Abstract
Encephalitozoon cuniculi is an obligate intracellular microsporidian parasite that infects a wide range of mammalian hosts. The present study investigated the prevalence of E. cuniculi in different animal hosts from different provinces of Egypt (Alexandria, Behera, and Assuit) using serological (IFAT and ELISA) and molecular (PCR) assays. A total of 324 serum and 274 urine samples were collected from seven different species of animals (cattle, buffaloes, sheep, goat, rabbit, dog, and rat). The results of serological examination confirmed the occurrence of antibodies against E. cuniculi in 38.9 % (126 out of 324) of the examined animals. The significant (P < 0.01) highest positivity was observed in goats (67 %) followed by buffaloes, rabbits, dogs, rat, and cattle (46.42, 41, 40, 36.2, and 28.1 %, respectively), while the least was recorded in sheep (9 %). Behera province showed the highest (P < 0.01) infection rate (40.68 %) followed by Alexandria and Assuit (39.2 and 22.73 %, respectively). The infection rate was significantly higher (P < 0.01) in females (45.34 %) than that in males (30.47 %). Positive cases were observed in all age categories. The highest infection rate (64.66 %) was recorded in the age group 1–5 years and the least was recorded in the age group <1 year (34.85 %).On the other hand, only five positive out of 274 urine samples (1.82 %) were detected by PCR. Our study provides a wide database on prevalence and epidemiology of an ignored parasite (E. cuniculi) for the first time in Egypt.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Encephalitozoon cuniculi is an obligatory intracellular microsporidian parasite that can infect a wide range of mammals, including rodents, rabbits, horses, carnivores, and humans, in which the organism is known as an opportunistic pathogen of immunocompromised individuals (Künzel and Joachim 2010). E. cuniculi replicates within infected host cells, causing rupture of the cells and release of spores into the bloodstream. Infectious spores are excreted in the urine or feces of infected animals (Kasickova et al. 2009). E cuniculi infects the eyes, kidneys, and brain of rabbits, causing clinical signs such as cataracts, head tilt, paralysis, and death. In dogs, the disease is only seen in puppies. Clinical signs of E. cuniculi infection in pups are almost entirely neurological, and include blindness, ataxia, and seizures (Jordan 2005). The disease occurs as a result of horizontal transmission in the majority of animal hosts; however, vertical transmission is also known to occur in some host species. A characteristic feature of the infection induced by E. cuniculi in susceptible hosts is usually its chronic latent course, with higher mortality in young and older individuals. Latently infected hosts, given immunosuppression factors, will develop an acute, clinically apparent disease (Schmidt and Shadduck 1983).
Seroepidemiological evidence suggests that rabbits represent the natural host of E. cuniculi strain I (Mathis et al. 2005), serving as a source of infection for other animals and humans (Dipineto et al. 2008). E. cuniculi was also considered one of the infectious agents responsible for infertility and abortions of goats, and these animals may be potential sources of infection for people, especially for breeders attending these animals (Cislakova et al. 2001). In cattle, E. cuniculi was first described by Reetz (1995). The canine encephalitoonosis was reported in various parts of the world (Akerstedt 2003; Lindsay et al. 2009). The infection in dogs would present a zoonotic problem due to close dog-man relationship (Akerstedt 2002). E. cuniculi infection has been also reported in rodents in the Czech Republic, Iceland, and Switzerland (Muller-Doblies et al. 2002; Sak et al. 2011).
Diagnosis of E. cuniculi is difficult in vivo because many animals are subclinically infected. Previous studies showed high seroprevalence in rabbits, mostly without clinical manifestations (Harcourt-Brown and Holloway 2003; Dipineto et al. 2008). At necropsy, parasites may be identified by histological examination, mainly of the brain and kidney. In live animals, a number of methods have been applied to detect E. cuniculi infection. There are methods for detecting spores in urine or feces. The methods for detection include specific stains, for example, the chromotrope stain (Weber et al. 1992) and isolation of spores (Cox et al. 1972). These methods are specific, but not practically applicable as diagnostic methods in veterinary medicine (Pye and Cox 1977). Serology is the most important diagnostic tool for diagnosis of E. cuniculi infection in the living animal. Various diagnostic tests have been developed in order to detect antibodies to E. cuniculi. Indirect fluorescent antibody technique (IFAT) and enzyme-linked immunosorbent assay (ELISA) are the most common tests and correlate well with each other (Boot et al. 2000).
Molecular methods such as polymerase chain reaction (PCR) have the potential to be simple, rapid, and highly sensitive, but have so far been underdeveloped and underused in veterinary diagnostic practices. Previous attempts to optimize an assay for diagnosis of encephalitozoonosis have been insufficient, and it is not known if this lack of reported sensitivity of PCR is due to intermittent shedding of spores or due to an assay design that is unable to detect low numbers of spores that may be shed in the urine (Reabel 2012). Many factors as microsporidia genotype, host species, age, immune status, and mode of infection may influence the outcome of infection (Wasson and peper 2000).
To date, few studies have been carried out in order to assess the prevalence of E. cuniculi infection in farm animals throughout the world. In Egypt, information on the occurrence and epidemiology of E.cuniculi is still scarce. The aim of the present study was to evaluate the prevalence of E. cuniculi infection in seven different animal hosts (cattle, buffaloes, sheep, goat, rabbit, dog, and rat) in Egypt using serological and molecular assays and to assess association between seroprevalence of E. cuniculi and species, age, and sex of affected animals. To the best of our knowledge, this is the first report to describe the prevalence of E. cuniculi in these animals in Egypt.
Material and methods
Collection of samples
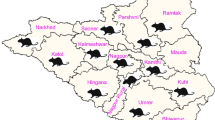
A total number of 324 blood and 274 urine samples were collected from animals of different host species (cattle, buffalo, sheep, goat, dogs, rat, and rabbit) (Table 1) from veterinary medical convoys, veterinary clinics, faculty hospital, and farms from different provinces of Egypt (Alexandria, Behera, and Assuit). The samples were collected in the period from November 2012 to March 2014. For each sampled animal, sex and age were recorded. With respect to sex, there were 105 males and 161 females used in the study. These animals were classified into three age groups: young (<1 year old), adults (1–5 years old), and old (>5 years old). The sample number of each sex and age group was shown in Table 2. All animals were apparently healthy. Blood samples were collected and centrifuged at 2000 rpm for 5 min and then serum was kept at −20 °C until used for serological tests. Collected urine samples were centrifuged at 12,000 rpm for 10 min and then supernatants were decanted, while pellets were kept at −20 °C for molecular tests.
E. cuniculi spores
Spores of E. cuniculi (rabbit strain isolate) kindly supplied by Dr. P. Deplazes (Institute of Parasitology, University of Zurich, Switzerland) were used as an antigen in serological tests.
Reference sera
A negative reference serum was kindly supplied by Lisa Bowers (Division of Microbiology, Tulane National Primate Research Center, Covington, LA, USA). A positive reference serum was obtained by experimental infection of rabbits with E. cuniculi spores. Sera were collected after 3 weeks postinfection (PI), stored at −20 °C, and used as the positive control.
Serological assays
All collected sera were tested using IFAT except the dog sera tested by ELISA.
Indirect immunofluorescent test
Sera of different animal hosts (cattle, buffalo, sheep, goat, rabbit, and rat) were investigated for antibodies of E.cuniculi using IFAT by the method described by Akerstedt (2002) and modified by Soliman and Selim (2005). Briefly, slides were washed with absolute alcohol and then kept till air dried. Using black nail varnish, eight small circles were made on each slide; each circle represents a well. Ten microliters of spore suspension (containing 80,000 spores of E.cuniculi) was added to each well on each slide, which were incubated at 37 °C for 30 min. Ten microliters of methanol was added to each well for antigen fixation, and then slides were kept for another 10 min at 37 °C. Slides were then rinsed with distilled water and kept till air dried. Serum was diluted 1:40 in PBS, and 20 μl of diluted serum was added to each well and then slides were incubated at 37 °C for 30 min. Slides were washed three times in PBS and then distilled water, and the slides were kept until air dried. Ten microliters of diluted fluorescein Isothiocyanate conjugated antibody (FITC) suitable for each host (Sigma, St. Louis, MO, USA) was added to each well. Slides were then incubated at 37 °C for 30 min. Slides were again rinsed three times in PBS and distilled water, then kept till air dried. Ten microliters of glycerin/PBS (1:9) was added to each well and kept till air dried. Slides were covered with a cover slip and examined at ×40 with an Olympus Immunofluorescent microscope. Positive and negative control sera were used as controls on each slide.
Enzyme-linked immunosorbent assay
Dog sera were examined by indirect ELISA as described by Akerstedt (2002). Briefly, Polystyrene microtitre plates (Immunoplate Maxisorb; Nunc, Roskilde, Denmark) were coated with the soluble antigen at a concentration of 2 μg/ml. Subsequently, the plates were incubated for 3 h at 37 °C, then washed with 200 μl PBS/Tween 20, and then treated with the blocking solution (3 % bovine serum albumin (BSA) with 0.05 % Tween 20). A total of 50 μl each of tested dog serum, negative control sera, and positive control sera were added to each well and then incubated at 37 °C for 1 h. After incubation, the plates were washed three times with 200 μl PBS/0.05 % Tween 20. After washing, horseradish peroxidase (HRP) conjugated goat poly anti-dog IgG (KOMABIOTECH) was diluted at 1:1000 in PBS-T and added to the plates (100 μl/well). Finally, the plates were washed three times with PBS/0.05 % Tween 20, and the enzyme activity of bound peroxidase was revealed by adding 100 μl of ortho-phenylenediamine substrate (OPD) (Laboratories Inc., San Diego, CA, USA) to each well. After incubation in darkness (45 min), the enzymatic color reaction was stopped by adding 100 μl of 1 M phosphoric acid to each well, and the optical density was read at 450 nm using a microplate reader (Corona Electrical, Japan). Cutoff value was calculated according to John (2009); Cutoff = Mean of negatives + (3 × SD of negatives).
Molecular assay
Extraction of DNA
DNA was extracted from urine samples of different animals according to the method described by Ghatak et al. (2013).
PCR
The PCR was performed using E. cuniculi-specific primers that amplified a 549-bp fragment of the small subunit ribosomal RNA (ssrRNA) gene (Csokai et al. 2009). The forward and reverse primers were (Ence F: 5′-ATGAGAAGTGATGTGTGTGCG-and (Ence R: 5′-TGCCATGCACTCACAGGCATC-3′). The PCR was performed using the following conditions: after initial denaturation of the DNA at 95 °C for 3 min, 35 cycles were run 95 °C for 30 s, 55 °C for 30 s, and 72 °C for 1 min with a 10-min 72 °C extension after the 35 cycles. Agarose gel electrophoresis was performed on PCR products, and the resulting bands were visualized using ethidium bromide and UV light. One positive and another negative control reaction without template DNA were also performed. The size of the PCR products was calculated using a 3000-bp ladder.
Statistical analysis
The data obtained were analyzed by the chi-square test to assess association between seroprevalence of E. cuniculi among different animal hosts and different parameters (species, age, and sex) by the aid of SAS software (SAS 2004).
Results
Seroprevalence of E.cuniculi in different animal hosts
The results of serological examination confirmed the occurrence of antibodies against E. cuniculi in all examined animal hosts in Egypt. E. cuniculi infection was detected in 126 out of 324 (38.9 %) of the examined animals. The infection rate of E. cuniculi was significantly different between different animal host species. The highest positivity was observed in goats (67 %) followed by buffaloes, rabbits, dogs, rat, and cattle (46.42, 41, 40, 36.2, and 28.1 %, respectively) while the least positivity was recorded in sheep (9 %) (Table 3). Seropositive animals were distributed in all of the provinces from which animals were sampled for this study. Behera province showed the highest percentage (40.68 %) followed by Alexandria and Assuit provinces (39.2 and 22.73 %, respectively) (Table 4). Comparison of the infection rate by sex showed that the highest infection rate was recorded in females 45.34 % (73 out of 161), then in males 30.47 % (32 out of 105) (Table 5). Positive cases were observed in all age categories. The highest infection rate (64.66 %) was recorded in the age group 1–5 years, followed by the age group >5 years (38.64 %), and the least was recorded in the age group <1 year (34.85 %) (Table 6). From statistical analysis, animal gender, age, and species had a significant effect on E. cuniculi infection.
Seroprevalence of E. cuniculi in each animal host according to sex and age
Table 7 demonstrates the rate of infection in each animal host species and its association with sex and age. The rate of infection in cattle was 28.12 % and was significantly higher (25 %) in females than in males (3.1 %). The highest infection rate was observed at ages more than 5 years (17.18 %) followed by ages less than 1 year (6.25 %) and finally the ages ranged from 1 to 5 years (4.69 %). The percentage of positivity in buffaloes reached 46.42 % that were 39.28 % in females and 7.14 % in males. In buffaloes, E. cuniculi infection was the highest among ages ranged from 1 to 5 years (25 %) and minimum in ages >5 years (10.71 %). Regarding goats, the rate of infection reached 67.74 %, and all tested and infected animals were females. Moreover, the highest infection rate was observed in ages ranged from 1 to 5 years (64.52 %) followed by ages less than 1 year (3.22 %). In sheep, the percentage of positive samples reached 9 %. All infected animals were females. The age of all infected animals ranged from 1 to 5 years. In case of rabbits, the percentage of positivity reached 41 % and its incidence in males (30.10 %) was higher than in females (10.90 %). In dogs, positivity reached 40 %, and it was equal in both males and females (20 %). The highest incidence was observed in ages less than 1 year (25 %) followed by ages more than 1 year (15 %).
The results in Table 8 showed that a total number of 58 rats (28 wild and 30 laboratory rats) were examined for E. cuniculi. From which, 21 rats were proven positively infected with E. cuniculi (36.20 %). Of these, 32.75 % were of wild rats while 3.45 % were of laboratory rats.
Molecular assays (PCR)
From urine samples collected from different animal hosts (n = 274), only five positive samples were detected with a percentage of 1.82 % including two samples from cattle with a percentage of 5.7 %, two from buffaloes with a percentage of 4.44 %, and only one from rabbits with a percentage of 2.85 %.
Discussion
In the present study, E. cuniculi was detected in different animal hosts including cattle, buffalo, sheep, goat, rabbit, dogs, and rats using serological and molecular assays. The serological survey showed a high prevalence of E. cuniculi infection (38.9 %) suggesting that this parasite may be endemic in farm animals, dogs, rabbits, and rats in Egypt. This high rate of infection may be explained by contamination of food or water sources with E. cuniculi spores as the main route of infection is through the ingestion of spores that are shed in the urine of infected animals (Wasson and Peper 2000).
Behera province showed the highest percentage (40.68 %) of infection rate followed by Alexandria and Assuit provinces (39.2 and 22.73 %, respectively); this might be due to environmental factors and husbandry. On the other hand, the highest infection rate was recorded in females 45.34 % (73 out of 161) and then in males 30.47 % (32 out of 105). Increased susceptibility in females might be returned to the suppression of immune functions associated with reproductive stress (Schuurs and Verheul 1989). Regarding age, the highest infection rate (64.66 %) was recorded in the age group 1–5 years, and the least infection rate was in the age group <1 year (27.11 %); this might be returned to age-dependent exposure to the parasite and/or predisposition to infection (Wilson et al. 2007).
In the present study, IFAT was used for detection of E. cuniculi antibodies in different animal hosts. Our findings were supported by Pakes et al. (1984) who showed that IFAT is very sensitive when compared to histological detection of E. cuniculi, intradermal test, or complement fixation test. Our results were also supported by Didier et al. (1998) who proved that IFAT was the gold standard for serological diagnosis of microsporidia infection. ELISA test was also used for detection of E. cuniculi antibodies in dogs in the present study. This finding was supported by the recommendation of Boot et al. (2000) who found that serology is the most important diagnostic tool for diagnosis of E. cuniculi infection in the living animals and that indirect fluorescent antibody technique (IFAT) and enzyme-linked immunosorbent assay (ELISA) are the most common tests and correlate well with each other.
Our results revealed that the prevalence of E. cuniculi infection in rabbits was 41 %. These results agreed with those of Meyer-Breckwoldt (1996) who found 84 out of 200 rabbits (42 %) in Germany had antibodies to E. cuniculi. These results agreed also with those of Ewringmann and Gobel (1999) who tested 227 pet rabbits brought to the Berlin Veterinary School Small Animal Clinic for E. cuniculi. Of these rabbits, 125 (45 %) had a positive antibody titer. This high prevalence might be due to bad hygiene, overcrowding, and urine contamination between rabbits (Hilali et al. 1991). In Turkey, Eroksuz et al. (1999) examined 150 rabbits from 15 different colonies both serologically (CIA) and pathomorphologically; seropositivity was 65.33 % which disagreed with our results. This might be due to different nonspecific diagnostic tests (Hunt et al. 1972). Seroprevalence investigations in Egypt have been published in our previous paper (Ashmawy et al. 2011) who revealed that E. cuniculi infection was detected in 15 % of tested rabbits. These different results from the present study might be attributed to the larger number of examined animals by Ashmawy et al. (2011) where they examined 240 rabbits. This is also in agreement with the conclusion of Malik et al. (1990) who stated that the prevalence of infection may vary strongly in the same country from one locality to another due to differences in certain ecological factors and the breeding system in these areas. On the other hand, Hilali et al. (1991) reported that 66–68 % of apparently healthy rabbits collected from different areas in Cairo province, Egypt, were seropositive for E. cuniculi which disagreed with our results. This might be returned to the different testing methods, where they used CIA to assess the seroprevalence of E. cuniculi which is known to be a nonspecific test (Akerstedt and Kapel 2003).
Concerning E. cuniculi infection in rats, the seroprevalence was 36.20 % (21 out of 58). These results accorded with those of Sak et al. (2011) who detected microsporidia in 34 % of M. m. musculus and 33 % of M. m. domesticus examined specimens. Our results disaccorded with those of Hersteinsson et al. (1993) in Iceland where infection rate was 4 and 9 % of Apodemus sylvaticus and Mus musculus animals, respectively. This may be due to different climatic and geographical conditions. Wild rodents spread infection through the release of spores into the environment (Sak et al. 2011). The spores of E. cuniculi survive for several months in the environment and are resistant to factors such as moisture and dryness (Mathis et al. 2005). Rodents are frequently found in agricultural areas and have many opportunities to come into contact with other wild animals, livestock, and humans. Thus, they are potential reservoirs of parasites (Murakoshi et al. 2013).
In dogs, infection rate of E. cuniculi was 40 %, which agreed with Lindsay et al. (2009) in Brazil who found that infection rate was 35.3 %. In Florida, Cray and Rivas (2013) found that infection rate was 21.6 % and Sasaki et al. (2011). This lower incidence might be due to better management and/or the examined animals might be recently infected and with very low IgG titers which cannot be detected (Bywater and Kellett 1978).
Infection rate in cattle was 28.12 %, in buffalo was 46.42, in goat was 67.74 %, and in sheep was 9 %. Our results agreed with those of Malcekova et al. (2010) who found that infection rate in sheep was 9 %. While disagreeing with those of Halánová et al. (1999) who found that 20 out of 55 cattle samples (36.3 %) in Slovakia were serologically positive. This might be due to different geographical, climatic conditions and breeding system.
Regarding PCR results, only five positive out of 274 urine samples (1.82 %) were detected. This result agreed with those of Hůrková and Modrý (2006) who found DNA of E. cuniculi in 3.28 % (2/61) martens and in one examined European otter (Lutra lutra). Our results also agreed with those of Jeklova et al. (2010) who found that, following experimental oral inoculation of high doses of E. cuniculi spores (107 spores), rabbit urine was analyzed by real-time PCR and DNA corresponding to low concentrations of spores (101 to 103 spores per ml) was detected consistently up to 18 weeks postinfection. When repeated with a low dosage of spores (103 spores), which is most likely to represent a natural infection, no DNA was detected. Reabel (2012) suggested that false negative PCR results are not because of intermittent shedding but occur due to low numbers of spores being shed that are below the detection threshold of the PCR assay. It is possible that domestic rabbits maintain a low level of infection because they are continuously exposed to low numbers of spores in the environment, which could be responsible for the low number of spores being shed in the urine. This suggestion confirmed by a study of Furuya (2009) who analyzed rabbit urine samples, spores were identified using immunofluorescent staining in three samples; however, only one of these samples was positive after PCR analysis. This could provide further evidence that current PCR assays cannot detect DNA corresponding to low numbers of spores shed in urine. This data could also indicate that the low dose of spores was not sufficient to overwhelm the host immune response allowing multiplication of organisms (Reabel 2012). In addition to that, experimental infection studies have reported that the shedding of spores in urine begins about 4 to 6 weeks after the initial infection occurs. Spores are shed consistently for approximately 1 to 3 months after the initial infection and intermittently and/or in smaller numbers (Jeklova et al. 2010). Moreover, urine has been found to be a mostly difficult specimen for PCR-based amplification due to the presence of many possible inhibitors (Yamaguchi-Shinozaki et al. 1992). For example, concentrations of 20 mM urea and above are inhibitory for PCR (Khan et al. 1991). Even though Ghosh and Weiss (2009) stated that molecular diagnostic methods involving PCR have the potential to be more rapid and sensitive than microscopy, serology, or cell culture, however, have yet to be utilized extensively in veterinary medicine.
It is obvious that serological tests revealed more reliable data about prevalence of E. cuniculi in different animal hosts than molecular tests; this is parallel to the data of Jordan et al. (2006) who showed that serology is the most important diagnostic tool for diagnosis of E. cuniculi infection in the living animals.
In conclusion, the serological positivity (38.9 %) recorded in our study within the chosen group of animals and in 100 % of the examined provinces indicates the wide distribution of E. cuniculi in Egypt and confirms that these animals may be reservoirs of E. cuniculi and thus potential sources for other animals and human. Therefore, we highlight the need to carry out further epidemiological and experimental work on E. cuniculi and developing preventive strategies for eradication of the infection. It is also advisable to accomplish screening serological checks for E. cuniculi infection in both symptomatic and asymptomatic animals.
References
Akerstedt J (2002) An indirect ELISA for detection of Encephalitozoon cuniculi infection in farmed blue foxes (Alopex lagopus). Acta Vet Scand 43:211–220
Akerstedt J (2003) Serological investigation of canine encephalitozoonosis in Norway. Parasitol Res 89:49–52
Akerstedt J, Kapel CM (2003) Survey for Encephalitozoon cuniculi in arctic foxes (Alopex lagopus) in Greenland. J Wildl Dis 39:228–232
Ashmawy KI, Abuakkada SS, Awad AM (2011) Seroprevalence of antibodies to Encephalitozoon cuniculi and Toxoplasma gondii in farmed domestic rabbits in Egypt. Zoonosis Public Health 58(5):357–364
Boot R, Hansen C, Nozari N, Thuis H (2000) Comparison of assays for antibodies to Encephalitozoon cuniculi in rabbits. Lab Anim 34(3):281–289
Bywater JEC, Kellett BS (1978) The eradication of Encephalitozoon cuniculi from a specific pathogen-free rabbit colony. Lab Anim Sci 28:402–404
Cislakova L, Literak I, Balent P, Hipikova V, Levkutova M, Travnicek M, Novotna A (2001) Prevalence of antibodies to Encephalitozoon cuniculi (Microsporida) in Angora goat—a potential risk of infection for breeders. Ann Agric Environ Med 8:289–291
Cox JC, Walden NB, Nairn RC (1972) Presumptive diagnosis of Nosema cuniculi in rabbits by immunofluorescence. Res Vet Sci 13:595–597
Cray C, Rivas Y (2013) Seroprevalence of Encephalitozoon cuniculi in dogs in the United States. J Parasitol 99(1):153–154
Csokai J, Joachim A, Gruber A, Tichy A, Pakozd A (2009) Diagnostic markers for encephalitozoonosis in pet rabbits. Vet Parasitol 163(1–2):18–26
Didier PJ, Didier ES, Snowden KS, Shadduck JA (1998) Encephalitozoonosis. In: Greene C (ed) Infectious diseases of the dog and cat. Saunders, Philadelphia, pp 465–470
Dipineto L, Rinaldi L, Santaniello A, Sensale M, Cuomo A, Calabria M, Menna LF, Fioretti A (2008) Serological survey for antibodies to Encephalitozoon cuniculi in pet rabbits in Italy. Zoonoses Public Health 55:173–175
Eroksuz Y, Eroksuz H, Ozer H, Cevik A, Unver O (1999) A survey of Encephalitozoon cuniculi infection in rabbit colonies in Elazig, Turkey: pathomorphologic and serologic (carbonimmunoassay test) studies. Isr J Vet Med 54:73–77
Ewringmann A, Göbel T (1999) Untersuchungen zur Klinik und Therapie der Encephalitozoonose beim Heimtierkaninchen. Kleintierpraxis 44:357–372
Furuya K (2009) Spore-forming microsporidian encephalitozoon: current understanding of infection and prevention in Japan. Jpn J Infect Dis 62:413–422
Ghatak S, Muthukumaran RB, Nachimuthu SK (2013) A simple method of genomic DNA extraction from human samples for PCR-RFLP analysis. J Biomol Tech 24(4):224–231
Ghosh K, Weiss LM (2009) Molecular diagnostic tests for microsporidia. Interdiscip Perspect Infect Dis, Article ID 926521: 1–13
Halánová M, Letková V, Macák V, Stefkovic M, Halán M (1999) The first finding of antibodies to Encephalitozoon cuniculi in cows in Slovakia. Vet Parasitol 82(2):167–171
Harcourt-Brown FM, Holloway HK (2003) Encephalitozoon cuniculi in pet rabbits. Vet Rec 152:427–431
Hersteinsson P, Gunnarsson ES, Hjartardottir S, Skirnisson K (1993) Prevalence of Encephalitozoon cuniculi antibodies in terrestrial mammals in Iceland, 1986 to 1989. J Wildlife Dis 29:341–344
Hilali M, Nassar AM, Ramadan EI (1991) Detection of encephalitozoonosis and toxoplasmosis among rabbits by carbon immunoassay. Vet Med J Giza 39:129–135
Hunt RD, King NW, Foster HL (1972) Encephalitozoonosis, evidence of verticaltransmission. J Infec Dis 126:212–214
Hůrková L, Modrý D (2006) PCR detection of Neospora caninum, Toxoplasma gondii andEncephalitozoon cuniculi in brains of wild carnivores. Vet Parasitol 137(1–2):150–154
Jeklova E, Leva L, Kovarcik K, Matiasovic J, Kummer V, Maskova J, Skoric M, Faldyna M (2010) Experimental oral and ocular Encephalitozoon cuniculi infection in rabbits. Parasitol 137(12):1749–1757
John RC (2009) The ELISA guidebook, 2nd ed Series: methods in molecular biology, pp 516–566
Jordan CN (2005) Encephalitozoon cuniculi: diagnostic test and methods of inactivation. MSc thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA
Jordan CN, Zajac AM, Lindsay DS (2006) Encephalitozoon cuniculi infection in rabbits. Compend Contin Educ Vet 28:108–116
Kasickova D, Sak B, Kvac M, Ditrich O (2009) Sources of potentially infectious human microsporidia: molecular characterization of microsporidia isolates from exotic birds in the Czech Republic, prevalence study and importance of birds in epidemiology of the human microsporidial infections. Vet Parasitol 165:125–130
Khan G, Kangro HO, Coates PJ, Heath RB (1991) Inhibitory effects of urine on the polymerase chain reaction for cytomegalovirus DNA. J Clinic Pathol 44:360–365
Künzel F, Joachim A (2010) Encephalitozoonosis in rabbits. Parasitol Res 106:299–309
Lindsay DS, Goodwin DG, Zajac AM, Corte´s-Vecino JA, Gennari SM, Rosypal AC, Dubey JP (2009) Serological survey for antibodies to Encephalitozoon cuniculi in ownerless dogs from urban areas of Brazil and Colombia. J Parasitol 95(3):760–763
Malcekova B, Halanova M, Sulinova Z, Molnár L, Ravaszova P, Adam J, Halán M, Valocky I, Baranovic M (2010) Seroprevalence of antibodies to Encephalitozoon cuniculi and Encephalitozoon intestinalis in humans and animals. ResVet Sci 89(3):358–361
Malik MA, Dressen DW, Cruz A (1990) Toxoplasmosis in sheep in North Eastern United States. J Am Vet Med Assoc 196:263–265
Mathis A, Weber R, Deplazes P (2005) Zoonotic potential of the microsporidia. Clin Microbiol Rev 18:423–445
Meyer-Breckwoldt A (1996) Epidemiological and clinical studies on encephalitozoonosis in dwarf rabbits. Thesis, Tierarztliche. Hochschule: Hannover, Germany
Muller-Doblies UU, Herzog K, Tanner I, Mathis A, Deplazes P (2002) First isolation and characterization of Encephalitozoon cuniculi from a free-ranging rat (Rattus norvegicus). Vet Parasitol 107(279):285
Murakoshi F, Fukuda Y, Matsubara R, Kato Y, Sato R, Sasaki T, Tada C, Nakai Y (2013) Detection and genotyping of Cryptosporidium spp. in large Japanese field mice, Apodemus speciosus. Vet Parasitol 196(1–2):184–188
Pakes SP, Shadduck JA, Feldman DB, Moore JA (1984) Comparison of tests for the diagnosis of spontaneous encephalitozoonosis in rabbits. Lab Anim Sci 34:356–359
Pye D, Cox JC (1977) Isolation of Encephalitozoon cuniculi from urine samples. Lab Anim 11:234
Reabel S (2012) Molecular diagnostic methods for detection of Encephalitozoon cuniculi in pet rabbits. Msc thesis in Pathobiology, University of Guelph, Guelph, Ontario, Canada
Reetz J (1995) Microsporidien als ursache von aborten beim rind. TieraÈrztl Umschau 50:550–554
Sak B, Kvac M, Kvetonova D, Albrecht T, Pialek J (2011) The first report on natural Enterocytozoon bieneusi and Encephalitozoon spp. infections in wild East European House Mice (Mus musculus musculus) and West-European House Mice (M. m. domesticus) in a hybrid zone across the Czech Republic Germany border. Vet Parasitol 178(3–4):246–250
SAS (2004) Statistical analysis system. SAS User’s Guide SAS Incorporation Institute
Sasaki M, Yamazaki A, Haraguchi A, Tatsumi M, Ishida K, Ikadai H (2011) Serological survey of Encephalitozoon cuniculi infection in Japanese dogs. J Parasitol 97(1):16–19
Schmidt EC, Shadduck JA (1983) Murine encephalitozoonosis model for studying the host parasite relationship of a chronic infection. Inf Immun 40:936–942
Schuurs AH, Verheul HA (1989) Effect of gender and steroids on the immune response. J Steroid Biochem 35:157–172
Soliman R, Selim S (2005) Practical immunology, 1st edn. Faculty of Veterinary Medicine, Cairo University Press, Egypt
Wasson K, Peper RL (2000) Mammalian microsporidiosis. Vet Pathol 37:113–128
Weber R, Bryan RT, Owen RL, Wilcox CM, Gorelkin NO, Visvesvara GS (1992) Improved light microscopical detection of microsporidia spores in stool and duodenal aspirates. N Engl J Med 326:161–166
Wilson K, Bjornstad ON, Dobson AP, Merler S, Poglayen G, Randolph SE, Read AF, Skorping A (2007) Heterogeneities in macroparasite infections: patterns and processes. In: Hudson PJ, Rizzoli A, Grenfell BT, Heesterbeek H, Dobson AP (eds) The ecology of wildlife disease. Oxford University Press, New York, pp 6–44
Yamaguchi-Shinozaki K, Koizumi M, Urao S, Shinozaki K (1992) Molecular cloning and characterization of one cDNAs for genes that are responsive to desiccation in Arabidopsis thaliana: sequence analysis of one cDNA clone that encodes a putative transmembrane channel protein. Plant Cell Physiol 33:217–224
Acknowledgments
The study was funded by the Alexandria University Research Fund (AURF), Grants, Innovation and Technology Transfer Center (GITTC), Research Enhancement Program (ALEX REP), 2011–2012 (project code, AGRV-11). The authors are indebted to Dr. Rafiq Soliman, professor of Microbiology, Faculty of Veterinary Medicine, Cairo University for his effort and valuable help.
Ethical standards
All investigations comply with the current laws of the countries in which they were performed.
Conflict of interest
The authors declare that they have no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abu-Akkada, S.S., Ashmawy, K.I. & Dweir, A.W. First detection of an ignored parasite, Encephalitozoon cuniculi, in different animal hosts in Egypt. Parasitol Res 114, 843–850 (2015). https://doi.org/10.1007/s00436-014-4247-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-014-4247-4