Abstract
The objective of this study was to investigate the parents’ attitudes towards and identify the possible factors associated with pandemic H1N1 vaccine uptake that was recommended to children between 6 months and 5 years of age. A questionnaire exploring the attitudes of parents to H1N1 vaccine was given to parents of children 6 through 60 months of age attending to Akdeniz and Gazi University Hospitals’ well-child departments between 15 November 2009 and 15 January 2010. The questionnaire included questions on demographic characteristics, parental perception of the severity of the pandemic, the presence of anyone in their environment who suffered from pandemic influenza, their decision on whether or not to vaccinate their child, the factors that influenced them during decision-making process and possible factors that might have influenced the opponents of their decision. Those who accepted to get their children vaccinated got it immediately, free of charge. Out of 611 parents who responded the questionnaire 226 (36.7%) had their children vaccinated. Parental education period of less than 12 years, not being a close relative of a health care worker, not having a relative who suffered from the disease, having a child younger than 36 months, being influenced by the relatives’ opinions or from the politicians or from the media all decreased vaccine acceptance. Factors that were most significantly associated with vaccine refusal were thinking that the pandemic was exaggerated (OR 9.44, 95% CI 4.28–20.82) and believing that other preventive measures were more effective than H1N1 vaccine (OR 15.61, 95% CI 7.37–33.08). Lessons learned from influenza H1N1/2009 pandemic may help national authorities, health care providers and media on how to keep the public well informed and find ways of better risk–benefit communication with the parents on vaccines.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
On 11 June 2009, the World Health Organization (WHO) declared the global spread of a novel influenza A (H1N1) pandemic. As of 1 August 2010, worldwide, more than 214 countries and overseas territories or communities have been reported to have confirmed cases of pandemic influenza H1N1 2009, including over 18,449 deaths [41]. In Turkey, a country that has nearly 74 million inhabitants, 656 deaths have occured due to influenza A (H1N1) during the pandemic.
After the procurement of monovalent pandemic H1N1 vaccine, the Turkish Ministry of Health started vaccination of target groups in November 2009. Initially, the focus was primarily on healthcare workers, children between the ages of 6 months through 5 years, pregnant or postpartum women, and people with underlying chronic health disorders. As more H1N1 vaccines became available, all child age groups as well as the general public were offered the vaccine. Vaccines were funded by the state and provided to the vaccinees at no cost. Vaccination was not mandatory for any of the groups. Although the government originally contracted with different vaccine companies for 44 million doses of pandemic vaccines, only 3 million doses could be administered.
Even though the good safety and immunogenicity profiles of pandemic vaccines have been acknowledged many times by international and national organizations, professional groups and scientific communities, their potential adverse effects have gathered much media attention in Turkey [3, 6, 12–14, 16, 19, 39]. Squalene-containing adjuvants have especially been largely debated and continuously questioned. In addition to some medical doctors and people dealing with alternative medicine, high-level politicians (including the prime minister and former health minister) were also involved in the discussions, and explained their own reasons for not getting the vaccine, on TV channels and newspapers.
Many countries ended up with low pandemic H1N1 vaccine coverage rates [4, 31, 32, 38]. So far, several studies have been published about the public response to these vaccines and factors that might have led to low vaccination rates [18, 25, 32, 35, 38]. Many of them searched intention to get the vaccine before vaccines were available and found that perception of the public about the severity of the pandemic or the disease, the cultural characteristics of eastern and western societies, different media coverages of pandemic vaccines according to political leanings of media groups, concerns about vaccine safety, health provider characteristics, or not receiving seasonal influenza vaccine had an effect on low vaccine coverage rates [10, 18, 26]. Most of those studies also emphasized that whether intention to get the vaccine would translate to behavior was not known then [8]. Recently, a limited number of studies have been published on parental attitudes towards pandemic vaccines when the vaccines were readily available [25, 37]. The aim of this study was to investigate the parents’ attitudes and identify the possible factors associated with pandemic H1N1 vaccine refusal that was recommended to children between 6 months and 5 years of age in Turkey.
Materials and methods
Participants
This study was conducted in well-child departments of two university hospitals (Gazi and Akdeniz University). Both departments serve to follow-up healthy children and vaccination of all children either healthy or in the risk groups, between 0 and 60 months of age.
Between 15 November 2009 and 15 January 2010, parents of all children 6 through 60 months of age were recommended to get their children vaccinated against pandemic influenza A/H1N1. Those who approved to get their children vaccinated got it free of charge. A questionairre was given to the parents regardless of whether they approved or refused the implementation of the vaccine to their children.
Questionnaire
The questionnaire included 14 questions on the following items: (a) demographics (b) parents’ perception on how pandemic flu can be prevented, (c) the presence of anyone in their environment who suffered from the pandemic, (d) whether they approve the immunization of their child against pandemic influenza, or not, (e) the factors that influenced them about their decision on approving or disapproving the vaccine (this question included a list of possible factors with a 3 point scale as “very much”, “somewhat”, or “none” for each item), (f) whether the programs on media, statements of the politicians, or the opinions of their relatives had effected their attitude towards vaccination (this question also had a three-point scale as above), and (g) in their opinion what possible factors might have influenced the others who just think the opposite decision is right (this question was asked regardless of the acceptance or refusal of the vaccine by the parent).
Statistical methods
The data were analyzed using the statistical package SPSS 13.0 for Windows (SPSS Inc., Chicago, IL, USA). Continuous variables are presented as mean ± SD, categorical variables are presented as%. In statistical analysis, chi-square test and logistic regression were performed to assess the independent effects of the predictors on acceptance of H1N1 vaccination. Statistical significance was considered as p < 0.05. For backward binary logistic regression, analysis factors that may decrease the acceptance of vaccine were evaluated by two models. In model ,1 the demographic factors were accepted as the risk factors for vaccine disapproval (i.e., parents younger than 35 years of age, parental education span less than 12 years, not having a close relative who is a health worker, age of the child ≤36 months, female child and having no close contact that had had the disease). In model 2, social factors such as being influenced by the news on the media, or from the attitudes of the politicians or the relatives, thinking that the measures other than vaccination are more effective for preventing the disease, and believing that the intensity of the pandemic is exaggerated were accepted as the risk factors for vaccine refusal.
The study was approved by the ethics committee of Gazi University.
Results
Of 611 children, between the ages of 6 to 60 months, whose families accepted to participate in the study 304 (49.7%) were male and 307 (50.3%) were female. Families of 226 (36.9%) accepted the vaccination of their children with pandemic H1N1 vaccine. The median age of the children was 18 months (range 6–60 months). The mean ages of the mothers and fathers were 30.6 ± 5.0 and 33.84 ± 5.5 years, respectively. A total of 131 (21.2%) parents stated that there were at least one person around them who suffered from the pandemic, most of them (44.6%) being the colleagues from workplace.
The demographic characteristics of the families with regard to vaccination status are summarized in Table 1. Parental education period of less than 12 years (p = 0.002), or not being a relative of a health care worker (p = 0.011) increased the refusal of the vaccine nearly twice. Having a child younger than 36 months (p = 0.012, OR 1.81, 95% CI 1.14–2.89) and not having a relative who suffered from the disease (p = 0.043, OR 1.55, 95% CI 1.04–2.38) were also associated with vaccine refusal.
Parental responses to questions about social factors that might have effected the acceptance of the vaccine are summarized in Table 2. The families who expressed that they were influenced by the relatives’ opinions were nearly four times and from the media or politicians were nearly five times more likely to refuse the vaccine for their children (p 0.0001). Most dramatic falls in the vaccine acceptance rates were seen in the children of the parents who thought that the severity of the pandemic was exaggerated (p = 0.0001, Odds Ratio 9.44, 95% CI 4.28–20.82) or who considered measures (like washing the hands, keeping away from crowded places, etc.) other than vaccination were more effective for preventing the disease (p = 0.0001, odds ratio 15.61, 95% CI 7.37–33.08).
Parents of the children who accepted or refused the vaccine were asked to rate a list of factors (on a three-point scale) that had affected them while deciding on the vaccine. Their responses are summarized in Table 3. Severity of the pandemic, physician’s recommendation on favor of the vaccine, and confidence on the effectiveness of the vaccine were the three most highly rated factors by the parents who approved the vaccine. Those parents who refused the vaccine most commonly stated that they were afraid of the adverse effects, measures other than vaccination can be more effective in averting the disease and that they did not believe there is a pandemic.
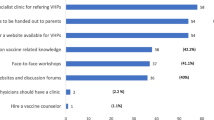
In order to reflect different views from the perspective of the society, we asked the parents what possible factors might have affected the opponents of their decision. The answers to this question are summarized in Table 4. Parents who accepted to get their children vaccinated thought that the others who did not accept it were either very much concerned about the side effects (37.8%), or were influenced by the media (24.4%) or they were ignorant about the vaccine (17.8%). On the other hand, parents who rejected the vaccine stated that the families who accepted the vaccine were wrongly believing that vaccination was the best way of protection (33.6%), they were exaggerating the pandemic (21.4%), or they were ignorant about the vaccine (19.5%).
Discussion
Vaccines are the most effective and easy way of protecting against certain diseases. It is the parents who decide whether their children will get a vaccine or not. It has been previously shown that as their level of education and access to information increase, parents are more willing to question the vaccines and medications that are offered to their children and want to participate more in the decision-making process [27]. Their attitudes are also known to be influenced by medical and social factors.
Although pandemic H1N1 vaccines were safe and effective, immunization rates remained low across all countries, including Turkey [31, 32, 34, 38]. According to the Turkish Ministry of Health, pandemic vaccine coverage among children younger than 14 years of age remained at 3.1%. Demographic characteristics of the families largely contributed to vaccine acceptance or refusal, even during a pandemic period. In our study group, parents were more likely to vaccinate older children. This may either be caused by parental perception about younger children being more prone to vaccine side effects or the belief that it is easier to keep younger children at home. As the child grows older, the frequency of attending to nursery increases which may in turn change the parent’s perception about the vulnerability of the child to the disease. A study carried out in Canada, on barriers to vaccination of the children, showed that parental beliefs including babies are too small, immature, or fragile to handle immunizations might have an effect on low vaccine coverage rates [24]. On the other hand, age-specific seasonal influenza vaccine coverage rates of children aged between 6 and 59 months in the USA during 2008–2009 have revealed a low coverage rate among children who were older. This difference was attributed to the more frequent physician visits of the infants which may result in a higher chance of completing the vaccine series [5].
Our results, like many others, have revealed that children of parents with a higher educational level were more likely to be vaccinated [7, 11]. This may be due to a more effective risk–benefit communication between the health care staff and the more educated parents, about personal risks versus the safety and effectiveness of the preventive maneuver [29]. Parents with a higher education may also be more prone to accept scientific knowledge instead of non-expert opinions on the media. Vaccine uptake of children with parents who have a health care worker friend or relative was higher. Since access to pandemic vaccines was made very easy to the general public, this difference is unlikely to result from the barriers in obtaining the vaccine.
Many others have shown that concerns about vaccine safety is the most important risk factor for low vaccine acceptance [10, 18, 22]. Parents who refused the vaccine have stated their fears during the study. Also, those who accepted the vaccine rightly emphasized that parents who refused the vaccine were afraid of the side effects. Such fears about vaccine safety may lead to dramatic falls in vaccine acceptance rates. The decrease in vaccine coverage rate of whole cell pertussis vaccine in Japan due to concerns about encephalopathy, low MMR acceptance rates due to rumors about autism, and low uptake of hepatitis B vaccines containing thiomersal are well-known examples, some of which even led to outbreaks [15, 20, 23, 27]. The same association between parental fears and low uptake of seasonal inluenza vaccine is also shown [1, 8].
While seasonal influenza vaccines were in use for several years, for certain risk groups in Turkey, their adverse effects were not questioned as much as the pandemic ones. Since many news on the media laid emphasis on allegedly “fast-track approval” of the vaccine, which was though not faster than the seasonal ones, possibly the public was predisposed to pay more attention on the potential adverse effects [2]. Another debate was on the adjuvant (squalene) and preservative (thiomersal) contents of the vaccine, which were both brand new topics for Turkish media and the public. We believe that all of these factors have contributed to the perception of the public that this was a totally new vaccine which suddenly emerged with inadequate vaccine safety testing. Studies have shown that sometimes it is hard for the society to accept a new vaccine. For example, a qualitative study on rotavirus vaccine acceptance of the parents showed that even though they believed that the disease was severe and the vaccine was effective, only 35% would accept vaccination of their infants with it [28].
Setbon et al. [35] revealed that the low intention to get the pandemic vaccine in France was related to the perception that the pandemic was not serious. Other studies also showed that willingness to vaccination decreases when the perceived risk of the disease or complications are low [10, 18, 22].
Physicians had a positive effect on the parents who accepted the vaccine. The role of health care providers is crucial in vaccine risk and benefit communication with the parents [27, 33]. Other studies on vaccines to prevent otitis media and on seasonal influenza vaccines revealed that parents value their physicians recommendations [9, 17]. However, the providers’ perception of vaccines also contribute to vaccine uptake. A study by Salmon et al. [33] revealed that vaccine providers of unvaccinated children had lesser confidence in vaccine safety and more propensity to believe that multiple vaccines weaken child’s immune system, compared to the providers of the vaccinated children. Since the providers of both vaccinated and unvaccinated children were the same in our study group, we believe that other social and demographic factors have contributed to this result.
Parents who believed that infection can be prevented with other precautionary measures such as nutrition, hand washing, keeping away from crowded places were 15 times less likely to vaccinate their children. This shows how important it is to explain to the public why vaccines are more effective in providing specific profection against certain diseases. Parents of unvaccinated children more frequently stated that they were influenced by their friends’ and relatives’ attitudes towards the vaccine. Others have also shown that parents have a tendency to rely on general opinion of the public on accepting or refusing a vaccine [21]. This tendency have been summarized as “science by consensus” (i.e., if many people make the same claim, it must be true). Maybe for some of the parents, science by consensus was more assuring than science by evidence [36].
Under extraordinary conditions that threatens public health, such as a pandemic, the media has the social responsibility of keeping the subject in their agenda with thorough discussions based on true information in order to increase the level of knowledge, awareness, and attention on the topic. Results of the present study showed that media and statements of the politicians, which are not adequate sources of scientific evidence, can largely affect the decisions of parents on a medical intervention. In France, while the favorable comments of scientific papers and medical doctors on pandemic vaccine, newspapers and Google were not so favorable about it and sometimes even worrying [26]. Nougairede et al. [26] revealed that the source of public information was an important determinant of vaccine acceptance. A study regarding health care workers’ acceptance of vaccine in Greece, also showed that the professionals who used expert opinions, disease control centers, or medical journals as the source of information were more likely to get the vaccine compared to those who used media as the source [30]. In a recent review by Poland [29], the population’s mistrust on the governmental health reports (leading to lack of concern regarding personal risk) added to the concerns regarding vaccine safety, which is mostly fueled by media in Turkey, were exhibited to be important risk factors for low vaccine uptake.
Out of 656 deaths that occured due to the pandemic, 578 were during the massive availability of vaccines in Turkey. Among the 3 million vaccinees, none died or permanently sequeled by the vaccine. Should there be a more serious pandemic, the consequences of being unimmunized would be more dramatic. Lessons learned from this pandemic may help national authorities, health care providers, and media on how to keep the public well informed and find ways of better risk–benefit communication with the parents on vaccines. Otherwise, uninformed discussions on any public health intervention, including but not restricted to old and new vaccines, may lead to unintended outcomes. Lastly, we would like to emphasize that continued refusal of the parents after adequate discussions based on existing evidence should be respected, unless the child bears a risk of serious harm.
Limitations of the study
Many studies show that prior receipt of seasonal influenza vaccine is strongly correlated with pandemic vaccine acceptance. In Turkey, seasonal vaccines are provided for free only to the risk groups with underlying chronic conditions, others who wish to be vaccinated need to pay for it. Thus, it was impossible to look for a possible correlation between seasonal and pandemic influenza vaccine uptake in our study group.
Another limitation of the study is that people who accepted the vaccine were overly represented in this study compared to the general public. We believe that this was due to the design of our study. Since it was carried out in two vaccine centers, probably, many of the attendees were already willing to get the vaccine when they admitted to the two centers. Other explanations for this discrepancy can be better education level of the parents in the study group compared to the general public, prefering a hospital-based setting instead of primary care or private offices for this “new” vaccine, and provider characteristics such as being more able to spend time on vaccine issues and guidance compared to general practitioners overwhelmed by the workload during the pandemic [7, 11, 27, 33, 40].
References
Allred NJ, Shaw KM, Santibanez TA, Rickert DL, Santoli JM (2005) Parental vaccine safety concerns: results from the national ımmunization survey, 2001–2002. Am J Prev Med 28:221–224
Brown KF, Kroll JS, Hudson MJ, Ramsay M, Green J, Vincent CA et al (2010) Omission bias and vaccine rejection by parents of healthy children: implications for the influenza A/H1N1 vaccination programme. Vaccine 28:4181–4185
Caillet C, Durrieu G, Jacquet A, Faucher A, Ouaret S, Perrault-Pochat MC, et al. (2011). Safety surveillance of influenza A (H1N1)v monovalent vaccines during the mass vaccination campaign in France. Eur J Clin Pharmacol (December 14, epub ahead of print). doi:10.1007/s00228-010-0961-8.
Centers for Disease Control and Prevention (CDC) (2010) Interim results: state-specific influenza A (H1N1) 2009 monovalent vaccination coverage—United States, October 2009–January 2010. MMWR 59:363–368
Centers for Disease Control and Prevention (CDC) (2009) Influenza vaccination coverage among children aged 6 months–18 years—eight immunization information system sentinel sites, United States, 2008–09 influenza season. MMWR 58:1059–1062
Clark TW, Pareek M, Hoschler K, Dillon H, Nicholson KG, Groth N et al (2009) Trial of 2009 influenza A (H1N1) monovalent MF59-adjuvanted vaccine. N Engl J Med 361:2424–2435
Daley MF, Crane LA, Chandramouli V, Beaty BL, Barrow J, Allred N et al (2006) Influenza among healthy young children: changes in parental attitudes and predictors of immunization during the 2003 to 2004 influenza season. Pediatrics 117:e268–e277
Daley MF, Crane LA, Chandramouli V, Beaty BL, Barrow J, Allred N et al (2007) Misperceptions about influenza vaccination among parents of healthy young children. Clin Pediatr (Phila) 46:408–417
Dubé E, De Wals P, Gilca V, Boulianne N, Ouakki M, Lavoie F et al (2009) New vaccines offering a larger spectrum of protection against acute otitis media: will parents be willing to have their children immunized? Int J Pediatr Otorhinolaryngol 73:987–991
Eastwood K, Durrheim DN, Jones A, Butler M (2010) Acceptance of pandemic (H1N1) 2009 influenza vaccination by the Australian public. Med J Aust 192:33–36
Elliott C, Farmer K (2006) Immunization status of children under 7 years in the Vikas Nagar area, North India. Child Care Health Dev 32:415–421
European Medicines Agency (2009). Press release at 20 November 2009. European Medicines Agency reaffirms efficacy and safety of pandemic H1N1 pandemic vaccines. Ref: EMEA/748707/2009.
European Medicines Agency (2009). Pandemic pharmacovigilance weekly update, status at 27 November 2009. Ref. EMEA/784681/2009.
European Medicines Agency (2010). Twenty-second pandemic pharmacovigilance update at 19 August 2010. Ref. EMEA/527985/2010.
Flynn M, Ogden J (2004) Predicting uptake of MMR vaccination: a prospective questionnaire study. Br J Gen Pract 54:526–530
Greenberg ME, Lai MH, Hartel GF, Wichems CH, Gittleson C, Bennet J et al (2009) Response to a monovalent 2009 influenza A (H1N1) vaccine. N Engl J Med 361:2405–2413
Hemingway CO, Poehling KA (2004) Change in recommendation affects influenza vaccinations among children 6 to 59 months of age. Pediatrics 114:948–952
Horney JA, Moore Z, Davis M, MacDonald PD (2010) Intent to receive pandemic influenza A (H1N1) vaccine, compliance with social distancing and sources of information in NC, 2009. PLoS ONE 5:e11226
Johansen K, Nicoll A, Ciancio BC, Kramarz P (2009). Pandemic influenza A (H1N1) 2009 vaccines in the European Union. Euro Surveill 14:pii=19361. (VS, VE)
Kimura M (1991) Japanese clinical experiences with acellular pertussis vaccines. Dev Biol Stand 73:5–9
Leask J, Macartney K (2008) Parental decisions about vaccination: collective values are important. J Paediatr Child Health 44:534–535
Leo HL, Clark SJ, Butchart AT, Singer DC, Clark NM, Davis MM (2010) 2009 seasonal and H1N1 influenza vaccination compliance in asthmatic children and adults. J Allergy Clin Immunol 126:166–168
Luman ET, Fiore AE, Strine TW, Barker LE (2004) Impact of thimerosal-related changes in hepatitis B vaccine birth-dose recommendations on childhood vaccination coverage. JAMA 29:2351–2358
Niederhauser VP, Markowitz M (2007) Barriers to immunizations: multiethnic parents of under- and unimmunized children speak. J Am Acad Nurse Pract 19:15–23
Nikolakopoulou NM (2010) H1N1 vaccine acceptance among parents in Greece. Int J Adolesc Med Health 22:339–340
Nougairède A, Lagier JC, Ninove L, Sartor C, Badiaga S, Botelho E et al (2010) Likely correlation between sources of information and acceptability of A/H1N1 swine-origin influenza virus vaccine in Marseille, France. PLoS One 5:e11292
Omer SB, Salmon A, Orenstein WA, de Hart MP, Halsey N (2009) Vaccine refusal, mandatory immunization, and the risks of vaccine-preventable diseases. N Engl J Med 360:1981–1988
Patel MM, Janssen AP, Tardif RR, Herring M, Parashar UD (2007) A qualitative assessment of factors influencing acceptance of a new rotavirus vaccine among health care providers and consumers. BMC Pediatr 7:32
Poland GA (2010) The 2009–2010 influenza pandemic: effects on pandemic and seasonal vaccine uptake and lessons learned for seasonal vaccination campaigns. Vaccine 285:D3–D13
Rachiotis G, Mouchtouri VA, Kremastinou J, Gourgoulianis K, Hadjichristodoulou C (2010). Low acceptance of vaccination against the 2009 pandemic influenza A(H1N1) among healtcare workers in Greece. Euro Surveill 15
Rizzo C, Rota MC, Bella A, Giannitelli S, De Santis S, Nacca G, Pompa MG, Vellucci L, Salmaso S, Declich S (2010). Response to the 2009 influenza A(H1N1) pandemic in Italy. Euro Surveill. 15(49)
Rodríguez-Rieiro C, Esteban-Vasallo MD, Domínguez-Berjón MF, Astray-Mochales J, Iniesta-Fornies D, Barranco-Ordoñez D et al (2011) Coverage and predictors of vaccination against 2009 pandemic H1N1 influenza in Madrid, Spain. Vaccine 29:1332–1338
Salmon DA, Pan WK, Omer SB, Navar AM, Orenstein W, Marcuse EK et al (2008) Vaccine knowledge and practices of primary care providers of exempt vs vaccinated chidren. Hum Vaccin 4:286–291
Schwarzinger M, Verger P, Guerville MA, Aubry C, Rolland S, Obadia Y et al (2010) Positive attitudes of French general practitioners towards A/H1N1 influenza-pandemic vaccination: a missed opportunity to increase vaccination uptakes in the general public? Vaccine 28:2743–2748
Setbon M, Raude J (2010) Factors in vaccination intention against the pandemic influenza A/H1N1. Eur J Public Health 20:490–494
Smith MJ, Marshall GS (2010) Navigating parental vaccine hesitancy. Pediatr Ann 39:476–482
Torun SD, Torun F, Catak B (2010) Healthcare workers as parents: attitudes toward vaccinating their children against pandemic influenza A/H1N1. BMC Public Health 10:596
Van-Tam J, Lambert PH, Carrasco P, Tschanz B, Leppo K (2010). Review of Switzerland’s H1N1 immunization strategy. Final Report. Available online: http://www.bag.admin.ch
Valencio M, Kissling E, Cohen JM, Oroszi B, Barret AS, Rizzo C et al (2011) Estimates of pandemic vaccine effectiveness in Europe, 2009–2010: results of influenza monitoring vaccine effectiveness in Europe (I-MOVE) multicenter case-control study. PLoS Med 9(1):e1000388. doi:10.1371/journal.pmed.1000388
Vaux S, Van Cauteren D, Guthmann JP, Strat YL, Vaillant V, de Valk H, Levy-Bruhl D (2011) Influenza vaccination coverage against seasonal and pandemic influenza and their determinants in France: a cross sectional survey. BMC Public Health 11:30
World Health Organization. Pandemic (H1N1) 2009—Update 112. URL. http://www.who.int/csr/don/2010_08_06/en/index.html, date of last access 31 August 2010.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Akıs, S., Velıpasaoglu, S., Camurdan, A.D. et al. Factors associated with parental acceptance and refusal of pandemic influenza A/H1N1 vaccine in Turkey. Eur J Pediatr 170, 1165–1172 (2011). https://doi.org/10.1007/s00431-011-1425-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-011-1425-6