Abstract
Diagnostic pathology activities are largely based on fixation of tissues in 4% formaldehyde, which has recently been re-classified as a carcinogenic compound and banned in several countries. Hospitals that do not have in-house pathology services need to send surgical and biopsy specimens to referral centers. These are generally transferred in liquid containers, under suboptimal safety conditions, as accidental spillage of potentially dangerous substances may occur. A safe, innovative, two-step procedure for pathology sample transportation is presented. Formalin-fixed material from ten surgical cases was dissected (including surrogate biopsies) and preserved in liquid-free plastic bags under vacuum for up to 30 days and subsequently processed for conventional histology, several immunohistochemical markers, and molecular tests (e.g., RAS mutation). The data were compared with the corresponding routine analyses. Formalin-fixed specimens after up to 30 days under vacuum storage gave equivalent results compared to standard histopathological slides and molecular tests, regarding both hematoxylin-eosin, immuno-stained slides and also nucleic acid extracted for molecular tests. The proposal of under-vacuum sealing pathology specimens that were previously formalin fixed can be adopted to transfer liquid-free biopsy and surgical specimens to referral pathology services. In fact, it is easy to perform, less expensive (both plastic bags and domestic-type vacuum chamber machines are at affordable costs), and above all is fully safe and adequate in the pre-analytical processing of pathology specimens.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A significant number of clinics and community hospitals in different parts of the world, and particularly in remote areas, have no in-house pathology services. It derives that surgical specimens and biopsy samples require transportation to the referral pathology center for microscopic analysis. In histopathology, the diagnostic process follows several steps, including crucial pre-analytical procedures that may deeply affect the accuracy of the final pathology report and basically depend on timely fixation of samples, as well as on an efficient transfer of material to pathology services [1, 2]. In countries with poor health facilities or remote hospital services, the transportation may imply long distances and usually specimens are transferred immersed in liquid formalin to avoid wasting of the biological properties. Often, samples are shipped by couriers or ordinary mail systems, and along this process when liquids are shipped, the safety conditions are suboptimal, especially if containing potentially dangerous substances in the solution (i.e., formaldehyde) [3].
Since the original description of its tissue fixation properties by Blum in 1893 [4], fixation in aqueous formaldehyde solution (formalin) is the most widely used preservation method in routine pathology. In general, immersion of the fresh specimen in liquid formalin for 6 up to 72 h gives an optimal tissue and cellular preservation, and degradation or autolysis is definitely avoided. In the daily practice, however, the pathology samples may remain immersed in liquid formalin until processing, sometimes even several days.
Due to its toxicity [5, 6] and its recent re-classification as a potentially carcinogenic compound [7, 8], the use of formalin has been banned or drastically reduced in several countries, including different work places, industry, and hospitals [the European Community (EC) regulation no. 1907/2006 was updated by the European Union (EU) Commission Regulation no. 895/2014 and is effective in the EU since August 2017]. At present, however, there is no valid alternative accepted by the scientific community and formalin is still ordinarily used worldwide, since the balance between the diagnostic and scientific importance (i.e., patient’s health) and the protection of workers is set on a risk reduction basis defined as the “As Low As Reasonably Achievable (Risk)” principle. In Italy, as in other countries of the European Union, this also results in strict obligations to protect the health of workers that manage formaldehyde. Therefore, it is essential to implement every possible technical, organizational, and procedural action to minimize the risk to the worker’s health [9]. In line with such scenario is the possibility of avoiding immediate formalin fixation in the surgical theater and rather favors the transfer of fresh pathology specimens under vacuum conditions and low temperatures, as adopted in our hospital [10].
Apart from the abovementioned harmful effects, another disadvantage of formalin (as of all liquid substances) is that during specimen transportation, an accidental spillage of the liquid may occur. For safety reasons, proper packaging, handling, and storage of formalin-containing samples are therefore of critical importance during all transportation steps. In clinics and community hospitals, especially those located in remote areas, where it is common practice to immerse tissues in formalin and send the container by post to the reference pathology laboratory, this procedure is cumbersome and potentially harmful. We designed an innovative procedure, intended to make the transportation of formalin-fixed specimens safer and lighter, thus ultimately cheaper.
We here show that the under-vacuum preservation in liquid-free plastic bags of already appropriately formalin-fixed tissues allows to safely store and transport pathology specimens at room temperature in a time frame at least up to 30 days. In order to test the feasibility of the procedure, we evaluated the quality of preservation of biopsy and surgical specimens, first routinely fixed in formalin and then removed from the liquid and stored in plastic bags under vacuum for variable periods of time.
Materials and methods
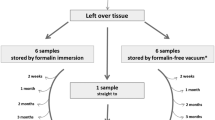
Ten specimens of different origins and diseases were randomly selected from the gross specimen room of the pathology division of the University of Torino in August 2017. All these cases had been fixed in formaldehyde for 2 to 5 days after surgical resection and represented the excess tissue to be eliminated after the case was reported and signed out. These cases included lung adenocarcinoma (4), colorectal carcinoma (1), colorectal carcinoma with polyposis (1), bladder carcinoma (1), breast carcinoma (1), gastroduodenal cancer (1), and a spleen (1) removed along with pancreatectomy. The description of surgical specimens included in this study is summarized in Table 1. A portion of the formalin-fixed surgical specimen, including part of the tumor (whenever present) and normal parenchyma, measuring approximately 7 × 8 × 3 cm was dissected and immediately anonymized by a pathology staff member not involved in the current study. The samples were then placed, liquid-free, into a plastic bag of the appropriate size. After recording only the date of sampling and the type of tissue and disease, the dedicated plastic bag was processed in the TissueSAFE Biospecimen vacuum sealing system (Milestone, Sorisole, Bergamo, Italy) (Fig. 1). The same procedure was repeated on another, similar sample of the same tissues using commercial plastic bags (size 25 × 30 cm, thickness 0.15 mm, by ORVED, Musile di Piave, Venice, Italy). These latter bags were processed for vacuum sealing using a commercial vacuum chamber machine, of the type normally used for foods (CVE 420 Vacuum sealer, VALKO, Bottanuco, Bergamo, Italy).
Two-step procedure for safe transportation under vacuum of liquid-free pathology specimens. Formalin fixation is followed by vacuum preservation (surgical specimen and surrogate biopsy sample in a vial to protect the small fragment). After transferred to pathology laboratory, the plastic bag under vacuum is opened for tissue sampling and histopathological analysis (step 3)
Samples were stored under vacuum for a variable time ranging from 7 to 30 days, either at room or at higher temperatures (up to 35 °C), in order to mimic possible environmental circumstances during transportation. Furthermore, some cases were shipped in an ordinary envelope by ordinary mail to our own address within the city (turnaround time of 7 days). During the entire duration of the study, the under-vacuum conditions were monitored and found to be maintained in all samples.
After 7 days, the cases were processed as follows: each sealed bag was opened, a representative fragment was processed for paraffin embedding, and routine hematoxylin and eosin staining was performed, following standard procedures (Leica ASP 300 processor and automated Leica ST5020 Multistainer, Leica Microsystems, Wetzlar, Germany) [11]. Then, the plastic bags were sealed again under vacuum, stored at room or higher temperatures, and the same procedure was repeated after 30 days with a new sampling of an adjacent tumor/tissue area. Finally, the bags were sealed again and stored (Fig. 1).
Finally, on four neoplastic cases (three lung carcinomas, one breast carcinoma), at the time of dissection of the portion for vacuum sealing, we performed needle biopsies with a cutting needle (Biopsy needle, Biopsybell, Modena, Italy) and also simulated an interventional endoscopy procedure on a surgical specimen of colonic cancer within polyposis by resecting small fragments of a vegetating tumor. In needle biopsies, we obtained approximately 2-cm long tissue cores that were simultaneously sealed under vacuum in a separate plastic bag, for the purpose of simulating the transportation of small biopsies with this newly devised system, as well. To protect the tissue and to avoid losing of small fragments in the plastic bags during the vacuum procedure, the cores were stored in a small Eppendorf vial (Fig. 1).
All such samples, obtained after 7 and 30 days from each case, as well as the original hematoxylin and eosin (H&E) stained slides, were comparatively evaluated (independently) by three of us (JM, GB, MP) with no knowledge of the time of sampling. The diagnostic quality of the glass slide was evaluated on whole H&E-stained sections, assessing the staining quality in general (clarity, uniformity, crispness) and in particular adequate nuclear and cytoplasmic staining, as well as tissue architecture and the cellular details.
Multicore tissue microarrays
For each case, tumor slides were evaluated to select representative areas for the multicore tissue microarray (TMA) construction. Using the Galileo TMA CK 3500 tissue array (Integrated Systems Engineering Srl, Milan, Italy), tissue cylinders with a diameter of 1 mm were punched from the specific areas of the “donor” block and brought into the “recipient” paraffin block, as previously described [12].
Immunohistochemical analysis
Using TMA blocks, we performed immunohistochemical (IHC) analysis using the following commonly used markers: TTF1, CD20, CD3, CEA, EMA, CD31, Vimentin, cytokeratin (CK), Ki67, S100, CD45, p40, CD68, p53, and mucin stain for each case. The IHC protocol is summarized in Table 2. Positive and negative controls were included for each immunohistochemical run.
Extraction, quantification, and quality assessment of DNA
Genomic DNA was isolated from three 10-μm-thick paraffin-embedded tissue sections obtained from selected cases (namely one breast and one colon carcinoma samples stored for 7 and 30 days under vacuum). Sections were deparaffinized in xylene followed by three incubations in 100% ethanol. After overnight incubation at 56 °C with proteinase K, DNA was isolated using the QIAamp DNA FFPE Tissue Kit (Qiagen) according to manufacturer’s protocol. DNA concentration was assessed using a NanoDrop spectrophotometer (Thermo Fisher Scientific). DNA quality was assessed by PCR, as previously described [13, 14]. Briefly, 100 ng of genomic DNA was analyzed for each sample. The PCR reaction was performed with four primer sets that produce 100, 200, 300, and 400 bp fragments from non-overlapping target sites in the GAPDH gene (chr12) on a PTC-100 Peltier Thermal Cycler (MJ Research, Inc.). The PCR products were separated by electrophoresis on ethidium bromide-stained 2% agarose gels. To reduce the risk of contamination from previously amplified products, separate areas were used for DNA isolation, amplification, and electrophoresis.
Extraction, quantification, and quality assessment of RNA
Total RNA was isolated from three 10-μm-thick paraffin-embedded tissue sections.
Sections were deparaffinized in xylene followed by three incubations in 100% ethanol. After overnight incubation at 55 °C with proteinase K, RNA was isolated using the Roche FFPET RNA Isolation Kit (Roche) according to manufacturer’s instructions. Total RNA concentration was assessed using a NanoDrop spectrophotometer (Thermo Fisher Scientific).
RNA integrity was assessed with Agilent 2100 Bioanalyzer (Agilent Technologies), using Agilent RNA 6000 Nano Kit, as previously described [9, 15]. In brief, the size distribution of the RNA fragments was calculated from Agilent 2100 Bioanalyzer readings using a Smear Analysis with a 200-nt threshold: the percentage of RNA fragments > 200 nt in size (DV200 metric) was recorded. In addition, RNA integrity number (RIN) was noted for each analyzed sample.
Gene mutation analysis
The DNAs were amplified according to the manufacturer’s instructions on a Labcycler (SensoQuest GmbH, Germany), using the CE-IVD Myriapod Colon Status kit (Diatech Pharmacogenetics, Jesi, Italy) based on a multiplex PCR. The amplified DNAs were submitted to a Shrimp Alkaline Phosphatase digestion (SAP reaction) to remove the excess of free nucleotides. Finally, the purified DNAs underwent a reaction of primer extension (iPLEX) with oligonucleotides flanking each gene position under study and with dideoxynucleotide terminators of known mass to discriminate wild-type from mutated genotypes, by using a matrix-assisted laser desorption ionization time-of-flight mass spectrometry. Amplified DNAs were dispensed on a supplied solid chip prior to be scanned on a MassARRAY instrument (Sequenom Inc., San Diego, CA, USA). The Myriapod Colon Status kit identifies 58, 54, 23, and 66 nucleotide substitutions in the KRAS, NRAS, BRAF, and PIK3CA genes, respectively [16].
Results
The under-vacuum sealing procedures performed with the certified instruments from Milestone (TissueSAFE) and using the official plastic bags produced by the same company provided an excellent preservation of the samples and of the vacuum conditions. Since the specimens here investigated were already fixed in formalin, the risk of spillover of fresh blood was obviously non-existing and indeed no other liquid was present in the plastic bags. In addition, when the same procedure was repeated using commercial plastic bags of slightly lower thickness (0.15 mm) and a vacuum chamber machine of the type used in food industry or in domestic environments, the quality of tissue preservation and vacuum maintenance was globally overlapping. Despite the higher reliability of specifically designed plastic bags and vacuum apparatus, this preliminary comparative analysis of the efficacy of the newly devised combined two step procedure was a compromise in order to assess the applicability of the new proposal also in extreme conditions, as those commonly encountered in remote hospitals and/or in low-income countries.
The independent and blind evaluation of the three hematoxylin and eosin stained slides corresponding to the original slide, the one taken after 7 days under vacuum and that after 30 days, gave comparable results among the three observers with the final conclusion that the three samples were indistinguishable. This unanimous impression was shared for both surgical specimens and for the surrogate biopsies (core needle or “pseudo-endoscopic” fragments of a polyp) taken from the formalin-fixed specimens and sealed under vacuum in similar conditions. The staining quality of H&E sections was evaluated as satisfactory and equivalent compared to routine slides in all aspects, including clarity, uniformity, and crispness of the glass slides, as well as staining of nuclear and cytoplasmic details. Tissue architecture and cellular elements were well preserved. The slides obtained from under-vacuum tissue did not show any difference regarding diagnostic utility compared to original slides (Fig. 2).
Examples of pre-fixed surgical or surrogate biopsy specimens, stored under vacuum for 7 to 30 days and then processed for light microscopy or immunohistochemistry. Hematoxylin- and eosin-stained slides of duodenum (a, b, after 7 days of under-vacuum preservation), surrogate biopsy of a fragment of colonic adenoma found in a surgical specimen (c, after 7 days of under-vacuum preservation), surrogate core biopsy in a resected breast invasive ductal carcinoma (d, and high magnification of the insert in e, after 30 days of under-vacuum preservation). Vimentin staining in a sample of breast carcinoma confirms antigen preservation after 30 days under vacuum (f) [original magnifications: a, c, d × 40; b, e, f × 200]
All tissues included in this study had the expected positive or negative expression of tested markers (TTF1, CD20, CD3, CEA, EMA, CD31, Vimentin, CK, Ki67, S100, CD45, p40, CD68, p53, and mucin stain).
Quantification and quality assessment of DNA and RNA
To evaluate the preservation of nucleic acids, DNA and RNA were extracted from standard-fixed and under-vacuum stored samples. The PCR assay providing information about the size range of fragments was used to assess DNA quality. In all the vacuum-stored specimens, DNA amplification was consistently successful at 100 and 200 bp and also occasionally at higher length (one sample after 7 days storage allowed the amplification of GAPDH up to 300 bp). Also, RNA quality, determined by Agilent Bioanalyzer traces, was satisfactory: the percentage of RNA fragments greater than 200 nt (DV200) was superior to 90% in all under-vacuum samples (Fig. 3a, b). Additionally, RIN for breast cancer sample after 7-day and 30-day storage was recorded at 2.50 and 2.10, respectively, while for the colon cancer, sample resulted at 2.70 for both storage periods.
a DNA amplification was found at 100 and 200 bp and also occasionally at higher length (one sample after 7 days storage allowed the amplification of GAPDH up to 300 bp) [MW, molecular weight; 1, colon cancer sample after 7-day storage; 2, colon cancer sample after 30-day storage; 3, breast cancer sample after 7-day storage; 4, breast cancer sample after 30-day storage; C+ positive control; C− negative control]. b The percentage of RNA fragments greater than 200 nt (DV200) superior to 90% was found in all under-vacuum samples [sample 1, breast cancer case; sample 2, colon cancer case]
Gene mutation analysis
In the case of colorectal carcinoma, the occurrence of KRAS/NRAS/BRAF/PIK3CA gene mutations was evaluated using Sequenom analysis, and a mutation at codon 61 of NRAS gene (c.182A>G, p.Gln61Arg, p.Q61R) and at codon 542 of PIK3CA gene (c.1624G>A, p.Glu542Lys, p.E542K) was detected, while KRAS and BRAF genes resulted as wild type. These results were compared with routine samples that identified the same mutations.
Discussion
The quality of pathology slides for the purpose of histological diagnosis of infectious or of non-neoplastic diseases, as well as of tumors, is crucial, and pre-analytical steps of the specimen histopathological management play a pivotal role in this process. We have here devised a procedure for improving the critical step of the transfer of fixed pathology specimens, and we show that the under-vacuum sealing in liquid-free plastic bags of previously appropriately formalin-fixed tissues allows to safely store and transport pathology specimens at room temperature in a time frame up to at least 2 months, provided that vacuum conditions are maintained. This procedure results to be simple and safe and offers definite advantages in terms of cost and speed of transfer.
Two established facts prompted us to explore alternative ways of transferring pathology specimens under safe conditions, while preserving the diagnostic standard guaranteed by appropriate formalin-based fixation. Not all hospitals and health centers in the world have indoor pathology services which imply that surgical pathology and biopsy specimens are regularly shipped or transported to referral centers. In addition, despite the fact that formalin remains the optimal tissue preservation solution (for the purposes of histocytological, immunohistochemical, and molecular diagnostic workup), it is well known that this aldehyde is hazardous (being associated to irritant properties), and it has recently been moved by the European Community to the category of forbidden, potentially cancerogenic reagents (see also Statement by the “Pre-analytical Tissue Condition” Working Group of the European Society of Pathology on Formalin banning in Europe in 2016) [7,8,9].
Regarding the first point, poor access to specialized health services in rural and remote areas is an actual and significant problem for people living in those regions. Among others, histopathological examination may be a relevant issue for the therapeutic strategy, and pre-analytical conditions, including tissue fixation, handling, and specimen transportation are crucial to provide accurate pathology reports [17,18,19]. Different methods and timing for tissue transfer to referral pathology laboratories occur throughout the world. In general, the most common transportation system is based on containers with variable amounts of liquid formalin. An alternative, as adopted in the Detroit area [20], is the transfer of fresh samples in vacuum-sealed plastic bags, but this requires affordable distances and cold (4 °C) conditions throughout.
In low-income countries, pathology services are mostly concentrated in capital or large cities and the organization of transportation of specimens from remote areas is a complicate issue. Apart from alternative fixatives, formalin itself may be difficult to purchase in large amounts, and the transfer of liquid-containing materials through long distances is cumbersome and hazardous [3, 21, 22]. The procedure here proposed can be adopted at low costs with a minimal amount of formalin required for the first fixation step and the use of a single plastic bag having a cost of a few cents of US dollar. The vacuum chamber machine, specifically devised in order to meet safety requirements, needs to be acquired in each hospital, while the cost of domestic instruments does not exceed 150 USD. In addition, the shipping of the light, liquid-free bags as post letter (rather than heavy parcels) results to be cheaper, faster, and safer.
The proposed procedure helps to restrict the use of formalin to the surgical theater, only (apart from the receiving pathology laboratory), with no formalin present in the transportation step, thus preventing potential liquid leakage from the containers and dispersion of a cancerogenic agent in the environment. Based on up to date findings [23,24,25], the current proposal represents then a compromise that reasonably combines the advantages of preserving both, the tissues and the environment. Regarding tissues, the quality of histopathological and immunophenotypical staining, as well as that of nucleic acids for molecular tests, was equivalent to that present in the original histological material, in both 7-day and 30-day under-vacuum storage conditions. In particular, immunohistochemical markers provided the expected reactivity in all tissues examined, and extracted DNA and RNA were found at an acceptable length and amount.
A final comment is related to technical issues of the devised procedure. Regarding plastic bags, we decided to test the feasibility of the current project on both certified containers made of polyvinyl chloride/PVC, as those provided for the TissueSAFE machine (Milestone), as well as commercial plastic bags of adequate and comparable thickness and resistance, together with a commercial vacuum chamber machine used for domestic purposes or in food industry, having costs not exceeding 150 USD. We demonstrated that previously formalin-fixed tissue samples can be safely stored and transported in both scenarios. As a consequence, this newly devised, two-step, procedure for fixing and safely shipping pathology specimens can be adapted to different situations, in different parts of the world, either a network of remote hospitals lacking pathology services or single remote health centers in low-income countries that are often forced to ship their pathology materials to referral centers, sometimes even out of their country. The lack of resources and limited financial availability may fit with the proposed transportation procedure, guaranteeing safe and optimal conditions in the pre-analytical steps of the histopathological diagnostic workup. Regarding sample size, this is not a limiting issue since the crucial point is an adequate formalin fixation prior to preparing the specimen for vacuum processing. Small specimens, such as core biopsies, can safely be shipped under vacuum, once protected in a plastic vial, while for large specimens, care should be taken to dissect the surgical specimen in such a way to respect the anatomical relationships between normal tissue and the lesion (tumor) and possibly in slices having a recommended thickness of approximately 2–3 cm, as recommended for proper fixation.
In conclusion, we show that the sealing under-vacuum conditions of previously and adequately formalin-fixed pathology specimens can safely be adopted to transfer biopsy and surgical specimens from remote areas to the referral pathology services. This newly devised, two-step procedure is based on low-cost technology in terms of instruments, timing, and personnel’s education, which renders this method easily affordable and above all fully safe and adequate in the pre-analytical processing of pathology specimens.
References
Hewitt SM, Lewis FA, Cao Y, Conrad RC, Cronin M, Danenberg KD, Goralski TJ, Langmore JP, Raja RG, Williams PM, Palma JF, Warrington JA (2008) Tissue handling and specimen preparation in surgical pathology: issues concerning the recovery of nucleic acids from formalin-fixed, paraffin-embedded tissue. Arch Pathol Lab Med 132:1929–1935. https://doi.org/10.1043/1543-2165-132.12.1929
Khoury T, Sait S, Hwang H, Chandrasekhar R, Wilding G, Tan D, Kulkarni S (2009) Delay to formalin fixation effect on breast biomarkers. Mod Pathol 22:1457–1467. https://doi.org/10.1038/modpathol.2009.117
Adesina A, Chumba D, Nelson AM, Orem J, Roberts DJ, Wabinga H, Wilson M, Rebbeck TR (2013) Improvement of pathology in sub-Saharan Africa. Lancet Oncol 14:e152–e157. https://doi.org/10.1016/S1470-2045(12)70598-3
Fox CH, Johnson FB, Whiting J, Roller PP (1985) Formaldehyde fixation. J Histochem Cytochem 33:845–853. https://doi.org/10.1177/33.8.3894502
Cheng J, Zhang L, Tang Y, Li Z (2016) The toxicity of continuous long-term low-dose formaldehyde inhalation in mice. Immunopharmacol Immunotoxicol 38:495–501. https://doi.org/10.1080/08923973.2016.1248844
Han RT, Back SK, Lee H, Lee J, Kim HY, Kim HJ, Na HS (2016) Formaldehyde-induced aggravation of pruritus and dermatitis is associated with the elevated expression of Th1 cytokines in a rat model of atopic dermatitis. PLoS One 11:e0168466. https://doi.org/10.1371/journal.pone.0168466
European Parliament and Council of European Union (2008) Regulation (EC) No 1272/2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006. Off J Eur Union L 353:1–1355
International Agency for Research and Cancer, IARC (2012) Monographs on the evaluation of carcinogenic risks to humans. Chemical Agents and Related Occupations 100F:401–430 Lyon
Bussolati G, Annaratone L, Berrino E, Miglio U, Panero M, Cupo M, Gugliotta P, Venesio T, Sapino A, Marchio C (2017) Acid-free glyoxal as a substitute of formalin for structural and molecular preservation in tissue samples. PLoS One 12:e0182965. https://doi.org/10.1371/journal.pone.0182965
Bussolati G, Chiusa L, Cimino A, D’Armento G (2008) Tissue transfer to pathology labs: under vacuum is the safe alternative to formalin. Virchows Arch 452:229–231. https://doi.org/10.1007/s00428-007-0529-x
Fischer AH, Jacobson KA, Rose J, Zeller R (2008) Hematoxylin and eosin staining of tissue and cell sections. CSH Protoc. pdb.prot4986. doi: https://doi.org/10.1101/pdb.prot4986
Sapino A, Marchiò C, Senetta R, Castellano I, Macrì L, Cassoni P, Ghisolfi G, Cerrato M, D’Ambrosio E, Bussolati G (2006) Routine assessment of prognostic factors in breast cancer using a multicore tissue microarray procedure. Virchows Arch 449:288–296 doi.org/10.1007/s00428-006-0233-2
Marchiò C, Lambros MB, Gugliotta P, Di Cantogno LV, Botta C, Pasini B, Tan DS, Mackay A, Fenwick K, Tamber N, Bussolati G, Ashworth A, Reis-Filho JS, Sapino A (2009) J Pathol 219:16–24. doi: https://doi.org/10.1002/path.2574, Does chromosome 17 centromere copy number predict polysomy in breast cancer? A fluorescence in situ hybridization and microarray-based CGH analysis
van Beers EH, Joosse SA, Ligtenberg MJ, Fles R, Hogervorst FB, Verhoef S, Nederlof PM (2006) A multiplex PCR predictor for a CGH success of FFPE samples. Br J Cancer 94:333–337
Illumina (2014) Expression analysis of FFPE samples. Illumina Technical Note: RNA Sequencing
Mariani S, Bertero L, Osella-Abate S, Di Bello C, Francia di Celle P, Coppola V, Sapino A, Cassoni P, Marchiò C (2017) Extreme assay sensitivity in molecular diagnostics further unveils intratumour heterogeneity in metastatic colorectal cancer as well as artifactual low-frequency mutations in the KRAS gene. Br J Cancer 117:358–366. https://doi.org/10.1038/bjc.2017.170
Berg D, Malinowsky K, Reischauer B, Wolff C, Becker KF (2011) Use of formalin-fixed and paraffin-embedded tissues for diagnosis and therapy in routine clinical settings. Methods Mol Biol 785:109–122. https://doi.org/10.1007/978-1-61779-286-1_8
Denoux Y, Blanc-Vincent MP, Simony-Lafontaine J, Verriele-Beurrier V, Briffod M, Voigt JJ (2002) Standards, options and recommendations: good practice for the management and shipment of histological and cytopathological cancer specimens. Bull Cancer 89:401–409
van der Loos CM (2007) A focus on fixation. Biotech Histochem 82:141–154. https://doi.org/10.1080/10520290701375302
Zarbo RJ (2015) Histologic validation of vacuum sealed, formalin-free tissue preservation, and transport system. Recent Results Cancer Res 199:15–26. https://doi.org/10.1007/978-3-319-13957-9_2
Adeyi OA (2011) Pathology services in developing countries: the West African experience. Arch Pathol Lab Med 135:183–186. https://doi.org/10.1043/2008-0432-CCR.1
Gopal S, Krysiak R, Liomba G (2013) Building a pathology laboratory in Malawi. Lancet Oncol 14:291–292. https://doi.org/10.1016/S1470-2045(13)70109-8
Duong A, Steinmaus C, McHale CM, Vaughan CP, Zhang L (2011) Reproductive and developmental toxicity of formaldehyde: a systematic review. Mutat Res 728:118–138. https://doi.org/10.1016/j.mrrev.2011.07.003
Zhang L, Tang X, Rothman N, Vermeulen R, Ji Z, Shen M, Qiu C, Guo W, Liu S, Reiss B, Freeman LB, Ge Y, Hubbard AE, Hua M, Blair A, Galvan N, Ruan X, Alter BP, Xin KX, Li S, Moore LE, Kim S, Xie Y, Hayes RB, Azuma M, Hauptmann M, Xiong J, Stewart P, Li L, Rappaport SM, Huang H, Fraumeni JF, Smith MT, Lan Q (2010) Occupational exposure to formaldehyde, hematotoxicity, and leukemia-specific chromosome changes in cultured myeloid progenitor cells. Cancer Epidemiol Biomark Prev 19:80–88. https://doi.org/10.1158/1055-9965.EPI-09-0762
Di Novi C, Minniti D, Barbaro S, Zampirolo MG, Cimino A, Bussolati G (2010) Vacuum-based preservation of surgical specimens: an environmentally-safe step towards a formalin-free hospital. Sci Total Environ 408:3092–3095. https://doi.org/10.1016/j.scitotenv.2010.04.022
Author information
Authors and Affiliations
Contributions
All individuals listed as co-authors of this manuscript qualify for every one of the four following criteria:
● Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work
● Drafting the work or revising it critically for important intellectual content
● Final approval of the version to be published
● Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Metovic, J., Bertero, L., Musuraca, C. et al. Safe transportation of formalin-fixed liquid-free pathology specimens. Virchows Arch 473, 105–113 (2018). https://doi.org/10.1007/s00428-018-2383-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-018-2383-4