome of oysters. Mar Pollut Bull 164:111991. https://doi.org/10.1016/j.marpolbul.2021.111991
Sehgal RNM (2010) Deforestation and avian infectious diseases. J Exp Biol 213:955–960. https://doi.org/10.1242/jeb.037663
Strand MR (2017) The gut microbiota of mosquitoes. In: Arthropod vector: controller of disease transmission, Volume 1. Elsevier, pp 185–199
Tomé B, Pereira A, Harris DJ et al (2019) A paradise for parasites? Seven new haemogregarine species infecting lizards from the Canary Islands. Parasitology 146:728–739. https://doi.org/10.1017/S0031182018002160
van den Hurk AF, Hall-Mendelin S, Pyke AT et al (2012) Impact of Wolbachia on infection with chikungunya and yellow fever viruses in the mosquito vector Aedes aegypti. PLoS Negl Trop Dis 6:e1892. https://doi.org/10.1371/journal.pntd.0001892
Vaughan JA, Turell MJ (1996) Facilitation of Rift Valley fever virus transmission by Plasmodium berghei sporozoites in Anopheles stephensi mosquitoes. Am J Trop Med Hyg 55:407–409. https://doi.org/10.4269/ajtmh.1996.55.407
Videvall E, Marzal A, Magallanes S et al (2021) The uropygial gland microbiome of house sparrows with malaria infection. J Avian Biol 52:e02686. https://doi.org/10.1111/jav.02686
Wang M, An Y, Gao L et al (2021) Glucose-mediated proliferation of a gut commensal bacterium promotes Plasmodium infection by increasing mosquito midgut pH. Cell Rep 35:108992. https://doi.org/10.1016/j.celrep.2021.108992
Weiss B, Aksoy S (2011) Microbiome influences on insect host vector competence. Trends Parasitol 27:514–522
Wells K, Flynn R (2022) Managing host-parasite interactions in humans and wildlife in times of global change. Parasitol Res. https://doi.org/10.1007/s00436-022-07649-7
World Health Organization (2014) World health statistics 2014. World Health Organization. https://apps.who.int/iris/handle/10665/112738
World Health Organization (2020) World Malaria Report 2020. World Health Organization. https://www.who.int/news-room/fact-sheets/detail/malaria
Wu P, Sun P, Nie K et al (2019) A gut commensal bacterium promotes mosquito permissiveness to arboviruses. Cell Host Microbe 25:101-112.e5. https://doi.org/10.1016/j.chom.2018.11.004
Zeevi D, Korem T, Godneva A et al (2019) Structural variation in the gut microbiome associates with host health. Nature 568:43–48. https://doi.org/10.1038/s41586-019-1065-y
Acknowledgements
We are grateful to Prof. Érika Martins Braga for her helpful comments on an earlier version of this manuscript. We also thank the insightful comments of three anonymous reviewers and the editor, Dr Xing-Quan Zhu.
Funding
Daniela de Angeli Dutra was supported by a doctoral scholarship from the University of Otago, New Zealand. Priscila Madi Salloum is supported by a grant from the Marsden Fund to Robert Poulin and Dr. Fátima Jorge.
Author information
Authors and Affiliations
Contributions
DdeAD and RP conceived the idea and designed the study. DdeAD wrote the manuscript with input from all other authors. All authors contributed critically to the drafts.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
All authors consented to participate of this research.
Consent for publication
All authors gave final approval for publication.
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: Xing-Quan Zhu
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Angeli Dutra, D., Salloum, P.M. & Poulin, R. Vector microbiome: will global climate change affect vector competence and pathogen transmission?. Parasitol Res 122, 11–17 (2023). https://doi.org/10.1007/s00436-022-07734-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-022-07734-x