Abstract
Of pulmonary adenocarcinomas, about 25–30 % of cases is of a mucinous type. Mucinous adenocarcinomas are regarded as more aggressive compared to their non-mucinous counterparts. Invasive mucinous adenocarcinoma, colloid, and enteric adenocarcinomas are variants within adenocarcinomas. We investigated 76 invasive mucinous adenocarcinomas, including colloid variants, for predominant and secondary patterns, their different form of mucin storage and release, expression of cytokeratin 7 and 20, TTF1 and CDX2, MUC1, 2, and 5AC proteins, p14 and p16 proteins, possible rearrangements for EML4ALK and ROS1, as well as KRAS mutational status, and correlated this with survival. For comparison, 259 non-mucinous adenocarcinomas were selected. Overall survival for invasive mucinous adenocarcinomas corrected for T and N stage was not different from their non-mucinous counterpart. Most were of an acinar pattern. Neither pattern, nor type of mucin storage and release, such as luminal, extracellular, or goblet cell type had any influence on survival. Of adenocarcinomas expressing CK20, all but one expressed TTF1 either strongly or at least focally, and 8 co-expressed CDX2 focally. Most mucinous adenocarcinomas expressed either MUC1 or MUC5AC proteins, but rarely MUC2, while a few cases co-expressed both or all three. Loss of p16 expression correlated with worse outcome. KRAS mutation was found in 56 % of mucinous adenocarcinomas. Mutational status was neither correlated with architectural pattern nor survival. Codon 12 mutations were most frequent, and one case presented with KRAS mutations in codon 12 and 61. Goblet cell variants of mucinous adenocarcinomas presented predominantly with codon 12 mutations, while all colloid variants had KRAS mutation. Two cases had EML4 and ALK1 rearranged; ROS1 rearrangement was not found. Mucinous adenocarcinomas behave similar to non-mucinous variants. TNM stage is the most important factor followed by p16 loss predicting overall survival.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lung cancer is the most common cancer in men worldwide and the number one cancer leading to death in developed and developing countries [8]. Worldwide, there are more than a million lung cancer related deaths per year due to late detection of the primary tumor and early metastasis [10, 15, 40]. Invasive pulmonary adenocarcinomas (AC) have been reclassified according to their predominant architecture into lepidic, acinar, papillary, micropapillary, cribriform, and solid [34, 36], and in anyone of them, mucin production can be found. Invasive mucinous adenocarcinoma has been defined in the new classification as having abundant mucin and present with either columnar or goblet cell morphology. It might present with a lepidic, acinar, papillary, micropapillary, cribriform or solid pattern, which might have prognostic value on its own [22]. In the new WHO classification, solid AC is defined as either “with mucin production” or expressing adenocarcinoma markers in contrast to the previous edition [34, 37]. Invasive mucinous together with enteric and colloid AC are listed as variants. Signet ring cell AC is no longer a subtype of AC but is considered a variant with signet ring features [35, 37]; mucinous cystadenocarcinoma has been merged with colloid AC. Within the AC category, approximately one third differentiate along the mucinous pathway. Mucinous differentiation is already present in the precursor lesion as atypical goblet cell proliferation [31] (Fig. 1). Mucinous AC originate most probably from basal/peripheral stem cells, which have retained the capacity to produce different types of mucins, such as fucosylated and sialylated carbohydrates, acidic sulfated carbohydrates, and also glycosylated lipoproteins [3].
Although it is generally assumed that when stratified by stage, mucinous AC behaves more aggressively than non-mucinous AC, cytomorphological and architectural features have not been evaluated and correlated with prognosis and genetic aberrations [1, 6, 22]. This study intends to address these issues in a series of 76 cases from a single institution.
Material and methods
Between 2002 and 2012, we identified 335 consecutive cases of surgically resected pulmonary AC, and of these, 76 were classified as mucinous AC. Mucinous AC was defined by positive mucicarmine and Alcian blue staining of at least 80 % of tumor cells, which is more precise than “abundant mucin”, and by either columnar or goblet cell morphology or characteristics of enteric or colloid variant. The tumor tissue blocks were retrieved from the Lung Biobank. Two cases were excluded because of insufficient tissue. Two other cases were excluded due to the lack of clinical data. A single case of surgically resected enteric AC was detected and also excluded from the analysis as the sample size was too small for analysis. In the remaining 71 cases of mucinous AC, immunohistochemistry and molecular analysis could be performed. This study was approved by the Ethical Committee of the Medical University (24–135 ex 11/12). Patient characteristics are listed in Table 1 (and in more detail Supplementary Table 1).
Four micrometer sections were cut from formalin-fixed paraffin-embedded tissue blocks and stained with hematoxyline & eosin (H&E), Mucicarmine, and Alcian blue at pH 2.5. Two pathologists (UGM, HHP) reevaluated all mucinous adenocarcinomas and classified them on whole tumor sections using the architectural patterns as acinar, papillary, micropapillary, cribriform, and solid. We also distinguished colloid patterns and AC with signet ring cells. Colloid AC was defined by a diffuse spreading without forming a pseudocapsule while cystadenocarcinomas present with a pseudocapsule. Secondary and tertiary patterns were noted in terms of surface percentage, in increments of 20 % for primary and secondary patterns. Four types of mucin storage and secretion were recognized. The goblet cell type shows mucin stored in large supranuclear vacuoles and secreted apically into acinar lumina (luminal/apical secretion). The columnar cell type shows mucin stored in very small vacuoles and again secreted apically into acinar lumina. A third type is seen in colloid AC and cystadenocarcinomas with tumor cells, having lost their basoapical orientation, storing mucin in small cytoplasmic vacuoles but secreting it basolaterally into the stroma, creating the appearance of tumor cells floating in mucin (basolateral secretion). A fourth type is seen in signet ring cell AC, with single tumor cells forming intracytoplasmic lumina into which mucin is secreted (intracytoplasmic luminal secretion).
A tissue microarray was produced from the same 71 tissue blocks that had been used for the molecular analysis. At least three cores of tumor and one core of uninvolved normal lung tissue clearly separated from the tumor area were chosen and inserted into a recipient paraffin block. For immunohistochemistry 4 mμ sections were incubated with antibodies for cytokeratin (CK) 7 and 20, TTF1, CDX2, MUC1, MUC2, MUC5AC, p14ARF, p15INK4B, p16INK4A, ALK1, and ROS1 (Table 2). Immunohistochemical stains were semi-quantitatively evaluated by intensity score (0−1 + −2 + −3+) multiplied by the percentage of positively stained tumor cells in 10 % increments (H-score 0–300)—a mean was created from the three cores. To assess validity of tissue microarrays (TMA) sampling, full sections from 20 randomly chosen cases were stained for MUC1, TTF1, p14, p16, and ALK1.
Whole tumor tissue sections from 71 adenocarcinomas (all pre-treatment) were used for the analysis of KRAS mutations. Four to six micrometer thick sections were cut and dewaxed in xylene, and the marked tumor areas were scrapped from the slides. Genomic DNA was extracted using EZ1 DNA investigator Kit (Qiagen, Hilden, Germany) from paraffin-embedded tissues. For KRAS mutation analysis, the Therascreen® KRAS PYRO® Kit (PyroMark Q24; Qiagen, Hilden, Germany), which detects mutations at codons 12, 13, and 61, was used according to the manufacturer’s instructions.
For EML4ALK rearrangement, tissue sections were primarily stained by an ALK1 antibody (Table 2). This antibody positively stained three cases, which were evaluated by fluorescence in situ hybridization (FISH) analysis using the FISH break-apart probe from Vysis (Abbott Laboratories, Abbott Park, IL, USA). ROS1 translocations were assessed using an antibody for ROS1 (Table 5). Since all cases were negative for ROS1, no further evaluation by FISH was done.
Clinical data
The clinical data of 71 patients with mucinous and of 259 patients with non-mucinous adenocarcinoma of the lung (a total of 330 patients) were collected retrospectively from the Division of Thoracic and Hyperbaric Surgery, Medical University Graz. Staging was available for all cases. Data included age, gender, smoking status (pack years), date of surgery, postoperative course, recurrence, disease free (overall) or progression free survival, metastasis (including location), death of disease, and other non-tumor related diseases (comorbidities). Patients were grouped according to the UICC/TNM staging system into: Stages IA-IV (Table 1 and Supplementary Tables 1 and 2).
Statistical analysis
Survival rates were calculated according to the Kaplan-Meier method, while survival differences between groups were compared by the log-rank test. Survival rates were adjusted for confounding risk factors by Cox regression and tested by the Wald test criterion.
Statistical analysis of the collected clinical data was performed by R software (www.r-project.org) showing the results graphically and in tabular as well as standard error, lower 95 % CI, and upper 95 % CI. P values below 0.05 were considered as statistically significant.
Expression of AC markers CK7, CK20, TTF1, CDX2, MUC1, MUC2, MUC5AC, p14, and p16 was compared between the different variants of the mucinous ACs using paired t test.
Results
Patterns of mucinous adenocarcinomas
Mucinous ACs showed a predominance of acinar (43/71), followed by papillary patterns (10/71). Other patterns were found in few cases (Fig. 2a–i). Two cases presented with a predominant signet ring cell pattern, and in one of them, the whole tumor was composed of signet ring cells. Most of the ACs showed secondary and even tertiary patterns, similar to what is known from the non-mucinous ACs (Tables 3 and 4).
Pattern variations in mucinous adenocarcinomas: a acinar, b papillary, c micropapillary, d cribriform, e colloid, f signet ring cell; bar 50 μm; Alcian blue stain shows different patterns of mucin storage and secretion, g colloid (basolateral), h apical/luminal), i combined apical and basolateral; goblet cell type, j mucicarmine stain, k Alcian blue stain, bar 50 μm
Immunohistochemistry
Some differences were found between full section and TMA staining results. For p14 and p16 staining, the difference between full section and TMA scores was <10 %. For MUC1, 4 cases showed differences (2 cases with higher and 2 with a lower percentage than in the TMA). TTF1 staining showed the highest difference with full sections showing in 8/20 cases a higher percentage of positive tumor cells. However, this did not substantially change the results of the statistical analysis: P = 0.075 in the TMA versus P = 0.064 in full sections.
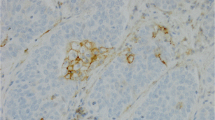
Reactivity for CK7 was found in all cases, in most a 100 % of tumor cells expressed this cytokeratin, but in 9 tumors only focally, and in these with a co-expression for CK20. In most of these cases, CK20 was found in <15 % of tumor cells, but 3 cases showed >40 % positivity (Fig. 3a–b).
Eight cases expressed CDX2, in 7 in less than 10 % of tumor cells, but in a single case in almost 50 %. This was the only case without expression of TTF1. In all other cases a majority of carcinoma cells expressed TTF1 (Fig. 3c-d). Co-expression of CDX2 and TTF1 within single tumor cells was not seen. Expression of CDX2 was always associated with CK20 positivity.
Luminal/apical mucin predominated in acinar ACs. A goblet cell pattern was found in 24, and a basolateral mucin secretion was seen in all colloid ACs (Fig. 2g–k). A combination of apical and basolateral mucin secretion was encountered in 33 cases (Tables 3 and 4). Goblet cell types of mucinous ACs showed always an apical/luminal mucin secretion (Fig. 2j–k). MUC1 antibodies stained 39 and MUC2 antibodies 7, whereas MUC5AC antibodies stained 50 AC. In cases with goblet cell differentiation, either MUC1 or MUC5AC was seen; in a few cases, both MUC proteins were expressed (Fig. 4a–d).
In 27 mucinous AC, staining for p16INK4A was negative while in 19 cases, a low H-score (<40) was found. In 48 cases moderate and in 20 cases, a high expression of p14ARF was found while in 5 cases, the p14ARF H-score was low (<40). (Supplementary Table 2). Immunohistochemistry for p15 was not reproducible and was not further evaluated.
KRAS mutation
KRAS mutations were detected in 40 out of 71 cases (56 %), most commonly in codon 12. Mutations on codon 13 were found in 5 cases, and in codon 61 in another 5 cases, one of them with double mutation G12D and Q61R (Table 5). There was no correlation of KRAS mutation and any of the structural patterns, and neither were correlations found with stage (Table 6 and Supplementary Table 2). KRAS codon 12 mutations were present in 16/26 mucinous ACs of goblet cell type, while only one presented with codon 13 mutation. KRAS mutation status was not different between AC with acinar, papillary, micropapillary, cribriform, and signet ring cell morphotypes. However, all colloid AC were KRAS mutant (Table 6).
EML4ALK1 rearrangement
Out of the three cases positive for immunohistochemistry, two were positive for FISH analysis, showing split signals in at least 30 % of tumor cells. All were negative when immunohistochemically stained for ROS1 (Supplementary Table 2).
Survival analysis
We analyzed 330 mucinous and non-mucinous AC patients for overall survival and correlated this with the T and N stages. A statistically significant difference was found between T stages (I + II versus III + IV) as well as N stages (N0 versus N1 + N2; log-rank test P = 0.00067; Fig. 5a, b). When clinical stages I-IV were taken together, overall survival was neither different between mucinous and non-mucinous AC nor different between different T-stages (Supplementary Fig. 1). When stratified for clinical stage (low stage IA, IB, IIA and high stage IIB, IIIA, IIIB), no differences in survival were found between mucinous and non-mucinous AC: clinically high-stage patients had a significantly worse course regardless of being mucinous or non-mucinous AC (log-rank test; P = 0.000073; Fig. 5c–d).
Survival analysis of all adenocarcinoma patients according to their T and N status (a, b). As expected, higher T as well as N stages correlated significantly with worse outcome (overall survival). A comparison of mucinous and non-mucinous adenocarcinomas grouped into low (c) and high stages (d) did not show any survival benefit for non-mucinous adenocarcinomas. Low stage IAB, IIA; high stage IIB, IIIAB
Lymph node (LN) metastases were equally common in mucinous and non-mucinous AC, and stratification according to N status did not reveal differences in outcome (data not shown). Goblet cell versus non-goblet cell types of AC and presence or absence of signet ring cells were not associated with differences in survival (Suppl Fig. 2).
A non-significant trend for a stage-independent correlation between loss of p16INK4A and worse prognosis (Fig. 6a) was noted, whereas p14ARF staining did not correlate with survival. Similarly, a non-significant trend for a correlation between high TTF1 expression and better survival was found (P = 0.075, Fig. 6b).
Survival analysis of mucinous adenocarcinomas for p16 loss versus positive staining (a), and TTF1 (b): Cases stained for TTF1 were grouped into low (“neg”) or high (“pos”); low/neg was defined by intensity of 1+/2+ and less 50 % of tumor cells, high/pos was defined by intensity of 3+ in ≥50 % of tumor cells
Discussion
Mucinous differentiation in a pulmonary adenocarcinoma seems to occur early on in carcinogenesis, most probably already in the precursor lesion [3]. Several molecular alterations have been identified in pre-neoplastic goblet cell proliferations, including over-expression of MUC2 and 5AC and cytokines such as interleukin (IL) 4, IL4-receptor, IL9, 13, and 17 [2–4, 13, 23, 31, 32], pointing to an involvement of inflammatory mediators in the development in mucinous adenocarcinomas. Our finding that in KRAS mutated AC, in contrast to EGFR mutated AC, angiogenesis is selectively suppressed via STAT3 whereas IL8 acts proangiogenic supports the view that mucinous and non-mucinous AC are really different at the molecular level [8]. Only few studies have compared behavior and overall survival between mucinous and non-mucinous ACs. Yoshizawa reported a worse prognosis for mucinous types, when applying the new adenocarcinoma classification, but demonstrated a similar behavior for mucinous and non-mucinous ACs in a subsequent report [44, 45]. Caretta et al. found a worse outcome in patients with mucinous AC in stage IA and IB. However, they studied only a small group of patients and mixed cases of invasive as well as in-situ ACs (formerly called bronchioloalveolar) [1]. Riquet et al. found that solid AC with mucin production has worse prognosis than solid AC without mucin production [22]. This evokes a problem of the previous classification: solid AC was defined by the presence of mucin producing tumor cells, whereas solid carcinomas without mucin producing cells were classified as undifferentiated or large cell carcinoma [35]. In the new adenocarcinoma classification for resection specimen, solid carcinomas without mucin production but expressing TTF1 are also classified as solid AC [34, 37]. Solid ACs by themselves have a worse prognosis compared to well-differentiated ACs, regardless of being mucinous or non-mucinous. Not surprisingly, in the Rossi study, bronchioloalveolar AC of mucinous type (which probably correspond to in-situ ACs) had a more favorable course than invasive AC [24].
We found mucinous and non-mucinous ACs stratified by stage and lymph node involvement to behave similar. Outcome of mucinous AC was not different between those with or without goblet cells (Supplementary Fig. 2). When stratified for stage, outcome of mucinous AC was not different between those with or without signet ring cells, including the two cases with a dominant or pure signet ring cell presence (Supplementary Fig. 2). This is in contrast to the study by Rossi, which however was based on only two cases [24]. So in contrast to signet ring cell carcinomas of the stomach, pulmonary ones do not confer an unfavorable prognosis.
TTF1 expression was found in almost every mucinous AC, most often associated with CK7 expression. Only a minority expressed CK20 and CDX2. However, all except one case also co-expressed TTF1 in subsets of the tumor cells, pointing to different clones either expressing TTF1 and CK7 or CDX2 and CK20, similar to the findings of other studies [24, 27, 38, 43] but in contrast to the study by Shah et al. [28]. Previous published studies investigated small number of cases, and in addition, AIS cases were included [24, 43], which makes survival analysis inconclusive.
All mucinous ACs showed acidic types of mucins (Alcian blue positive at pH 2.5). Rossi et al. as well as Yatabe et al. reported MUC 2 in goblet cell types and MUC5AC in the other variants [24, 43]. In another study, MUC1 expression was found in ACs without goblet cell morphology [15], whereas Kunii confirmed MUC2 and MUC5AC expression [11]. In the study by Kunii et al., MUC2 and MUC5AC expression together with HNF4α were observed in cases negative for TTF1. In our study, the number of TTF1 negative mucinous ACs was quite high, which might point to differences between a Caucasian and an Asian population, and will need further investigation [11]. We found MUC 1, 2, or 5AC staining in all ACs. In contrast to previous studies, MUC 1 and 5AC were the most common types, either as the dominant MUC protein or co-expressed in subsets of ACs, with only two cases co-expressing all three MUC proteins. A higher propensity of MUC1 expression was seen in mucinous ACs with goblet cell morphology; however, it was also found in a few cases without goblet cells in contrast to the study of Maeshima [14]. Several AC with goblet cell morphology in our cases co-expressed MUC5AC. Expression of MUC genes early during carcinogenesis might induce precursor cell proliferation and their differentiation into a mucinous type. In contrast to the study of Tsutsumida et al., we did not find MUC expression to be correlated with aggressive behavior [39]. However, Tsutsumida focused on micropapillary ACs, which are known to behave worse compared to other types. Consequently, the reported aggressive behavior is likely based on this pattern and not MUC expression.
In recent investigations, different functions of TTF1 have been discussed: in non-mucinous AC, TTF1 upregulates downstream surfactant genes [19], pointing to some inflammation regulating functions. TTF1 (also known as NKX2-1) is a transcription factor and prevents gastric differentiation in fetal lung. If downregulated in lung AC, it is associated with poor differentiation, and in experimental models, low expression is associated with the emergence of a gastric tumor type [30]. However, these models do not present as signet ring cell type ACs. This fits well with TTF1 expression in all our cases. TTF1 also inhibits metastasis by binding to SREBF2 promoter, leading to repression of miR33a and finally upregulation of prometastatic HMGA2 [20]. In another study, TTF1 caused the expression of MUC2 in CDX2 positive mucinous ACs, which also acts as a transcriptional regulator [16]. We did not assess whether or not TTF1 also regulates expression of other MUCs. TTF1 is a marker of better differentiation and outcome not only in non-mucinous ACs but also in mucinous ACs: intense TTF1 expression in >50 % of tumor cells was tended to be associated with better overall survival, although this did not reach statistical significance (Fig. 6b).
EML4ALK1 is not preferentially rearranged in mucinous ACs, as only 2/74 mucinous acinar ACs showed a rearrangement of EML4ALK1, both carrying wild-type sequences for KRAS. A third case positively stained for ALK1 showed a codon 12 mutation of the KRAS gene. Rearrangement for ROS1 was not found in any of our cases.
KRAS mutation was the most frequent finding in our series of mucinous ACs (56 % in comparison to 20–25 % for non-mucinous variants) in accordance with previous studies [5, 14, 26, 43, 47]. KRAS mutation status was not related to mucinous AC subtype, such as goblet cell variant or AC with signet ring cells. Remarkable features of mucinous AC with goblet cell components were the predominance of KRAS mutation in codon 12 and KRAS mutation in all colloid AC. In other mucinous AC, mutations were randomly distributed (Table 6 and Supplementary Table 2). This is in contrast to the studies of Yatabe [43] and Maeshima [14], which found no KRAS mutations in their cases with goblet cell morphology, probably reflecting the overall low percentage of KRAS mutations in Southeast Asian populations.
An important question is at which time KRAS mutations occur during carcinogenesis [12, 25, 33, 41]. Comparison of human mucinous AC and KRAS mutant induced AC in mice suggests that KRAS mutation is most probably a second or third step in human AC carcinogenesis: Mouse ACs induced by KRAS mutant alleles never develop a mucinous phenotype but most often, a non-mucinous papillary AC [7, 17]. In addition in mice, KRAS mutation does not result in an invasive phenotype, and additional genetic hits are needed to induce invasion and metastasis [7, 17]. Inflammatory factors play a role in the development of precursor lesions of mucinous ACs. Inflammatory cytokines have been shown to modulate goblet cell proliferation and transformation. These might be one of the early abnormalities leading to precursor lesions with mucinous differentiation [4, 9, 18, 31, 42, 46] and might precede KRAS mutation. Inflammatory mediators also play an important role in adenocarcinoma progression, as IL8 was identified to promote angiogenesis [7].
Loss of function or reduced protein expression for p14ARF and p16INK4A was seen in 43 and 1 cases, respectively, in some associated with KRAS mutation, in others not. Loss of function of p16 (absent or reduced protein expression) was more common in ACs with decreased overall survival, but did not reach statistical significance. Loss of p14ARF protein expression was not related to survival. Both proteins act as mitosis checkpoint control proteins, sending cell with defective DNA either into repair or into apoptosis. Whereas p14ARF acts at the G1 transition, p16INK4B cooperates with p53 at the G2 checkpoint. Since patients with mucinous ACs are predominantly cigarette smokers, which also carry a high percentage of p53 mutations, combined loss of function of p53 and p16INK4A seems important in the genesis of mucinous AC.
Almost 41 % of mucinous ACs in our series had no identified driver mutation (59 % KRAS and EML4ALK). These cases will need further investigation using large cancer gene panels. Additional genetic hits need to be identified in KRAS mutated ACs since invasion and metastasis might be regulated by other genes [17]. Analysis of downstream signaling pathways in KRAS mutated ACs is urgently needed. Subtyping of patterns in mucinous AC although not associated with survival is recommended: patterns might be associated with different genetic background, as in goblet cell and colloid variants of AC.
Targeted therapy for patients with a KRAS mutated mucinous AC need to be considered with caution: although inhibitors for downstream kinases of the RAS pathway are available [21, 29], mutated KRAS can signal downstream not only into the MAPK-ERK pathway but also into RAL, PI3K, or PLCέ pathways [17]. So a blockade of just one downstream pathway might not be efficient.
References
Carretta A, Canneto B, Calori G, Ceresoli GL, Campagnoli E, Arrigoni G, Vagani A, Zannini P (2001) Evaluation of radiological and pathological prognostic factors in surgically-treated patients with bronchoalveolar carcinoma. Eur J Cardiothorac Surg 20:367–371
Chen Y, Thai P, Zhao YH, Ho YS, DeSouza MM, Wu R (2003) Stimulation of airway mucin gene expression by interleukin (IL)-17 through IL-6 paracrine/autocrine loop. J Biol Chem 278:17036–17043
Copin MC, Buisine MP, Devisme L, Leroy X, Escande F, Gosselin B, Aubert JP, Porchet N (2001) Normal respiratory mucosa, precursor lesions and lung carcinomas: differential expression of human mucin genes. Front Biosci 6:D1264–D1275
Dabbagh K, Takeyama K, Lee HM, Ueki IF, Lausier JA, Nadel JA (1999) IL-4 induces mucin gene expression and goblet cell metaplasia in vitro and in vivo. J Immunol 162:6233–6237
Dacic S, Shuai Y, Yousem S, Ohori P, Nikiforova M (2010) Clinicopathological predictors of EGFR/KRAS mutational status in primary lung adenocarcinomas. Mod Pathol 23:159–168
Dammrich J, Vollmers H, Silber R, Buchwald J, Papadopoulos T, Mullerhermelink H (1997) Postoperative prognosis of pulmonary adenocarcinoma subtypes correlates with HLA-DR expression and the number of tumor-infiltrating cells. Oncol Rep 4:59–64
Grabner B, Schramek D, Mueller KM, Moll HP, Svinka J, Hoffmann T, Bauer E, Blaas L, Hruschka N, Zboray K, Stiedl P, Nivarthi H, Bogner E, Gruber W, Mohr T, Zwick RH, Kenner L, Poli V, Aberger F, Stoiber D, Egger G, Esterbauer H, Zuber J, Moriggl R, Eferl R, Gyorffy B, Penninger JM, Popper H, Casanova E (2015) Disruption of STAT3 signalling promotes KRAS-induced lung tumorigenesis. Nat Commun 6:6285
Hansen J (1998) Common cancers in the elderly. Drugs Aging 13:467–478
Harkema JR, Wagner JG (2002) Non-allergic models of mucous cell metaplasia and mucus hypersecretion in rat nasal and pulmonary airways. Novartis Found Symp 248:181–197, discussion 197-200, 277-182
Kato T, Ishikawa K, Aragaki M, Sato M, Okamoto K, Ishibashi T, Kaji M (2012) Angiolymphatic invasion exerts a strong impact on surgical outcomes for stage I lung adenocarcinoma, but not non-adenocarcinoma. Lung Cancer 77:394–400
Kunii R, Jiang S, Hasegawa G, Yamamoto T, Umezu H, Watanabe T, Tsuchida M, Hashimoto T, Hamakubo T, Kodama T, Sasai K, Naito M (2011) The predominant expression of hepatocyte nuclear factor 4alpha (HNF4alpha) in thyroid transcription factor-1 (TTF-1)-negative pulmonary adenocarcinoma. Histopathology 58:467–476
Licciulli S, Avila JL, Hanlon L, Troutman S, Cesaroni M, Kota S, Keith B, Simon MC, Pure E, Radtke F, Capobianco AJ, Kissil JL (2013) Notch1 is required for Kras-induced lung adenocarcinoma and controls tumor cell survival via p53. Cancer Res 73:5974–5984
Louahed J, Toda M, Jen J, Hamid Q, Renauld JC, Levitt RC, Nicolaides NC (2000) Interleukin-9 upregulates mucus expression in the airways. Am J Respir Cell Mol Biol 22:649–656
Maeshima A, Miyagi A, Hirai T, Nakajima T (1997) Mucin-producing adenocarcinoma of the lung, with special reference to goblet cell type adenocarcinoma: immunohistochemical observation and Ki-ras gene mutation. Pathol Int 47:454–460
McWilliams A, MacAulay C, Gazdar AF, Lam S (2002) Innovative molecular and imaging approaches for the detection of lung cancer and its precursor lesions. Oncogene 21:6949–6959
Mesquita P, Almeida R, Van Seuningen I, David L (2004) Coordinated expression of MUC2 and CDX-2 in mucinous carcinomas of the lung can be explained by the role of CDX-2 as transcriptional regulator of MUC2. Am J Surg Pathol 28:1254–1255
Musteanu M, Blaas L, Zenz R, Svinka J, Hoffmann T, Grabner B, Schramek D, Kantner HP, Muller M, Kolbe T, Rulicke T, Moriggl R, Kenner L, Stoiber D, Penninger JM, Popper H, Casanova E, Eferl R (2012) A mouse model to identify cooperating signaling pathways in cancer. Nat Methods 9:897–900
Park KS, Korfhagen TR, Bruno MD, Kitzmiller JA, Wan H, Wert SE, Khurana Hershey GK, Chen G, Whitsett JA (2007) SPDEF regulates goblet cell hyperplasia in the airway epithelium. J Clin Invest 117:978–988
Pelizzoli R, Tacchetti C, Luzzi P, Strangio A, Bellese G, Zappia E, Guazzi S (2008) TTF-1/NKX2.1 up-regulates the in vivo transcription of nestin. Int J Dev Biol 52:55–62
Rice SJ, Lai SC, Wood LW, Helsley KR, Runkle EA, Winslow MM, Mu D (2013) MicroRNA-33a mediates the regulation of high mobility group AT-hook 2 gene (HMGA2) by thyroid transcription factor 1 (TTF-1/NKX2-1). J Biol Chem 288:16348–16360
Riely GJ, Johnson ML, Medina C, Rizvi NA, Miller VA, Kris MG, Pietanza MC, Azzoli CG, Krug LM, Pao W, Ginsberg MS (2011) A phase II trial of Salirasib in patients with lung adenocarcinomas with KRAS mutations. J Thorac Oncol 6:1435–1437
Riquet M, Foucault C, Berna P, Assouad J, Dujon A, Danel C (2006) Prognostic value of histology in resected lung cancer with emphasis on the relevance of the adenocarcinoma subtyping. Ann Thorac Surg 81:1988–1995
Rose MC, Piazza FM, Chen YA, Alimam MZ, Bautista MV, Letwin N, Rajput B (2000) Model systems for investigating mucin gene expression in airway diseases. J Aerosol Med 13:245–261
Rossi G, Murer B, Cavazza A, Losi L, Natali P, Marchioni A, Migaldi M, Capitanio G, Brambilla E (2004) Primary mucinous (so-called colloid) carcinomas of the lung: a clinicopathologic and immunohistochemical study with special reference to CDX-2 homeobox gene and MUC2 expression. Am J Surg Pathol 28:442–452
Sakamoto H, Shimizu J, Horio Y, Ueda R, Takahashi T, Mitsudomi T, Yatabe Y (2007) Disproportionate representation of KRAS gene mutation in atypical adenomatous hyperplasia, but even distribution of EGFR gene mutation from preinvasive to invasive adenocarcinomas. J Pathol 212:287–294
Sakuma Y, Matsukuma S, Yoshihara M, Nakamura Y, Noda K, Nakayama H, Kameda Y, Tsuchiya E, Miyagi Y (2007) Distinctive evaluation of nonmucinous and mucinous subtypes of bronchioloalveolar carcinomas in EGFR and K-ras gene-mutation analyses for Japanese lung adenocarcinomas: confirmation of the correlations with histologic subtypes and gene mutations. Am J Clin Pathol 128:100–108
Sato K, Ueda Y, Shikata H, Katsuda S (2006) Bronchioloalveolar carcinoma of mixed mucinous and nonmucinous type: immunohistochemical studies and mutation analysis of the p53 gene. Pathol Res Pract 202:751–756
Shah RN, Badve S, Papreddy K, Schindler S, Laskin WB, Yeldandi AV (2002) Expression of cytokeratin 20 in mucinous bronchioloalveolar carcinoma. Hum Pathol 33:915–920
Smit EF, Dingemans AM, Thunnissen FB, Hochstenbach MM, van Suylen RJ, Postmus PE (2010) Sorafenib in patients with advanced non-small cell lung cancer that harbor K-ras mutations: a brief report. J Thorac Oncol 5:719–720
Snyder EL, Watanabe H, Magendantz M, Hoersch S, Chen TA, Wang DG, Crowley D, Whittaker CA, Meyerson M, Kimura S, Jacks T (2013) Nkx2-1 represses a latent gastric differentiation program in lung adenocarcinoma. Mol Cell 50:185–199
Stacher E, Ullmann R, Halbwedl I, Gogg-Kammerer M, Boccon-Gibod L, Nicholson AG, Sheppard MN, Carvalho L, Franca MT, Macsweeney F, Morresi-Hauf A, Popper HH (2004) Atypical goblet cell hyperplasia in congenital cystic adenomatoid malformation as a possible preneoplasia for pulmonary adenocarcinoma in childhood: a genetic analysis. Hum Pathol 35:565–570
Takeyama K, Dabbagh K, Lee HM, Agusti C, Lausier JA, Ueki IF, Grattan KM, Nadel JA (1999) Epidermal growth factor system regulates mucin production in airways. Proc Natl Acad Sci U S A 96:3081–3086
Thunnissen FB, Prinsen C, Hol B, Van der Drift M, Vesin A, Brambilla C, Montuenga L, Field JK (2012) Smoking history and lung carcinoma: KRAS mutation is an early hit in lung adenocarcinoma development. Lung Cancer 75:156–160
Travis WD, Brambilla E, Burke AP, Marx A, Nicholson AG (2015) WHO classification of tumours of the lung, pleura, thymus and heart. IARC, WHO Press, Geneva
Travis WD, Brambilla E, Müller-Hermelink HK, Harris CC (2004) Pathology and genetics of tumours of the lung, pleura, thymus and heart IARC WHO classification of tumours. IARC Press, Lyon, 10
Travis WD, Brambilla E, Noguchi M, Nicholson AG, Geisinger K, Yatabe Y, Powell CA, Beer D, Riely G, Garg K, Austin JH, Rusch VW, Hirsch FR, Jett J, Yang PC, Gould M (2011) International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society: international multidisciplinary classification of lung adenocarcinoma: executive summary. Proc Am Thorac Soc 8:381–385
Travis WD, Brambilla E, Noguchi M, Nicholson AG, Geisinger KR, Yatabe Y, Beer DG, Powell CA, Riely GJ, Van Schil PE, Garg K, Austin JH, Asamura H, Rusch VW, Hirsch FR, Scagliotti G, Mitsudomi T, Huber RM, Ishikawa Y, Jett J, Sanchez-Cespedes M, Sculier JP, Takahashi T, Tsuboi M, Vansteenkiste J, Wistuba I, Yang PC, Aberle D, Brambilla C, Flieder D, Franklin W, Gazdar A, Gould M, Hasleton P, Henderson D, Johnson B, Johnson D, Kerr K, Kuriyama K, Lee JS, Miller VA, Petersen I, Roggli V, Rosell R, Saijo N, Thunnissen E, Tsao M, Yankelewitz D (2011) International association for the study of lung cancer/American thoracic society/European respiratory society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 6:244–285
Tsuta K, Ishii G, Nitadori J, Murata Y, Kodama T, Nagai K, Ochiai A (2006) Comparison of the immunophenotypes of signet-ring cell carcinoma, solid adenocarcinoma with mucin production, and mucinous bronchioloalveolar carcinoma of the lung characterized by the presence of cytoplasmic mucin. J Pathol 209:78–87
Tsutsumida H, Nomoto M, Goto M, Kitajima S, Kubota I, Hirotsu Y, Wakimoto J, Hollingsworth MA, Yonezawa S (2007) A micropapillary pattern is predictive of a poor prognosis in lung adenocarcinoma, and reduced surfactant apoprotein A expression in the micropapillary pattern is an excellent indicator of a poor prognosis. Mod Pathol 20:638–647
Tyczynski JE, Bray F, Parkin DM (2003) Lung cancer in Europe in 2000: epidemiology, prevention, and early detection. Lancet Oncol 4:45–55
Wistuba II, Gazdar AF (2006) Lung cancer preneoplasia. Annu Rev Pathol 1:331–348
Yamauchi K, Piao HM, Nakadate T, Shikanai T, Nakamura Y, Ito H, Mouri T, Kobayashi H, Maesawa C, Sawai T, Ohtsu H, Inoue H (2009) Enhanced goblet cell hyperplasia in HDC knockout mice with allergic airway inflammation. Allergol Int 58:125–134
Yatabe Y, Koga T, Mitsudomi T, Takahashi T (2004) CK20 expression, CDX2 expression, K-ras mutation, and goblet cell morphology in a subset of lung adenocarcinomas. J Pathol 203:645–652
Yoshizawa A, Motoi N, Riely GJ, Sima CS, Gerald WL, Kris MG, Park BJ, Rusch VW, Travis WD (2011) Impact of proposed IASLC/ATS/ERS classification of lung adenocarcinoma: prognostic subgroups and implications for further revision of staging based on analysis of 514 stage I cases Mod Pathol
Yoshizawa A, Sumiyoshi S, Sonobe M, Kobayashi M, Fujimoto M, Kawakami F, Tsuruyama T, Travis WD, Date H, Haga H (2013) Validation of the IASLC/ATS/ERS lung adenocarcinoma classification for prognosis and association with EGFR and KRAS gene mutations: analysis of 440 Japanese patients. J Thorac Oncol 8:52–61
Zhang X, Zhang Y, Tao B, Wang D, Cheng H, Wang K, Zhou R, Xie Q, Ke Y (2012) Docking protein Gab2 regulates mucin expression and goblet cell hyperplasia through TYK2/STAT6 pathway. FASEB J 26:4603–4613
Zhang Y, Sun Y, Pan Y, Li C, Shen L, Li Y, Luo X, Ye T, Wang R, Hu H, Li H, Wang L, Pao W, Chen H (2012) Frequency of driver mutations in lung adenocarcinoma from female never-smokers varies with histologic subtypes and age at diagnosis. Clin Cancer Res 18:1947–1953
Addendum
A recently published study by Shim et al. focused also on a similar cohort of mucinous adenocarcinomas. The authors found similar survival data as well as a high prevalence of KRAS mutations, thus confirming our findings. Importantly they had significantly low mutations in TP53, which raises the question which proteins other than p53 cooperates with p16 (J.Thoracic Oncol. 2015 Aug;10(8):1156-62).
Conflict of interest
This investigation was in part supported by an unrestricted grant from Hofman La Roche to HHP. No conflict of interest is to be declared in connection with this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Geles, A., Gruber-Moesenbacher, U., Quehenberger, F. et al. Pulmonary mucinous adenocarcinomas: architectural patterns in correlation with genetic changes, prognosis and survival. Virchows Arch 467, 675–686 (2015). https://doi.org/10.1007/s00428-015-1852-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-015-1852-2