Abstract
The Gleason grading system remains one of the most powerful prognostic factors in prostate cancer and is the dominant method around the world in daily practice. It is based solely on the glandular architecture performed at low magnification. The Gleason grading system should be performed in needle core biopsies and radical prostatectomy specimens where it shows a reasonable degree of correlation between both specimens, and most importantly, it remains vital in the treatment decision-making process. This review summarizes the current status of Gleason grading in prostate cancer, incorporating recent proposals for the best contemporary practice of prostate cancer grading.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Gleason grading system for prostate cancer, named after Donald F. Gleason, is the predominant grading system around the world [21–23]. The Gleason grading system is based on glandular architecture which can be divided into five patterns of growth (also known as grades) with decreasing differentiation [10, 21–23]. The primary and secondary pattern or grade, i.e., the most prevalent and the second most prevalent pattern or grade is added to obtain a Gleason score or sum that is to be reported (see Appendix) [10, 21]. Nuclear atypia or cytoplasmic features are not evaluated. An important issue is that the initial grading of prostate carcinoma should be performed at low magnification using a 4× or 10× lens [16, 18]. Then one may proceed wit high-power objectives to look for rare fused glands or a few individual cells. Gleason grading of prostate cancer has changed over the years to adapt to needle biopsies and radical prostatectomy specimens that were unavailable at time when Gleason proposed his system [3, 4, 9, 14, 18, 19, 31, 32, 35, 36, 39, 48]. Issues on the application of the Gleason grading system in prostate cancer have been dealt with in recent consensus conferences organized under the auspices of the Uropathology Working Group (European Society of Pathology, ESP), the European Society of Uropathology (a full section office member of the European Association of Urology, EAU), and the Italian Society of Urology (SIU; Florence, Italy, June 2003; Palermo, Sicily, June 2005) [9, 39], World Health Organization (Lyon, France, December 2002; and Stockholm, Sweden, September 2004) [3, 14], and International Society of Urologic Pathology (San Antonio, TX, USA, March 2005) [18]. The authors of this review have been involved in the organization and participated in and contributed to these conferences. Important recommendations follow.
Gleason score as prognostic and predictive factor
Gleason grading remains as one of the most significant factors in the clinical decision-making activity both in needle biopsy specimens and after radical prostatectomy is performed; that is, it predicts pathological stage, margin status, biochemical failure, local recurrences, lymph node, disease progression, or distant metastasis after prostatectomy [3, 5–8, 11–13, 31, 32, 35]. Radiation therapy, radical prostatectomy, and other therapies are initially based on the Gleason score; in practice, Gleason scores of 7–10 are associated with worse prognosis, meanwhile Gleason scores of 5–6 are associated with lower progression rates after therapy [17, 19, 20]. In recent years, Gleason score has been included in clinical nomograms which are used with increasing frequency to predict disease progression [25–27, 30, 40, 44–48].
Reporting Gleason scores in prostate needle biopsies
Once carcinoma is detected in the needle biopsy, further descriptive information regarding the tumor type, Gleason score, and amount of cancer is necessary for management of the patient [35, 38]. As a recently indicated, the highest Gleason score was selected by most urologists to determine their treatment plan regardless of the overall involvement (Table 1) [35, 38, 45]. Some issues that need clarification follow.
Gleason scores 2–4
The diagnosis of Gleason scores 2–4 on needle biopsies should be made “rarely, if ever,” and the reasons are compelling [18]: (1) Gleason scores 2–4 cancer is extraordinarily rare in needle biopsies as compared to transurethral resection (TURP) specimens, (2) poor reproducibility among experts [1, 2, 16], (3) the correlation with the prostatectomy score is poor, and (4) “low” Gleason scores of 2–4 may misguide clinicians into believing that there is an indolent tumor [20]. A recent consensus stated that a Gleason score of 1+1=2 is a grade that should not be diagnosed, regardless of the type of specimen with extremely rare exception, since most of these cases in the era of Gleason would today be referred to as adenosis (atypical adenomatous hyperplasia) because of improvement in recognizing basal cells [17, 18, 29]. Cribriform morphology is not allowed within Gleason pattern 2 [5].
Gleason pattern 3
Cribriform pattern 3 applies to round, well-circumscribed glands of the same size of normal glands, but only rare cribriform lesions satisfy these criteria. Therefore, the majority of cribriform patterns should be diagnosed as Gleason pattern 4. “Individual cells” would not be allowed within Gleason pattern 3 [5, 18, 19].
Gleason pattern 4 in Gleason score 7 tumors
The importance of the percentage of Gleason 4 pattern in Gleason score 7 tumors accumulates rapidly [18, 19, 34, 35, 40, 47, 49]. In recently generated nomograms, patients with Gleason score 4+3 vs 3+4 are stratified differently [30]. Whether or not the percentage of pattern 4 tumor should be included in the report remains optional at present time. Small, ill-defined glands with poorly formed glandular lumina also warrant the diagnosis of Gleason pattern 4 as stated by a recent consensus [18].
Gleason pattern 5
Comedonecrosis when seen in solid nests or cribriform masses should be regarded as Gleason pattern 5. However, the definition of comedonecrosis requires intraluminal necrotic cells and/or nuclear debris (karyorrhexis) [18].
Tertiary pattern
Another important change recently incorporated in current practice is the recognition and reporting of the tertiary pattern in needle biopsies [34, 43]. This includes tumors with patterns 3, 4, and 5 in various proportions on a biopsy. Tertiary patterns are uncommon, but when the worst Gleason grade is the tertiary pattern, it should influence the final Gleason score, and therefore, the primary pattern and the highest grade should be recorded following the rule of “the most and the worst” [18]. For example, a case with primary Gleason pattern 3, secondary pattern 4, and tertiary pattern 5 should be assigned a Gleason score of 8. These tumors should be classified overall as high grade (Gleason scores 8–10) [43, 46].
Needle biopsy with different cores showing different grades
This is related to a case when one or more of the cores shows pure high-grade cancer (i.e. Gleason score 4+4=8), and the other cores show pattern 3 (3+3, 3+4, or 4+3) cancer [18, 24, 27, 35]. If one reports the grades of each core separately, whereby the highest grade tumor (Gleason score 8) would typically be the one selected by the clinician as the grade of the entire case. Others give instead an overall score for the entire case. For example, in a case with Gleason score 4+4=8 on one core with pattern 3 (3+3=6, 3+4=7, 4+3=7), on other cores, the overall score for the entire case would be Gleason score 4+3=7 or 3+4=7, depending on whether pattern 4 or 3 predominated. Likewise, it was demonstrated that when one core is Gleason score 4+4=8 with other cores having pattern 3, the pathological stage at radical prostatectomy is comparable to cases with all needle cores having Gleason score 4+4=8 [3, 6–8, 18, 19]. The use of the highest core grade of the given case in cases where there are multiple cores of different grades advocated in current tables and nomograms gives additional support for giving cores a separate grade rather than an overall score for the entire case. This is the rationale of a recent survey in which 81% of urologists used the highest Gleason score on a positive biopsy, regardless of the overall percentage involvement, to determine treatment [45]. Consequently, it has been recommended to assign individual Gleason scores to separate cores as long as the cores were submitted in separate containers or the cores were in the same container yet specified by the urologist as to their location (i.e., by different color inks). In addition, one has the option to also give an overall score at the end of the case [33, 35].
In cases where a container contains multiple pieces of tissue and one cannot be sure if one is looking at an intact core, it is recommended that one should only give an overall score for that container [18].
Reporting Gleason scores in radical prostatectomies
In these specimens, one should assign the Gleason score based on the primary and secondary patterns with a comment if present on the tertiary pattern (Table 1) [3, 18, 19]. Specific issues follow.
Gleason scores 2–4
Gleason scores 2–4 are rarely seen as the grade of the main tumor in radical prostatectomies performed for stages T1c or T2 disease. These tumors are typically seen in multifocal incidental adenocarcinoma of the prostate found within the transition zone in TURP specimens [17–19]. The situation where Gleason scores 2–4 tumor represents the major tumor at radical prostatectomy performed for tumor incidentally found on TURP (stages T1a and T1b) is uncommon. In one study, Gleason scores 2–4 were the grade of the main tumor in 2% of the radical prostatectomy specimens; this represents a disproportionate number of T1a and T1b tumors, as compared to what would be currently seen in today’s practice. All men with only Gleason scores 2–4 tumor at radical prostatectomy are cured [18, 19].
Gleason scores 5–6
It is important to recognize that the majority of tumors with Gleason scores 5–6 are cured after radical prostatectomy [18, 26].
Gleason score 7
Tumors with a Gleason score of 7 have a significantly worse prognosis than those with a Gleason score of 6. Given the adverse prognosis associated with Gleason pattern 4, one would expect that whether a tumor with Gleason score 3+4 vs 4+3 would influence prognosis [26]. There have been several studies addressing Gleason score 3+4 as compared to Gleason score 4+3 at radical prostatectomy, with somewhat conflicting results. In one study, they reported no significant survival advantage for Gleason pattern 3+4 over 4+3, but other study showed that Gleason score 3+4 vs 4+3 correlated with both stage and progression in men with serum PSA values less than 10 ng/ml and organ-confined disease [30, 47, 48]. Several other investigations have shown that Gleason score 4+3 has a worse prognosis. This is an issue in which much work needs to be done.
Gleason scores 8–10
Gleason scores 8–10 may account for only 7% of the grades seen at radical prostatectomy, but these patients have highly aggressive tumors with advanced stage such that they are not amenable to surgical therapy alone. Overall, patients with Gleason scores 8–10 at radical prostatectomy have a 15% chance of having no evidence of disease at 15 years following surgery [26, 33].
Percent Gleason pattern 4/5
The proportion of high-grade tumor as the preferred method for grading prostate cancer has been proposed, as it is predictive for progression at the extremes (greater than 70% or less than 20% of pattern 4/5) [12, 13]. It has been recently demonstrated that classifying tumors based on the combined percent of pattern 4/5 is more predictive than stratifying patients into Gleason score alone. Therefore, it has been recommended these data be included in the report [13].
Tertiary Gleason pattern
In radical prostatectomy, a higher proportion of cases are found to contain more than two grades, and over 50% of them contain at least three different grades [11, 18]. The progression rate of Gleason scores 5–6 tumor with a tertiary component of Gleason pattern 4 is almost the same as those of pure Gleason score 7 tumors. Gleason score 7 tumors with tertiary pattern 5 experience progression rates following radical prostatectomy approximating pure Gleason score 8 tumors [40, 43]. On the other hand, no such significance could be seen in cases of Gleason (4+4) score 8 with tertiary pattern 5; since Gleason score 8 tumors are already aggressive, the existence of pattern 5 elements adds no difference. These tumors should be graded routinely (primary and secondary patterns) with a comment in the report noting the presence of the tertiary element. In the setting of high-grade cancer (scores 8–10), one should ignore lower-grade patterns if they occupy less than 5% of the area of the tumor [35, 36, 38, 39].
Tumors with one predominant pattern and a small percentage of higher-grade tumor
Some controversy still exist on how to grade tumors, which are over 95% of one pattern, where there is only a very small percentage of higher-grade tumor. For example, a tumor composed of >95% Gleason pattern 3 and <5% pattern 4, some experts would assign a Gleason score 3+3=6, as it has been proposed that one needs over 5% of a pattern to be present for it to be incorporated within the Gleason score. Others might grade the tumor as Gleason score 3+4=7. It seems that the existence of a high-grade component, even if it constitutes less than 5% of the whole tumor, has a significant adverse influence [18, 20].
Radical prostatectomy specimens with separate tumor nodules
Radical prostatectomy specimens should be processed in an organized fashion where one can make some assessment as to whether one is dealing with a dominant nodule or separate tumor nodules and assign a separate grade to each dominant tumor nodule(s). Most often, the dominant nodule is the largest tumor, which is also the tumor associated with the highest stage and grade [35].
Grading variants and variations of adenocarcinoma of the prostate
A number of architectural or cytologic variations of carcinoma of the prostate have been described [15, 28, 35, 41, 42, 50]. These are descriptive terms used to assist pathologists avoid diagnostic pitfalls. Most of the times, one should grade the tumor solely based on the underlying architecture with some exceptions (Table 2). Variants of prostate cancer include ductal, mucinous (colloid), mucinous signet-ring cell, small cell carcinoma, sarcomatoid carcinoma, and some others (Table 2). The former three are diagnoses tenable only on examination of radical prostatectomy or TURP specimens. If seen in needle biopsy specimens, the diagnostic terminologies used are adenocarcinoma of prostate with ductal features, adenocarcinoma of prostate with signet-ring cell features, and adenocarcinoma of prostate with mucinous (colloid) differentiation. Small cell carcinoma and sarcomatoid carcinoma may be diagnosed on needle biopsies. Ductal adenocarcinoma should be graded as Gleason score 4+4=8 while retaining the diagnostic term of ductal adenocarcinoma to denote their unique clinical and pathological features. There is no consensus on how mucinous (colloid) carcinoma should be scored [18]. Some authors suggest that a Gleason score of 8 is to be assigned, while others recommend ignoring mucin and grading the tumor based on the underlying architectural pattern. Small cell, sarcomatoid, and mucinous signet-ring cell carcinomas should not be assigned a Gleason grade. It has been suggested that the rare pleomorphic giant cell carcinoma of the prostate should be assigned a pattern 5 (Table 2) [18, 28, 37].
Sources of discrepancies between needle biopsy and radical prostatectomy Gleason scores
Several studies have addressed the correlation between Gleason scores in needle biopsies and corresponding radical prostatectomy specimens [35]. In a recent meta-analysis from 18 studies, exact correlation of Gleason scores was found in 43% of cases and correlation plus or minus one Gleason score unit in 77% of cases. Undergrading of carcinoma in needle biopsy is the most common problem occurring in 42% of all reviewed cases, and overgrading is present in up to 15% of cases. In general, adverse findings on needle biopsy accurately predict adverse findings in the radical prostatectomy, whereas favorable findings on the needle biopsy do not necessarily predict favorable findings in the radical prostatectomy [35, 49].
Sampling error
This is related to the small sample of tissue inherent to needle biopsy [35]. The most common type of sampling error occurs when there is a higher-grade component present within the radical prostatectomy, which is not sampled on needle biopsy. This typically occurs when a needle biopsy tumor is graded as Gleason score 3+3=6, and in the prostatectomy exists a Gleason pattern 4 which was not sampled on the biopsy, thus resulting in a prostatectomy Gleason 3+4=7. In some instances, undergrading results from an attempt to grade very tiny areas of carcinoma, so-called minimal or limited adenocarcinoma. Overgrading can result from sampling error in cases where the high-grade pattern is present in needle biopsy, but it may only represent a very minor element in the radical prostatectomy specimen [35].
Borderline cases
There are some cases in the original description of the Gleason grading system that are right at the interface between two different patterns leading to interobserver and possibly intraobserver variability [21, 35].
Pathology error
A common pathology error is seen when pathologists assign a Gleason score of <4 on a needle biopsy which in fact was Gleason score 5–6 or higher. When there is a limited focus of small glands of cancer on needle biopsy, by definition, this is a Gleason pattern 3 or higher [35]. Gleason pattern 3 consists of small glands with an infiltrative pattern. Biopsying truly low-grade adenocarcinoma of the prostate could not result in just a few neoplastic glands but rather would be more extensive, as low-grade adenocarcinoma grows as nodules of closely packed rather than infiltrating glands. Undergrading may result from difficulty in recognizing an infiltrative growth pattern or failing to recognize the presence of small areas of gland fusion [35].
Pathologists’ experience
With experience, pathologists recognize grading pitfalls; in particular, the fact that Gleason scores of 4 and lower are almost nonexistent in needle biopsy situation. Furthermore, small areas of fusion in the presence of a predominantly grade 3 background are recognized and will yield a Gleason score of 7 [35, 46].
Intraobserver and interobserver variability
Intraobserver variability of Gleason grade and exact agreement was reported in 43–78% of cases, and agreement within plus or minus one Gleason score unit was reported in 72–87% of cases. Gleason wrote that he duplicated exactly his previous histological scores approximately 50% times. Highly variable levels of interobserver agreement on Gleason scores have also been reported, with ranges of 36–81% for exact agreement and 69–86% within plus or minus one Gleason score unit [45–49].
Conclusions
The Gleason grading system is a powerful tool to predict response to therapy. The needle biopsy Gleason score correlates with virtually all other pathological and clinical parameters after radical prostatectomy. The Gleason score assigned to the tumor at radical prostatectomy is the most powerful predictor of progression and is very useful in stratifying patients for therapy. However, still significant deficiencies in the practice of this grading system exist. An important issue frequently missed by practicing pathologists is that the initial grading of prostate carcinoma should be performed at low magnification using a 4× or 10× lens. Keeping this in mind might help pathologists avoid overgrade or undergrade prostate cancer in daily practice. Improvements in Gleason grading reproducibility can be achieved by recognizing problematic areas and educating physicians via meetings, courses, website tutorials, and publications.
References
Allsbrook WC Jr, Mangold KA, Johnson MH, Lane RB, Lane CG, Amin MB, Bostwick DG, Humphrey PA, Jones EC, Reuter VE, Sakr W, Sesterhenn IA, Troncoso P, Wheeler TM, Epstein JI (2001) Interobserver reproducibility of Gleason grading of prostatic carcinoma: urologic pathologists. Hum Pathol 32:74–80
Allsbrook WC Jr, Mangold KA, Johnson MH, Lane RB, Lane CG, Epstein JI (2001) Interobserver reproducibility of Gleason grading of prostatic carcinoma: general pathologist. Hum Pathol 32:81–88
Amin M, Boccon-Gibod L, Egevad L, Epstein JI, Humphrey PA, Mikuz G, Newling D, Nilsson S, Sakr W, Srigley JR, Wheeler TM, Montironi R (2005) Prognostic and predictive factors and reporting of prostate carcinoma in prostate needle biopsy specimens. Scand J Urol Nephrol Suppl 216:20–33
Amin MB, Grignon DJ, Humphrey PA, Srigley JR (2004) Gleason grading of prostate cancer: a contemporary approach. Lippincott Williams & Wilkins, Philadelphia, PA
Amin MB, Schultz DS, Zarbo RJ (1994) Analysis of cribriform morphology in prostatic neoplasia using antibody to high-molecular-weight cytokeratins. Arch Pathol Lab Med 118:260–264
Augustin H, Eggert T, Wenske S, Karakiewicz PI, Palisaar J, Daghofer F, Huland H, Graefen M (2004) Comparison of accuracy between the Partin tables of 1997 and 2001 to predict final pathological stage in clinically localized prostate cancer. J Urol 171:177–181
Babaian RJ, Troncoso P, Bhadkamkar VA, Johnston DA (2001) Analysis of clinicopathologic factors predicting outcome after radical prostatectomy. Cancer 91:1414–1422
Bailar JC 3rd, Mellinger GT, Gleason DF (1966) Survival rates of patients with prostatic cancer, tumor stage, and differentiation–preliminary report. Cancer Chemother Rep 50:129–136
Boccon-Gibod L, van der Kwast TH, Montironi R, Bono A (2004) Handling and pathology reporting of prostate biopsies. Eur Urol 46:177–181
Bostwick DG (1994) Gleason grading of prostatic needle biopsies. Correlation with grade in 316 matched prostatectomies. Am J Surg Pathol 18:796–803
Bostwick DG, Grignon DJ, Hammond ME, Amin MB, Cohen M, Crawford D, Gospadarowicz M, Kaplan RS, Miller DS, Montironi R, Pajak TF, Pollack A, Srigley JR, Yarbro JW (2000) Prognostic factors in prostate cancer. College of American Pathologists Consensus Statement 1999. Arch Pathol Lab Med 124:995–1000
Chan TY, Partin AW, Walsh PC, Epstein JI (2000) Prognostic significance of Gleason score 3+4 versus Gleason score 4+3 tumor at radical prostatectomy. Urology 56:823–827
Cheng L, Koch MO, Juliar BE, Daggy JK, Foster RS, Bihrle R, Gardner TA (2005) The combined percentage of Gleason patterns 4 and 5 is the best predictor of cancer progression after radical prostatectomy. J Clin Oncol 23:2911–2917
Eble JN, Sauter G, Epstein JI, Sesterhenn IA (2004) Pathology and genetics. Tumors of the urinary system and male genital organs. IARC, Lyon
Egan AJ, Lopez-Beltran A, Bostwick DG (1997) Prostatic adenocarcinoma with atrophic features: malignancy mimicking a benign process. Am J Surg Pathol 21:931–935
Egevad L, Allsbrook WC Jr., Epstein JI (2005) Current practice of Gleason grading among genitourinary pathologists. Hum Pathol 36:5–9
Epstein JI (2000) Gleason score 2–4 adenocarcinoma of the prostate on needle biopsy: a diagnosis that should not be made. Am J Surg Pathol 24:477–478
Epstein JI, Allsbrook WCJ, Amin MB, Egevad LL, and The ISUP Grading Committee (2005) The 2005 International Society of Urological Pathoogy (ISUP) consensus conference on Gleason grading of prostatic carcinoma. Am J Surg Pathol 29:1228–1242
Epstein JI, Amin M, Boccon-Gibod L, Egevad L, Humphrey PA, Mikuz G, Newling D, Nilsson S, Sakr W, Srigley JR, Wheeler TM, Montironi R (2005) Prognostic factors and reporting of prostate carcinoma in radical prostatectomy and pelvic lymphadenectomy specimens. Scand J Urol Nephrol Suppl:34–63
Epstein JI, Partin AW, Sauvageot J, Walsh PC (1996) Prediction of progression following radical prostatectomy. A multivariate analysis of 721 men with long-term follow-up. Am J Surg Pathol 20:286–292
Gleason DF (1966) Classification of prostatic carcinomas. Cancer Chemother Rep 50:125–128
Gleason DF (1977) Histologic grading and clinical staging of prostatic carcinoma. In: Tannenbaum M (eds) Urologic pathology: the prostate. Lea & Feibiger, Philadelphia, PA
Gleason DF, Mellinger GT (1974) Prediction of prognosis for prostatic adenocarcinoma by combined histological grading and clinical staging. J Urol 111:58–64
Grober ED, Tsihlias J, Jewett MA, Sweet JM, Evans AJ, Trachtenberg J, Robinette M, Nam RK (2004) Correlation of the primary Gleason pattern on prostate needle biopsy with clinico-pathological factors in Gleason 7 tumors. Can J Urol 11:2157–2162
Humphrey PA (2003) Prostate pathology. ASCP Press, Chicago, IL
Kattan MW, Eastham JA, Wheeler TM, Maru N, Scardino PT, Erbersdobler A, Graefen M, Huland H, Koh H, Shariat SF, Slawin KM, Ohori M (2003) Counseling men with prostate cancer: a nomogram for predicting the presence of small, moderately differentiated, confined tumors. J Urol 170:1792–1797
Kunz GM Jr, Epstein JI (2003) Should each core with prostate cancer be assigned a separate Gleason score? Hum Pathol 34:911–914
Lopez-Beltran A, Eble JN, Bostwick DG (2005) Pleomorphic giant cell carcinoma of the prostate. Arch Pathol Lab Med 129:683–685
Lopez-Beltran A, Qian J, Montironi R, Luque RJ, Bostwick DG (2005b) Atypical adenomatous hyperplasia (adenosis) of the prostate: DNA ploidy analysis and immunophenotype. Int J Surg Pathol 13:167–173
Makarov DV, Sanderson H, Partin AW, Epstein JI (2002) Gleason score 7 prostate cancer on needle biopsy: is the prognostic difference in Gleason scores 4+3 and 3+4 independent of the number of involved cores? J Urol 167:2440–2442
Mazzucchelli R, Lopez-Beltran A, Scarpelli M, Montironi R (2002) Predictive factors in prostate needle biopsy. Pathologica 94:331–337
Mazzucchelli R, Santinelli A, Lopez-Beltran A, Scarpelli M, Montironi R (2002) Evaluation of prognostic factors in radical prostatectomy specimens with cancer. Urol Int 68:209–215
Mian BM, Troncoso P, Okihara K, Bhadkamkar V, Johnston D, Reyes AO, Babaian RJ (2002) Outcome of patients with Gleason score 8 or higher prostate cancer following radical prostatectomy alone. J Urol 167:1675–1680
Mills SE, Fowler JE Jr (1986) Gleason histologic grading of prostatic carcinoma. Correlations between biopsy and prostatectomy specimens. Cancer 57:346–349
Montironi R, Mazzuccheli R, Scarpelli M, Lopez-Beltran A, Fellegara G, Algaba F (2005) Gleason grading of prostate cancer in needle biopsies or radical prostatectomy specimens: contemporary approach, current clinical significance and sources of pathology discrepancies. BJU Int 95:1146–1152
Montironi R, Mazzucchelli R, Stramazzotti D, Scarpelli M, Lopez Beltran A, Bostwick DG (2005) Basal cell hyperplasia and basal cell carcinoma of the prostate: a comprehensive review and discussion of a case with c-erbB-2 expression. J Clin Pathol 58:290–296
Montironi R, Mazzucchelli R, van der Kwast T (2003) Morphological assessment of radical prostatectomy specimens. A protocol with clinical relevance. Virchows Arch 442:211–217
Montironi R, van der Kwast T, Boccon-Gibod L, Bono AV (2003b) Handling and pathology reporting of radical prostatectomy specimens. Eur Urol 44:626–636
Montironi R, Scarpelli M, Lopez Beltran A (2004) Carcinoma of the prostate: inherited susceptibility, somatic gene defects and androgen receptors. Virchows Arch 444:503–508
Mosse CA, Magi-Galluzzi C, Tsuzuki T, Epstein JI (2004) The prognostic significance of tertiary Gleason pattern 5 in radical prostatectomy specimens. Am J Surg Pathol 28:394–398
Pacelli A, Lopez-Beltran A, Egan AJ, Bostwick DG (1998) Prostatic adenocarcinoma with glomeruloid features. Hum Pathol 29:543–546
Pan CC, Chiang H, Chang YH, Epstein JI (2000) Tubulocystic clear cell adenocarcinoma arising within the prostate. Am J Surg Pathol 24:1433–1436
Pan CC, Potter SR, Partin AW, Epstein JI (2000) The prognostic significance of tertiary Gleason patterns of higher grade in radical prostatectomy specimens: a proposal to modify the Gleason grading system. Am J Surg Pathol 24:563–569
Partin AW, Kattan MW, Subong EN, Walsh PC, Wojno KJ, Oesterling JE, Scardino PT, Pearson JD (1997) Combination of prostate-specific antigen, clinical stage, and Gleason score to predict pathological stage of localized prostate cancer. A multi-institutional update. JAMA 277:1445–1451
Rubin MA, Bismar TA, Curtis S, Montie JE (2004) Prostate needle biopsy reporting: how are the surgical members of the Society of Urologic Oncology using pathology reports to guide treatment of prostate cancer patients? Am J Surg Pathol 28:946–952
Rubin MA, Dunn R, Kambham N, Misick CP, O’Toole KM (2000) Should a Gleason score be assigned to a minute focus of carcinoma on prostate biopsy? Am J Surg Pathol 24:1634–1640
Sakr WA, Tefilli MV, Grignon DJ, Banerjee M, Dey J, Gheiler EL, Tiguert R, Powell IJ, Wood DP (2000) Gleason score 7 prostate cancer: a heterogeneous entity? Correlation with pathologic parameters and disease-free survival. Urology 56:730–734
Srigley JR, Amin M, Boccon-Gibod L, Egevad L, Epstein JI, Humphrey PA, Mikuz G, Newling D, Nilsson S, Sakr W, Wheeler TM, Montironi R (2005) Prognostic and predictive factors in prostate cancer: historical perspectives and recent international consensus initiatives. Scand J Urol Nephrol Suppl 216:8–19
Steinberg DM, Sauvageot J, Piantadosi S, Epstein JI (1997) Correlation of prostate needle biopsy and radical prostatectomy Gleason grade in academic and community settings. Am J Surg Pathol 21:566–576
Tran TT, Sengupta E, Yang XJ (2001) Prostatic foamy gland carcinoma with aggressive behavior: clinicopathologic, immunohistochemical, and ultrastructural analysis. Am J Surg Pathol 25:618–623
Acknowledgement
This work was supported by the Grant FIS 03/0952 (Madrid, Spain).
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Gleason pattern 1
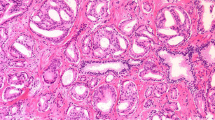
Nodule of separate, closely packed glands which do not infiltrate into adjacent benign prostatic tissue is very well circumscribed. The glands are of intermediate size and approximately equal in size and shape. The nucleus is typically small, and cytoplasm frequently is abundant and pale-staining. Nuclear and cytoplasm appearances are not taken into account in diagnosis. This pattern is exceedingly rare and usually seen in transition zone cancers (Fig. 1).
Gleason pattern 2
This pattern is characterized with round to oval glands with smooth ends. The glands are more loosely arranged and not quite as uniform in size and shape as those of Gleason pattern 1. There may be minimal invasion by neoplastic glands into the surrounding non-neoplastic prostatic tissue. The glands are of intermediate size and larger than those in Gleason pattern 1. The variation in glandular size and separation between glands is less than that seen in pattern 3. Although not evaluated in Gleason grading, the cytoplasm of Gleason pattern 2 cancers is abundant and pale-staining (Fig. 1). Gleason pattern 2 is usually seen in transition zone cancers but may occasionally be found in the peripheral zone.
Gleason pattern 3
This pattern is the most common pattern but is morphologically heterogeneous. The glands are infiltrative, and the distance between them is more variable than that in patterns 1 and 2. Malignant glands often infiltrate between adjacent non-neoplastic glands. The glands of pattern 3 vary in size and shape and are often angular (Fig. 1). Small glands are typical for pattern 3, but there may also be large and irregular glands. Each gland has an open lumen and is circumscribed by stroma. Cribriform pattern 3 is rare and difficult to distinguish morphologically from cribriform high-grade prostatic intraepithelial neoplasia (PIN). The latter shows the presence of basal cells. These are lacking in cribriform pattern 3 prostate cancer. This heterogeneous expression of Gleason grade 3 raised an initial subdivision in patterns A, B, and C, respectively.
Gleason pattern 4
Glands appear fused, cribriform, or they may be poorly defined and small. Fused glands are composed of a group of glands that are no longer completely separated by stroma (Fig. 1). The edge of a group of fused glands is scalloped, and there are occasional thin strands of connective tissue within this group. The hypernephroid pattern described by Gleason is a rare variant of fused glands with clear or very pale-staining cytoplasm. Cribriform pattern 4 glands are large or they may be irregular with jagged edges. As opposed to fused glands, there are no strands of stroma within a cribriform gland. Most cribriform invasive cancers should be assigned a pattern 4 rather than pattern 3. Poorly defined glands do not have a lumen that is completely encircled by epithelium.
Gleason pattern 5
This pattern is characterized with an almost complete loss of glandular lumina which are only occasionally present. The epithelium forms solid sheets, solid strands, or single cells invading the stroma; comedonecrosis may be present (Fig. 1). Care must be applied when assigning a Gleason pattern 4 or 5 to limited cancer on needle biopsy to exclude an artifact of tangential sectioning of lower-grade cancer.
Rights and permissions
About this article
Cite this article
Lopez-Beltran, A., Mikuz, G., Luque, R.J. et al. Current practice of Gleason grading of prostate carcinoma. Virchows Arch 448, 111–118 (2006). https://doi.org/10.1007/s00428-005-0102-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-005-0102-4