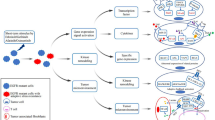
Abstract
Purpose
Epidermal growth factor receptor (EGFR) mutation is a prominent driver of lung cancer. Tyrosine kinase inhibitors (TKIs) have shown efficacy in treating EGFR-mutant lung cancer, but the emergence of drug resistance poses a significant challenge. Recent research has highlighted solute carrier family 12 member 8 (SLC12A8) as one of the highly upregulated genes in various cancer types. However, its oncogenic function remains largely unexplored.
Methods
343 consecutive lung cancer patients were prospectively recruited and were followed for over 10 years. SLC12A8 expression in lung cancer tissues was measured by qPCR and was associated with patient survival. The association of SLC12A8 with TKI resistance was studied in in vitro EGFR-mutant lung cancer cell line as well as in in vivo xenograft tumor model. High-throughput kinome screening was employed to investigate SLC12A8-mediated oncogenic signaling pathway in lung cancer.
Results
SLC12A8 is a predictive biomarker of poor prognosis in lung cancer, particularly in patients with EGFR mutations. SLC12A8 overexpression diminishes the effectiveness of TKIs in EGFR-mutant lung cancer, resulting in treatment failure and disease progression. More importantly, SLC12A8-induced TKI resistance is mediated by the PDK1/AKT signaling axis, while silencing SLC12A8 expression inhibits oncogenic PDK1/AKT signaling, restoring TKI sensitivity in lung cancer cells.
Conclusion
SLC12A8 mediates TKI resistance in EGFR-mutant lung cancer via PDK1/AKT axis. These findings not only advance our understanding of the molecular mechanisms driving TKI resistance, but also offer novel alternative strategies for the treatment of lung cancer.







Similar content being viewed by others
Data availability
Data will be made available on request.
References
Alessi DR, James SR, Downes CP, Holmes AB, Gaffney PR, Reese CB et al (1997) Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Balpha. Curr Biol 7(4):261–269. https://doi.org/10.1016/s0960-9822(06)00122-9
Cross DA, Ashton SE, Ghiorghiu S, Eberlein C, Nebhan CA, Spitzler PJ et al (2014) AZD9291, an irreversible EGFR TKI, overcomes T790M-mediated resistance to EGFR inhibitors in lung cancer. Cancer Discov 4(9):1046–1061. https://doi.org/10.1158/2159-8290.CD-14-0337
Cui JJ (2014) A new challenging and promising era of tyrosine kinase inhibitors. ACS Med Chem Lett 5(4):272–274. https://doi.org/10.1021/ml500091p
Goan YG, Liu PF, Chang HW, Chen HC, Chen WC, Kuo SM et al (2019) Kinome-wide screening with small interfering RNA identified polo-like kinase 1 as a key regulator of proliferation in oral cancer cells. Cancers. https://doi.org/10.3390/cancers11081117
Grozio A, Mills KF, Yoshino J, Bruzzone S, Sociali G, Tokizane K et al (2019) Slc12a8 is a nicotinamide mononucleotide transporter. Nat Metab 1(1):47–57. https://doi.org/10.1038/s42255-018-0009-4
Hsu WH, Yang JC, Mok TS, Loong HH (2018) Overview of current systemic management of EGFR-mutant NSCLC. Ann Oncol 29(1):i3–i9. https://doi.org/10.1093/annonc/mdx702
Ito S, Fujino Y, Ogata S, Hirayama-Kurogi M, Ohtsuki S (2018) Involvement of an orphan transporter, SLC22A18, in cell growth and drug resistance of human breast cancer MCF7 cells. J Pharm Sci 107(12):3163–3170. https://doi.org/10.1016/j.xphs.2018.08.011
Kang E, Kim K, Jeon SY, Jung JG, Kim HK, Lee HB et al (2022) Targeting CLK4 inhibits the metastasis and progression of breast cancer by inactivating TGF-beta pathway. Cancer Gene Ther 29(8–9):1168–1180. https://doi.org/10.1038/s41417-021-00419-0
Kim SY (2018) Cancer energy metabolism: shutting power off cancer factory. Biomol Ther (seoul) 26(1):39–44. https://doi.org/10.4062/biomolther.2017.184
Kim JY, Bai Y, Jayne LA, Hector RD, Persaud AK, Ong SS et al (2020) A kinome-wide screen identifies a CDKL5-SOX9 regulatory axis in epithelial cell death and kidney injury. Nat Commun 11(1):1924. https://doi.org/10.1038/s41467-020-15638-6
Kobayashi S, Boggon TJ, Dayaram T, Janne PA, Kocher O, Meyerson M et al (2005) EGFR mutation and resistance of non-small-cell lung cancer to gefitinib. N Engl J Med 352(8):786–792. https://doi.org/10.1056/NEJMoa044238
Lavoro A, Falzone L, Tomasello B, Conti GN, Libra M, Candido S (2023) In silico analysis of the solute carrier (SLC) family in cancer indicates a link among DNA methylation, metabolic adaptation, drug response, and immune reactivity. Front Pharmacol 14:1191262. https://doi.org/10.3389/fphar.2023.1191262
Li AR, Chitale D, Riely GJ, Pao W, Miller VA, Zakowski MF et al (2008) EGFR mutations in lung adenocarcinomas: clinical testing experience and relationship to EGFR gene copy number and immunohistochemical expression. J Mol Diagn 10(3):242–248. https://doi.org/10.2353/jmoldx.2008.070178
Li L, Xia J, Cui R, Kong B (2021a) Solute carrier family 12 member 8 impacts the biological behaviors of breast carcinoma cells by activating TLR/NLR signaling pathway. Cytotechnology 73(1):23–34. https://doi.org/10.1007/s10616-020-00439-y
Li SL, Li ZF, Cao QW, Wang WZ (2021b) SLC12A8 plays a key role in bladder cancer progression and EMT. Open Med (wars) 16(1):58–67. https://doi.org/10.1515/med-2021-0013
Liu Q, Yu S, Zhao W, Qin S, Chu Q, Wu K (2018) EGFR-TKIs resistance via EGFR-independent signaling pathways. Mol Cancer 17(1):53. https://doi.org/10.1186/s12943-018-0793-1
Melosky B, Kambartel K, Hantschel M, Bennetts M, Nickens DJ, Brinkmann J et al (2022) Worldwide prevalence of epidermal growth factor receptor mutations in non-small cell lung cancer: a meta-analysis. Mol Diagn Ther 26(1):7–18. https://doi.org/10.1007/s40291-021-00563-1
Midha A, Dearden S, McCormack R (2015) EGFR mutation incidence in non-small-cell lung cancer of adenocarcinoma histology: a systematic review and global map by ethnicity (mutMapII). Am J Cancer Res 5(9):2892–911. https://www.ncbi.nlm.nih.gov/pubmed/26609494
Paez JG, Janne PA, Lee JC, Tracy S, Greulich H, Gabriel S et al (2004) EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 304(5676):1497–1500. https://doi.org/10.1126/science.1099314
Saito Y, Soga T (2021) Amino acid transporters as emerging therapeutic targets in cancer. Cancer Sci 112(8):2958–2965. https://doi.org/10.1111/cas.15006
Sapio L, Di Maiolo F, Illiano M, Esposito A, Chiosi E, Spina A et al (2014) Targeting protein kinase A in cancer therapy: an update. EXCLI J 13:843–55. https://www.ncbi.nlm.nih.gov/pubmed/26417307.
Tan FL, Ooi A, Huang D, Wong JC, Qian CN, Chao C et al (2010) p38delta/MAPK13 as a diagnostic marker for cholangiocarcinoma and its involvement in cell motility and invasion. Int J Cancer 126(10):2353–2361. https://doi.org/10.1002/ijc.24944
Tang Z, Li C, Kang B, Gao G, Li C, Zhang Z (2017) GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res 45(W1):W98–W102. https://doi.org/10.1093/nar/gkx247
Walter AO, Sjin RT, Haringsma HJ, Ohashi K, Sun J, Lee K et al (2013) Discovery of a mutant-selective covalent inhibitor of EGFR that overcomes T790M-mediated resistance in NSCLC. Cancer Discov 3(12):1404–1415. https://doi.org/10.1158/2159-8290.CD-13-0314
Wu SG, Shih JY (2018) Management of acquired resistance to EGFR TKI-targeted therapy in advanced non-small cell lung cancer. Mol Cancer 17(1):38. https://doi.org/10.1186/s12943-018-0777-1
Yang B, Ma YB, Chu SH (2018) Silencing SATB1 overcomes temozolomide resistance by downregulating MGMT expression and upregulating SLC22A18 expression in human glioblastoma cells. Cancer Gene Ther 25(11–12):309–316. https://doi.org/10.1038/s41417-018-0040-3
Zhang Q, Liu Y, Chen P, Shi X, Liu Y, Shi L et al (2021) Solute carrier family 12 member 8 (SLC12A8) is a potential biomarker and related to tumor immune cell infiltration in bladder cancer. Bioengineered 12(1):4946–4961. https://doi.org/10.1080/21655979.2021.1962485
Zheng H, Long G, Zheng Y, Yang X, Cai W, He S et al (2022) Glycolysis-related SLC2A1 is a potential pan-cancer biomarker for prognosis and immunotherapy. Cancers. https://doi.org/10.3390/cancers14215344
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. FH, JC and CGJ performed material preparation, data collection and analysis. FH, WZ and TZ wrote the first draft of this manuscript and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interests
The authors declare no conflict of interest.
Ethics approval
The human reserch was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee at the First Affiliated Hospital of Nanchang University. Animal studies were approved by of the Research Ethic Committee at the First Affiliated Hospital of Nanchang University and were performed in accordance with institutional guidelines of the Animal Care and Use Committee.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent to publish
The authors affirm that human research participants provided informed consent for publication of the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, F., Cui, J., Wan, J. et al. SLC12A8 mediates TKI resistance in EGFR-mutant lung cancer via PDK1/AKT axis. J Cancer Res Clin Oncol 149, 16729–16739 (2023). https://doi.org/10.1007/s00432-023-05416-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-05416-4