Abstract
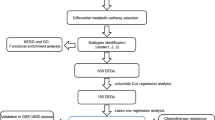
Although hepatocellular carcinoma (HCC) is rather frequent, little is known about the molecular pathways underlying its development, progression, and prognosis. In the current study, we comprehensively analyzed the deferentially expressed metabolism-related genes (MRGs) in HCC based on TCGA datasets attempting to discover the potentially prognostic genes in HCC. The up-regulated MRGs were further subjected to analyze their prognostic values and protein expressions. Twenty-seven genes were identified because their high expressions were significant in OS, PFS, DFS, DSS, and HCC tumor samples. They were then used for GO, KEGG, methylation, genetics changes, immune infiltration analyses. Moreover, we established a prognostic model in HCC using univariate assays and LASSO regression based on these MRGs. Additionally, we also found that SLC38A1, an amino acid metabolism closely related transporter, was a potential prognostic gene in HCC, and its function in HCC was further studied using experiments. We found that the knockdown of SLC38A1 notably suppressed the growth and migration of HCC cells. Further studies revealed that SLC38A1 modulated the development of HCC cells by regulating PI3K/AKT/mTOR signaling via glutamine mediated energy metabolism. In conclusion, this study identified the potentially prognostic MRGs in HCC and uncovered that SLC38A1 regulated HCC development and progression by regulating PI3K/AKT/mTOR signaling via glutamine mediated energy metabolism, which might provide a novel marker and potential therapeutic target in HCC.









Similar content being viewed by others
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Altman BJ, Stine ZE, Dang CV (2016) From Krebs to clinic: glutamine metabolism to cancer therapy. Nat Rev Cancer 16(10):619–634
Anderson PM, Lalla RV (2020) Glutamine for amelioration of radiation and chemotherapy associated mucositis during cancer therapy. Nutrients 12(6):1675
Böhme-Schäfer I, Lörentz S, Bosserhoff AK (2022) Role of amino acid transporter SNAT1/SLC38A1 in human melanoma. Cancers 14(9):2151
Boyd SD (2013) Diagnostic applications of high-throughput DNA sequencing. Annu Rev Pathol 8:381–410
Bradley RK, Anczuków O (2023) RNA splicing dysregulation and the hallmarks of cancer. Nat Rev Cancer 23(3):135–155
Certo M, Tsai CH, Pucino V, Ho PC, Mauro C (2021) Lactate modulation of immune responses in inflammatory versus tumour microenvironments. Nat Rev Immunol 21(3):151–161
Counihan JL, Grossman EA, Nomura DK (2018) Cancer metabolism: current understanding and therapies. Chem Rev 118(14):6893–6923
Cruzat V, Macedo Rogero M, Noel Keane K, Curi R, Newsholme P (2018) Glutamine: metabolism and immune function, supplementation and clinical translation. Nutrients 10(11):1564
DeBerardinis RJ, Chandel NS (2016) Fundamentals of cancer metabolism. Sci Adv 2(5):e1600200
Desvergne B, Michalik L, Wahli W (2006) Transcriptional regulation of metabolism. Physiol Rev 86(2):465–514
Dolina JS, Van Braeckel-Budimir N, Thomas GD, Salek-Ardakani S (2021) CD8(+) T cell exhaustion in cancer. Front Immunol 12:715234
El Jabbour T, Lagana SM, Lee H (2019) Update on hepatocellular carcinoma: pathologists’ review. World J Gastroenterol 25(14):1653–1665
Friedensohn S, Khan TA, Reddy ST (2017) Advanced methodologies in high-throughput sequencing of immune repertoires. Trends Biotechnol 35(3):203–214
Inoue T, Tanaka Y (2020) Novel biomarkers for the management of chronic hepatitis B. Clin Mol Hepatol 26(3):261–279
Johnson P, Zhou Q, Dao DY, Lo YMD (2022) Circulating biomarkers in the diagnosis and management of hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol 19(10):670–681
Kim H (2011) Glutamine as an immunonutrient. Yonsei Med J 52(6):892–897
Kim YC, Guan KL (2015) mTOR: a pharmacologic target for autophagy regulation. J Clin Investig 125(1):25–32
Kim MH, Kim H (2017) The roles of glutamine in the intestine and its implication in intestinal diseases. Int J Mol Sci 18(5):1051
Kim KH, Lee MS (2014) Autophagy—a key player in cellular and body metabolism. Nat Rev Endocrinol 10(6):322–337
Koch A, Joosten SC, Feng Z, de Ruijter TC, Draht MX, Melotte V, Smits KM, Veeck J, Herman JG, Van Neste L et al (2018) Analysis of DNA methylation in cancer: location revisited. Nat Rev Clin Oncol 15(7):459–466
Kyrysyuk O, Wucherpfennig KW (2023) Designing cancer immunotherapies that engage T cells and NK cells. Annu Rev Immunol 41:17–38
Liu L, Su S, Ye D, Yu Z, Lu W, Li X (2022) Long non-coding RNA OGFRP1 regulates cell proliferation and ferroptosis by miR-299-3p/SLC38A1 axis in lung cancer. Anticancer Drugs 33(9):826–839
Marsan E, Baulac S (2018) Review: mechanistic target of rapamycin (mTOR) pathway, focal cortical dysplasia and epilepsy. Neuropathol Appl Neurobiol 44(1):6–17
Meng H, Cao Y, Qin J, Song X, Zhang Q, Shi Y, Cao L (2015) DNA methylation, its mediators and genome integrity. Int J Biol Sci 11(5):604–617
Meng H, Niu R, Huang C, Li J (2022) Circular RNA as a novel biomarker and therapeutic target for HCC. Cells 11(12):1948
Moore LD, Le T, Fan G (2013) DNA methylation and its basic function. Neuropsychopharmacology 38(1):23–38
Mossmann D, Park S, Hall MN (2018) mTOR signalling and cellular metabolism are mutual determinants in cancer. Nat Rev Cancer 18(12):744–757
Nault JC, Villanueva A (2021) Biomarkers for hepatobiliary cancers. Hepatology (baltimore, MD) 73(Suppl 1):115–127
Nilsson RH, Anslan S, Bahram M, Wurzbacher C, Baldrian P, Tedersoo L (2019) Mycobiome diversity: high-throughput sequencing and identification of fungi. Nat Rev Microbiol 17(2):95–109
Nishikawa H, Osaki Y (2013) Non-B, non-C hepatocellular carcinoma (review). Int J Oncol 43(5):1333–1342
Pai JA, Satpathy AT (2021) High-throughput and single-cell T cell receptor sequencing technologies. Nat Methods 18(8):881–892
Palomeras S, Diaz-Lagares Á, Viñas G, Setien F, Ferreira HJ, Oliveras G, Crujeiras AB, Hernández A, Lum DH, Welm AL et al (2019) Epigenetic silencing of TGFBI confers resistance to trastuzumab in human breast cancer. Breast Cancer Res BCR 21(1):79
Pavlova NN, Thompson CB (2016) The emerging hallmarks of cancer metabolism. Cell Metab 23(1):27–47
Pipis M, Rossor AM, Laura M, Reilly MM (2019) Next-generation sequencing in Charcot-Marie-Tooth disease: opportunities and challenges. Nat Rev Neurol 15(11):644–656
Qureshi T, Bjørkmo M, Nordengen K, Gundersen V, Utheim TP, Watne LO, Storm-Mathisen J, Hassel B, Chaudhry FA (2020) Slc38a1 conveys astroglia-derived glutamine into GABAergic interneurons for neurotransmitter GABA synthesis. Cells 9(7):1686
Sangro B, Sarobe P, Hervás-Stubbs S, Melero I (2021) Advances in immunotherapy for hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol 18(8):525–543
Sankaran VG, Gallagher PG (2013) Applications of high-throughput DNA sequencing to benign hematology. Blood 122(22):3575–3582
Schlosser S, Tümen D, Volz B, Neumeyer K, Egler N, Kunst C, Tews HC, Schmid S, Kandulski A, Müller M et al (2022) HCC biomarkers—state of the old and outlook to future promising biomarkers and their potential in everyday clinical practice. Front Oncol 12:1016952
Siegel RL, Miller KD, Wagle NS, Jemal A (2023) Cancer statistics, 2023. CA Cancer J Clin 73(1):17–48
Strobel EJ, Yu AM, Lucks JB (2018) High-throughput determination of RNA structures. Nat Rev Genet 19(10):615–634
Takarada T, Ogura M, Nakamichi N, Kakuda T, Nakazato R, Kokubo H, Ikeno S, Nakamura S, Kutsukake T, Hinoi E et al (2016) Upregulation of Slc38a1 gene along with promotion of neurosphere growth and subsequent neuronal specification in undifferentiated neural progenitor cells exposed to theanine. Neurochem Res 41(1–2):5–15
Tapiero H, Mathé G, Couvreur P (2002) Tew KD: II. Glutamine and glutamate. Biomed Pharmacother 56(9):446–457
Tewari D, Patni P, Bishayee A, Sah AN, Bishayee A (2022) Natural products targeting the PI3K-Akt-mTOR signaling pathway in cancer: a novel therapeutic strategy. Semin Cancer Biol 80:1–17
Thommen DS, Schumacher TN (2018) T cell dysfunction in cancer. Cancer Cell 33(4):547–562
Tsuchiya N, Sawada Y, Endo I, Saito K, Uemura Y, Nakatsura T (2015) Biomarkers for the early diagnosis of hepatocellular carcinoma. World J Gastroenterol 21(37):10573–10583
Vahidi Y, Faghih Z, Talei AR, Doroudchi M, Ghaderi A (2018) Memory CD4(+) T cell subsets in tumor draining lymph nodes of breast cancer patients: a focus on T stem cell memory cells. Cell Oncol (dordrecht) 41(1):1–11
van der Lee M, Kriek M, Guchelaar HJ, Swen JJ (2020) Technologies for pharmacogenomics: a review. Genes 11(12):1456
van der Leun AM, Thommen DS, Schumacher TN (2020) CD8(+) T cell states in human cancer: insights from single-cell analysis. Nat Rev Cancer 20(4):218–232
Wang W, Wei C (2020) Advances in the early diagnosis of hepatocellular carcinoma. Genes Dis 7(3):308–319
Wang B, Wu G, Zhou Z, Dai Z, Sun Y, Ji Y, Li W, Wang W, Liu C, Han F et al (2015) Glutamine and intestinal barrier function. Amino Acids 47(10):2143–2154
Wang Z, Qin H, Liu S, Sheng J, Zhang X (2023) Precision diagnosis of hepatocellular carcinoma. Chin Med J 136(10):1155–1165
Wen N, Cai Y, Li F, Ye H, Tang W, Song P, Cheng N (2022) The clinical management of hepatocellular carcinoma worldwide: a concise review and comparison of current guidelines: 2022 update. Biosci Trends 16(1):20–30
Xie G, Dong H, Liang Y, Ham JD, Rizwan R, Chen J (2020) CAR-NK cells: a promising cellular immunotherapy for cancer. EBioMedicine 59:102975
Xue W, Dong B, Wang Y, Xie Y, Li P, Gong Z, Niu Z (2022) A novel prognostic index of stomach adenocarcinoma based on immunogenomic landscape analysis and immunotherapy options. Exp Mol Pathol 128:104832
Yang B, Wang JQ, Tan Y, Yuan R, Chen ZS, Zou C (2021) RNA methylation and cancer treatment. Pharmacol Res 174:105937
Yang M, Lu Y, Piao W, Jin H (2022) The translational regulation in mTOR Pathway. Biomolecules 12(6):802
Yu J, Chen X, Li J, Wang F (2021) CircRUNX1 functions as an oncogene in colorectal cancer by regulating circRUNX1/miR-485-5p/SLC38A1 axis. Eur J Clin Invest 51(7):e13540
Zhang H, Zhang Y, Dong J, Zuo S, Meng G, Wu J, Wei J (2021) Recombinant adenovirus expressing the fusion protein PD1PVR improves CD8(+) T cell-mediated antitumor efficacy with long-term tumor-specific immune surveillance in hepatocellular carcinoma. Cell Oncol (dordrecht) 44(6):1243–1255
Zucman-Rossi J, Villanueva A, Nault JC, Llovet JM (2015) Genetic landscape and biomarkers of hepatocellular carcinoma. Gastroenterology 149(5):1226-1239.e1224
Funding
There is no special funding for this article.
Author information
Authors and Affiliations
Contributions
Hua-guo Feng and Bin Xiong conceived the project, conducted the experiments, and prepared the manuscript. Chuan-xin Wu and Guo-chao Zhong assisted in the experiments. Jian-ping Gong and Chun-mu Miao performed the data analysis. Hua-guo Feng and Chun-mu Miao prepared figures, Chuan-xin Wu, Jian-ping Gong, and Bin Xiong edited manuscript. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Animal research (ethics)
This research did not involve animal experiments.
Ethics approval and informed consent
This research did not involve Ethical consent. Sequencing data of HCC patients from public databases (TCGA and ICGC datasets).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
432_2023_5360_MOESM1_ESM.tif
Supplementary file1 Supplementary Figure S1. Identification of HCC molecular subtypes based on TCGA database using DEGs. (A) The cumulative distribution function (CDF) Delta area curve of TCGA-LIHC samples. (B) The cumulative distribution function (CDF) curves, which is able to describe the probability distribution of a real random variable, and established using consensus clustering approach. (C) The 371 HCC samples were split into 2 clusters by the consensus clustering matrix (k = 2). (D) Color-coded heatmap related to the consensus matrix for k= 2 obtained by using consensus clustering. C1 has 104 HCC samples and C2 has 267 HCC samples. (E) The overall survival (OS) analysis of the two groups. G1 has 104 HCC samples and G2 has 267 HCC samples. (F) The progression-free survival (PFS) analysis of the two groups. G1 has 104 HCC samples and G2 has 267 HCC samples (TIF 4349 KB)
432_2023_5360_MOESM2_ESM.tif
Supplementary file2 Supplementary Figure S2. Identification of differentially expressed genes (DEGs) in the above-certified two HCC groups and functional enrichment analysis. (A) Heatmap of DEGs in the two HCC groups. G1 has 104 HCC samples and G2 has 267 HCC samples. (B) Volcano map of DEGs. (C and D) KEGG (Kyoto Encyclopedia of Genes and Genomes) analyses of up- and down-regulated DEGs. (E and F) GO (Gene Ontology) analyses of up- and down-regulated DEGs (TIF 5720 KB)
432_2023_5360_MOESM3_ESM.tif
Supplementary file3 Supplementary Figure S3. The expression analyses of SLC38A1 in pan-cancers. (A). SLC38A1 mRNA expression in pan-cancers which is analyzed by using TIMER 2.0 databse. (B) The protein expression of SLC38A1 in multiple cancer types which is analyzed by using HPA database. (C) SLC38A1 methylation analysis across TCGA cancer types which is analyzed by using GSCA database. (D) SLC38A1 mutation landscape in pan-cancers which is analyzed by using TIMER 2.0 databse (TIF 4132 KB)
432_2023_5360_MOESM4_ESM.tif
Supplementary file4 Supplementary Figure S4. Immune cell infiltration analyses of SLC38A1 in TCGA cancer types. The correlation between SLC38A1 expression and the levels of infiltration of CD4+ T cells, CAF, HSC, γδ T cells, MDSC, NKT, regulatory T cells (Tregs), B cells, neutrophils, monocytes, macrophages, dendritic cells (DC), NK cells, Mast cells, and CD8+ T cells in TCGA cancers (TIF 5213 KB)
432_2023_5360_MOESM5_ESM.tif
Supplementary file5 Supplementary Figure S5. Expression and prognosis analyses of SLC38A1 in HCC. (A) mRNA expression of SLC38A1 in HCC based on TCGA database. (B) Protein expression of SLC38A1 in HCC using UALCAN database. (C) The immunohistochemistry analysis of SLC38A1 expression in HCC tumor samples using HPA database. (D) SLC38A1 expression and survival status of HCC patients from TCGA datasets. (E) The overall survival analysis of SLC38A1 high and low expression groups. (F) The ROC curves with AUC values at 1-year, 3-year and 5-year (TIF 3609 KB)
432_2023_5360_MOESM6_ESM.tif
Supplementary file6 Supplementary Figure S6. Sankey diagrams presents the SLC38A1 expression and clinicopathological characteristics, and immune interacting network construction. (A-B) The correlation between SLC38A1 high and low expression and clinicopathological characteristics (age, gender, grade, pT stage, pN stage, pM stage, survival status). (C) The immune interacting network between SLC38A1 and kinds of immune cells in HCC (TIF 1423 KB)
432_2023_5360_MOESM7_ESM.tif
Supplementary file7 Supplementary Figure S7. The SLC38A1-gene interaction network and functional enrichment analyses of SLC38A1 co-expression genes. (A) Gene-gene interaction of SLC38A1 with other genes was generated by GeneMANIA. (B-D) GO analyses of SLC38A1 co-expression genes. BP, biological process, CC, cellular component, MF, molecular function. (E) KEGG analyses of SLC38A1 co-expression genes. KEGG, Kyoto Encyclopedia of Genes and Genomes (TIF 4164 KB)
432_2023_5360_MOESM8_ESM.tif
Supplementary file8 Supplementary Figure S8. SLC38A1 expression in HCC cancer cell lines. (A) SLC38A1 expression in 946 cell lines of various cancer types using CCLE database. (B) The SLC38A1 expression in 25 HCC cell lines based on CCLE dataset. (C) The qRT-PCR detected the SLC38A1 mRNA levels in SNU-449, SNU-423, SNU-398, JHH-2, SMMC-7721, Huh-7, HepG2, Hep3B HCC cell lines (TIF 1752 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Feng, Hg., Wu, Cx., Zhong, Gc. et al. Integrative analysis reveals that SLC38A1 promotes hepatocellular carcinoma development via PI3K/AKT/mTOR signaling via glutamine mediated energy metabolism. J Cancer Res Clin Oncol 149, 15879–15898 (2023). https://doi.org/10.1007/s00432-023-05360-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-05360-3