Abstract
Purpose
Non-small cell lung cancer (NSCLC) accounts for about 85% in all cases of lung cancer. In recent years, molecular targeting drugs for NSCLC have been developed rapidly. The epidermal growth factor receptor-tyrosine kinase inhibitors (EGFR-TKIs) have changed the paradigm of cancer therapy from empirical cytotoxic chemotherapy to molecular-targeted cancer therapy. Currently, there are three generations of EGFR-TKIs, all of which have achieved good efficacy in clinical therapy. However, most patients developed drug resistance after 6–13 months EGFR-TKIs treatment. Therefore, a comprehensive understanding of EGFR-TKIs resistance mechanisms is of vital importance for clinical management of NSCLC.
Methods
Relevant data and information about the topic were obtained by searching PubMed (Medline), Web of Science and Google Scholar using the subject headings, such as “NSCLC”, “EGFR-TKIs resistance”, “EGFR mutations”, “human epidermal growth factor receptor-2 (HER2/erbB-2)”, “hepatocyte growth factor (HGF)”, “vascular endothelial growth factor (VEGF)”, “insulin-like growth factor 1 (IGF-1)”, “epithelial–mesenchymal transition (EMT)”, “phosphatase and tensin homolog (PTEN)”, “RAS mutation”, “BRAF mutation”, “signal transducer and activator of transcription 3 (STAT3)”, and “tumor microenvironment”, etc.
Results
The mechanisms for EGFR-TKIs resistance include EGFR mutations, upregulation of HER2, HGF/c-MET, VEGF IGF1, EMT and STAT3 pathways, mutations of PTEN, RAS and BRAF genes, and activation of other by-pass pathways. These mechanisms are interconnected and can be potential targets for the treatment of NSCLC.
Conclusion
In this review, we discuss the mechanisms of EGFR-TKIs drug resistance and the clinical strategies to overcome drug resistance from the perspective of EGFR-TKIs combined treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The most predominant pathological type of lung cancer is non-small cell lung cancer (NSCLC), which accounts for about 85% in all cases (Molina et al. 2008; Zappa and Mousa 2016). Fortunately, there is an increasing number of oncogenic driver mutations discovered in NSCLC, accompanied with an expanding spectrum of clinical signal transduction pathway inhibitors targeting these mutations, offers a considerable opportunity to improve the prognosis of patients. The discovery of epidermal growth factor receptor (EGFR) mutations is one of the most striking findings in NSCLC. Until now, the EGFR tyrosine kinases inhibitors (TKIs) has successfully changed the paradigm of cancer therapy from empirical cytotoxic chemotherapy to molecular-targeted cancer therapy (Lee 2017).
At present, there are three generations of EGFR-TKIs proved to be effective in clinical trials. The representative first-generation TKIs are gefitinib and erlotinib, which play an anti-tumor role by reversibly competing with ATP, binding to the tyrosine kinase domain through EGFR intracellular protein tyrosine kinase (Huang and Fu 2015; Seshacharyulu et al. 2012). Different from the first generation, the second-generation TKIs, such as dacomitinib and afatinib, are irreversible EGFR-TKIs, which can irreversibly inhibit three different members of the ERBB family, including EGFR, HER2 and HER4 (Subramaniam et al. 2014). Osimertinib, the third generation TKI, can irreversibly inhibit tyrosine kinase phosphorylation as well as reverse the T790M resistance mutation of patients (Cross et al. 2014). Though these EGFR-TKIs are effective in the treatment of EGFR mutated NSCLC, most patients developed drug resistance after 6 to 13 months, especially with the treatment of first/second-generation TKIs (Mok et al. 2009; Wu et al. 2015, 2017; Yang et al. 2016). Therefore, overcoming drug resistance has become an urgent clinical need to improve the survival of patients harbored EGFR mutations. A comprehensive understanding of the drug resistance mechanism of TKIs targeting NSCLC will be very beneficial to the follow-up drug research and the optimization of clinical strategies. In this review, the resistance mechanisms of EGFR-TKIs are discussed from the perspective of different targets. We also concern about the clinical strategies to overcome TKIs resistance in the current clinical research, and put forward to the possible direction of basic research as well as clinical development.
Gene alterations related to drug resistance
“On-target” and “off-target” have been classified as two main targeting resistance mechanisms proposed by Rotow and Bivona (2017). In detail, the alteration of primary target of the drug signifies on-target resistance, which decreased the inhibition effects of drugs when they interact with targets. Off-target resistance means changes of other related signaling pathways. We mainly elaborated on the molecular mechanisms of resistance of EGFR-TKIs targeted therapy for NSCLC based on these two levels in this review.
On-target resistance mechanism
EGFR mutations
HER family of receptor tyrosine kinases includes four members, respectively are EGFR (HER1 or ErbB1), HER2 (nue/ErbB2), HER3 (ErbB3) and HER4 (ErbB4) (Wheeler et al. 2008). EGFR mutations are limited to 4 exons (exons 18–21), and can be divided into 3 types (deletions, insertions and missense point mutations) (Shigematsu and Gazdar 2006). Most EGFR mutations occur in exon 19 (exon19 deletion, Del19) and exon 21 (exon 21 substitution, L858R) (Yang et al. 2020; Yasuda et al. 2012). These two mutations result in impaired endocytosis of EGFR, increasing the tyrosine phosphorylation. T790M mutation is another important mutation in clinic. It is the most common resistant mechanism to TKIs (Kobayashi et al. 2005). Studies have showed that T790M mutation could cause acquired resistance to first/second generation TKIs in 50–60% patients (Kosaka et al. 2006). At position 790 (T790M) in exon 20, threonine is replaced by methionine in specific kinase domain. Threonine 790 has been designated as a “gatekeeper” residue (Kon et al. 2014), and its mutation leads to the conformational change of EGFR kinase region, which makes TKI drugs unable to approach the active center of tyrosine kinase, weakening the reversible binding with TKI drugs. Table 1 lists the common resistance mechanisms of EGFR-mutant NSCLC. Actually, apart from single gene mutation, the important roles of hybrid mutations and secondary mutations playing in drug resistance have been gradually realized in recent years. The combination type of mutations is related to the prognosis of patients, and also essential to the change of therapeutic strategy. For instance, a study found that NSCLC patients harboring Del19 mutation was more likely to develop T790M secondary mutation than those with L858R mutants (Ke et al. 2017). More evidence revealed that patients of Del19 with T790M had a superiority of overall survival. This result may be caused by the different interaction sites with drug between Del19 and L858R (Furuyama et al. 2013). Both of the mutations are located near the active site cleft of kinase. Del19 removes several residues from the loop leading into the αC-helix, while L858R lies in the activation loop of the kinase. This distinction results in different phosphorylation inhibition degree of gefitinib of EGFR, Akt and Erk1/2 in Del19 cells than in L858R cells (Zhu et al. 2008).
There are also some patients with other mutations, such as L747S (Yamaguchi et al. 2014a), D761Y (Balak et al. 2006) and T854A (Bean et al. 2008), which reduce the sensitivity of the mutant EGFR to bind to EGFR-TKIs as well. With the development of medicinal measurement technique and the awareness of important role that gene mutation plays in TKI drug resistance, there is an increasing number of new mutations have been detected and reported in recent years, including some rare insertion mutations (Woo et al. 2014). However, their specific mechanism is still not clear.
Off-target resistance mechanism
An overview of off-target resistance on type, frequency, molecular mechanism and possible targeted therapy are summarized in Table 2. More details are included in each part.
HER2 amplification
Takezawa et al. (2012) firstly reported that HER2 amplification was a new mechanism of acquired resistance to EGFR-TKIs in EGFR-mutant NSCLC tumors (Takezawa et al. 2012). Interestingly, in human model of NSCLC, EGFR-T790M mutation and HER2 amplification are often mutually exclusive. However, Shtiegman et al. (2007) proved that the amplification of HER2 could help the EGFR-L858R mutation individual cells escape ubiquitination by c-Cbl and enhance ligand-mediated receptor desensitization (Shtiegman et al. 2007). Activated ERK1/2 signaling pathway has been demonstrated as an important cause of drug resistance caused by HER2 amplification (Scrima et al. 2017). In recent years, the promising effect of trastuzumab against HER2 mutation are being widely explored. For instance, the American National Comprehensive Cancer Network (NCCN) has included trastuzumab and afatinib as potential therapy options for NSCLC patients with HER2 mutation (Eberlein et al. 2015). Besides, a phase II trial enrolled 24 patients for treatment with trastuzumab and paclitaxel (de Langen et al. 2018). The results revealed that this combination treatment has a better objective response rate and median duration of response than taxane monotherapy. Another ongoing phase II study of trastuzumab deruxtecan in HER2-overexpressing patients also showed obvious antitumor activity so far (Nakagawa et al. 2021). However, the risk and safety still need further estimation.
HGF/c-MET pathway
C-MET is the receptor for hepatocyte growth factor (HGF). Studies have found that 5% to 10% of patients developed acquired resistance to TKI due to c-MET gene amplification (Lei et al. 2020; Zhang et al. 2019b). C-MET amplification is one of discovered causes of gefitinib resistance, driving ErbB3 dependent activation of PI3K. This pathway is thought to be specific to EGFR/ErbB family receptors and independent of EGFR kinase activity (Engelman et al. 2007). Combinatorial therapy should be considered for these patients with resistant NSCLC carrying c-MET amplification (Wang et al. 2019). For instance, the c-MET inhibitor onartuzumab with erlotinib combination treatment has reported a benefit in a randomized phase II trial for c-MET immunohistochemistry (IHC)-positive patients (Spigel et al. 2013). Activation of HGF/c-MET signaling pathway was also sporadic reported about acquired resistance to TKIs by affecting the c-MET gene. Evidence showed HGF induced gefitinib resistance of lung adenocarcinoma cells with EGFR-activating mutations by restoring the PI3K/Akt signaling pathway via phosphorylation of c-MET in an ErbB3-independent manner (Yano et al. 2008). Therefore, combined therapy with EGFR and HGF-c-MET inhibitors may overcome the drug resistance and prolong the effective time of EGFR-TKI treatment.
VEGF pathway
Vascular endothelia growth factor (VEGF) is essential in the progress of both physiological and pathological angiogenesis. VEGF receptor 2 (VEGFR2) is a kind of receptor tyrosine kinase which primarily mediates the VEGF signals, and it is also related to VEGF-induced cell proliferation in NSCLC (Devery et al. 2015). It has been well documented that in NSCLC, high expression of VEGFR2 often related to poor prognosis, since VEGF/VEGFR2 has potential to drive tumor cell proliferation through an autocrine signaling pathway (Pau et al. 2009; Seto et al. 2006). Besides, the enhanced VEGF level is often associated with EGFR-TKIs resistance (Larsen et al. 2011). A preclinical model has demonstrated simultaneous use of EGFR and VEGFR2 inhibitors were effective in antitumor treatment, supporting that combination strategy has a significant potential in future EGFR-TKIs resistance therapy (Tonra et al. 2006). A study included 311 Chinese NSCLC patients compared the combination of erlotinib and VEGF inhibitor bevacizumab to erlotinib (Le et al. 2021). Patients in combined therapy group had progression-free survival (PFS) improvement of 6.7 months at the primary endpoint. Other evidence revealed that patients with acquired resistance to EGFR-TKIs may also get profit of apatinib by its inducing effect on cell cycle arrest and VEGFR signaling pathway inhibition (Song et al. 2019).
IGF1 pathway
Insulin-like growth factor 1(IGF1) is an intermediate of many growth hormone responses, and has potential to stimulate the growth of some type of cancers. IGF1 receptor (IGF1R) is a tyrosine kinase-containing heterotetramer on cell surface, which mediated the biological activities of the IGFs (Werner et al. 2019). Activation of IGF1R signaling pathway is responsible for afatinib resistance in NSCLC patients harboring the T790M mutation (Lee et al. 2016). In addition, NSCLC cells with gefitinib-resistance are also related to the activity of IGF1R, since cell lines that were already resistant to gefitinib were found to have increased total-IGF1R and phosphorylated-IGF1R expression (Denduluri et al. 2015). Guerard et al. (2018) discovered that amphiregulin induced the nuclear accumulation of IGF1R. As a result, cell cycle was arrested through p21 (WAF1/CIP1) upregulation, and the induction of apoptosis was prevented in response to gefitinib (Guerard et al. 2018). Another study showed that hypoxia activated IGF1R, stimulating the growth of NSCLC stem cells which were already resistant to gefitinib (Murakami et al. 2014). This result suggests that IGF1R pathway might be a promising target for overcoming gefitinib resistance in NSCLC.
EMT
The epithelial-mesenchymal transition (EMT) phenotype is a malignant cancer phenotype characterized by augmented invasion, metastasis and chemoresistance (Kurimoto et al. 2016). Studies have demonstrated that many cytokines induced acquired resistance to EGFR-TKIs by promoting the occurrence of EMT (Xue et al. 2017; Yamaguchi et al. 2014b). Suda et al. (2011) added EMT-inducing ligand transforming growth factor β (TGF-β) to HCC4006 lung cancer cells in vitro, both morphological changes of cells and resistance to erlotinib were observed after 2 weeks treatment (Suda et al. 2011). It is reported that Forkhead box protein M1 (FOXM1), pyruvate dehydrogenase kinase 4 (PDK4) and Connexin 26 (Cx26) may be the essential regulators of EMT and are associated with TKIs resistance (Kong et al. 2014; Yang et al. 2015). Induction of EMT via the activation of the TGF-β/Smad canonical signaling pathway has been reported to be involved in the EGFR-TKIs induced resistance (Zhang et al. 2019a). TGF-β2 signaling links with EGFR-TKI drug-escape, cancer cells undergoing early adaptive drug escape are susceptible to TGF-β2 inhibition (Thiagarajan et al. 2016). In addition, activation of TGF-β-mediated signaling was sufficient to induce EMT occurrence and an increased secretion level of TGF-β-dependent interleukin 6 (IL-6) unleashed previously addicted lung tumor cells from their EGFR dependency (Yao et al. 2010). Thus, precise inhibition of both EGFR and TGF-β is a possible way to overcome drug resistance. Yochum et al. (2019) demonstrated that genetic silencing of TWIST1, the transcription factors for EMT, can inhibit the growth of EGFR-mutant NSCLC cells (Yochum et al. 2019). Overexpression of TWIST1 leads to resistance to erlotinib and AZD9291 in NSCLC patients. Inhibition of TWIST1 may be helpful to slow down the occurrence of EGFR-TKI resistance.
PTEN mutation
Phosphatase and tension homolog (PTEN) inactivation can specifically raise EGFR activity. By disintegrating newly formed ubiquitin ligase Cbl complexes, PTEN impairing the ligand-induced ubiquitylation and degradation of the activated receptor (Jin et al. 2018). A recent study demonstrated that EGFR-mutant patients harboring PTEN deletion had a shorter PFS and overall survival (OS) than those with intact PTEN. Moreover, PTEN deletion was identified as a cause of resistance to EGFR-TKIs in advanced NSCLC patients with EGFR sensitive mutations (Gkountakos et al. 2019). PI3K/Akt signaling pathway is regulated by PTEN deletion negatively, and it was found that CpG hyper methylation of the PTEN gene contributed to the decreased expression of PTEN during acquired resistance to gefitinib or erlotinib (Maeda et al. 2015). Up-regulation of PTEN provides a new idea for improving drug resistance of EGFR-TKIs. Hopkins et al. (2013) found a translational variant leading to a longer PTEN protein (Hopkins et al. 2013). This kind of special protein is a membrane-permeable lipid phosphatase which is able to enter other cells and antagonize PI3K signaling. There are also some other possible methods to increase PTEN expression level. For instance, statins was suggested to promote the activity of transcription factor peroxisome proliferator-activated receptor-γ, consequently upregulating PTEN mRNA levels (Teresi et al. 2006). Regulating micro-RNA is another way which deserves trying to control PTEN expression. For example, highly expressed microRNA-21 (Zhang and Peng 2017), microRNA-25-3p (Sun et al. 2021) and microRNA-103a-3p (Li et al. 2021) are responsible for the proliferation and invasion of NSCLC cells via targeting PTEN. Controlling these microRNAs expression may resume the function of PTEN and re-sensitize the drug-resistant cancer cells to EGFR-TKIs.
RAS mutation
K-RAS is a member of the RAS family. Researchers used a mass spectrometry to detect genetic mutations related to EGFR-TKIs resistant lung cancer, and found that K-RAS was one of the drug resistance mutations (Tian et al. 2017). 5.9% K-RAS mutation could be found in EGFR mutated lung adenocarcinoma patients who developed acquired resistance to erlotinib (Cardona et al. 2017). K-RAS inhibitors have been under clinical or pre-clinical investigation for cancers with K-RAS mutation. Sotorasib, a small molecule inhibitor of K-RAS, was administered orally once daily to 129 patients (59 with NSCLC) harboring K-RAS p.G12C mutation. The results showed 32.2% of the NSCLC patients had a considerable control, leading to a median PFS of 4 months (Hong et al. 2020). Other compound such as KYA1797K (Park et al. 2019) also showed significant efficacy in animal models, more details are under further study.
BRAF mutation
According to statistics, approximately 1% patients of lung cancers with resistance to EGFR-TKIs were found to have mutations in BRAF (Ohashi et al. 2012). The BRAF mutations consists of three types, BRAF-V600E, BRAF-G469A/V, and BRAF-D594G, in which BRAF-V600E accounts for 50% in total (Tissot et al. 2016). Ho et al. (2017) has reported that activation of downstream MEK-ERK signaling pathway was the reason why BRAF-V600E mutation caused osimertinib resistance (Ho et al. 2017). Evidence suggests that BRAF inhibition demonstrated antitumor activity in NSCLC, and strategy combined BRAF and MEK inhibitors has a great potential to achieve a superior efficacy (Trojaniello et al. 2019; Tu et al. 2015). For example, in a clinical cohort, researchers found that trametinib ± dabrafenib, LXH254 and lifirafenib had a potent inhibition effect on both BRAF V600E and non-V600 mutant NSCLC cell lines (Negrao et al. 2020).
STAT3 gene expression signal activation
High expression and sustained activation of signal transducer and activator 3 of transcription (STAT3) can promote cancer cell proliferation and tumor growth (Hu et al. 2019). Lee et al. (2014) found that a tremendous number of drug-treated ‘‘oncogene-addicted’’ cancer cells engaged a positive feedback loop leading to STAT3 activation, promoting cell survival as a result. They also proved the role of STAT3 following erlotinib treatment in limiting the overall drug response (Lee et al. 2014). Specifically, inhibition of an oncogenic kinase in “addicted” cancer cells by transient STAT3 activation through IL-6R and EGFR, significantly contributes to resistance of the cell population to drug treatment and consequently limits the efficacy of these agents. Another study also demonstrated that EGFR inhibition resulted in the adaptive activation of both STAT3 and Src-YAP1 signaling, potentially operating downstream of IL-6, to promote cell survival and limit the initial response to EGFR-TKIs treatment in EGFR-mutant lung cancer (Chaib et al. 2017). Moreover, STAT3 polymorphism rs4796793 is possible to be a potential biomarker for the prediction of prognosis and susceptibility in lung cancer patients (Gong et al. 2019). MicroRNA-218 was proved to be a tumor suppressor in lung cancer by regulating IL-6/STAT3 signaling pathway (Yang et al. 2017). These results revealed that in the future, we may have more ways associated with STAT3 to predict tumor progression, which is of great significance for the evaluation and adjustment of patient’s treatment strategy.
By-pass pathway
A study demonstrated that TNF1-driven adaptive response is detected in NSCLCs with EGFR activating mutations. Activation of EGFR signaling leads to downregulation of TNF mRNA rapidly, and the inhibition of EGFR signaling resulted in ascending TNF mRNA stability by decreasing miR-21 levels. Meanwhile, NF-κB is activated by TNF, then increases the transcription of TNF mRNA in a feed-forward loop in return, which may be a major mechanism of resistance to EGFR inhibition (Gong et al. 2018). Aberrant activation of NF-κB upregulates the transcription of numerous genes involved in cancer cell proliferation, survival, metastasis and therapeutic resistance (Li and Sethi 2010). For example, NF-κB takes part in initial EGFR inhibitor treatment, promoting tumor cell survival because EGFR oncogene inhibition induces the production of EGFR-TRAF2-RIP1-IKK complex (Blakely et al. 2015), which is related to an NF-κB-mediated transcriptional survival program. Bivona et al. (2011) had proved that the inhibition of NF-κB pathway can enhance or restore erlotinib sensitivity (Bivona et al. 2011). This result indicates a possible way to overcome EGFR-TKIs resistance.
Tumor microenvironment
In recent years, immunosuppressive effects of EGFR mutations have been reported in tumor microenvironment, such as regulatory cells (Tregs) (Mascia et al. 2016), immunoregulatory cytokines (Jia et al. 2019), and exosomes (Poggio et al. 2019). Actually, the immunosuppressive environment caused by TGF-β, IL-10 and IL-35 secretion from Tregs in tumors actively attenuates and subverts the antitumor immune responses of CD4+ T cells, CD8+ T cells and natural killer cells (Chin and Wang 2016; Frydrychowicz et al. 2017). Moreover, studies have shown that the level of amphiregulin in plasma in patient with NSCLC counts for poor prognosis (Chang et al. 2011). Another research found that amphiregulin could mediate suppressive function of Tregs via the EGFR/glycogen synthase kinase (GSK)-3/forkhead box P3 (Foxp3) axis (Wang et al. 2016).
Apicella et al. (2018) found that under long-term treatment with TKIs, glycolysis and lactic acid production increased in EGFR- or MET-addicted cancer cells (Apicella et al. 2018). Interestingly, the secreted lactate is the key molecule to instruct cancer-associated fibroblasts to produce HGF activating MET-dependent signaling in cancer cells, leading to sustained resistance to TKIs. EGFR-TKIs have been shown to down-regulate the expression of programmed death ligand (PD-L1), a prognostic indicator related to immunotherapy, in selected NSCLC with sensitive EGFR mutations that expressed high level of PD-L1(Tang et al. 2015). Actually, erlotinib up-regulated major histocompatibility complex (MHC)-I and MHC-II proteins in interferon (IFN)-γ treated keratinocytes but abrogated IFN-γ-induced expression of PD-L1 (Im et al. 2016). CD47 and calreticulin, those kinds of proteins associated with immune recognition, could affect the clearance of tumor cells by phagocytosis, contributing to cancer cell immune escape and adaptive drug resistance. In addition, Liu et al. (2019) observed that hypofractionated (high dose with a low frequency treatment) EGFR TKI treatment is more potent than standard strategy (Liu et al. 2019). The hypofractionated EGFR TKI triggers greater innate sensing for type I IFN and CXCL10 production through the Myd88 signaling pathway to enhance tumor-specific T cell infiltration and reactivation. This result provides evidence of proper combination of EGFR TKIs and immunotherapy in future treatment exploration.
Clinical strategy to overcome EGFR-TKIs drug resistance
Though the third generation of TKIs as monotherapy can make an improvement sometimes when the resistance of the first and second TKIs appears (Tan et al. 2018), it still lacks long-term efficacy due to drug resistance. Therefore, combination of EGFR-TKIs with other treatments are considered an effective strategy to overcome drug resistance. A series of recent studies have shown that EGFR-TKIs combination therapy has shown survival benefits. Relevant ongoing clinical trials about EGFR-TKIs combined with other treatments are listed in Table 3.
Many clinical studies were carried out to evaluate the effect of first-line combination EGFR-TKIs plus chemotherapy 10 years ago. However, the results of these studies were negative (Rebuzzi et al. 2020). The main problem is before experiments, the type and status of EGFR mutation of patients were not detected and selected. Therefore, patients who are not likely to respond to EGFR-TKIs were also included in these studies, leading to unsatisfied results. Recent clinical studies showed that the combined treatment of EGFR-TKIs and chemotherapy exhibited significant clinical benefits. Han et al. (2017) found that the PFS and OS of patients with lung adenocarcinoma harboring sensitive EGFR mutations in the combination group received gefitinib and pemetrexed/carboplatin were longer than that of patients in the chemotherapy or gefitinib alone (Han et al. 2017). Similarly, Noronha et al. (2019) demonstrated that patients accepted gefitinib with additional pemetrexed-carboplatin chemotherapy got a significantly prolonged PFS and OS (Noronha et al. 2019). Hosomi et al. (2020) conducted a phase III study to evaluate the efficacy of EGFR-TKI combined with platinum doublet chemotherapy in untreated advanced NSCLC patients with EGFR mutations (Hosomi et al. 2019). Compared with gefitinib alone, carboplatin combined with gefitinib improved PFS in patients with EGFR mutations. However, the OS of this kind of treatment requires further validation.
EGFR-TKIs combined with antiangiogenic therapy are also extensively investigated in recent years. Seto et al. (2014) demonstrated that a prolonged median PFS in the treatment of erlotinib plus bevacizumab (16.0 months) compared with erlotinib alone (9.7 months) (Seto et al. 2014). Another study also confirmed that bevacizumab plus erlotinib combination therapy improved PFS compared with erlotinib alone in patients with EGFR-positive NSCLC (Saito et al. 2019). Besides, Nakagawa et al. (2019) demonstrated in the ramucirumab plus erlotinib treated group, patients have a longer PFS (19.4 months) than in the placebo plus erlotinib treated group (12.4 months) (Nakagawa et al. 2019). Ramucirumab is an antagonist of VEGFR, which can specifically bind VEGFR2 and block the coordination of VEGF ligand, VEGF-A, VEGF-C and VEGF-D (Clarke and Hurwitz 2013). Because of the interconnections between VEGF and EGFR, apatinib (a highly selective VEGFR2 inhibitor) can significantly enhance the anti-tumor effect of gefitinib in T790M positive mutant cells (Li et al. 2017).
Other specific mutation inhibitors combined with EGFR-TKIs are reported as well besides of those mentioned above. For example, the combination of EGFR-TKIs gefitinib with the demethylating agent 5-aza-2'deoxycytidine (5-AZA) and the histone deacetyl-transferase (HDAC) inhibitor Trichostatin A (TSA) may be an effective strategy aimed at PTEN inactivation lung cancer (Noro et al. 2007). MEK inhibitors combined with EGFR-TKIs are also explored (Martinelli et al. 2017). Eberlein et al. (2015) reported combination therapy of osimertinib and the MEK inhibitor selumetinib could prevent the emergence of drug resistance in PC9 cells and delayed drug resistance in H1975 cells (Eberlein et al. 2015). Day et al. (2019) demonstrated that the knockdown of TNFAIP8 inhibited EGF and IGF-1-stimulated migration in NSCLC cells. This discovery indicated TNFAIP8 and its effectors may be exploited to realize EGFR and IGF1R regulation, improving the efficacy of molecular-targeted therapies as a result (Day et al. 2019). Jia et al. (2016) tested the activity of a new type of allosteric inhibitor EAI045, which can inhibit L858R/T790M-mutant EGFR with nanomolar potency (Jia et al. 2016). Okura et al. (2020) conducted a small group of clinical trials to verify the efficacy of a novel AXL inhibitor ONO-7475, which showed that ONO-7475 suppressed the emergence and maintenance of tolerant cells to the initial EGFR-TKIs (Okura et al. 2020). These findings suggest that combined strategy of EGFR-TKI and other signaling pathway inhibitors is a promising therapeutic approach in the future.
Future perspectives
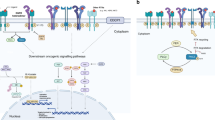
Target therapy has been a revolution in the management of advanced cancers, particularly NSCLC using EGFR-TKIs. However, drug resistance shortly occurs as expected. During the treatment of EGFR-TKIs, cancer cells develop drug resistance owing to EGFR on-target and off-target mechanisms (Fig. 1). Tumor microenvironment also plays imperative roles in EGFR-TKI drug resistance. Although EGFR-TKIs combination therapy has delayed the occurrence and development of drug resistance to some extent, there is large space to be improved and developed. For instance, combinatory treatment of EGFR-TKIs combined with inhibitors of MET, EMT and PI3K/Akt pathways, and antibodies against IGF1R, IL-6 and VEGF should be extensively explored. Owing to the cutting-edge technologies, such as whole-genome sequencing and RNA sequencing, in accurate detection and diagnosis of cancers, precision medicine and individualized therapy are conceived in clinical application. The disease types, genotypes, mutations in specific genes and immune status of patients should be comprehensively analyzed before individualized treatment of lung cancers. Nevertheless, the researches for more in-depth and detailed molecular mechanisms involved in the disease and drug resistance of targeted therapy of NSCLC are still highly demanded to transform NSCLC into a chronic disease.
Data availability
Not applicable.
Code availability
Not applicable.
References
Apicella M et al (2018) Increased lactate secretion by cancer cells sustains non-cell-autonomous adaptive resistance to MET and EGFR targeted therapies. Cell Metab 28:848−865
Balak MN et al (2006) Novel D761Y and common secondary T790M mutations in epidermal growth factor receptor—mutant lung adenocarcinomas with acquired resistance to kinase inhibitors. Clin Cancer Res 12:6494–6501
Bean J et al (2008) Acquired resistance to epidermal growth factor receptor kinase inhibitors associated with a novel T854A mutation in a patient with EGFR-mutant lung adenocarcinoma. Clin Cancer Res 14:7519–7525
Bivona TG et al (2011) FAS and NF-kappa B signalling modulate dependence of lung cancers on mutant EGFR. Nature 471:523–526
Blakely CM et al (2015) NF-kappa B-activating complex engaged in response to EGFR oncogene inhibition drives tumor cell survival and residual disease in lung cancer. Cell Rep 11:98–110
Cardona AF et al (2017) Acquired resistance to erlotinib in EGFR mutation-positive lung adenocarcinoma among Hispanics (CLICaP). Target Oncol 12:513–523
Chaib I et al (2017) Co-activation of STAT3 and YES-associated protein 1 (YAP1) Pathway in EGFR-Mutant NSCLC. J Nat Cancer Instit 109. https://doi.org/10.1093/jnci/djx014
Chang MH et al (2011) Clinical impact of amphiregulin expression in patients with epidermal growth factor receptor (EGFR) wild-type nonsmall cell lung cancer treated with EGFR-tyrosine kinase inhibitors. Cancer 117:143–151
Chin AR, Wang SE (2016) Cancer-derived extracellular vesicles: the ‘soil conditioner’ in breast cancer metastasis? Cancer Metastasis Rev 35:669–676
Clarke JM, Hurwitz HI (2013) Targeted inhibition of VEGF receptor 2: an update on ramucirumab. Expert Opin Biol Ther 13:1187–1196
Cross DAE et al (2014) AZD9291, an irreversible EGFR TKI, overcomes T790M-mediated resistance to EGFR inhibitors in lung cancer. Cancer Discov 4:1046–1061
Day TF et al (2019) Dual targeting of EGFR and IGF1R in the TNFAIP8 knockdown non-small cell lung cancer cells. Mol Cancer Res 17:1207–1219
de Langen AJ et al (2018) Trastuzumab and paclitaxel in patients with EGFR mutated NSCLC that express HER2 after progression on EGFR TKI treatment. Br J Cancer 119:558–564
Denduluri SK et al (2015) Insulin-like growth factor (IGF) signaling in tumorigenesis and the development of cancer drug resistance. Genes Dis 2:13–25
Devery AM et al (2015) Vascular endothelial growth factor directly stimulates tumour cell proliferation in non-small cell lung cancer. Int J Oncol 47:849–856
Dong J et al (2010) Polymorphisms in EGFR and VEGF contribute to non-small-cell lung cancer survival in a Chinese population. Carcinogenesis 31:1080–1086
Eberlein CA et al (2015) Acquired resistance to the mutant-selective EGFR Inhibitor AZD9291 is associated with increased dependence on RAS signaling in preclinical models. Cancer Res 75:2489–2500
Engelman JA et al (2007) MET amplification leads to gefitinib resistance in lung cancer by activating ERBB3 signaling. Science 316:1039–1043
Frydrychowicz M et al (2017) The dual role of Treg in cancer. Scand J Immunol 86:436–443
Furuyama K et al (2013) Sensitivity and kinase activity of epidermal growth factor receptor (EGFR) exon 19 and others to EGFR-tyrosine kinase inhibitors. Cancer Sci 104:584–589
Gkountakos A et al (2019) PTEN in lung cancer: dealing with the problem, building on new knowledge and turning the game around. Cancers 11:1141. https://doi.org/10.3390/cancers11081141
Gong K et al (2018) TNF-driven adaptive response mediates resistance to EGFR inhibition in lung cancer. J Clin Investig 128:2500–2518
Gong WJ et al (2019) STAT3 rs4796793 contributes to lung cancer risk and clinical outcomes of platinum-based chemotherapy. Int J Clin Oncol 24:476–484
Guerard M et al (2018) Nuclear translocation of IGF1R by intracellular amphiregulin contributes to the resistance of lung tumour cells to EGFR-TKI. Cancer Lett 420:146–155
Han BH et al (2017) Combination of chemotherapy and gefitinib as first-line treatment for patients with advanced lung adenocarcinoma and sensitive EGFR mutations: a randomized controlled trial. Int J Cancer 141:1249–1256
Hasako S et al (2018) TAS6417, a novel EGFR inhibitor targeting Exon 20 insertion mutations. Mol Cancer Ther 17:1648–1658
Ho CC et al (2017) Acquired BRAF V600E mutation as resistant mechanism after treatment with osimertinib. J Thorac Oncol 12:567–572
Hong DS et al (2020) KRAS (G12C) inhibition with sotorasib in advanced solid tumors. N Engl J Med 383:1207–1217
Hopkins BD et al (2013) A secreted PTEN phosphatase that enters cells to alter signaling and survival. Science 341:399–402
Hosomi Y et al (2019) Gefitinib alone versus gefitinib plus chemotherapy for non-small-cell lung cancer with mutated epidermal growth factor receptor: NEJ009 Study. J Clin Oncol 38:115−123
Hu YS et al (2019) STAT3: a potential drug target for tumor and inflammation. Curr Top Med Chem 19:1305–1317
Huang LH, Fu LW (2015) Mechanisms of resistance to EGFR tyrosine kinase inhibitors. Acta Pharmaceutica Sinica B 5:390–401
Im JS et al (2016) Immune-modulation by epidermal growth factor receptor inhibitors: implication on anti-tumor immunity in lung cancer. PLoS ONE 11:e0160004. https://doi.org/10.1371/journal.pone.0160004
Jia Y et al (2016) Overcoming EGFR(T790M) and EGFR(C797S) resistance with mutant-selective allosteric inhibitors. Nature 534:129−134
Jia YJ et al (2019) EGFR-targeted therapy alters the tumor microenvironment in EGFR-driven lung tumors: implications for combination therapies. Int J Cancer 145:1432–1444
Jin G et al (2010) PTEN mutations and relationship to EGFR, ERBB2, KRAS, and TP53 mutations in non-small cell lung cancers. Lung Cancer 69:279–283
Jin Y et al (2018) Mechanisms of primary resistance to EGFR targeted therapy in advanced lung adenocarcinomas. Lung Cancer 124:110–116
Ke EE et al (2017) A higher proportion of the EGFR T790M mutation may contribute to the better survival of patients with exon 19 deletions compared with those with L858R. J Thorac Oncol 12:1368–1375
Kempf E et al (2016) KRAS oncogene in lung cancer: focus on molecularly driven clinical trials. Eur Respir Rev
Kobayashi S et al (2005) EGFR mutation and resistance of non-small-cell lung cancer to gefitinib. N Engl J Med 352:786–792
Kon S et al (2014) Altered trafficking of mutated growth factor receptors and their associated molecules: implication for human cancers. Cell Logist 4:e28461. https://doi.org/10.4161/cl.28461
Kong FF et al (2014) FOXM1 regulated by ERK pathway mediates TGF-beta 1-induced EMT in NSCLC. Oncol Res 22:29–37
Kosaka T et al (2006) Analysis of epidermal growth factor receptor gene mutation in patients with non-small cell lung cancer and acquired resistance to gefitinib. Clin Cancer Res 12:5764–5769
Kurimoto R et al (2016) Drug resistance originating from a TGF-beta/FGF-2-driven epithelial-to-mesenchymal transition and its reversion in human lung adenocarcinoma cell lines harboring an EGFR mutation. Int J Oncol 48:1825–1836
Larsen AK et al (2011) Targeting EGFR and VEGF(R) pathway cross-talk in tumor survival and angiogenesis. Pharmacol Ther 131:80–90
Le X et al (2021) ARTEMIS highlights VEGF inhibitors as effective partners for EGFR TKIs in EGFR mutant NSCLC. Cancer Cell 39:1178–1180
Lee DH (2017) Treatments for EGFR-mutant non-small cell lung cancer (NSCLC): the road to a success, paved with failures. Pharmacol Ther 174:1–21
Lee HJ et al (2014) Drug resistance via feedback activation of stat3 in oncogene-addicted cancer cells. Cancer Cell 26:207–221
Lee Y et al (2016) Inhibition of IGF1R signaling abrogates resistance to afatinib (BIBW2992) in EGFR T790M mutant lung cancer cells. Mol Carcinog 55:991–1001
Lei L et al (2020) Potential mechanism of primary resistance to icotinib in patients with advanced non-small cell lung cancer harboring uncommon mutant epidermal growth factor receptor: a multi-center study. Cancer Sci 111:679–686
Li F, Sethi G (2010) Targeting transcription factor NF-kappa B to overcome chemoresistance and radioresistance in cancer therapy. Biochimica Biophysica Acta Rev Cancer 1805:167–180
Li F et al (2017) Apatinib enhances antitumour activity of EGFR-TKIs in non-small cell lung cancer with EGFR-TKI resistance. Eur J Cancer 84:184–192
Li HX, et al (2021) MicroRNA-103a-3p promotes cell proliferation and invasion in non-small-cell lung cancer cells through akt pathway by targeting PTEN. Biomed Res Int 7590976. https://doi.org/10.1155/2021/7590976
Liu ZD et al (2019) Hypofractionated EGFR tyrosine kinase inhibitor limits tumor relapse through triggering innate and adaptive immunity. Sci Immunol 38. https://doi.org/10.1126/sciimmunol.aav6473
Maeda M et al (2015) CpG hypermethylation contributes to decreased expression of PTEN during acquired resistance to gefitinib in human lung cancer cell lines. Lung Cancer 87:265–271
Martinelli E et al (2017) Cancer resistance to therapies against the EGFR-RAS-RAF pathway: the role of MEK. Cancer Treat Rev 53:61–69
Mascia F et al (2016) Cell autonomous or systemic EGFR blockade alters the immune-environment in squamous cell carcinomas. Int J Cancer 139:2593–2597
Mok TS et al (2009) Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med 361:947–957
Molina JR et al (2008) Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clin Proc 83:584–594
Murakami A et al (2014) Hypoxia increases gefitinib-resistant lung cancer stem cells through the activation of insulin-like growth factor 1 receptor. PLoS ONE
Nakagawa K et al (2019) Ramucirumab plus erlotinib in patients with untreated, EGFR-mutated, advanced non-small-cell lung cancer (RELAY): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 20:1655–1669
Nakagawa K et al (2021) Trastuzumab deruxtecan in HER2-overexpressing metastatic non-small cell lung cancer: interim results of DESTINY-Lung01. J Thorac Oncol 16:S109–S110
Negrao MV et al (2020) Molecular landscape of BRAF-mutant NSCLC reveals an association between clonality and driver mutations and identifies targetable non-V600 driver mutations. J Thorac Oncol 15:1611–1623
Noro R et al (2007) PTEN inactivation in lung cancer cells and the effect of its recovery on treatment with epidermal growth factor receptor tyrosine kinase inhibitors. Int J Oncol 31:1157–1163
Noronha V et al (2019) Phase III randomized trial comparing gefitinib to gefitinib with pemetrexed-carboplatin chemotherapy in patients with advanced untreated EGFR mutant non-small cell lung cancer (gef vs gef plus C). J Clin Oncol 37. https://doi.org/10.1200/JCO.2019.37.15_suppl.9001
Ohashi K et al (2012) Lung cancers with acquired resistance to EGFR inhibitors occasionally harbor BRAF gene mutations but lack mutations in KRAS, NRAS, or MEK1. Proc Natl Acad Sci USA 109:E2127–E2133
Okura N et al (2020) ONO-7475, a Novel AXL inhibitor, suppresses the adaptive resistance to initial EGFR-TKI treatment in EGFR-mutated non-small cell lung cancer. Clin Cancer Res 26:2244–2256
Park J et al (2019) A Ras destabilizer KYA1797K overcomes the resistance of EGFR tyrosine kinase inhibitor in KRAS-mutated non-small cell lung cancer. Sci Rep. https://doi.org/10.1038/s41598-018-37059-8
Pau ECD et al (2009) Prognostic significance of the expression of vascular endothelial growth factors A, B, C, and D and their receptors R1, R2, and R3 in patients with nonsmall cell lung cancer. Cancer 115:1701–1712
Poggio M et al (2019) Suppression of exosomal PD-L1 induces systemic anti-tumor immunity and memory. Cell 177:414−427
Qian X et al (2016) Circulating cell-free DNA has a high degree of specificity to detect exon 19 deletions and the single-point substitution mutation L858R in non-small cell lung cancer. Oncotarget 7:29154–29165
Rebuzzi SE et al (2020) Combination of EGFR-TKIs and chemotherapy in advanced EGFR mutated NSCLC: review of the literature and future perspectives. Crit Rev Oncol Hematol 146:102820. https://doi.org/10.1016/j.critrevonc.2019.102820
Rotow J, Bivona TG (2017) Understanding and targeting resistance mechanisms in NSCLC. Nat Rev Cancer 17:637–658
Saito H et al (2019) Erlotinib plus bevacizumab versus erlotinib alone in patients with EGFR-positive advanced non-squamous non-small-cell lung cancer (NEJ026): interim analysis of an open-label, randomised, multicentre, phase 3 trial. Lancet Oncol 20:625–635
Scrima M et al (2017) Aberrant signaling through the HER2-ERK1/2 pathway is predictive of reduced disease-free and overall survival in early stage non- small cell lung cancer (NSCLC) patients. J Cancer 8:227–239
Seshacharyulu P et al (2012) Targeting the EGFR signaling pathway in cancer therapy. Expert Opin Ther Targets 16:15–31
Seto T et al (2006) Prognostic value of expression of vascular endothelial growth factor and its flt-1 and KDR receptors in stage I non-small-cell lung cancer. Lung Cancer 53:91–96
Seto T et al (2014) Erlotinib alone or with bevacizumab as first-line therapy in patients with advanced non-squamous non-small-cell lung cancer harbouring EGFR mutations (JO25567): an open-label, randomised, multicentre, phase 2 study. Lancet Oncol 15:1236–1244
Shigematsu H, Gazdar AF (2006) Somatic mutations of epidermal growth factor receptor signaling pathway in lung cancers. Int J Cancer 118:257–262
Shtiegman K et al (2007) Defective ubiquitinylation of EGFR mutants of lung cancer confers prolonged signaling. Oncogene 26:6968–6978
Sierra JR, Tsao M-S (2011) c-MET as a potential therapeutic target and biomarker in cancer. Ther Adv Med Oncol 3:S21–S35
Song YA et al (2019) Apatinib preferentially inhibits PC9 gefitinib-resistant cancer cells by inducing cell cycle arrest and inhibiting VEGFR signaling pathway. Cancer Cell Int 19:117. https://doi.org/10.1186/s12935-019-0836-8
Spigel DR et al (2013) Randomized phase II trial of onartuzumab in combination with erlotinib in patients with advanced non-small-cell lung cancer. J Clin Oncol 31:4105–4114
Subramaniam D et al (2014) Irreversible multitargeted ErbB family inhibitors for therapy of lung and breast cancer. Curr Cancer Drug Targets 14:775–793
Suda K et al (2011) Epithelial to mesenchymal transition in an epidermal growth factor receptor-mutant lung cancer cell line with acquired resistance to erlotinib. J Thorac Oncol 6:1152–1161
Sun B et al (2021) MicroRNA-25-3p promotes cisplatin resistance in non-small-cell lung carcinoma (NSCLC) through adjusting PTEN/PI3K/AKT route. Bioengineered 12:3219–3228
Swanton C et al (2006) Her2-targeted therapies in non-small cell lung cancer. Clin Cancer Res 12:4377S-4383S
Takezawa K et al (2012) HER2 amplification: a potential mechanism of acquired resistance to EGFR inhibition in EGFR-mutant lung cancers that lack the second-site EGFR(T790M) mutation. Cancer Discov 2:922–933
Tan CS et al (2018) Third generation EGFR TKIs: current data and future directions. Mol Cancer 17:29. https://doi.org/10.1186/s12943-018-0778-0
Tang YN et al (2015) The association between PD-L1 and EGFR status and the prognostic value of PD-L1 in advanced non-small cell lung cancer patients treated with EGFR-TKIs. Oncotarget 6:14209–14219
Teresi RE et al (2006) Increased PTEN expression due to transcriptional activation of PPAR gamma by Lovastatin and Rosiglitazone. Int J Cancer 118:2390–2398
Thiagarajan PS et al (2016) Transcriptomic-metabolomic reprogramming in EGFR-mutant NSCLC early adaptive drug escape linking TGF beta 2-bioenergetics-mitochondrial priming. Oncotarget 7:82013–82027
Tian HX et al (2017) Establishment of a novel method for screening epidermal growth factor receptor tyrosine kinase inhibitor resistance mutations in lung cancer. Chin Med J 130:1446–1453
Tissot C et al (2016) Clinical characteristics and outcome of patients with lung cancer harboring BRAF mutations. Lung Cancer 91:23–28
Tonra JR et al (2006) Synergistic antitumor effects of combined epidermal growth factor receptor and vascular endothelial growth factor receptor-2 targeted therapy. Clin Cancer Res 12:2197–2207
Trojaniello C et al (2019) Encorafenib in combination with binimetinib for unresectable or metastatic melanoma with BRAF mutations. Expert Rev Clin Pharmacol 12:259–266
Tu NN et al (2015) BRAF alterations as therapeutic targets in non-small-cell lung cancer. J Thorac Oncol 10:1396–1403
Wang SH et al (2016) Amphiregulin confers regulatory T cell suppressive function and tumor invasion via the EGFR/GSK-3 beta/Foxp3 Axis. J Biol Chem 291:21085–21095
Wang QM et al (2019) MET inhibitors for targeted therapy of EGFR TKI-resistant lung cancer. J Hematol Oncol 12:63. https://doi.org/10.1186/s13045-019-0759-9
Werner H et al (2019) Investigational IGF1R inhibitors in early stage clinical trials for cancer therapy. Expert Opin Investig Drugs 28:1101–1112
Wheeler DL et al (2008) Mechanisms of acquired resistance to cetuximab: role of HER (ErbB) family members. Oncogene 27:3944–3956
Woo HS et al (2014) Epidermal growth factor receptor (EGFR) exon 20 mutations in non-small-cell lung cancer and resistance to EGFR-tyrosine kinase inhibitors. Invest New Drugs 32:1311–1315
Wu YL et al (2015) First-line erlotinib versus gemcitabine/cisplatin in patients with advanced EGFR mutation-positive non-small-cell lung cancer: analyses from the phase III, randomized, open-label, ENSURE study. Ann Oncol 26:1883–1889
Wu YL et al (2017) Dacomitinib versus gefitinib as first-line treatment for patients with EGFR-mutation-positive non-small-cell lung cancer (ARCHER 1050): a randomised, open-label, phase 3 trial. Lancet Oncol 18:1454–1466
Xue YJ et al (2017) Evolution from genetics to phenotype: reinterpretation of NSCLC plasticity, heterogeneity, and drug resistance. Protein Cell 8:178–190
Yamaguchi F et al (2014a) Acquired resistance L747S mutation in an epidermal growth factor receptor-tyrosine kinase inhibitor-naive patient: a report of three cases. Oncol Lett 7:357–360
Yamaguchi N et al (2014b) Dual ALK and EGFR inhibition targets a mechanism of acquired resistance to the tyrosine kinase inhibitor crizotinib in ALK rearranged lung cancer. Lung Cancer 83:37–43
Yang J et al (2015) Reciprocal positive regulation between Cx26 and PI3K/Akt pathway confers acquired gefitinib resistance in NSCLC cells via GJIC-independent induction of EMT. Cell Death Dis 6:e1829. https://doi.org/10.1038/cddis.2015.197
Yang JCH et al (2016) Effect of dose adjustment on the safety and efficacy of afatinib for EGFR mutation-positive lung adenocarcinoma: post hoc analyses of the randomized LUX-Lung 3 and 6 trials. Ann Oncol 27:2103–2110
Yang Y et al (2017) MicroRNA-218 functions as a tumor suppressor in lung cancer by targeting IL-6/STAT3 and negatively correlates with poor prognosis. Mol Cancer 16:141. https://doi.org/10.1186/s12943-017-0710-z
Yang GJ et al (2020) EGFR exon 20 insertion mutations in Chinese advanced non-small cell lung cancer patients: molecular heterogeneity and treatment outcome from nationwide real-world study. Lung Cancer 145:186–194
Yano S et al (2008) Hepatocyte growth factor induces gefitinib resistance of lung adenocarcinoma with epidermal growth factor receptor-activating mutations. Can Res 68:9479–9487
Yao Z et al (2010) TGF-beta IL-6 axis mediates selective and adaptive mechanisms of resistance to molecular targeted therapy in lung cancer. Proc Natl Acad Sci USA 107:15535–15540
Yasuda H et al (2012) EGFR exon 20 insertion mutations in non-small-cell lung cancer: preclinical data and clinical implications. Lancet Oncol 13:E23–E31
Yochum ZA et al (2019) Targeting the EMT transcription factor TWIST1 overcomes resistance to EGFR inhibitors in EGFR-mutant non-small-cell lung cancer. Oncogene 38:656–670
Yu HA et al (2014) Poor response to erlotinib in patients with tumors containing baseline EGFR T790M mutations found by routine clinical molecular testing. Ann Oncol 25:423–428
Zappa C, Mousa SA (2016) Non-small cell lung cancer: current treatment and future advances. Transl Lung Cancer Res 5:288–300
Zhang X-b, Peng F (2017) Expression level of microRNA-21 in peripheral blood and cancer tissues of patients with non-small cell lung cancer and its mechanism of involvement in cancer suppression by regulating PTEN protein. Zhongguo Laonianxue Zazhi 37:5571–5573
Zhang Y et al (2019a) The canonical TGF-beta/Smad signalling pathway is involved in PD-L1-induced primary resistance to EGFR-TKIs in EGFR-mutant non-small-cell lung cancer. Respir Res 20:164. https://doi.org/10.1186/s12931-019-1137-4
Zhang Z et al (2019b) Impact of MET alterations on targeted therapy with EGFR-tyrosine kinase inhibitors for EGFR-mutant lung cancer. Biomark Res 7:27. https://doi.org/10.1186/s40364-019-0179-6
Zhu JQ et al (2008) Better survival with EGFR exon 19 than exon 21 mutations in gefitinib-treated non-small cell lung cancer patients is due to differential inhibition of downstream signals. Cancer Lett 265:307–317
Funding
This work was supported by the Macao Science and Technology Development Fund (070/2017/A2, 0024/2020/A1), the Research Fund of the University of Macau (MYRG2018-00176-ICMS), Macao Young Scholars Program (AM201911) and the 2020 Guangdong Provincial Science and Technology Innovation Strategy Special Fund (Guangdong-Hong Kong-Macau Joint Lab) (2020B1212030006).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to this work.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, Y., Wang, H. & He, C. Drug resistance of targeted therapy for advanced non-small cell lung cancer harbored EGFR mutation: from mechanism analysis to clinical strategy. J Cancer Res Clin Oncol 147, 3653–3664 (2021). https://doi.org/10.1007/s00432-021-03828-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-021-03828-8