Abstract
Previous studies demonstrated that no significant relationships exist between salivary and serum IL-6 in resting conditions and following exercise and that appropriate saliva collection procedures allow to avoid analytical drawbacks. This investigation aimed to: (a) compare the effects of two methods of saliva collection on IL-6 assay; (b) search for correlation between salivary and serum IL-6 in resting and post-exercise conditions; (c) evaluate the IL-6 response to isometric contractions. Seventeen sedentary subjects and fifteen athletes underwent one blood and two salivary draws: saliva was collected chewing on cotton salivettes and using a plastic straw (SA method and ST method, respectively). Afterwards, the athletes only completed a fatiguing isometric exercise of the knee extensors and blood and saliva were sampled after the exercise. In the entire group (n = 32), ST method produced higher IL-6 levels than SA method and serum sampling. The exercise elicited significant responses of lactate, serum IL-6, salivary IL-6 (by ST method): salivary IL-6 values using the ST collection method were higher at each sampling point than with the SA method. The correlation analyses applied to both resting levels in the entire group and absolute changes above baseline in the athlete group showed that: (1) no significant relationships exist between serum and salivary IL-6 levels; (2) the greater the salivary IL-6 measurement, the higher the resultant inaccuracy of the SA method; (3) significant correlations exist between isometric force and mechanical fatigue during exercise and peaks of lactate and serum IL-6. These data provided demonstration of a cotton-interference effect for the results of salivary IL-6 assay and confirmed the lack of significant correlation between salivary and serum IL-6 in resting and post-exercise conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Substantial research interest in recent years has focused on the remarkable ability of contracting skeletal muscle to increase circulating levels of interleukin-6 (IL-6) (Fischer 2006; Pedersen and Febbraio 2005; Pedersen and Hoffman-Goetz 2000; Pedersen et al. 2001; Petersen and Pedersen 2005; Steensberg et al. 2000, 2001).
In a previous issue of this journal (Minetto et al. 2005), we examined the serum and salivary IL-6 responses to different protocols of acute exercise (spinning activity and isokinetic muscle contractions): we found no significant correlations between salivary and serum IL-6 levels, possibly in relation to analytical drawbacks relative to the salivary assay. In fact, we used a method of absorbing salivary samples based on cotton salivettes and it is known from Shirtcliff et al. (2001) that immunoassay of salivary samples collected using cotton-based materials may be affected by inaccuracy: cotton-related elevations in assay results might be due to the presence of an interfering substance in the cotton, whereas cotton-related reductions in assay results raise the possibility that some of the molecules in the sample are adhering to the cotton fibers. However, IL-6 was not evaluated by Shirtcliff et al. (2001) and, to our knowledge, no subsequent study has investigated the existence of a cotton-interference effect for the results of salivary IL-6 assayed in resting and post-exercise conditions.
Moreover, we identified the lack of significant relationships between the systemic (muscular) and the salivary routes of IL-6 production as a further possible explanation of the observed lack of correlation between salivary and serum IL-6 (Minetto et al. 2005). Several sources of salivary IL-6 were identified in the oral cavity, whereas the precise cellular sources of IL-6 within skeletal muscles and blood remain not completely defined. A fiber-type specificity of the contraction-related IL-6 production has been reported, but the data are conflicting (Fischer et al. 2004; Hiscock et al. 2004; Penkowa et al. 2003; Plomgaard et al. 2005): taken together, the available evidences suggest that both type I and II muscle fibers have the capacity to synthesize IL-6, depending on the level of motor unit recruitment and hence on mode and level of muscle contraction. It was also demonstrated that IL-6 that appears in the circulation during exercise could potentially come from tendons and adipose tissue (Fischer 2006) and from the brain as well (Pedersen and Febbraio 2005).
In the present work, serum and salivary IL-6, and lactate levels were analyzed before a fatiguing isometric exercise, immediately after and throughout the subsequent rest time of 2 h, aiming to: (a) compare the effects of two different methods of salivary sample collection on IL-6 assay; (b) search for correlation between salivary and serum IL-6 levels in resting conditions and following strenuous exercise; (c) evaluate the IL-6 response to isometric contractions and search for correlation among IL-6 levels, mechanical performance, and lactate response.
Point (a) aims to evaluate the existence of a cotton-interference effect for the results of salivary IL-6; point (b) aims to re-examine the previously reported lack of significant relationship between salivary and serum IL-6 after improvement of the saliva collection procedure with the consequent correction of the cotton-interference effect; point (c) aims to characterize the IL-6 response to a mode of acute exercise that has not yet been evaluated in previous studies.
Materials and methods
Subjects
Seventeen sedentary (<1 h per week of physical activity for the last 3 years) subjects of both genders (seven females and ten males) and fifteen male athletes participated in the study (Table 1 summarizes their characteristics). They underwent a medical examination and a standard set of blood tests.
The athlete group consisted of: two marathon runners, two long-distance cyclists, six triathletes, one body builder, one rugby player, three martial art competitors. They were all elite athletes competing at national or international level. Median (1st quartile–3rd quartile) value of training history was 6 (4–14) years and median (1st quartile–3rd quartile) value of training volume over the last 3 years was 9 (7–9) h/week.
None of the subjects was a current smoker, or was receiving any medication. They were instructed not to perform exercise 24 h before the experiments which were always carried out in the morning (hours 10:00–13:00) to rule out the possible confounding effects of circadian variability (Vgontzas et al. 1999). All the subjects were informed about risks and discomfort via verbal and written information. The study was approved by the Regional Ethics Committee and was in accordance with the Declaration of Helsinki.
Experimental procedure
All the subjects underwent a single blood and two salivary draws, in randomized order. The saliva was collected adopting two different methods: spitting out 3–5 ml of saliva through a short plastic straw into a collection vial and chewing on a cotton salivette (Sarstedt, Nümbrecht, Germany) over a 1-min period, according to the manufacturer’s instructions. The salivette assembly consists of a cotton swab (similar to dental cotton) and two plastic tubes that fit one inside the other. These two methods will be indicated in the following as straw-based saliva collection method and salivette-based saliva collection method and abbreviated as ST method and SA method, respectively.
Afterwards, the athletes only completed a fatiguing exercise of intermittent isometric contractions of the lower limbs to evaluate the post-exercise increases in serum and salivary IL-6 and in capillary lactate.
For each individual experimental session, the following protocol was adopted:
Basal reference trial (Pre): After an adequate warm-up consisting of light aerobic activity on a bicycle ergometer (15 min cycling at 75–100 W) and lower body static stretches, each athlete was positioned on the isometric dynamometer mounted on a horizontal leg press machine. The appropriate knee angle (120°) was obtained by sliding the leg press seat and checked using a manual goniometer. The athlete was required to perform three isometric maximal voluntary contractions (MVCs) of the knee extensors (5-s long with 3 min of recovery between each MVC and the next) and the highest of the three MVC values was taken as reference.
Fatiguing exercise (EXE): After 5 min of recovery from the last MVC, each athlete performed eight sets of 20 intermittent isometric contractions of the knee extensor muscle groups (3-s contraction/1-s relaxation), with a rest period of 2 min between each set. The athlete was asked to maintain the contraction level at a constant value of about 90% MVC. The complete exercise routine (a total of 160 isometric contractions) was completed in about 25 min. Visual feedback on the force level was provided to the subject during both MVCs and intermittent contractions and strong verbal encouragement was provided throughout by the same investigator.
End of the fatiguing exercise (Post) and recovery: Blood and saliva for IL-6 determinations were sampled immediately after the termination of EXE (Post) and 30, 60, 90, 120 min thereafter. Capillary blood samples for lactate analysis were taken during (Intermediate, Int, after the fourth set), immediately after the termination of EXE (Post) and 30, 60 min thereafter.
Instrumentation and measurements
The isometric dynamometer (Ergometer by Globus Italia s.r.l., Codognè, Italy) consisted of two force transducers (load cells) connected to an external unit composed of a microprocessor and a feedback system. As represented in Fig. 1a, the athlete performed the exercise in a seated position and the load cells were placed over the support plane of the feet. Force data were sampled at 2,048 samples/s, digitized by a 12 bit A/D converter and stored on a disk of a personal computer.
a The isometric leg press device used in the study. b Example of time course of force during one set of 20 intermittent isometric contractions from one athlete (3-s contraction/1-s relaxation): the dotted line fits the intercepts (grey circles) of the regressions within each of the 20 isometric contractions
Serum and salivary IL-6 concentrations were measured by commercially available ELISA (Quantikine High Sensitivity human IL-6 immunoassay, R&D Systems, Abingdon, UK—dynamic range: 0.156–10 pg/ml). Dilution (1:4) was chosen for salivary IL-6 measurements, as previously validated (Minetto et al. 2005). All ELISA readings were performed by Microreader Instrument ELx808-I (Bio-Tek Instruments, Inc., Winooski, Vermont, USA). Absorbance was measured at 490 and 630 nm. Data were analyzed by KC4 v3.0 software (Bio-Tek Instruments).
All samples from each individual subject were tested in duplicate in the same assay run. Blood and saliva samples were centrifuged at 3,000 RPM for 15 min at room temperature (for the SA method, centrifugation is needed to separate the saliva from the cotton tube) and stored at −20°C until assayed. All salivary samples were subjected to a single freeze–thaw cycle to break down mucopolysaccharides that can interfere with pipetting (Kirschbaum and Hellhammer 1989) and on the day of the assay were centrifuged at 3,000 RPM for 15 min to remove particulate matter.
Minimum detectable concentration was 0.01 pg/ml for serum assay and 0.06 pg/ml for salivary assay. Intra- and inter-assay coefficients of variation for serum and salivary assays were below 3.5 and 5%, respectively.
Finger-prick blood samples were obtained from each athlete to evaluate lactate concentration using the Lactate Pro (Arkray, Inc., Kyoto, Japan) hand-held portable analyzer. Previous research has demonstrated high accuracy and reliability of this measuring device (Pyne et al. 2000).
Data management and statistical analysis
Mechanical variables
The studied mechanical variables were: the peak extension force value recorded during the isometric exercise (MVCISM, N) and the maximal rate of change of the extension force produced during the whole isometric exercise (SLOPEISM, N/contraction): for each repetition of the eight isometric sets, a regression among the force values was done and the intercept calculated. Intercept and slope of the regression line fitted over the 20 intercepts provided the values of MVC and SLOPE for each set (Fig. 1b). The highest (in absolute value) values of intercept and slope were chosen, among all values of the eight sets of exercise, as MVCISM and SLOPEISM.
Statistical analysis
Normal distribution of the data was tested by Shapiro–Wilk test: because data were not normally distributed, non parametric tests were used.
Between-group analysis of age and anthropometric characteristics was performed by means of the Mann–Whitney U test.
Friedman’s ANOVA and Dunn’s post hoc test were used for the comparison of the different assays of resting serum and salivary IL-6 in the entire group of 32 subjects.
Friedman’s ANOVA was also adopted to determine whether there were any significant differences over time among the samples in the group of athletes who performed the fatiguing isometric exercise. If a significant difference was present a Dunn’s test was used to locate the difference.
The relationships both between serum and salivary IL-6 levels and between the two measurements of salivary IL-6 were assessed by means of the Spearman rank correlation analysis applied to the resting (pre-exercise, Pre) levels in the two populations (n = 32). Additionally, a Bland–Altman plot was made showing the differences between the two measurements of resting salivary IL-6 against their means to check whether the error of measurement was independent of the magnitude of the mean score (Bland and Altman 1986): the strength of the relationship between the differences and their means was evaluated by the Spearman test.
Moreover, in the athlete group only (n = 15), the non parametric Wilcoxon paired test was adopted to compare the IL-6 measurements obtained with the two methods at each sampling time and the Spearman test was applied to the absolute serum and salivary changes above baseline (peak-Pre differences). Finally, Spearman test was also adopted to assess the correlation between biochemical and mechanical variables.
Statistical analysis was performed with the Statistica 6 for Windows software package (Statsoft Inc., Tulsa, OK, USA). Bland-Altman plot was performed with the MedCalc 9 software (MedCalc, Mariakerke, Belgium).
All values are reported as median and interquartile range. Threshold for statistical significance was set to P = 0.05.
Results
In the entire group of sedentary subjects and athletes (n = 32), ST method produced higher (P < 0.001) salivary IL-6 levels with respect to both SA method and serum sampling. The median (1st quartile–3rd quartile) IL-6 resting values were: serum, 0.6 (0.5–0.9) pg/ml; saliva by SA method, 0.7 (0.2–1.8) pg/ml; saliva by ST method, 4.0 (2.0–7.4) pg/ml.
No significant correlations were found in the entire group of 32 subjects between resting serum and salivary IL-6 levels (Fig. 2).
Absence of significant correlations between serum and salivary IL-6 levels in the entire group (n = 32) of sedentary subjects (white circles) and athletes (black circles) evaluated in resting conditions. ST method straw-based saliva collection method, SA method salivette-based saliva collection method
On the contrary, a significant positive correlation was observed in the entire group of 32 subjects between resting salivary IL-6 levels obtained by the two saliva collection methods (Fig. 3a: r = 0.72, P < 0.001).
a Significant positive correlation (r = 0.72, P < 0.001) between resting salivary IL-6 levels obtained by the two saliva collection methods in the entire group (n = 32) of sedentary subjects (white circles) and athletes (black circles). b Bland-Altman plot of IL-6 measurements obtained with the two saliva collection methods: mean difference (±1.96 SD) is reported and the regression line of the differences is drawn (r = 0.85, P < 0.001). White symbols represent the differences of sedentary subjects, black symbols represent the differences of athletes
Bland-Altman plot showed (Fig. 3b): (1) a mean difference between the two measurements of resting IL-6 of 3.8 pg/ml (±1.96 SD: 8.1 pg/ml), thus suggesting that the two methods of saliva collection cannot be used interchangeably; (2) a significant positive correlation between differences and means of the two measurements (r = 0.85, P < 0.001), thus indicating that the variability in salivary IL-6 values (that is the inaccuracy of IL-6 measurements made on saliva collected with the SA method) was dependent on the magnitude of IL-6 measurement.
The protocol of fatiguing isometric exercise was completed by all the athletes. The median (1st quartile–3rd quartile) values of the studied mechanical variables were: MVCISM, 1633.0 (1273.5–1985.9) N; SLOPEISM, −0.75 (−1.42 to −0.56) N/contraction.
The fatiguing isometric exercise elicited significant responses of the following variables: lactate (Fig. 4a), serum IL-6 (Fig. 4b), salivary IL-6 (by ST method) (Fig. 4c).
Lactate (a), serum IL-6 (b), and salivary IL-6 (c) responses to fatiguing isometric exercise in the athlete group (n = 15). The values are represented as median and interquartile range. Statistically significant differences of each sampling point with respect to baseline values are highlighted (§ P < 0.001, *P < 0.01, $ P < 0.05). Pre pre-exercise, Int intermediate (after the fourth set of exercise); Post immediately after the exercise; 30, 60, 90, 120 min after the termination of the exercise. ST method straw-based saliva collection method, SA method salivette-based saliva collection method
The comparison of the two saliva collection methods showed significant IL-6 differences at each sampling point of the experimental procedure [ST method vs. SA method, median (1st quartile–3rd quartile), Pre: 5.0 (2.9–7.0) pg/ml vs. 0.6 (0.2–1.8) pg/ml, P < 0.001; Post: 7.1 (3.3–15.9) pg/ml vs. 1.1 (0.4–3.3) pg/ml, P < 0.01; 30 min: 7.1 (3.6–13.3) pg/ml vs. 1.2 (0.2–2.4) pg/ml, P < 0.001; 60 min: 5.9 (3.0–12.8) pg/ml vs. 0.9 (0.4–1.9) pg/ml, P < 0.001; 90 min: 4.8 (2.8–15.3) pg/ml vs. 1.2 (0.4–2.3) pg/ml, P < 0.001; 120 min: 4.2 (3.3–12.3) pg/ml vs. 1.4 (0.8–2.5) pg/ml, P < 0.01].
No significant correlations were found in the group of 15 athletes between serum and salivary changes above baseline levels (data not shown), whereas a weak correlation was found between the salivary changes above baseline obtained with the two IL-6 measurements (r = 0.52, P = 0.05).
Finally, significant correlations were found in the athlete group between mechanical and biochemical variables, as follows: MVCISM and SLOPEISM (r = −0.55, P = 0.03); lactate peak and SLOPEISM (r = −0.55, P = 0.03); lactate peak and IL-6 peak (r = 0.60, P = 0.02); IL-6 peak and MVCISM (r = 0.51, P = 0.04); IL-6 peak and SLOPEISM (r = −0.51, P = 0.05). Briefly, the greater isometric force and mechanical fatigue during exercise, the greater lactate production and circulating IL-6 levels.
Discussion
The main findings of this study are: (1) the demonstration that the two saliva collection methods provide IL-6 measurements which are correlated but cannot be used interchangeably, as a possible consequence of a cotton-interference effect for the results of salivary IL-6 assay: ST method produced higher salivary IL-6 levels with respect to SA method in both resting and post-exercise conditions and the greater the IL-6 measurement, the higher the resultant inaccuracy of the SA method; (2) the evidence of the lack of correlation between serum and (true) salivary IL-6 levels in both experimental conditions.
The demonstration of a cotton-interference effect represents an original observation that can partly account for the results of our previous study showing no significant relationship between serum and salivary IL-6 levels in response to different protocols of acute exercise (Minetto et al. 2005). In keeping with Shirtcliff et al. (2001), this cotton-interference effect can be explained as a consequence of the adhesion of IL-6 to the cotton fibers of the salivette and provides further support to the conclusions of their paper: “cotton may potentially interfere with immunoassay results. Appropriate collection procedures need to be taken into consideration for different types of markers”.
Alternatively, the lower IL-6 concentration observed with the cotton swab method could be due to a higher saliva flow rate as a consequence of the chewing of the swab. Since we did not measure the salivary flow rate and its possible variations due to the collection method, we cannot reject this alternative explanation of the experimental findings. However, salivary flow stimulation and the consequent dilution effect associated with the use of a cotton swab should be constant and independent of the amount of IL-6 in saliva. On the contrary, we observed that the differences between the two measurements increased with increases of salivary IL-6 amount. Moreover, we are not aware of previous studies which showed that the two methods of saliva collection herein adopted can differently modulate the salivary flow rate. It is well known that the salivary reflex is significantly activated during mastication, but the same should be true during the spitting condition. In fact, it was demonstrated that the threshold for parotid salivary reflex is very low: the reflex can be evoked by masticatory forces as low as 5% of comfortable chewing (Anderson et al. 1996). These low levels of masticatory (masseter) activity resulted in ability to stimulate the periodontal ligament mechanoreceptors, which have a major role in the parotid response to both chewing (Jensen Kjeilen et al. 1987) and empty clenching (Anderson et al. 1996).
The lack of correlation between serum and salivary IL-6 indicates that the mechanisms responsible for IL-6 release in saliva are independent of those regulating serum IL-6 levels in both resting conditions (adipose tissue and immune productions) and in response to exercise: as already mentioned in the Introduction, IL-6 that appears in the circulation during the exercise could potentially come from tendons, adipose tissue and the brain as well as from contracting muscle fibres and immune cells. This different regulation of IL-6 levels in the two different compartments is not surprising, considering that different sources of salivary IL-6 were identified in the oral cavity: acinar cells of the salivary glands (Boumba et al. 1995; Fox et al. 1994; Yao et al. 2005), oral epithelial cells (Formanek et al. 1998, 1999), periodontal ligament cells (Okada et al. 1997; Yamamoto et al. 2006), and gingival fibroblasts (Okada et al. 1997; Parkar et al. 1998). All these sources are probably regulated by specific mechanisms which can differ also with respect to those regulating muscle IL-6 production.
Consistently, Aleksandra Nielsen et al. (2005) evaluated 22 patients with inflammatory bowel disease and 19 healthy controls and found a significant correlation between saliva and plasma IL-6 concentrations only in the subgroup of seven patients with ulcerative colitis, whereas the control group exhibited no correlation between salivary and plasma IL-6. Interestingly, in this study the subjects collected the saliva spitting out into polypropylene containers without additives: the salivary IL-6 concentrations of the 19 healthy subjects (median 6.3 ng/l, range 3.0–32.5 ng/l) were comparable to the levels we found in our study.
Briefly, our data indicate that a reliable analysis of the IL-6 response to acute exercise can be made only by serial blood sampling and that no significant exercise-elicited increase in IL-6 levels can be detected in the salivary compartment after a short duration exercise: however, this finding cannot rule out that a positive association between serum and saliva IL-6 might become evident if a long duration exercise protocol (that causes higher rises in serum IL-6 concentration) is performed.
Even if many studies provided evidence of significant IL-6 increase in blood during and after exercise (Fischer 2006; Pedersen and Febbraio 2005; Pedersen and Hoffman-Goetz 2000; Pedersen et al. 2001; Petersen and Pedersen 2005; Steensberg et al. 2000, 2001), to our knowledge this is the first report that evaluated the serum IL-6 levels in response to a protocol of intermittent isometric contractions. We observed that IL-6 peak levels occurred 1 h after the termination of the exercise: this delayed increase allows us to exclude that plasma volume changes played a role in determining the observed cytokine response. Moreover, post-exercise IL-6 change presented a dynamics similar to the previously described cytokine increase in response to isokinetic exercise (Croisier et al. 1999; Minetto et al. 2006). However, in these previous papers the IL-6 peak levels were higher with respect to the values we measured in response to isometric contractions: this observation suggests that the mode of muscle contraction must be considered as an important modulator factor of the contraction-related IL-6 production. Notwithstanding this, it is presently unknown if sarcomere shortening can trigger IL-6 production mechanisms which differ with respect to the production elicited by static contractions.
With regard to serum IL-6 changes as a function of exercise intensity, we found significant correlations among force production, mechanical fatigue, lactate, and IL-6: these findings can be considered as confirmations of the well established association between exercise intensity (and level of motor unit recruitment as well) and IL-6 production, but cannot provide further insights in the debate about the fiber-type specificity of the contraction-related IL-6 production (Fischer et al. 2004; Hiscock et al. 2004; Penkowa et al. 2003; Plomgaard et al. 2005).
Moreover, a recent review of the literature has confirmed that exercise duration (rather than intensity or mode) is most important in determining the magnitude of the circulating IL-6 response to exercise (Fischer 2006). Consistently, in a pilot (unpublished) study of IL-6 evaluation in five athletes submitted to a protocol of four sets of 20 intermittent isometric contractions (80 contractions with duration and recoveries equal to the present protocol) we found serum IL-6 peak values three times lower than the values reported herein. Accordingly, the amount of IL-6 increase following submaximal long-duration (dynamic) exercises is much higher with respect to short-duration exercises (Margeli et al. 2005; Pedersen and Hoffman-Goetz 2000; Pedersen et al. 2001; Petersen and Pedersen 2005). The observed significant relationship between IL-6 release and muscle glycogen store both at rest and after exercise suggested that IL-6 may act as a carbohydrate sensor (Helge et al. 2003; Steensberg et al. 2001): on these bases, the volume effect on the amount of IL-6 production could be explained as a function of glycogen store depletion.
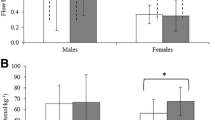
Another interesting aspect of the exercise-triggered IL-6 increase is the high inter-individual variability of IL-6 peak values that was observed in the present study, as well as in previous reports (Minetto et al. 2006; Ostrowski et al. 1998). The biological reasons why athletes can have different degrees of IL-6 increase after completing similar strenuous exercises begin to be uncovered: a recent and very interesting study showed the existence of an association between IL-1 gene variants and post-exercise IL-6 mRNA content in vastus lateralis biopsies collected before and after acute resistance exercise (Dennis et al. 2004). Moreover, Edwards et al. (2006) found a higher IL-6 response to a strenuous cycling exercise in a group of females compared to age-matched males, thus suggesting the existence of a gender dimorphism in the cytokine response to exercise.
Referring to the aims listed in the Introduction, the conclusions can be summarized as follows: (a) significant influences of the collection method were apparent on salivary IL-6 levels in both resting and post-exercise conditions; (b) no correlation was found between salivary and serum IL-6 values; (c) a significant increase in serum IL-6 levels was found after the exercise due to the muscular cytokine production: serum IL-6 levels were found to correlate with both mechanical variables and post-exercise lactate increase.
References
Aleksandra Nielsen A, Nederby Nielsen J, Schmedes A, Brandslund I, Hey H (2005) Saliva Interleukin-6 in patients with inflammatory bowel disease. Scand J Gastroenterol 40:1444–1448
Anderson DJ, Hector MP, Linden RW (1996) The effects of unilateral and bilateral chewing, empty clenching and simulated bruxism, on the masticatory-parotid salivary reflex in man. Exp Physiol 81:305–312
Bland JM, Altman DG (1986) Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1:307–310
Boumba D, Skopouli FN, Moutsopoulos HM (1995) Cytokine mRNA expression in the labial salivary gland tissues from patients with primary Sjogren’s syndrome. Br J Rheumatol 34:326–333
Croisier JL, Camus G, Venneman I, Deby-Dupont G, Juchmes-Ferir A, Lamy M, Crielaard JM, Deby C, Duchateau J (1999) Effects of training on exercise-induced muscle damage and interleukin 6 production. Muscle Nerve 22:208–212
Dennis RA, Trappe TA, Simpson P, Carroll C, Huang BE, Nagarajan R, Bearden E, Gurley C, Duff GW, Evans WJ, Kornman K, Peterson CA (2004) Interleukin-1 polymorphisms are associated with the inflammatory response in human muscle to acute resistance exercise. J Physiol 560:617–626
Edwards KM, Burns VE, Ring C, Carroll D (2006) Individual differences in the interleukin-6 response to maximal and submaximal exercise tasks. J Sports Sci 24:855–862
Fischer CP (2006) Interleukin-6 in acute exercise and training: what is the biological relevance? Exerc Immunol Rev 12:6–33
Fischer CP, Hiscock NJ, Penkowa M, Basu S, Vessby B, Kallner A, Sjoberg LB, Pedersen BK (2004) Supplementation with vitamins C and E inhibits the release of interleukin-6 from contracting human skeletal muscle. J Physiol 558:633–645
Formanek M, Knerer B, Temmel A, Thurnher D, Millesi W, Kornfehl J (1998) Oral keratinocytes derived from the peritonsillar mucosa express the proinflammatory cytokine IL-6 without prior stimulation. J Oral Pathol Med 27:202–206
Formanek M, Knerer B, Kornfehl J (1999) Cytokine expression of human oral keratinocytes. ORL J Otorhinolaryngol Relat Spec 61:103–107
Fox RI, Kang HI, Ando D, Abrams J, Pisa E (1994) Cytokine mRNA expression in salivary gland biopsies of Sjogren’s syndrome. J Immunol 152:5532–5539
Helge JW, Stallknecht B, Pedersen BK, Galbo H, Kiens B, Richter EA (2003) The effect of graded exercise on IL-6 release and glucose uptake in human skeletal muscle. J Physiol 546:299–305
Hiscock N, Chan MH, Bisucci T, Darby IA, Febbraio MA (2004) Skeletal myocytes are a source of interleukin-6 mRNA expression and protein release during contraction: evidence of fiber type specificity. FASEB J 18:992–994
Jensen Kjeilen JC, Brodin P, Aars H, Berg T (1987) Parotid salivary flow in response to mechanical and gustatory stimulation in man. Acta Physiol Scand 131:169–175
Kirschbaum C, Hellhammer DH (1989) Salivary cortisol in psychobiological research: an overview. Neuropsychobiology 22:150–169
Margeli A, Skenderi K, Tsironi M, Hantzi E, Matalas AL, Vrettou C, Kanavakis E, Chrousos G, Papassotiriou I (2005) Dramatic elevations of interleukin-6 and acute-phase reactants in athletes participating in the ultradistance foot race spartathlon: severe systemic inflammation and lipid and lipoprotein changes in protracted exercise. J Clin Endocrinol Metab 90:3914–3918
Minetto M, Rainoldi A, Gazzoni M, Terzolo M, Borrione P, Termine A, Saba L, Dovio A, Angeli A, Paccotti P (2005) Differential responses of serum and salivary interleukin-6 to acute strenuous exercise. Eur J Appl Physiol 93:679–686
Minetto MA, Rainoldi A, Gazzoni M, Ganzit GP, Saba L, Paccotti P (2006) Interleukin-6 response to isokinetic exercise in elite athletes: relationships to adrenocortical function and to mechanical and myoelectric fatigue. Eur J Appl Physiol 98:373–382
Okada N, Kobayashi M, Mugikura K, Okamatsu Y, Hanazawa S, Kitano S, Hasegawa K (1997) Interleukin-6 production in human fibroblasts derived from periodontal tissues is differentially regulated by cytokines and a glucocorticoid. J Periodontal Res 32:559–569
Ostrowski K, Hermann C, Bangash A, Schjerling P, Nielsen JN, Pedersen BK (1998) A trauma-like elevation of plasma cytokines in humans in response to treadmill running. J Physiol 15:889–894
Parkar M, Tabona P, Newman H, Olsen I (1998) IL-6 expression by oral fibroblasts is regulated by androgen. Cytokine 10:613–619
Pedersen BK, Hoffman-Goetz L (2000) Exercise and the immune system: regulation, integration, and adaptation. Physiol Rev 80:1055–1081
Pedersen BK, Febbraio M (2005) Muscle-derived interleukin-6 – a possible link between skeletal muscle, adipose tissue, liver, and brain. Brain Behav Immun 19:371–376
Pedersen BK, Steensberg A, Fischer C, Keller C, Ostrowski K, Schjerling P (2001) Exercise and cytokines with particular focus on muscle-derived IL-6. Exerc Immunol Rev 7:18–31
Penkowa M, Keller C, Keller P, Jauffred S, Pedersen BK (2003) Immunohistochemical detection of interleukin-6 in human skeletal muscle fibers following exercise. FASEB J 17:2166–2168
Petersen AM, Pedersen BK (2005) The anti-inflammatory effect of exercise. J Appl Physiol 98:1154–1162
Plomgaard P, Penkowa M, Pedersen BK (2005) Fiber type specific expression of TNF-alpha, IL-6 and IL-18 in human skeletal muscles. Exerc Immunol Rev 11:53–63
Pyne DB, Boston T, Martin DT, Logan A (2000) Evaluation of the Lactate Pro blood lactate analyser. Eur J Appl Physiol 82:112–116
Shirtcliff EA, Granger DA, Schwartz E, Curran MJ (2001) Use of salivary biomarkers in biobehavioral research: cotton-based sample collection methods can interfere with salivary immunoassay results. Psychoneuroendocrinology 26:165–173
Steensberg A, van Hall G, Osada T, Sacchetti M, Saltin B, Klarlund Pedersen B (2000) Production of interleukin-6 in contracting human skeletal muscles can account for the exercise-induced increase in plasma interleukin-6. J Physiol 529:237–242
Steensberg A, Febbraio MA, Osada T, Schjerling P, van Hall G, Saltin B, Pedersen BK (2001) Interleukin-6 production in contracting human skeletal muscle is influenced by pre-exercise muscle glycogen content. J Physiol 537:633–639
Vgontzas AN, Papanicolaou DA, Bixler EO, Lotsikas A, Zachman K, Kales A, Prolo P, Wong ML, Licinio J, Gold PW, Hermida RC, Mastorakos G, Chrousos GP (1999) Circadian interleukin-6 secretion and quantity and depth of sleep. J Clin Endocrinol Metab 84:2603–2607
Yamamoto T, Kita M, Kimura I, Oseko F, Terauchi R, Takahashi K, Kubo T, Kanamura N (2006) Mechanical stress induces expression of cytokines in human periodontal ligament cells. Oral Dis 12:171–175
Yao C, Wei W, Li X, Hosoi K (2005) Acute phase protein induction by experimental inflammation in the salivary gland. J Oral Pathol Med 34:364–367
Acknowledgments
We wish to thank Prof. E. Ghigo (Division of Endocrinology and Metabolism, Department of Internal Medicine, University of Turin, Turin, Italy) and Prof. R. Merletti (LISiN, Department of Electronics, Polytechnic of Turin, Turin, Italy) for their constructive criticisms in the manuscript editing and M. Gollin and P. Bertano (SUISM, University of Turin, Turin, Italy) for valuable assistance in the sample collection.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Minetto, M.A., Gazzoni, M., Lanfranco, F. et al. Influence of the sample collection method on salivary interleukin–6 levels in resting and post-exercise conditions. Eur J Appl Physiol 101, 249–256 (2007). https://doi.org/10.1007/s00421-007-0484-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-007-0484-x