Abstract
Purpose
There is no approved targeted therapy for chordoma at present. Although several preclinical studies have implied the potential applicability of CDK4/6 inhibitor for this rare tumor, no clinical evidence has been documented so far. The purpose of this study was to elucidate the therapeutic efficacy of CDK4/6 inhibitor for chordoma.
Methods
The next generation sequencing (as for whole-exome sequencing, WES assay) and immunohistochemical (IHC) staining of the chordoma tissue from a patient with an advanced lesion were performed before treatment. Then, the patient was treated with Palbociclib for 4 months until progression occurred in the 5th month. Surgical resection was implemented and the tumor tissue was obtained postoperatively for assessment of molecular alterations.
Results
Molecular features of the tumor before medical treatment suggested applicability of CDK4/6 inhibitor and the patient showed partial response (PR) according to Choi Criteria after 4 months treating with Palbociclib until progression occurred. Then, a drastic molecular alteration of the tumor as represented by emergence of dramatic E2F amplification, which is known to induce CDK4/6 independent cell-cycle entry and progression after treatment, was detected. The findings in this patient demonstrated tumor evolution under drug pressure.
Conclusion
The findings of the present study suggest the feasibility of Palbociclib for the clinical treatment of chordoma, and imply the necessity of combination therapies rather single drug administration due to the quick resistance of the tumor to Palbociclib treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chordoma is a rare malignancy derived from remnants of the notochord, commonly occurring in the axial skeleton. Because of the radio- and chemo-resistance of chordoma, radical resection, sometimes combined with high-dose radiation or the proton radiotherapy remains to be the mainstay of treatment to ensure long-term survival (Stacchiotti and Sommer 2015). However, the local recurrent rate is high due to the anatomical limitation and rarity of adjuvant therapeutic options(Kerekes et al. 2019). Although clinical trials for targeted therapies, such as apatinib (Liu et al. 2020), lapatinib (Stacchiotti et al. 2013), imatinib and everolimus (Stacchiotti et al. 2012, 2018) have been conducted in patients with advanced chordoma, only a small portion of patients possessing the corresponding alterations of molecular targets can benefit. Therefore, elucidation of therapeutic markers harboring in most cases of chordoma will largely facilitate improving current chordoma treatment.
As one of the most prevalent genetic alterations in chordoma, homo- (11.25–30% in chordoma) or heterozygous loss of chromosome 9p21 (about 70% in chordoma) with loci of cyclin-dependent kinase inhibitor 2A/2B (CDKN2A/2B), has been described in several cohorts (Bai et al. 2021; Le et al. 2011; Tarpey et al. 2017). Alterations of CDKN2A/2B, are known to play essential tumor-suppressing roles in a variety of tumors,, making this pathway an attractive therapeutic target (Helsten et al. 2016; Kato et al. 2015). Previous studies suggested that chordoma cells exhibiting a loss of CDKN2A (p16) may trigger universal activation of CDK4/6, implying the potential applicability of CDK4/6 inhibitor in chordoma treatment (Liu et al. 2018) (Passeri et al. 2022). However, no clinical evidence has ever been reported for the effectiveness of CDK inhibitors in the treatment of chordoma.
Here, we present the first case of repeatedly recurrent chordoma in a patient who partially responded to Palbociclib therapy for 4 months and then developed drug resistance within a month. Molecular landscapes of the tumor tissues before and after Palbociclib treatment were obtained by IHC and WES for investigating potential responsive and drug-resistant markers indicating the administration or withdrawal of CDK4/6 inhibitor.
Case report
A 50-year-old man was admitted to our hospital with a history of repeatedly recurrent cervical chordoma. The treatment timeline of this patient is presented in Fig. 1. The lesion involving C2 was first detected when the patient complained of cervical pain and weakness of the lower limbs 7 years ago. The pathological result postoperatively suggested that the lesion was conventional type. Local recurrence occurred one year after partial resection of the tumor, and was treated with conventional radiotherapy. 15 months after the 2nd surgery, the tumor progression led to cervical pain and incomplete paralysis of the extremities. The patient was then referred to our center, where magnetic resonance imaging (MRI) scan revealed a 70 × 47 × 36 mm, well-defined paraspinal pathological mass with bone erosion and remodeling extending to the transverse processes and part of the vertebra bodies, and with a widening of their intervertebral foramina on C2 to C4. A staged posterior-anterior surgery was performed using the 3D printed vertebral body for anterior reconstruction. The pathological result remained to be conventional chordoma. To enhance marginal control, heavy proton therapy was delivered 1 month after surgery. However, local recurrence was detected once again 8 months later during the follow-up period. The tumor was removed and the tumor tissue specimen was obtained. Considering a relative tumor-free surgical margin was achieved in the surgery, adjuvant treatment was not delivered postoperatively. Unfortunately, tumor progression was detected 12 months after operation, when MRI scan showed a 69 × 60 × 73mm pathological mass on the right side (Fig. 2). The patient’s chief complaint was swelling pain of the neck. Tissue obtained from the last surgery were subjected to WES and IHC assays for detection of 719 COSMIC cancer gene alterations (Sondka et al. 2018) (Fig. 3C and Tables 1, 2). Previous studies had observed genetic (Wang et al. 2016) and protein (Cottone et al. 2020) loss of CDKN2A-p16 in different chordoma cohorts, and we also detected a heterozygous deletion that involved the depletion of CDKN2A/2B/2C and gain of CCND2 and CDK6 (Table 2). Meanwhile, IHC staining also showed a loss of CDKN2A-p16, a high expression of CDK6 and positive CCND1 (cyclin D1) in this patient, and nearly half of the cells exhibited phosphorylated Rb1.
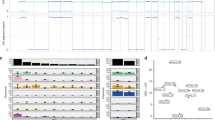
Molecular overview before and after Palbociclib treatment. A Protein expression of CDK6, CDKN2A-p16, Rb1-phosphor, and CCND1 of tumor tissues before and after treatment; B Somatic copy number alterations (SCNAs) before and after treatment. A alleles are depicted in red, B alleles in blue; C Mutant genes before and after treatment
After multidisciplinary evaluation, Palbociclib was selected to facilitate tumor resection. It was administered orally at a dose of 125 mg daily for 21 days, paused for 7 days, and then continued with the next cycle. In the first few days, the patient reported an obvious relief of the swelling pain of the neck. The follow-up MRI scan performed after 4 months after treatment showed shrinkage of the tumor volume and signs of necrosis. According to the Choi Criteria(van der Veldt et al. 2010), partial response (PR) of the lesion was defined. MRI also showed a decreased rate of enhancement and partial recession of tumor infiltration compared to the pre-treatment MRI (Fig. 2). Blood routine, liver/kidney function, and blood coagulation function were examined biweekly, showing no significant adverse event. However, the patient reported a relapse of the cervical pain during the fifth cycle of treatment. MRI was performed immediately, showing re-progression of the lesion causing compression of the spinal cord (Fig. 2). Hence, surgery was inevitable, and the surgically resected tumor specimen was sent for second WES and IHC assays.
After Palbociclib treatment until re-progression occurred, a proportion of negative CDK6 cells appeared for IHC staining (Fig. 3A), but more gains of CDK6 (CNt from 3 to 4) and CCND1 (CNt from 2 to 4) was observed at genetic level (Table 2). CDKN2A-p16 protein became positive which is consistent with genetic alteration. No significant difference was observed for phosphorylated Rb1 before and after treatment. WES assay showed less chromosomal deletions (including disappearance of chromosome 9p) and more gains and amplifications after treatment when comparing the genetic profiles (Fig. 3B). Besides, some mutations disappeared and others emerged, and the frequencies of the mutant genes shared by both tissues from before and after treatment were significantly reduced after treatment (Fig. 3C; Table 1). Notably, the copy number of E2F1 dramatically increased from the wild type (CNt 2; A allele 1, B allele 1) to amplification (CNt 15; A allele 8, B allele 7) which may imply drug resistance (Huang et al. 2022) (Table 2). Routine postoperative follow-up observation showed no local recurrence 6 months thereafter.
Discussion
In this study, we firstly reported the response and development of drug resistance to Palbociclib applied in a patient with advanced chordoma. As the tumor tissue specimens were obtained both before and after treatment, molecular portraits reflecting the tumor evolutionary process under medical pressure or the spatial heterogeneity of the tumor mass were captured in this case.
Although no specific parameter correlative to the efficacy of CDK4/6 inhibitors has been reported, amplification of CDK4/6, CCND1, D2, and/or D3, and alterations in CDKN2A/B are suggested as putative markers to predict the response from CDK4/6 inhibitors (Kato et al. 2021). It is reported that co-deletion of CDKN2A and CDKN2C confer Palbociclib-sensitivity in glioblastoma (Wiedemeyer et al. 2010) and soft tissue sarcoma, demonstrating that amplification of CDK4 benefits from the CDK4/6 inhibitor (Mangat et al. 2018). Broto et al. have also assessed the role of Palpociclib in sarcomas overexpressing CDK4 but not CDKN2A and achieved promising results excluding liposarcoma (Martin-Broto et al. 2023). In our case, the tumor specimen presented heterozygous deletions of CDKN2A, CDKN2B and CDKN2C (Table 2), and gains of CDK6 and CCND2. CDKN2A-p16 protein was not detected,accompanied with the positive expression of CDK6 and CCND1 (Fig. 3A). Thus, the CDK4/6 inhibitor Palbociclib was administered to the patient. After 4-cycle treatment, PR of the lesion was defined according to the Choi Criteria. However, the tumor continued progressing thereafter. The relatively short duration of treatment response in our case might be attributed to several reasons. Firstly, the WES assay was not performed right after Palbociclib treatment after the third surgery. The delay of adjuvant targeted therapy might impair the efficacy of treatment. Secondly, the tissue sample analyzed by WES assay might not represent the whole tumor mass due to the spatial heterogeneity, which may result in inaccurate assessment of the Palbociclib sensitivity. In addition, the current biomarkers analyzed might not be precise enough for the prediction of Palbociclib efficacy.
The results of WES assay and IHC staining of the tissues obtained from the third and the fourth surgery were compared to further explore inherent mechanism of alteration in drug sensitivity. Although the protein expression of CDK6 slightly decreased after treatment, the former heterozygous deletion of CDKN2A/2B/2C vanished, and CCND1/2/3 and CDK6 obtained more gains than before as shown by WES assay (Table 2). The divergent genetic landscapes suggest a dramatic change in tumor composition. Since both tumor tissues were taken from the same location, the possibility of spatial heterogeneity could be ruled out. Considering the alteration of sensitivity to the drug, the genetic discrepancy might be the result of tumor evolution under drug pressure.
It is worthy to mention that E2F1 amplification emerged after drug resistance (Table 2), which is considered to be associated with the resistance of CDK4/6 inhibitors due to the compensation of CDK4/6 independent CyclinE-CDK2 cell-cycle entry and progression (Álvarez-Fernández and Malumbres 2020). Previous studies suggest that CDK2 ablation may rescue the sensitivity of resistant cells to CDK4/6 inhibitors in vitro (Gong et al. 2017; Harbour et al. 1999). Our results also suggest that the combined administration of additional drugs such as specific CDK2 inhibitor may provide better outcomes compared with the use of the CDK4/6 inhibitor alone (Kato et al. 2021). Additionally, previous studies suggested that CDK4/6 inhibitors could augment response to immune-checkpoint therapy (Álvarez-Fernández and Malumbres 2020; Fassl et al. 2022). In our case, we observed an increase of tumor mutational burden (TMB) (from 0.80 to 1.23 mutations/Mb), which is widely admitted as a biomarker for response to immunotherapy across multiple cancer types (Samstein et al. 2019).
In summary, our data provide the first clinical evidence and comprehensive genomic profiling including the potential response, and assumed drug resistance markers during application of the CDK4/6 inhibitor in a chordoma patient. The tumor evolution detected in this case demonstrated that, chordoma patients whose tumors harbor potentially sensitizing alterations to G1/S cell-cycle signaling pathway may benefit from CDK4/6 inhibitor, and also implies necessity of combination therapies because of the possibility of drug-resistance generated by single drug administration. Moreover, this study also highlighted the role of next generation of sequencing (NGS) analysis in daily clinical practice for tumors with less mutations (Racanelli et al. 2020). However, considering the limited case number and retrospective nature of this report, the evidence level of this result is relatively low. Besides, as there is a paucity of feasible biomarkers for the evaluation of CDK4/6 inhibitor sensitivity in chordoma (Passeri et al. 2022), another limitation of this research includes that the criteria for application of Palbociclib in the current case might be controversial. Hence, the precise prediction of Palbociclib sensitivity might be undermined, and the spatial heterogeneity of the tumor cannot be completely ruled out, which possibly lead to the reduced duration of treatment response. Indeed, larger prospective trials on Palbociclib treatment and identification of drug-targeted biomarkers in chordoma are required for an unmet need to expand the limited therapeutic options of chordoma.
Data availability
The data are available from the corresponding author on reasonable request. The sequencing data are deposited in the Genome Sequence Archive (GSA) for human under accession number PRJCA017336 (https://ngdc.cncb.ac.cn/gsa-human).
References
Álvarez-Fernández M, Malumbres M (2020) Mechanisms of sensitivity and resistance to CDK4/6 inhibition. Cancer Cell 37:514–529. https://doi.org/10.1016/j.ccell.2020.03.010
Bai J, Shi J, Li C, Wang S, Zhang T, Hua X, Zhu B, Koka H, Wu H-H, Song L, Wang D, Wang M, Zhou W, Ballew BJ, Hicks B, Mirabello L, Parry DM, Zhai Y, Li M, Du J, Wang J, Zhang S, Liu Q, Zhao P, Gui S, Goldstein AM, Zhang Y, Yang XR (2021) Whole genome sequencing of skull-base chordoma reveals genomic alterations associated with recurrence and chordoma-specific survival. Nat Commun 12:757. https://doi.org/10.1038/s41467-021-21026-5
Cottone L, Eden N, Usher I, Lombard P, Ye H, Ligammari L, Lindsay D, Brandner S, Pižem J, Pillay N, Tirabosco R, Amary F, Flanagan AM (2020) Frequent alterations in p16/CDKN2A identified by immunohistochemistry and FISH in chordoma. J Pathol Clin Res 6:113–123. https://doi.org/10.1002/cjp2.156
Fassl A, Geng Y, Sicinski P (2022) CDK4 and CDK6 kinases: from basic science to cancer therapy. Science. https://doi.org/10.1126/science.abc1495
Gong X, Litchfield LM, Webster Y, Chio L-C, Wong SS, Stewart TR, Dowless M, Dempsey J, Zeng Y, Torres R, Boehnke K, Mur C, Marugán C, Baquero C, Yu C, Bray SM, Wulur IH, Bi C, Chu S, Qian H-R, Iversen PW, Merzoug FF, Ye XS, Reinhard C, de Dios A, Du J, Caldwell CW, Lallena MJ, Beckmann RP, Buchanan SG (2017) Genomic aberrations that activate D-type cyclins are associated with enhanced sensitivity to the CDK4 and CDK6 inhibitor abemaciclib. Cancer Cell 32:761-776.e6. https://doi.org/10.1016/j.ccell.2017.11.006
Harbour JW, Luo RX, Dei Santi A, Postigo AA, Dean DC (1999) Cdk phosphorylation triggers sequential intramolecular interactions that progressively block Rb functions as cells move through G1. Cell 98:859–869. https://doi.org/10.1016/s0092-8674(00)81519-6
Helsten T, Kato S, Schwaederle M, Tomson BN, Buys TPH, Elkin SK, Carter JL, Kurzrock R (2016) Cell-cycle gene alterations in 4,864 tumors analyzed by next-generation sequencing: implications for targeted therapeutics. Mol Cancer Ther 15:1682–1690. https://doi.org/10.1158/1535-7163.MCT-16-0071
Huang J, Zheng L, Sun Z, Li J (2022) CDK4/6 inhibitor resistance mechanisms and treatment strategies (review). Int J Mol Med. https://doi.org/10.3892/ijmm.2022.5184
Kato S, Schwaederle M, Daniels GA, Piccioni D, Kesari S, Bazhenova L, Shimabukuro K, Parker BA, Fanta P, Kurzrock R (2015) Cyclin-dependent kinase pathway aberrations in diverse malignancies: clinical and molecular characteristics. Cell Cycle 14:1252–1259. https://doi.org/10.1080/15384101.2015.1014149
Kato S, Okamura R, Adashek JJ, Khalid N, Lee S, van Nguyen, Sicklick JK, Kurzrock R (2021) Targeting G1/S phase cell-cycle genomic alterations and accompanying co-alterations with individualized CDK4/6 inhibitor-based regimens. JCI Insight. https://doi.org/10.1172/jci.insight.142547
Kerekes D, Goodwin CR, Ahmed AK, Verlaan J-J, Bettegowda C, Abu-Bonsrah N, Sciubba DM (2019) Local and distant recurrence in resected sacral chordomas: a systematic review and pooled cohort analysis. Global Spine J 9:191–201. https://doi.org/10.1177/2192568217741114
Le LP, Nielsen GP, Rosenberg AE, Thomas D, Batten JM, Deshpande V, Schwab J, Duan Z, Xavier RJ, Hornicek FJ, Iafrate AJ (2011) Recurrent chromosomal copy number alterations in sporadic chordomas. PLoS One 6:e18846. https://doi.org/10.1371/journal.pone.0018846
Liu T, Shen JK, Choy E, Zhang Y, Mankin HJ, Hornicek FJ, Duan Z (2018) CDK4 expression in chordoma: a potential therapeutic target. J Orthop Res 36:1581–1589. https://doi.org/10.1002/jor.23819
Liu C, Jia Q, Wei H, Yang X, Liu T, Zhao J, Ling Y, Wang C, Yu H, Li Z, Jiao J, Wu Z, Yang C, Xiao J (2020) Apatinib in patients with advanced chordoma: a single-arm, single-centre, phase 2 study. Lancet Oncol 21:1244–1252. https://doi.org/10.1016/S1470-2045(20)30466-6
Mangat PK, Halabi S, Bruinooge SS, Garrett-Mayer E, Alva A, Janeway KA, Stella PJ, Voest E, Yost KJ, Perlmutter J, Pinto N, Kim ES, Schilsky RL (2018) Rationale and design of the targeted agent and profiling utilization registry (TAPUR) study. JCO Precis Oncol. https://doi.org/10.1200/PO.18.00122
Martin-Broto J, Martinez-Garcia J, Moura DS, Redondo A, Gutierrez A, Lopez-Pousa A, Martinez-Trufero J, Sevilla I, Diaz-Beveridge R, Solis-Hernandez MP, Carnero A, Perez M, Marcilla D, Garcia-Foncillas J, Romero P, Fernandez-Jara J, Lopez-Lopez D, Arribas I, Hindi N (2023) Phase II trial of CDK4/6 inhibitor palbociclib in advanced sarcoma based on mRNA expression of CDK4/ CDKN2A. Signal Transduct Target Ther 8:405. https://doi.org/10.1038/s41392-023-01661-8
Passeri T, Dahmani A, Masliah-Planchon J, El Botty R, Courtois L, Vacher S, Marangoni E, Nemati F, Roman-Roman S, Adle-Biassette H, Mammar H, Froelich S, Bièche I, Decaudin D (2022) In vivo efficacy assessment of the CDK4/6 inhibitor palbociclib and the PLK1 inhibitor volasertib in human chordoma xenografts. Front Oncol 12:960720. https://doi.org/10.3389/fonc.2022.960720
Racanelli D, Brenca M, Baldazzi D, Goeman F, Casini B, de Angelis B, Guercio M, Milano GM, Tamborini E, Busico A, Dagrada G, Garofalo C, Caruso C, Brunello A, Pignochino Y, Berrino E, Grignani G, Scotlandi K, Parra A, Hattinger CM, Ibrahim T, Mercatali L, de Vita A, Carriero MV, Pallocca M, Loria R, Covello R, Sbaraglia M, Dei Tos AP, Falcioni R, Maestro R (2020) Next-generation sequencing approaches for the identification of pathognomonic fusion transcripts in sarcomas: the experience of the Italian ACC Sarcoma Working Group. Front Oncol 10:489. https://doi.org/10.3389/fonc.2020.00489
Samstein RM, Lee C-H, Shoushtari AN, Hellmann MD, Shen R, Janjigian YY, Barron DA, Zehir A, Jordan EJ, Omuro A, Kaley TJ, Kendall SM, Motzer RJ, Hakimi AA, Voss MH, Russo P, Rosenberg J, Iyer G, Bochner BH, Bajorin DF, Al-Ahmadie HA, Chaft JE, Rudin CM, Riely GJ, Baxi S, Ho AL, Wong RJ, Pfister DG, Wolchok JD, Barker CA, Gutin PH, Brennan CW, Tabar V, Mellinghoff IK, DeAngelis LM, Ariyan CE, Lee N, Tap WD, Gounder MM, D’Angelo SP, Saltz L, Stadler ZK, Scher HI, Baselga J, Razavi P, Klebanoff CA, Yaeger R, Segal NH, Ku GY, DeMatteo RP, Ladanyi M, Rizvi NA, Berger MF, Riaz N, Solit DB, Chan TA, Morris LGT (2019) Tumor mutational load predicts survival after immunotherapy across multiple cancer types. Nat Genet 51:202–206. https://doi.org/10.1038/s41588-018-0312-8
Sondka Z, Bamford S, Cole CG, Ward SA, Dunham I, Forbes SA (2018) The COSMIC Cancer Gene Census: describing genetic dysfunction across all human cancers. Nat Rev Cancer 18:696–705. https://doi.org/10.1038/s41568-018-0060-1
Stacchiotti S, Sommer J (2015) Building a global consensus approach to chordoma: a position paper from the medical and patient community. Lancet Oncol 16:e71-83. https://doi.org/10.1016/S1470-2045(14)71190-8
Stacchiotti S, Longhi A, Ferraresi V, Grignani G, Comandone A, Stupp R, Bertuzzi A, Tamborini E, Pilotti S, Messina A, Spreafico C, Gronchi A, Amore P, Vinaccia V, Casali PG (2012) Phase II study of imatinib in advanced chordoma. J Clin Oncol 30:914–920. https://doi.org/10.1200/JCO.2011.35.3656
Stacchiotti S, Tamborini E, Lo Vullo S, Bozzi F, Messina A, Morosi C, Casale A, Crippa F, Conca E, Negri T, Palassini E, Marrari A, Palmerini E, Mariani L, Gronchi A, Pilotti S, Casali PG (2013) Phase II study on lapatinib in advanced EGFR-positive chordoma. Ann Oncol 24:1931–1936. https://doi.org/10.1093/annonc/mdt117
Stacchiotti S, Morosi C, Lo Vullo S, Casale A, Palassini E, Frezza AM, Dinoi G, Messina A, Gronchi A, Cavalleri A, Venturelli E, Morelli D, Pilotti S, Collini P, Brich S, Tamborini E, Mariani L, Casali PG (2018) Imatinib and everolimus in patients with progressing advanced chordoma: a phase 2 clinical study. Cancer 124:4056–4063. https://doi.org/10.1002/cncr.31685
Tarpey PS, Behjati S, Young MD, Martincorena I, Alexandrov LB, Farndon SJ, Guzzo C, Hardy C, Latimer C, Butler AP, Teague JW, Shlien A, Futreal PA, Shah S, Bashashati A, Jamshidi F, Nielsen TO, Huntsman D, Baumhoer D, Brandner S, Wunder J, Dickson B, Cogswell P, Sommer J, Phillips JJ, Amary MF, Tirabosco R, Pillay N, Yip S, Stratton MR, Flanagan AM, Campbell PJ (2017) The driver landscape of sporadic chordoma. Nat Commun 8:890. https://doi.org/10.1038/s41467-017-01026-0
van der Veldt AAM, Meijerink MR, van den Eertwegh AJM, Haanen JBAG, Boven E (2010) Choi response criteria for early prediction of clinical outcome in patients with metastatic renal cell cancer treated with sunitinib. Br J Cancer 102:803–809. https://doi.org/10.1038/sj.bjc.6605567
Wang L, Zehir A, Nafa K, Zhou N, Berger MF, Casanova J, Sadowska J, Lu C, Allis CD, Gounder M, Chandhanayingyong C, Ladanyi M, Boland PJ, Hameed M (2016) Genomic aberrations frequently alter chromatin regulatory genes in chordoma. Genes Chromosomes Cancer 55:591–600. https://doi.org/10.1002/gcc.22362
Wiedemeyer WR, Dunn IF, Quayle SN, Zhang J, Chheda MG, Dunn GP, Zhuang L, Rosenbluh J, Chen S, Xiao Y, Shapiro GI, Hahn WC, Chin L (2010) Pattern of retinoblastoma pathway inactivation dictates response to CDK4/6 inhibition in GBM. Proc Natl Acad Sci USA 107:11501–11506. https://doi.org/10.1073/pnas.1001613107
Acknowledgements
We thank the patient and his family for participating in this study, as well as the clinical data managers and other support staff for their assistance and help. No individuals were compensated for their contributions. We also appreciate the financial support from the National Natural Science Fund Project of China (NSFC No. 82141105; No. 81902731).
Funding
This study was funded by the National Natural Science Fund Project of China (NSFC No. 82141105 and No. 81902731).
Author information
Authors and Affiliations
Contributions
Conception and design of the study: SC. Financial support: JX. Provision of study materials or patients: NZ, MY, JJ and JX. Acquisition of data: SC, DY, NZ, and XL. Analysis of data: DY, SC, NZ, and QZ. Pathological identification: ZZ. Manuscript writing: SC and NZ. Final approval of manuscript: All authors. Accountable for all aspects of the work: All authors.
Corresponding authors
Ethics declarations
Conflict of interest
No potential conflicts of interest to report.
Informed consent
The investigators of this study had obtained informed consent from the patient for the use of his information for research purposes and for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhong, N., Yu, D., Yang, M. et al. A retrospective study on the mechanism underlying quick transfer from response to resistance in a repeated recurrent chordoma patient with molecular alterations treated with Palbociclib. J Cancer Res Clin Oncol 150, 95 (2024). https://doi.org/10.1007/s00432-023-05560-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00432-023-05560-x