Abstract
Purpose
Systemic chemotherapy is generally used for metastatic pancreatic cancer; however, pulmonary resection may be a treatment option for lung oligometastases from pancreatic cancer. The current study aimed to clarify the oncological outcomes and clinical benefits of pulmonary resection for lung metastases.
Methods
Of 510 patients who underwent pancreatic resection for pancreatic cancer, 44 patients with recurrence of isolated lung metastases and one patient with simultaneous lung metastases were evaluated.
Results
Of the 45 patients, 20 patients were selected as candidates for pulmonary resection based on clinical factors such as recurrence-free interval (RFI) from pancreatectomy to lung metastases, number of lung metastases, and serum CA19-9 level. The post-recurrent survival of patients with pulmonary resection was significantly better than that of patients without pulmonary resection. Fourteen of the 20 patients with pulmonary resection developed tumor recurrence with a median disease-free survival (DFS) of 15 months. Univariate analyses revealed that an RFI from pancreatectomy to lung metastases of ≥28 months was associated with better DFS after pulmonary resection. Of the 14 patients with an RFI of ≥28 months, pulmonary resection resulted in prolonged chemotherapy-free interval in 12 patients. Furthermore, repeat pulmonary resection for recurrent tumors after pulmonary resection led to further cancer-free interval in some cases.
Conclusions
Although many patients had tumor recurrence after pulmonary resection, pulmonary resection for lung metastases from pancreatic cancer may provide prolonged cancer-free interval without the need for chemotherapy. Pulmonary resection should be performed for the patients with a long RFI from pancreatectomy to lung metastases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pancreatic cancer is one of the most fatal malignancies with aggressive biology worldwide [1]. Because of a high recurrence rate after surgery, the prognosis of pancreatic cancer is dismal even after curative pancreatic resection [2]. However, patient survival differs according to the site of recurrence. Patients with lung metastases had a better prognosis than those with other hematogenous disseminations [2,3,4]. The lung is the most common site of recurrence among long-term survivors after pancreatectomy for pancreatic cancer [5]. Some studies have shown favorable biological features of lung metastases [6,7,8]. In contrast, the optimal treatment strategy for isolated lung metastases from pancreatic cancer has not been clarified.
Local treatment, including surgical resection, may be oncologically beneficial for oligometastases which are characterized by a limited number of metastases in an organ [9]. Systemic chemotherapy is commonly used and surgical resection is generally contraindicated for metastatic pancreatic cancer. Nevertheless, some studies have shown that pulmonary resection can be a treatment option in some patients with isolated lung metastases from pancreatic cancer as well as patients with lung metastases from colorectal cancer [10,11,12]. The criteria for resection of lung metastases have not been determined; however, recurrence-free interval (RFI) from primary lesion pancreatectomy to lung metastases may be an important factor for appropriately selecting patients with oligometastatic state of disease for surgical resection [13]. Growing evidence from retrospective studies has shown that pulmonary resection of isolated lung metastases is associated with long-term survival in some patients [14,15,16,17]. However, the effect of surgical resection on extending patient survival is uncertain because the selection of patients with relatively indolent diseases might cause survival benefits after pulmonary resection. Furthermore, oncological outcomes and clinical benefits of pulmonary resection for patients with lung metastases have not been clarified. We hypothesized that pulmonary resection may contribute to long-term disease control or cure of disease in some patients. This study aimed to elucidate the clinical benefits of surgical management for lung metastases from pancreatic cancer by performing a detailed investigation of postoperative oncological outcomes after pulmonary resection.
Materials and methods
Patients
We retrospectively analyzed the medical records of 510 consecutive patients with pancreatic cancer who underwent R0 or R1 pancreatic resection at the Department of General Surgery in Chiba University (Chiba, Japan) between January 2006 and December 2021. Pancreatic tumors were surgically resected using the standard method and technique, as described in previous studies [18, 19]. Pathological specimens were examined based on the TNM classification of Malignant Tumors, 8th Edition, of the Union for International Cancer Control. The resection margin status was microscopically evaluated and resection margin positive (R1) was defined as tumors with cancer cells at the transection line. The patients received adjuvant chemotherapy, which is the standard treatment, after pancreatectomy for 6 months. Regarding postoperative surveillance, the patients underwent routine blood tests including tumor markers every 3 months and contrast-enhanced chest and abdominal computed tomography (CT) every 6 months. Written informed consent was obtained from all patients according to the ethical standards of the 1975 Declaration of Helsinki. The study protocol was approved by the Committee on Human Research of Chiba University School of Medicine (approval code: #3302).
Detection and treatment of isolated lung metastases from pancreatic cancer
Tumor recurrence was commonly detected on CT during follow-up after pancreatectomy. If lung metastases were observed on CT, positron emission tomography was performed to rule out concomitant extrapulmonary recurrence and validate isolated lung metastases. The treatment strategy for isolated lung metastases (either pulmonary resection or systemic chemotherapy) was determined based on some clinical factors in each case. Furthermore, thoracic surgeons determined the indication of pulmonary resection and performed the surgery. Lung metastases from pancreatic cancer were pathologically diagnosed after pulmonary resection. Some patients received preoperative chemotherapy and/or adjuvant chemotherapy. The patients were followed up after treatment for isolated lung metastases until death or June 30, 2023. Patients who presented with tumor recurrence after pulmonary resection underwent repeat resection or received chemotherapy based on the timing and patterns of the recurrent tumor.
Statistical analyses
Differences in the clinicopathological features between the pulmonary resection and non-resection groups were assessed using the chi-squared test, Fisher’s exact test, or t-test. Survival after lung metastases or pulmonary resection was analyzed using the Kaplan-Meier method, and statistical significance was examined using the log-rank test. Data were analyzed using JMP (SAS institute, Cary, NC, the USA). All tests were two-tailed, p-values of <0.05 were used to denote statistical significance.
Results
Patients
Figure 1 shows the patient cohort in this study. Of the 510 patients with pancreatic cancer who underwent curative pancreatectomy, one patient with synchronous lung metastases underwent pulmonary resection after pancreatectomy for curative intent. Of the 396 patients who had tumor recurrence after pancreatectomy, 44 (11.1%) patients had isolated lung metastases from pancreatic cancer. Regarding treatment for metachronous isolated lung metastases, 19 patients underwent pulmonary resection, whereas 25 patients received chemotherapy and did not undergo pulmonary resection.
Treatment strategy for isolated lung metastases and its outcome
To identify the characteristics of patients who underwent pulmonary resection for isolated lung metastases from pancreatic cancer in this cohort, the clinicopathological features of the pulmonary resection group (n = 20, including one patient with synchronous lung metastases) and the non-resection group (n = 25) were investigated (Table 1). The results showed no significant difference in age, sex, and pathological factors for primary pancreatic cancer, except for resection margin status between the resection and non-resection groups. In contrast, the RFI from initial pancreatectomy to lung metastases of the resection group was significantly longer than that of the non-resection group. Furthermore, the number of lung metastases was limited in the resection group, and pulmonary resection tended to be performed for patients with unilateral lung metastases. The resection group had significantly lower serum CA19-9 levels at the time of lung metastases than the non-resection group. These results suggest that candidates for pulmonary resection were selected based on the clinical factors of lung metastases, rather than the primary lesion. Then, the prognosis of patients with isolated lung metastases was analyzed according to the treatment strategy using the Kaplan-Meier method (Fig. 2). The resection group had a significantly longer median post-relapse survival time than the non-resection group (51 months vs 26 months, respectively; p = 0.001), indicating that pulmonary resection is associated with long-term survival after metastases in some patients with isolated lung metastases.
Pulmonary resection and postoperative course
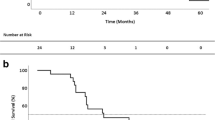
To investigate the details of pulmonary resection in our cohort, the clinical features of patients who underwent pulmonary resection for isolated lung metastases from pancreatic cancer were analyzed (Table 2). Of 20 patients, 15 developed lung metastases more than 2 years after the initial pancreatectomy. Furthermore, 15 patients presented with solitary lung metastasis and five patients had two metastases or more. Four patients received preoperative chemotherapy before pulmonary resection. The regimen administered as preoperative chemotherapy included gemcitabine (GEM) monotherapy (n = 1), S-1 monotherapy (n = 2), and nab-paclitaxel plus GEM (n = 1). Moreover, 60% of the patients underwent wedge resection, and no mortality occurred after pulmonary resection. All resected lung tumors were histologically diagnosed as lung metastases from pancreatic cancer by pathologists. When diagnosing lung metastases was difficult only by hematoxylin and eosin staining, immunohistochemical staining was also performed to confirm the lung metastases in some cases. After pulmonary resection, 12 patients received adjuvant chemotherapy. The regimen administered as adjuvant chemotherapy included GEM monotherapy (n = 4), S-1 monotherapy (n = 7), and nab-paclitaxel plus GEM (n = 1). At the time of analysis, 14 (70%) of 20 patients developed tumor recurrence after pulmonary resection for isolated lung metastases, and the median disease-free survival (DFS) time after pulmonary resection was 15 months (Supplementary Fig. 1). The types of recurrence after pulmonary resection included lung metastases (n = 9), pleural dissemination (n = 1), liver metastases (n = 1), ovarian metastases (n = 1), bone metastases (n = 1), and multiple-site metastases (n = 1). Regarding the treatment for recurrent tumor after pulmonary resection, five patients underwent repeat pulmonary resection for another lung metastases, and one patient underwent ovarian resection with curative intent. Meanwhile, other patients received chemotherapy or the best supportive care.
Clinical factors affecting DFS after pulmonary resection for isolated lung metastases
To identify the appropriate indication of pulmonary resection for isolated lung metastases, the association between clinical factors and DFS after pulmonary resection was evaluated by univariate analyses. No significant difference in DFS was observed based on the factors for primary pancreatic cancer such as tumor location, tumor differentiation, pT factor, pN factor, and resection margin status. Regarding clinical factors for lung metastases, the optimal cutoff value of RFI from pancreatectomy to lung metastases was 28 months based on receiver operating characteristic analyses of RFI associated with 1-year DFS. Patients with an RFI of ≥28 months had a significantly better DFS after pulmonary resection than those with an RFI of <28 months (Fig. 3a). In contrast, no significant difference in DFS was observed based on the number of lung metastases (Fig. 3b), serum CA19-9 level at the time of lung metastases (Fig. 3c), and induction of adjuvant chemotherapy after pulmonary resection (Fig. 3d). These results indicate that patients with a long RFI from pancreatectomy to lung metastases can be a good candidate for pulmonary resection.
a Disease-free survival rate after pulmonary resection for isolated lung metastases from pancreatic cancer stratified according to recurrence-free interval (RFI) from initial pancreatectomy to lung metastases. b Disease-free survival rate after pulmonary resection for isolated lung metastases from pancreatic cancer stratified according to the number of lung metastases. c Disease-free survival rate after pulmonary resection for isolated lung metastases from pancreatic cancer stratified according to serum CA19-9 level at the time of lung metastases. d Disease-free survival rate after pulmonary resection for isolated lung metastases from pancreatic cancer stratified according to the induction of adjuvant chemotherapy after pulmonary resection
Clinical benefits of pulmonary resection for lung metastases
Our results suggest that patients with a long RFI can receive some clinical benefits from pulmonary resection. Figure 4 shows the association between the interval from pancreatectomy to pulmonary resection and postoperative course after pulmonary resection in each patient undergoing pulmonary resection. As expected, four of six patients with an RFI of <28 months had early tumor recurrence, thereby resulting in continuous chemotherapy after lung metastases and poor prognosis (cases 1–3, and 5). In contrast, 13 of 14 patients with an RFI of ≥28 months did not have tumor recurrence within 12 months after pulmonary resection (cases 6–8, 10–12, and 14–20), and among them, 12 patients had a chemotherapy-free interval of ≥6 months after lung metastases. These results indicate that pulmonary resection may contribute prolonged chemotherapy-free interval in patients with a long RFI. Furthermore, among the patients with an RFI of ≥28 months in our all cohort, patients who underwent pulmonary resection had significantly longer median post-relapse survival time than patients who did not undergo pulmonary resection (68 months vs 9 months, respectively; p < 0.001).
All patients who underwent pulmonary resection. Yellow column: cancer-free interval, Red column: interval with recurrent tumor, Purple arrow: preoperative chemotherapy, Blue arrow: adjuvant chemotherapy. Red arrow: palliative chemotherapy, star: repeat resection for recurrence. AD: alive with disease, AW: alive without disease, DD: died of disease
Although many patients had tumor recurrence after pulmonary resection, six patients underwent repeat resection for recurrent tumors after pulmonary resection. Among them, three patients who underwent repeat pulmonary resection had a DFS time of 18 months or longer after repeat resection and achieved further chemotherapy-free interval as shown in Figure 4 (cases 11, 12, and 17). These results suggest that repeat pulmonary resection may further prolong the disease-free interval without the need for chemotherapy after repeat resection, which results in long-term survival in some patients.
Discussion
This study confirmed that pulmonary resection resulted in a favorable prognosis in some patients with isolated lung metastases from pancreatic cancer. Although most patients developed tumor recurrence after pulmonary resection, patients with an RFI of ≥28 months commonly experienced a longer cancer-free interval without the need for chemotherapy after pulmonary resection. The clinical benefits of pulmonary resection for lung metastases have not been clarified; however, this study demonstrated that pulmonary resection resulted in prolonged chemotherapy-free interval, which is beneficial for patients with metastatic pancreatic cancer. Furthermore, our study also showed that repeat pulmonary resection may further prolong the cancer-free interval without the need for chemotherapy in some patients with lung recurrence after pulmonary resection.
Despite the aggressive tumor behavior of metastatic pancreatic cancer, some reports showed that patients with lung metastases had better survival than those with recurrence at other sites, such as liver metastases and peritoneal dissemination [2,3,4]. In our analyses, 11.1% of patients with recurrence after pancreatectomy had isolated lung metastases, and some patients with isolated lung metastases had long-term survival after recurrence. These data indicate that lung metastases from pancreatic cancer have favorable biology. Previous studies have revealed that pancreatic cancer with lung recurrence commonly presents with well or moderately differentiated adenocarcinoma and minor microscopical venous invasion [20], and the percentage of SMAD4 loss is relatively low in lung metastases from pancreatic cancer [10]. Furthermore, lung metastases may have a specific tumor microenvironment in which a antitumor immune response is activated [7]. Although systemic chemotherapy is a standard treatment strategy for metastatic pancreatic cancer, resection of the metastatic tumor may be beneficial for some patients whose metastatic tumor is oligometastatic with favorable biology [12, 21, 22]. Taken together, the indication of pulmonary resection for isolated lung metastases and the clinical benefits of pulmonary resection must be well defined.
Previous reports have revealed that pulmonary resection is associated with long-term survival in some patients with lung metastases from pancreatic cancer [15,16,17]. However, no randomized controlled trial has evaluated whether surgical resection of lung metastatic tumors extends patient survival. Our data also showed that patients who underwent pulmonary resection had a significantly better post-relapse survival than those who did not undergo surgical resection. However, prudent patient selection was made based on the clinical factors of lung metastases. Additionally, compared with patients without pulmonary resection, those who underwent pulmonary resection had a significantly lower rate of R1 resection at pancreatectomy, which may reflect biologically less aggressive tumors [23]. Based on these results, the appropriate selection of candidates for pulmonary resection is important for long-term survival. Our study revealed that patients with an RFI of ≥28 months had prolonged DFS after pulmonary resection. As shown in previous reports [13, 24], the interval between pancreatectomy and lung metastases should be considered for the indication of surgical treatment. Even though patients who had pulmonary resection were selected based on their long RFI in our cohort, our postoperative surveillance detected that patients who underwent pulmonary resection commonly developed tumor recurrence after pulmonary resection with a median DFS of 15 months. These results indicate that pulmonary resection contributes to long-term disease control rather than cure. Furthermore, most patients with an RFI of ≥28 months could experience a prolonged chemotherapy-free interval after pulmonary resection. Considering that continuous systemic chemotherapy is recommended for metastatic pancreatic cancer, achieving a cancer-free interval without the need for chemotherapy is an important benefit of pulmonary resection for the patients with lung metastases.
This study revealed that the median DFS time after pulmonary resection was 15 months. In contrast, post-relapse survival time after lung metastases was 50 months among patients who underwent pulmonary resection. These results suggest that treatment for recurrent tumors after pulmonary resection is also important. Our data showed that lung was the most common recurrent site after pulmonary resection, and repeat pulmonary resection for lung recurrence prolonged the cancer-free interval without the need for chemotherapy after repeat resection in some cases (cases 11, 12, and 17). These results suggest that repeat pulmonary resection can be a promising treatment option for recurrent tumors after pulmonary resection. Because the DFS after pulmonary resection was relatively long in these cases, the interval from pulmonary resection to the detection of another lung metastasis may be an important factor for the indication of repeat pulmonary resection.
Multidisciplinary treatment is necessary for improving the prognosis of patients with advanced-stage pancreatic cancer [25, 26]. Previous studies reported that some patients received chemotherapy before and/or after pulmonary resection [13, 16, 27]. One of the roles of preoperative chemotherapy before pulmonary resection may be patient selection for the operation. This study revealed that all patients with an RFI of <28 months experienced early tumor recurrence leading to poor survival. Therefore, preoperative chemotherapy may be beneficial for patients with a short RFI after the initial pancreatectomy. In this study, only four (20%) patients received preoperative chemotherapy. Several patients with solitary lung metastases underwent upfront pulmonary resection possibly because second primary lung cancer could not be ruled-out as a preoperative differential diagnosis only by radiological examinations. Even though previous reports showed the role of adjuvant chemotherapy after pulmonary resection in extending patient survival [27], no significant difference in DFS after pulmonary resection was observed between patients with adjuvant chemotherapy and those without adjuvant chemotherapy in our analyses. The discrepancy in results may be caused by the difference in patient cohort and indication of pulmonary resection and adjuvant chemotherapy among various studies. Indeed, this study revealed that some patients with a relatively long RFI did not receive adjuvant chemotherapy after pulmonary resection but had long DFS (cases 8, 12, 15, 18, and 19). Adjuvant chemotherapy may eliminate minimal residual disease even after curative resection [28, 29]. Therefore, adjuvant chemotherapy after pulmonary resection should be performed on patients who cannot be categorized as having an oligometastatic state of the disease based on their clinical factors.
This study had some limitations. First, it was a retrospective study and was conducted at a single institution. Thus, selection bias may be present. The less aggressive biological features of lung metastases might influence the prognosis after pulmonary resection. Indeed, patients with an RFI of ≥28 months had significantly lower venous invasion of the primary tumor than patients with an RFI of <28 months in our cohort. However, our data clearly demonstrated that pulmonary resection lead to chemotherapy-free interval in the patients with metastatic pancreatic cancer. The effects of pulmonary resection on extending post-relapse survival should be elucidated in further studies. Second, a relatively small sample size was included in this study. However, this research is one of the largest case series on pulmonary resection for lung metastases from pancreatic cancer. Further studies should be conducted to validate the clinical role and indication of pulmonary resection.
Conclusion
Pulmonary resection may lead to prolonged disease-free interval without the need for chemotherapy in patients with isolated lung metastases from pancreatic cancer, though tumor recurrence was frequently observed after pulmonary resection. Pulmonary resection should be considered in some patients with a long RFI from pancreatectomy to lung metastases. Furthermore, repeat pulmonary resection can be a treatment option for recurrent tumors after pulmonary resection in selected patients.
References
Siegel RL, Miller KD, Fuchs HE, Jemal A (2022) Cancer statistics, 2022. CA Cancer J Clin 72(1):7–33
Groot VP, Rezaee N, Wu W, Cameron JL, Fishman EK, Hruban RH, Weiss MJ, Zheng L, Wolfgang CL, He J (2018) Patterns, timing, and predictors of recurrence following pancreatectomy for pancreatic ductal adenocarcinoma. Ann Surg 267(5):936–945
Zheng B, Ohuchida K, Yan Z, Okumura T, Ohtsuka T, Nakamura M (2017) Primary recurrence in the lung is related to favorable prognosis in patients with pancreatic cancer and postoperative recurrence. World J Surg 41(11):2858–2866
Wangjam T, Zhang Z, Zhou XC, Lyer L, Faisal F, Soares KC, Fishman E, Hruban RH, Herman JM, Laheru D et al (2015) Resected pancreatic ductal adenocarcinomas with recurrence limited in lung have a significantly better prognosis than those with other recurrence patterns. Oncotarget 6(34):36903–36910
Katz MH, Wang H, Fleming JB, Sun CC, Hwang RF, Wolff RA, Varadhachary G, Abbruzzese JL, Crane CH, Krishnan S et al (2009) Long-term survival after multidisciplinary management of resected pancreatic adenocarcinoma. Ann Surg Oncol 16(4):836–847
Reichert M, Bakir B, Moreira L, Pitarresi JR, Feldmann K, Simon L, Suzuki K, Maddipati R, Rhim AD, Schlitter AM et al (2018) Regulation of epithelial plasticity determines metastatic organotropism in pancreatic cancer. Dev Cell 45(6):696–711 e698
Sasaki T, Nishiwada S, Nakagawa K, Nagai M, Terai T, Hokuto D, Yasuda S, Matsuo Y, Doi S, Sho M (2022) Integrative analysis identifies activated anti-tumor immune microenvironment in lung metastasis of pancreatic cancer. Int J Clin Oncol 27(5):948-957
Kruger SF, Lohneis A, Abendroth A, Berger AW, Ettrich TJ, Waidmann O, Kapp M, Steiner B, Kumbrink J, Reischer A et al (2022) Prognosis and tumor biology of pancreatic cancer patients with isolated lung metastases: translational results from the German multicenter AIO-YMO-PAK-0515 study. ESMO Open 7(1):100388
Hellman S, Weichselbaum RR (1995) Oligometastases. J Clin Oncol 13(1):8–10
Arnaoutakis GJ, Rangachari D, Laheru DA, Iacobuzio-Donahue CA, Hruban RH, Herman JM, Edil BH, Pawlik TM, Schulick RD, Cameron JL et al (2011) Pulmonary resection for isolated pancreatic adenocarcinoma metastasis: an analysis of outcomes and survival. J Gastrointest Surg 15(9):1611–1617
Ripley RT, Downey RJ (2014) Pulmonary metastasectomy. J Surg Oncol 109(1):42–46
Sakaguchi T, Valente R, Tanaka K, Satoi S, Del Chiaro M (2019) Surgical treatment of metastatic pancreatic ductal adenocarcinoma: a review of current literature. Pancreatology 19(5):672–680
Thomas RM, Truty MJ, Nogueras-Gonzalez GM, Fleming JB, Vauthey JN, Pisters PW, Lee JE, Rice DC, Hofstetter WL, Wolff RA et al (2012) Selective reoperation for locally recurrent or metastatic pancreatic ductal adenocarcinoma following primary pancreatic resection. J Gastrointest Surg 16(9):1696–1704
Lovecek M, Skalicky P, Chudacek J, Szkorupa M, Svebisova H, Lemstrova R, Ehrmann J, Melichar B, Yogeswara T, Klos D et al (2017) Different clinical presentations of metachronous pulmonary metastases after resection of pancreatic ductal adenocarcinoma: Retrospective study and review of the literature. World J Gastroenterol 23(35):6420–6428
Yamashita K, Miyamoto A, Hama N, Asaoka T, Maeda S, Omiya H, Takami K, Doki Y, Mori M, Nakamori S (2015) Survival impact of pulmonary metastasis as recurrence of pancreatic ductal adenocarcinoma. Dig Surg 32(6):464–471
Yasukawa M, Kawaguchi T, Kawai N, Tojo T, Taniguchi S (2017) Surgical Treatment for pulmonary metastasis of pancreatic ductal adenocarcinoma: study of 12 cases. Anticancer Res 37(10):5573–5576
Stuart CM, Kirsch MJ, Zhuang Y, Meguid CL, Sugawara T, Colborn KL, Messersmith W, Lieu C, Gleisner AL, Del Chiaro M et al (2023) Pulmonary metastasectomy is associated with survival after lung-only recurrence in pancreatic cancer. Surgery 174(3):654–659
Konishi T, Takano S, Furukawa K, Takayashiki T, Kuboki S, Suzuki D, Sakai N, Hosokawa I, Mishima T, Ohtsuka M (2022) Impact of resection margin status on survival after operation for pancreatic head cancer with extrapancreatic nerve plexus invasion. J Surg Oncol 126(6):1038–1047
Hartwig W, Vollmer CM, Fingerhut A, Yeo CJ, Neoptolemos JP, Adham M, Andren-Sandberg A, Asbun HJ, Bassi C, Bockhorn M et al (2014) Extended pancreatectomy in pancreatic ductal adenocarcinoma: definition and consensus of the International Study Group for Pancreatic Surgery (ISGPS). Surgery 156(1):1–14
Hayashi K, Ono Y, Takamatsu M, Oba A, Ito H, Sato T, Inoue Y, Saiura A, Takahashi Y (2022) Prediction of recurrence pattern of pancreatic cancer post-pancreatic surgery using histology-based supervised machine learning algorithms: a single-center retrospective study. Ann Surg Oncol 29:4624–4634
Damanakis AI, Ostertag L, Waldschmidt D, Kutting F, Quaas A, Plum P, Bruns CJ, Gebauer F, Popp F (2019) Proposal for a definition of “oligometastatic disease in pancreatic cancer”. BMC Cancer 19(1):1261
Takeda T, Sasaki T, Okamoto T, Kasuga A, Matsuyama M, Ozaka M, Inoue Y, Takahashi Y, Saiura A, Sasahira N (2022) Outcomes of pancreatic cancer with liver oligometastasis. J Hepatobiliary Pancreat Sci 30(2):229–239
Kimbrough CW, St Hill CR, Martin RC, McMasters KM, Scoggins CR (2013) Tumor-positive resection margins reflect an aggressive tumor biology in pancreatic cancer. J Surg Oncol 107(6):602–607
Ilmer M, Schiergens TS, Renz BW, Schneider C, Sargut M, Waligora R, Weniger M, Hartwig W, Ceyhan GO, Friess H et al (2019) Oligometastatic pulmonary metastasis in pancreatic cancer patients: safety and outcome of resection. Surg Oncol 31:16–21
Sakaguchi T, Satoi S, Yamamoto T, Yamaki S, Sekimoto M (2020) The past, present, and future status of multimodality treatment for resectable/borderline resectable pancreatic ductal adenocarcinoma. Surg Today 50(4):335–343
Yoshitomi H, Takano S, Furukawa K, Takayashiki T, Kuboki S, Ohtsuka M (2019) Conversion surgery for initially unresectable pancreatic cancer: current status and unresolved issues. Surg Today 49(11):894–906
Homma Y, Endo I, Matsuyama R, Sho M, Mizuno S, Seyama Y, Hirano S, Aono T, Kitami C, Morita Y et al (2022) Outcomes of lung metastasis from pancreatic cancer: a nationwide multicenter analysis. J Hepatobiliary Pancreat Sci 29(5):552–561
Nissan A (2007) Minimal residual disease, the rationale for adjuvant therapy in epithelial malignancies. J Surg Oncol 96(3):185–187
Pantel K, Alix-Panabieres C (2019) Liquid biopsy and minimal residual disease - latest advances and implications for cure. Nat Rev Clin Oncol 16(7):409–424
Author information
Authors and Affiliations
Contributions
All authors contributed to the study concept and design. Material preparation, data collection and analyses were performed by Takanori Konishi and Shigetsugu Takano. Interpretation of data were performed by Takanori Konishi and Masayuki Ohtsuka. The first draft of the manuscript was written by Takanori Konishi. Critical revision of manuscript was performed by Shigetsugu Takano. All authors commented on previous versions of the manuscript. All authors read and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
Supplementary Fig. 1 Disease-free survival rate after pulmonary resection for isolated lung metastases from pancreatic cancer. (PPTX 37 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Konishi, T., Takano, S., Takayashiki, T. et al. Clinical benefits of pulmonary resection for lung metastases from pancreatic cancer. Langenbecks Arch Surg 409, 11 (2024). https://doi.org/10.1007/s00423-023-03198-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00423-023-03198-4