Abstract
Cryostat sections of bovine embryos of exactly known age (obtained from artificial insemination), ranging from 32 to 60 days post-insemination, were treated with a wide range of antibodies directed against cell surface antigens or lineage-specific factors in order to demonstrate different types of fetal blood cells and their precursors. An antibody specific to bovine c-kit (bk-1) stained not only presumptive haematopoietic stem cells in the dorsal aorta and the embryonic liver, but also a subpopulation of putative primordial germ cells in the gonadal anlage, the latter being further characterised by a positive labelling with the lectins STA, WFA and WGA and a histochemical reaction for alkaline phosphatase. The antibody against CD 45, commonly regarded as a pan-leukocyte marker, reacted in the bovine embryo with different types of blood cells, as well as with presumptive vasculogenetic cells and a subpopulation of putative primordial germ cells. CD 61 immunoreaction proved to be a useful tool for demonstrating megakaryocytopoiesis in the embryonic liver, in addition to the lumen of blood vessels and the mesonephros. Staining with BM-2 was restricted to a single population of medium-sized, round to oval cells, forming small groups within the parenchymal strands of the liver. Characterised furthermore by a U-shaped nucleus, this BM-2-positive cell type apparently represents a developmental stage in the granulopoietic lineage. B-lymphocytopoiesis in the bovine liver was detected with antibodies directed against WC-4 and IgM, but not until day 58 post-insemination. Using antibodies to CD 14, no positive results could be obtained in embryonic tissues, although anti-CD 14-positive macrophages were easily recognised in lymph nodes of adult bovines. The antibody against CD 68, however, identified two populations of primitive macrophages in our samples. One population was located in parenchymal strands of the embryonic liver, probably acting as nursing cells for haematopoietic foci, and the other was observed intravasally in the sinusoids of the liver, most probably representing primitive Kupffer cells.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In recent years, the development of a wide range of monoclonal antibodies directed against bovine cell surface molecules (see, for example, Naessens and Howard 1993; Rathkolb et al. 1997) and the crossreactivity of a number of antibodies specific for human leucocyte surface antigens (Jones et al. 1993; Ackermann et al. 1994; Brodersen et al. 1998) led to a number of meticulous studies on the adult bovine immune and haematopoietic systems (see, for example, Murakami et al. 1999; Van Kampen et al. 1999; Rebelatto et al. 2000). However, very little is known about the expression of blood cell surface antigens in bovine embryos and the immunohistochemical application of the corresponding antibodies in developmental studies. The aim of this investigation was, therefore, to test a number of antibodies for their suitability to discriminate between myeloid cells, macrophages, megakaryocytes and their precursor and stem cells in bovine embryos. The first haematopoietic stem cells with the ability of adult reconstitution are located in the aorta-gonad-mesonephros region (AGM) in vertebrate embryos and display a c-kit+/CD 34+/lineage (lin)− phenotype in mouse and man (Medvinsky and Dzierzak 1996; Sanchez et al. 1996; Labastie et al. 1998; Tavian et al. 1999). C-kit is a proto-oncogene which encodes a transmembrane tyrosine kinase receptor (Yarden et al. 1987). C-kit mutation of the W locus in the mouse affects gametogenesis, pigmentation, haematopoiesis and mast cell development during murine development (reviewed in Broudy 1997; Lyman and Jacobsen 1998). Consistent with these results, the expression of c-kit mRNA (Yarden et al. 1987; Geissler et al. 1988; Qiu et al. 1988; Nocka et al. 1989; Tan et al. 1990) and c-kit protein (Horie et al. 1991, 1992; Yoshinaga et al. 1991; Maeda et al. 1992) was detected in the ovary, testis, fetal liver, bone marrow, mast cells and skin. Additionally, the embryonic expression of c-kit includes a variety of tissues in which no obvious phenotypic changes have been detected in W mutant mice, for example, gut and central nervous system (Keshet et al. 1991; Motro et al. 1991).
Recently, the cDNA for bovine c-kit (bokit) has been cloned (Kubota et al. 1994) and six bovine-specific monoclonal antibodies (bk1–6) have been raised (Hikono et al. 1999). These antibodies have been used in flow cytometry for characterisation of bokit-positive fractions of bone marrow and peripheral blood (Hikono et al. 2001a, b). In the present study, we applied bk1–6 and a commercially available c-kit antibody with broad species crossreactivity, including ovine embryonic tissue (Tisdall et al. 1999). Haematopoietic stem cells and primordial germ cells (PGCs) are both expected to display c-kit immunoreactivity. Therefore, WFA, WGA, STA or alkaline phosphatase (AP) histochemistry was used to label putative PGCs (Wrobel and Süss 1998), and a bovine-specific antibody directed against the pan-leucocyte marker CD 45 was used to mark haematopoietic cells.
Morphological and functional differences between the embryonic and adult haematopoietic system are evident; most obvious is the formation of nucleated erythrocytes in the yolk sac or the development of fetal macrophages not via monocytes, but by intercalation of a specific precursor cell type designated as primitive macrophage (Faust et al. 1997; Naito et al. 1997). The availability of molecular tools such as specific antibodies allowed an accurate description of separate haematopoietic lineages and their precursors during different stages of human embryonic and fetal development (see, for example, Timens et al. 1990). On the other hand, little is known about species-specific differences in embryonic haematopoiesis. One should expect characteristic adaptations of the embryonic haematopoiesis depending on growth rate of the yolk sac, mode of placentation, morphological structure of the AGM region or the timeline for acquisition of immunological competence. For an analysis of ongoing lineage-specific blood cell proliferation and differentiation in the bovine embryo, we tested a wide range of antibodies (BM1, BM2, CD 11b, CD 14, CD 34, CD 45, CD 61, CD 68, c-kit, IgM, myeloperoxidase, WC-4) in order to discriminate between myelopoiesis, megakaryocytopoiesis, B-lymphopoiesis and macrophages.
Materials and methods
Embryo collection and tissue preparation
For the present study, we used 12 bovine embryos of known age. Heifers of the Fleckvieh breed were artificially inseminated at the end of oestrus. Embryos were collected at slaughter in a local abattoir. The age of the embryos was determined in days post-insemination (dpi) and ranged from 32 to 60 dpi (Table 1).
Immediately after collection, the fetal membranes and the abdomen were opened prior to fixation. The fixation was carried out in two steps. Fixative I (0.5 h) contained 4% paraformaldehyde, 15% v/v saturated picric acid and 0.1% glutaraldehyde in 0.1 M phosphate buffer, pH 7.4. Fixative II (1.5 h) had the same composition, but without glutaraldehyde. Following fixation, the complete embryos were washed (3×10 min) in 0.1 M phosphate buffer and transferred into a graded series of saccharose (10%, 20%, 30%, 40%). Embryos were oriented in the desired position on brass holders, enclosed in Tissue TEK OCT compound (Miles, Elkhart, IN, USA) and frozen in liquid nitrogen. Cryostat sections (12 μm thick) were mounted on gelatine/chrome-alum-coated slides and air dried for 2–3 min before further processing.
Histochemistry
Alkaline phosphatase was demonstrated using the methods of Gomori (1952) and McGadey (1970).
Lectin histochemistry was performed with biotinylated STA, biotinylated WFA and biotinylated WGA. (STA: Solanum tuberosum lectin; binding characteristics: β-d-GlcNac. WFA: Wisteria floribunda lectin; binding characteristics GalNac 1,6 Gal >αGalNac. WGA: Triticum vulgaris lectin; binding characteristics: [β-d-GlcNac]n linked 1→4 Neu Nac]. Source of lectins: Sigma, Deisenhofen, Germany.) All steps of lectin histochemistry were carried out in a moist chamber on cryostat sections surrounded by water-repellent PAP-PEN (Science Services, Munich, Germany) as follows: (1) blocking of endogenous peroxidase with 10% methanol and 3% H2O2; (2) rinsing (3×10 min) in PBS; (3) preincubation (45 min) with blocking buffer containing 10% fetal calf serum (Seromed, Munich, Germany) and 0.005% cold water fish gelatine (Biotrend, Köln, Germany) in PBS; (4) incubation with the optimal lectin concentrations (STA: 2.0 μg/ml; WFA: 0.5 μg/ml; WGA: 0.062 μg/ml) in blocking buffer overnight at 4°C; (5) rinsing as in step 2; (6) incubation in streptavidin-peroxidase (1:200; Jackson via Dianova, Hamburg, Germany) for 60 min at room temperature; (7) rinsing as in step 2; (8) developing with a DAB (diaminobenzidine tetrahydrochloride) kit (Immunotech, Hamburg, Germany) supplemented by 0.002% CoCl·6H2O and 0.04% NiCl2·6H2O (Kujat et al. 1993); (9) rinsing twice in PBS; (10) dehydration in graded alcohol, clearing in xylol and mounting in DPX (Serva, Heidelberg, Germany). Control sections were incubated: (1) without lectin and (2) with lectins that had been diluted with PBS containing 0.2 M of the adequate inhibitory sugar (Sigma). These control sections remained unstained.
Immunohistochemistry
Immunohistochemistry was carried out in a moist chamber on cryostat sections surrounded by water repellent PAP-PEN as follows: (1) blocking of endogenous peroxidase; (2) rinsing; (3) preincubation (see blocking, preincubation and rinsing for lectin histochemistry); (4) overnight incubation at 4°C with the primary antibody (0.01 μg/ml) in blocking buffer (see Table 2); (5) rinsing as in step 2; (6) incubation (60 min) in the secondary antibody, i.e. 1:100 goat anti-mouse/biotinylated IgG (Jackson) or 1:200 goat anti-rabbit/biotinylated IgG (Jackson); (7) rinsing as in step 2; (8) incubation in avidin-biotin-peroxidase complex, ABC (Vector via Linearis, Wertheim, Germany) after Hsu et al. (1981) for 60 min; (9) rinsing as in step 2; (10) developing as in step 8 of lectin histochemistry; (11) rinsing twice in PBS; (12) dehydration in ethanol and mounting in DPX. Control sections were incubated without primary antibody and remained unstained.
Double-labelling of putative PGCs with anti-CD 45 and WFA
Double-labelling of CD 45 and WFA was performed on selected sections in one 34-dpi embryo in order to provide direct evidence for the identification of CD 45-positive cells as putative PGCs. All steps were carried out in a moist chamber on cryostat sections surrounded by water-repellent PAP-PEN. First, anti-CD 45 immunohistochemistry was carried out according to immunohistochemistry steps 1–5. Then, sections were placed in peroxidase-coupled goat anti-mouse IgG (Jackson) diluted 1:100 in blocking buffer for 1 h at room temperature. After rinsing with PBS, CD 45 immunoreactivity was visualised with DAB/Ni/Co/H2O2 (for details see step 8 lectin histochemistry). After rinsing with PBS, sections were covered with a coverslip. Staining was immediately documented on a Zeiss Axiolab microscope equipped with a digital camera (Axio Cam MRc; Zeiss) using Zeiss MR grab software version 1.0. Next, the coverslip was removed and sections were incubated for 1 h with blocking buffer, followed by overnight incubation with biotinylated WFA (0.5 μg/ml in blocking buffer) at 4°C. After rinsing with PBS, sections were incubated with alkaline phosphatase-coupled ABC kit (ABC-AP; Vector) for 1 h. Lectin binding was visualised using a Vector Red alkaline phosphatase substrate kit I (Vector) as chromogen supplemented with levamisole for quenching endogenous alkaline phosphatase. Finally, sections were rinsed in A. dest. and mounted with Aquatex (Merck, Darmstadt, Germany). Staining was digitally documented. In control sections, both anti-CD 45 IgG and biotinylated WFA were omitted. These sections remained unstained.
Results
Identification of haematopoietic stem cells
C-kit immunoreactivity
From the one polyclonal (sc-168) and six monoclonal (bk1–6) antibodies directed against c-kit which were applied immunohistochemically only one, namely bk-1, produced a reliable staining of distinct cell types in a variety of bovine embryonic tissues. In the gonadal fold and early gonad, large round cells which often displayed characteristic finger-like processes were moderately to intensely stained with bk-1 (Fig. 1a, b). Similar cells in consecutive serial sections were positive with the lectins STA and WGA (Fig. 1c, d), thus representing PGCs. Blastema cells in the centre of the gonad showed weak to moderate immunostaining (Fig. 1a, b). However, bk-1 failed to demonstrate extragonadal PGCs in the paraaortic mesenchyme and especially in the coelomic bay, where numerous putative PGCs are expected to be present close to the gonadal fold.
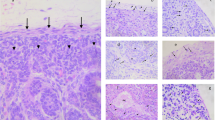
Female embryo, 45 days post-insemination (dpi). a, b C-kit. c STA. d WGA. Low power photomicrograph (a) and detail (b) of c-kit-labelled gonad. Putative primordial germ cells (PGCs) display finger-like processes and are strongly positive. The same cells are also stained with the lectins STA (c) and WGA (d). Furthermore, somatic cells in the interior of the gonad show a moderate c-kit immunoreactivity. a ×360; b–d ×580
Presumptive haematopoietic stem cells clustered together and attached to the wall of the dorsal aorta were intensely labelled with bk-1 (Fig. 2a, b). In the embryonic liver, bk-1-positive cells of various sizes occurred in the parenchymal strands (Fig. 3), most probably representing here also haematopoietic stem and progenitor cells.
Embryo 45 dpi, c-kit. C-kit-labelled cells are located in the parenchymal strands of the liver and most probably represent haematopoietic stem and progenitor cells. ×360
Embryo 45 dpi, c-kit. The podocytes (arrows) in the mesonephros are intensely stained. ×460
Embryo 34 dpi, c-kit. The epithelia of the oesophagus (E) and of the developing lung buds (L) are distinctly labelled. Capillary endothelia (arrow) and presumptive vasculogenetic cells (arrowhead) in the periphery of the lung buds display a moderate c-kit immunoreactivity. a ×145, b ×360
Moderate to intense immunostaining with bk-1 occurred in the nervous system, including the neural tube, the developing spinal ganglia and the peripheral spinal nerve (Fig. 2a). Podocytes of the bovine mesonephros displayed bk-1 immunoreactivity from 45 dpi onwards (Fig. 4), but were completely unstained in the developing metanephros. The endodermic epithelium of the gut, the developing lung buds (Fig. 5a, b) and the gall bladder also displayed intense bk-1 immunostaining. A network of capillaries with moderately to intensely stained endothelial cells as well as single cells with a stellate appearance due to multiple processes encircled the cranial embryonic gut, especially the lung buds (Fig. 5a, b).
The mesenchyme underlying the future cutis and subcutis, where melanoblasts are expected to be present, was completely devoid of bk-1-positive cells.
CD 34 immunoreactivity
The polyclonal antibody directed against CD 34 is known to display a broad species reactivity (mouse, rat, human; manufacturer’s specification). In our material, it labelled almost all intravasal blood cells. Therefore, it is not suited as a marker for bovine haematopoietic stem cells or blood cell precursors.
Identification of CD 45-positive cells
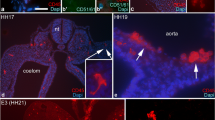
Antibodies directed against CD 45 are commonly used as pan-leucocyte markers (Figs. 6, 7, 8, 9, 10, 11). In the bovine embryo, CD 45-positive cells were frequently observed in peripheral blood (Fig. 6) and liver (Fig. 7), the latter being the principal site of haematopoiesis during the first gestational trimester. Labelled cells displayed various sizes and shapes, thus indicating different types of blood cells such as haematopoietic stem cells, megakaryocytes, macrophages and granulocytes. Surprisingly, anti-CD 45 also labelled non-blood cells in bovine embryos. In the gonadal anlage, CD 45-positive cells were identified as putative PGCs by their large round somata and typical finger-like processes (Fig. 9a). In consecutive sections, the same cell type was labelled with the alkaline phosphatase reaction product (Fig. 9b) and the lectin STA (Fig. 9c), respectively. Double-labelling of selected sections first with anti-CD 45 (Fig. 10a, d) and second with the lectin WFA (Fig. 10b, e, f) revealed that a subpopulation of intragonadal CD 45-positive cells was also stained with WFA (Fig. 10d, e), thus providing direct evidence for putative PGCs expressing CD 45. Besides CD 45+/WFA+ cells, also CD 45+/WFA− (Fig. 10d, e) putative PGCs, which displayed the same morphology as double-labelled cells and CD 45−/WFA+ (Fig. 10f) cells were present.
Embryo 32 dpi, CD 45 immunoreactivity (IR). Intravasally located CD 45-immunoreactive leucocytes (arrow) in the dorsal mesentery. The other blood cells in the vessel are negative. ×330
Embryo 34 dpi, CD 45-IR. In the liver, numerous CD 45-labelled cells are present. These cells are located predominantly in the parenchymal strands where they display different sizes and shapes. The larger and more irregular cells (arrows) most probably represent cells of the megakaryocytic lineage, whereas medium-sized and more ovoid-shaped cells (arrowheads) may belong to the lineage of granulopoietic cells. ×425
Embryo 34 dpi, CD 45-IR. The epithelium of the lung buds (L) is distinctly CD 45 positive. Smaller CD 45-immunoreactive cells are scattered in the surrounding mesenchyme and may include vasculogenetic cells. ×340
Embryo 34 dpi. a CD 45-IR. b Double-labelling with anti-CD 45 and WFA (same section as a). c Control processed according to the double-labelling protocol with omission of primary antibody and biotinylated lectin. d CD 45-IR (high power micrograph of a). e, f Double-labelling with anti-CD 45 and WFA (high power micrograph of b). Labelling of the same section with first anti-CD 45 (a, d) and then additionally with WFA (b, e, f) reveals three differently labelled types of putative PGCs: CD 45+/WFA− cells, CD 45+/WFA+ cells and CD 45−/WFA+ cells. Arrows in a, b point to a group of three CD 45+/WFA− and two CD 45+/WFA+ cells (enlarged in d, e). Arrowheads in a, b point to two CD 45−/WFA+ cells enlarged in f. a–c ×240; d–f ×1,100
Putative PGC-like cells outside the gonads, especially in the coelomic bay and the paraaortic mesenchyme, were also CD 45-immunoreactive (Fig. 11a). These extragonadal CD 45-positive cells often also displayed the typical finger-like processes of PGCs. Cells of similar phenotype and position were labelled in consecutive sections with the lectin STA (Fig. 11b), corroborating their identification as putative PGCs. However, PGC-like cells represent only a subpopulation of CD 45-positive cells within the mesenchyme, mesenteria and mediastinum. Here, a population of smaller and irregularly shaped cells (for example, the CD 45-positive cells encircling the lung buds; see Fig. 8) may also comprise additional cell types including precursors of megakaryocytes, macrophages and presumptive vasculogenetic cells.
Identification of megakaryocytes and platelets
Anti-human CD 61 (clone Y2/51) is an antibody with established crossreactivity to bovine megakaryocytes in adult tissues. In our embryonic material, it also labelled very large and irregularly shaped megakaryocytes as well as platelets and platelet aggregates. Labelled megakaryocytes were most common in the trabeculae of the liver (Fig. 12a), but also frequently occurred elsewhere, mainly in an intravasal location (Fig. 12b).
Embryo 32 dpi, CD 61-IR. a Large and irregularly shaped megakaryopoietic cells are observed in liver strands or close to the margins of liver sinusoids. b Intravasal megakaryocytes (arrows) are located within the perineural mesenchyme. Nt Neural tube. ×360
Embryo 34 dpi, BM-2-IR. BM-2-labelled granulopoietic progenitor cells are present in the parenchymal strands of the liver and arranged in small groups. Most BM-2-positive cells are of ovoid shape and medium size. Sometimes, a paler U-shaped nucleus (arrow), similar to that of myeloid cells, can be recognised at higher magnification (b). a ×360; b ×580
Platelets and small platelet aggregates were most abundant in blood vessels, and larger platelet aggregations occurred in the coelomic cavity (not shown), possibly due to injuries during opening of the abdomen prior to fixation.
Myeloid cells/granulocytes
Human-specific antibodies to CD 11b, BM-1 and myeloperoxidase showed no crossreactivity with bovine embryonic tissue. However, BM-2, an antibody directed against human granulocytes, labelled a distinct cell type in bovine liver strands (Fig. 13a). These BM-2-positive cells showed a round to oval soma of medium size. Labelling with BM-2 usually obscured the nucleus of the cell, but sometimes a slightly paler U-shaped nucleus was identified (Fig. 13b). Two to five BM-2-positive cells were often loosely grouped together, most likely indicating areas of granulopoiesis.
B-lymphopoietic cells
Labelling with B-lymphoid-specific antibodies directed against bovine IgM and bovine WC-4 occurred in the liver of embryos older than 57 dpi. Anti-WC-4 demonstrated a few medium-sized or larger, irregularly shaped cells restricted to the liver strands, which, however, resembled more a subpopulation of megakaryocytes than B-cell progenitors (Fig. 14a). Cells labelled with anti-IgM outnumbered the WC-4-positive cells. The former were medium-sized and displayed a slightly irregular, ovoid shape (Fig. 14b).
Embryo 60 dpi. a WC-4-IR. b IgM-IR. a Two populations of WC-4-IR cells can be identified in the embryonic liver: one fraction is large with irregular outer contours (arrows), the other smaller and of ovoid shape (arrowhead). b Cells of the smaller, ovoid fraction are also positive for IgM and most likely represent pre-B cells. ×580
Embryo 34 dpi, CD 68-IR. a Primitive macrophages are found in intravasal (arrows) and extravasal (arrowhead) localisation in the perineural mesenchyme. b In the liver, macrophages are numerous in the parenchymal strands, but are also located within the sinusoids. ×360
Identification of macrophages
CD 14-positive cells
Bovine-specific CD 14 (VPM 65) antibody labelled the macrophages in control sections of adult bovine tissue (lymph nodes), but failed to stain cells in embryonic tissue.
CD 68-positive cells
Human-specific CD 68 (EBM-11) antiserum is known for its crossreactivity to adult bovine tissue. This antibody also produced a specific staining of macrophages in our embryonic specimens. CD 68-labelled cells were irregularly shaped and often displayed processes and protrusions. Such cells occurred in an intravasal or extravasal localisation throughout the mesenchyme (Fig. 15a), but were more numerous in liver strands (Fig. 15b).
Discussion
The present study reports for the first time the expression pattern of surface molecules of bovine embryonic haematopoietic cells and PGCs, thus establishing the methodological basis for further investigations of normal development and pathological conditions in these two important cell lines.
C-kit
It is well established that haematopoiesis in the yolk sac and fetal liver starts from cells that display a c-kit+/lin− phenotype and give rise to c-kit−/lin+ mature cells (Ogawa et al. 1993). C-kit has been demonstrated in the haematopoietic tissues of the mouse (Orr-Urtreger et al. 1990; Motro et al. 1991; Ikuta and Weissman 1992; Palacios and Nishikawa 1992; Marcos et al. 1997) and human embryo (Teyssier-le Discorde et al. 1999). It has been shown that c-kit is essential for the proliferation of erythroid and myeloid progenitors in fetal mouse liver from day 12.5 of gestation on and for the generation of the stem cell pool that initiates bone marrow haematopoiesis (Ogawa et al. 1993). Therefore, c-kit is a useful marker for identifying haematopoietic progenitors in early embryonic and fetal organs. The bovine liver is the main site of fetal haematopoiesis and accordingly, intensely c-kit-labelled cells could be demonstrated in liver parenchymal strands. In this species as well, they most likely represent haematopoietic stem cells and progenitor cells. The latter are known to display c-kit immunoreactivity and additionally the characteristic lineage markers (Delassus et al. 1999).
In the bovine embryo, intensely c-kit-immunoreactive cells also occur in large clusters attached to the wall of the dorsal aorta. C-kit immunoreactivity has also been reported for aortic clusters of the mouse (Yoshida et al. 1998; Yokomizo et al. 2001) and c-kit mRNA has been detected in CD 34+ aortic clusters of human embryos (Labastie et al. 1998). These intraaortic clusters of spherical cells which adhere to the aortic endothelium are characterised as embryonic haematopoietic stem cells by their expression of typical surface antigens, transcription factors and oncogenes (for review see Péault et al. 2002). Their emergence at day 28 to day 38 in humans and at day 9.5 to day 11.5 in mice coincides with the appearance of functionally defined long-term culture-initiating cells (reviewed in Marshall and Thrasher 2001). Recently, the AGM region turned out to be a potent intraembryonic source of haematopoietic stem cells for fetal liver colonisation. However, not all the c-kit-positive cells in the AGM region are haematopoietic stem cells. Putative PGCs are also located in this region, especially in the gonadal folds. Cells with PGC-typical histochemical properties are observed in the periaortal mesenchyme and the coelomic bay and, to a lesser extent, in the dorsal mesentery (Wrobel and Süss 1998). While intragonadal bovine PGCs display a distinct c-kit immunoreactivity, the presumptive extragonadal PGCs are c-kit negative in this species. These findings contrast with those in the mouse, where c-kit mRNA has been detected in migrating PGCs of mice ranging from 10.5 to 12.5 dpi. Strong c-kit immunofluorescence has also been reported for migrating PGCs in the wall of the gut in 9.5 dpi mouse embryos (Bernex et al. 1996). The c-kit-negative putative extragonadal PGCs with unknown developmental potential in the AGM region of bovine embryos may probably never reach the gonads and undergo apoptosis due to the lack of trophic factors. C-kit is such a factor known to be important for survival and proliferation and perhaps also for the migration of PGCs (Pesce et al. 1993; Donovan 1994). As shown for the mouse, extragonadal PGCs located in the distal dorsal mesentery fail to reach the gonadal ridge (Molyneaux et al. 2001).
Paralleling findings in the mouse, c-kit immunoreactivity was additionally located in various bovine embryonic tissues, for example, the epithelia of gut, lung buds and gall bladder, and podocytes of the mesonephros, but the functional significance of c-kit in these sites has yet to be elucidated. The distinctly labelled capillary network encircling the developing lung buds is another interesting c-kit localisation because these vessels develop in mice by true vasculogenesis (Schachtner et al. 2000), i.e. via de novo differentiation of endothelial cells in the mesoderm. However, whether the c-kit-positive peribronchial capillaries in the bovine embryo also develop from individual angioblasts cannot be concluded from our material. This could be determined by careful examination of a close and uninterrupted series of bovine embryos at stages between 28 dpi (where no capillary network or individual angioblasts are present around the sprouting tracheal bud; unpublished observations) and 32 dpi (where the peribronchial capillary network is already established). The developing dorsal aortae in 22 dpi bovine embryos represent another site of vasculogenesis and are also c-kit positive (unpublished observation), pointing to c-kit as a general vasculogenetic marker. On the other hand, in the bovine embryo the c-kit antibody failed to label cells of the melanocytic lineage and interstitial cells of Cajal, both characterised by their distinct c-kit expression in the mouse (Manova and Bachvarova 1991; Opdecamp et al. 1997; Young 1999).
CD 45
CD 45 is commonly accepted as a pan-leucocytic marker, and antibodies directed against this surface molecule are widely used for discrimination of haematopoietic and non-haematopoietic cell lineages (see, for example., Labastie et al. 1998; Jaffredo et al. 2000). As expected, also in the bovine embryo, CD 45 labels not only leucocytes of different sizes and shapes in intravasal localisations and in the developing liver, but also megakaryocytes, haematopoietic stem and progenitor cells. Rather surprisingly, cells of the non-haematopoietic lineage were also labelled, with the putative PGCs being the most conspicuous. Recently, it has been shown by Crosby et al. (2002) that progenitor cells of the biliary epithelial cell lineage in adult mice share the same markers (CD 34, c-kit and CD 45) as haematopoietic precursors from the bone marrow and embryonic liver, indicating a more widespread distribution and a lower tissue specificity of CD 45 than previously thought. Identification of intragonadal CD 45+/WFA− cells as well as CD 45−/WFA+ and CD 45+/WFA+ cells may reflect a stepwise differentiation of putative PGCs. The presence of the CD 45+/WFA+ subpopulation of PGCs in the embryonic bovine gonad corresponds well with the results of studies demonstrating the transdifferentiation capacity of alkaline phosphatase/SSEA-1-positive PGCs in different murine culture systems into erythroid, myelomonocytic, multipotential and primitive haematopoietic stem cells (Rich 1995; Ohtaka et al. 1999). Additionally, PGCs and intraaortic clusters of haematopoietic stem cells can both be labelled with the lectin STA (unpublished observation). Thus far there is no direct evidence of PGCs differentiating into haematopoietic cells in vivo. However, indirect evidence has accumulated over several decades (for a review see Woodruff et al. 1995; Motzer et al. 1998; Saito et al. 1998). Numerous case reports dealing with the clonal analysis of extragonadal germ cell malignancies in the mediastinum or in the nervous system associated with haematological malignancies indicate that both types of malignant cell populations arose from a common precursor. Therefore, it has been concluded that extragonadal germ cell tumours represent aberrant germ cell migration to an “inappropriate” environment, which might result in a malignant transformation (see, for example, Chaganti et al. 1989; Heimdal et al. 1991; Scaravaglio et al. 1996). Additionally, under certain culture conditions, PGCs give rise to a population of cells which resemble pluripotent embryonic stem cells and are able to produce a wide variety of mature differentiated cell types including haematopoietic cells when injected into nude mice (Matsui et al. 1992). The apparent similarities between PGCs and haematopoietic stem cells have also led to the question of whether a common precursor may exist for these two cell types in normal development (De Felici and Pesce 1994). Arguments for such a common origin are derived from a clonal analysis of the extraembryonic mesoderm in the mouse embryo. While that part of the presumptive extraembryonic mesoderm which gives rise to blood islands is generally located more distally than the PGC precursors, there exists a small region of overlap in the extreme proximal part of the epiblast which gives rise to clones containing both types of cells (Lawson and Hage 1994). Nevertheless, direct evidence for a well-defined common haematopoietic stem cell/PGC precursor is still lacking. The markers shared by PGCs and putative haematopoietic stem cells could simply reflect the late final determination of the germ cell line in mammalian embryos, the so-called PGCs being a rather undifferentiated cell population with the capacity to still develop into tissues of all three germ layers (Durcova-Hills et al. 2001). Another possibility is that structural and functional similarities between the haematopoietic and PGC systems could have evolved in parallel because both cell types are subjected to similar problems, for example, homing, apoptosis, cluster formation during proliferation, etc.
Lineage markers
In this study, embryonic megakaryocytes and platelets have been successfully demonstrated with anti-CD 61 (clone Y2/51) which is an antibody with known species crossreactivity to adult bovine tissue (Brodersen et al. 1998). In the bovine embryo, megakaryocytes are most common in parenchymal liver strands, the main site of haematopoiesis during this gestational period. Surprisingly, developing megakaryocytes are also found in tissues which are generally not considered to be haematopoietic sites, for example, in blood vessels of the body wall and in the mesonephros. The CD 61 antibody used in this study can be recommended as a valuable and reliable tool for the in vivo study of embryonic, fetal and adult megakaryocytopoiesis.
The only antibody for myelopoietic/granulopoietic cells which yielded convincing results in this study was BM-2, a monoclonal antibody primarily developed against human granulocytes. BM-2 also reacts with early precursor forms of human myeloid cells and is recommended for the detection of myeloid leukaemias and granulocytic sarcomas (manufacturer’s information). In the bovine embryo, staining with BM-2 occurred solely in parenchymal liver strands and seemed to be restricted to a single population of medium-sized, round to oval cells. Due to the U-shaped nucleus of this cell type and the arrangement of the cells in small groups, the cells characterised by BM-2 may constitute granulopoietic nests. Occurrence of BM-2-positive cells in the liver coincides with the first appearance of myelocytes in bovine peripheral blood and liver (Winquist 1954). Since we were not able to detect BM-2-labelled myelocytes in peripheral blood and because of the uniform shape and size of BM-2-labelled cells in the liver, it can be concluded that BM-2 is expressed only in certain developmental stages of granulocytic precursors in the bovine embryo and not in mature granulocytes. Therefore, we consider BM-2 to be a useful marker for the detection and quantification of granulopoietic development.
B-lymphopoiesis in the bovine liver was detected with antibodies directed against WC-4 and IgM, but not until 58 dpi. Accordingly, Winquist (1954) described the first appearance of lymphocytes in peripheral blood in bovine embryos of 70 mm, corresponding to an age of approximately 60 dpi. WC-4 and IgM-labelled cells were scarce and displayed a large and irregularly shaped cell body. The same morphology has also been described in early mouse lymphopoiesis (Timens and Kamps 1997). Therefore, we consider WC-1 and especially IgM immunoreactivity as specific B-cell markers suited for the investigation of fetal B-lymphopoiesis.
The correct identification of different types of macrophages is of great importance in both veterinary diagnostics and experimental pathology. Therefore, markers specific for the monocytic lineage have been developed and well characterised for the bovine. Antibodies exist which direct against bovine CD 14 (Berthon and Hopkins 1996; Gupta et al. 1996; Sopp et al. 1996) and CD 68 (Ackermann et al. 1994; Zeng et al. 1996; Gutierrez et al. 1999). Using the antibody directed against CD 14, we were unable to detect macrophages in embryonic tissues, although these cells were easily recognised with anti-CD 14 in lymph nodes of adult bovines. Using anti-CD 68, numerous macrophages could be detected in the embryonic liver. The liver macrophages were identified as two populations, one being located in liver parenchymal strands where they may act as nursing cells in haematopoietic foci, and the other in the sinusoids, here most probably representing primitive Kupffer cells. CD 68 is also suited for the identification of an intravasal macrophage population and extravasal tissue macrophages in the bovine embryo, corroborating findings in adult cattle (Bertram 1986; Ackermann et al. 1994). Thus, CD 68 constitutes a reliable marker for bovine macrophages from the embryonic period through to the adult state. It may be used in the study of embryonic/fetal haematopoiesis for the detection of haematopoietic foci, and in developmental studies of different tissue macrophage populations, for example, Kupffer cells in the liver, or, at later developmental stages, alveolar macrophages in the lung.
References
Ackermann MR, DeBey BM, Stabel TJ, Gold JH, Register KB, Meehan JT (1994) Distribution of anti-CD68 (EBM11) immunoreactivity in formalin-fixed, paraffin-embedded bovine tissues. Vet Pathol 31:340–348
Bernex F, De Sepulveda P, Kress Ch, Elbaz C, Delois C, Panthier JJ (1996) Spatial and temporal patterns of c-kit-expressing cells in WlacZ/+ and WlacZ/WlacZ mouse embryos. Development 122:3023–3033
Berthon P, Hopkins J (1996) Ruminant cluster CD 14. Vet Immunol Immunopathol 52:245–248
Bertram TA (1986) Intravascular macrophages in lungs of pigs infected with Haemophilus pleuropneumoniae. Vet Pathol 23:681–691
Brodersen R, Bijlsma F, Gori K, Jensen KT, Chen W, Dominguez J, Haverson K, Moore PF, Saalmüller A, Sachs D, Slierendrecht WJ, Stokes C, Vainio O, Zuckermann F, Aasted B (1998) Analysis of the immunological cross reactivities of 213 well-characterized monoclonal antibodies with specificities against various leucocyte surface antigens of human and 11 animal species. Vet Immunol Immunopathol 64:1–13
Broudy VC (1997) Stem cell factor and hematopoiesis. Blood 90:1345–1364
Chaganti RSK, Ladanyi M, Samaniego F, Offit K, Reuter V, Jhanwar SC, Bosl GJ (1989) Leukemic differentiation of a mediastinal germ cell tumor. Genes Chromosomes Cancer 1:83–87
Crosby HA, Nijjar S, de Ville de Goyet J, Kelly DA, Strain AJ (2002) Progenitor cells of the biliary epithelial cell lineage. Cell Dev Biol 13:397–403
De Felici M, Pesce M (1994) Interactions between migratory primordial germ cells and cellular substrates in the mouse. In: Marsh J, Goode J (eds) Germline development (Ciba Foundation Symposium 182). Wiley, Chichester, pp 140–153
Delassus S, Titley I, Enver T (1999) Functional and molecular analysis of hematopoietic progenitors derived from the aorta-gonad-mesonephros region of the mouse embryo. Blood 94:1495–1503
Donovan PJ (1994) Growth factor regulation of mouse primordial germ cell development. Curr Top Dev Biol 29:189–225
Durcova-Hills G, Ainscough JFX, McLaren A (2001) Pluripotential stem cells derived from migrating primordial germ cells. Differentiation 68:220–226
Faust N, Huber MC, Sippel AE, Bonifer C (1997) Different macrophage populations develop from embryonic/fetal and adult hematopoietic tissues. Exp Hematol 25:432–444
Geissler EN, Ryan MA, Housman DE (1988) The dominant-white spotting (W) locus of the mouse encodes the c-kit proto-oncogene. Cell 55:185–192
Gomori G (1952) Microscopic histochemistry, principles and practice. University of Chicago Press, Chicago
Gupta VK, McConell I, Dalziel RG, Hopkins J (1996) Identification of the sheep homologue of the monocytic cell surface molecule, CD14. Vet Immunol Immunopathol 51:89–99
Gutierrez M, Forster FI, McConell SA, Cassidy JP, Pollock JM, Bryson DG (1999) The detection of CD2+, CD4+, CD8+, and WC1+ T lymphocytes, B cells and macrophages in fixed and paraffin embedded bovine tissue using a range of antigen recovery and signal amplification techniques. Vet Immunol Immunopathol 71:321–334
Heimdal K, Evensen SA, Stein AE, Fosså SD, Hirscberg H, Langholm R, Brøgger A, Møller P (1991) Karyotyping of a hematologic neoplasia developing shortly after treatment for cerebral extragonadal germ cell tumor. Cancer Genet Cytogenet 57:41–46
Hikono H, Ohta M, Kubota T, Zhou J-H, Inumaru S, Sakurai M (1999) Production and characterization of monoclonal antibodies that recognize bovine kit receptor. Vet Immunol Immunopath 68:101–112
Hikono H, Ohta M, Zhou J-H, Sakurai M (2001a) Expression and distribution of the kit receptor in bovine bone marrow cells. Am J Vet Res 62:974–977
Hikono H, Ohta M, Sakurai M, Momotani E (2001b) Expression of kit, the receptor for stem cell factor, in bovine peripheral blood. J Vet Med Sci 63:321–324
Horie K, Takakura K, Taii S, Narimoto K, Noda Y, Nishikawa S, Nakayama H, Fujita J, Mori T (1991) The expression of c-kit protein during oogenesis and early embryonic development. Biol Reprod 45:547–552
Horie K, Fujita J, Takakura K, Kanzaki H, Kaneko Y, Iwai M, Nakayama H, Mori T (1992) Expression of c-kit protein during placental development. Biol Reprod 47:614–620
Hsu SM, Raine L, Fanger H (1981) Use of avidin-biotin peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem 29:577–580
Ikuta K, Weissman IL (1992) Evidence that hematopoietic stem cells express mouse c-kit but do not depend on steel factor for their generation. Proc Natl Acad Sci U S A 89:1502–1506
Jaffredo T, Gautier R, Brajeul V, Dieterlen-Lièvre F (2000) Tracing the progeny of the aortic hemangioblast in the avian embryo. Dev Biol 224:204–214
Jones M, Cordell JL, Beyers AD, Tse AGD, Mason DY (1993) Detection of T and B ells in many animal species using cross-reactive anti-peptide antibodies. J Immunol 150:5429–5435
Keshet E, Lyman SD, Williams DE, Anderson DM, Jenkins NA, Copeland NG, Parada LF (1991) Embryonic RNA expression patterns of the c-kit receptor and cognate ligand suggest multiple functional roles in mouse development. EMBO J 10:2425–2435
Kubota T, Hikono H, Sasaki E, Sakurai M (1994) Sequence of a bovine c-kit proto-oncogene cDNA. Gene 141:305–306
Kujat R, Rose C, Wrobel KH (1993) The innervation of the bovine ductus deferens: comparison of a modified acetylcholinesterase-reaction with immunoreactivities of cholinacetyl-transferase and panneuronal markers. Histochemistry 99:231–239
Labastie MC, Cortés F, Roméo PH, Dulac C, Péault B (1998) Molecular identity of hematopoietic precursor cells emerging in the human embryo. Blood 92:3624–3635
Lawson KA, Hage WJ (1994) Clonal analysis of the origin of primordial germ cells in the mouse. In: Marsh J, Goode J (eds) Germline development (Ciba Foundation Symposium 182). Wiley, Chichester, pp 68–91
Lyman SD, Jacobsen SEW (1998) C-kit ligand and Flt3 ligand: stem/progenitor cell factors with overlapping yet distinct activities. Blood 91:1101–1134
Maeda H, Yamagata A, Nishikawa S, Yoshinga K, Kobayashi S, Nishi K, Nishikawa SI (1992) Requirement of c-kit for development of intestinal pacemaker system. Development 116:369–375
Manova K, Bachvarova RF (1991) Expression of c-kit encoded at the W locus of mice in developing embryonic germ cells and presumptive melanoblasts. Dev Biol 146:312–324
Marcos MAR, Morales-Alcelay S, Godin IE, Dieterlen-Lièvre F, Copin SG, Gaspar M-L (1997) Antigenic phenotype and gene expression pattern of lymphohemopoietic progenitors during early mouse ontogeny. J Immunol 158:2627–2637
Marshall C, Thrasher AJ (2001) The embryonic origins of human hematopoiesis. Br J Hematol 112:838–850
Matsui Y, Zsebo K, Hogan BLM (1992) Derivation of pluripotential embryonic stem cells from murine primordial germ cells in culture. Cell 70:841–847
McGadey J (1970) A tetrazolium method for non-specific alkaline phosphatase. Histochemie 23:180–184
Medvinsky A, Dzierzak E (1996) Definitive hematopoiesis is autonomously initiated by the AGM region. Cell 86:897–906
Molyneaux KA, Stallock J, Schaible K, Wylie C (2001) Time-lapse analysis of living mouse germ cell migration. Development 240:488–498
Motro B, Van der Kooy D, Rossant J, Teith A, Bernstein A (1991) Contiguous pattern of c-kit and Steel expression: analysis of mutation at the W and SI loci. Development 113:1207–1221
Motzer RJ, Amsterdam A, Prieto V, Sheinfeld J, Murty VVVS, Mazumdar M, Bosl GJ, Chaganti RSK, Reuter VE (1998) Teratoma with malignant transformation: diverse malignant histologies arising in men with germ cell tumors. J Urol 159:133–138
Murakami K, Inumaru S, Yokoyama T, Okada K, Sentsui H (1999) Expression of granulocyte-macrophage colony-stimulating factor (GM-CSF) receptor on B-1a cell from persistent lymphocytosis (PL) cows and lymphoma cell induced by bovine leukaemia virus. Vet Immunol Immunopathol 68:49–59
Naessens J, Howard CJ (1993) Second workshop on ruminant leucocyte antigens. Vet Immunol Immunopathol 39:1–10
Naito M, Hasegawa G, Takahashi K (1997) Development, differentiation, and maturation of Kupffer cells. Microsc Res Tech 39:350–364
Nocka K, Majumder S, Chabot B, Ray P, Cervone M, Bernstein A, Besmer P (1989) Expression of c-kit gene products in known cellular targets of W mutations in normal and W mutant mice: evidence for an impaired c-kit kinase in mutant mice. Genes Dev 3:816–826
Ogawa M, Nishikawa S, Yoshinaga K, Hayashi SI, Kunisada T, Nakao J, Kina T, Sudo T, Kodama H, Nishikawa SI (1993) Expression and function of c-kit in fetal hemopoietic progenitor cells: transition from the early c-kit-independent to the late c-kit-dependent wave of hemopoiesis in the murine embryo. Development 117:1089–1098
Ohtaka T, Matsui Y, Obinata M (1999) Hematopoietic development of primordial germ cell-derived mouse embryonic germ cells in culture. Biochem Biophys Res Commun 260:475–482
Opdecamp K, Nakayama A, Nguyen M-T, Hodgkinson CA, Pavan WJ, Arnheiter H (1997) Melanocyte development in vivo and in neural crest cell cultures: crucial dependence on the Mitf basic-helix-loop-helix-zipper transcription factor. Development 24:2377–2386
Orr-Urtreger A, Avivi A, Zimmer Y, Givol D, Yarden Y, Lonai P (1990) Developmental expression of c-kit, a protooncogene encoded by the W locus. Development 109:911–923
Palacios R, Nishikawa SI (1992) Developmentally regulated cell surface expression and function of the c-kit receptor during lymphocyte ontogeny in the embryo and adult mice. Development 115:1133–1147
Péault B, Oberlin E, Tavian M (2002) Emergence of hematopoietic stem cells in the human embryo. C R Biologies 325:1021–1026
Pesce M, Farrace MG, Piacentini M, Dolci S, De Felici M (1993) Stem cell factor and leukemia inhibitory factor promote primordial germ cell survival by suppressing programmed cell death (apoptosis). Development 118:1089–1094
Qiu F, Ray P, Brown K, Braker PE, Jhanwar S, Ruddle RH, Besmer P (1988) Primary structure of c-kit, a protooncogene encoded by the W locus. Development 109:911–923
Rathkolb B, Pohlenz JF, Wohlsein P (1997) Identification of leucocyte surface antigens in paraffin-embedded bovine tissues using modified formalin dichromate fixation. Histochem J 29:487–493
Rebelatto MC, Mead C, HogenEsch H (2000) Lymphocyte populations and adhesion molecule expression in bovine tonsils. Vet Immunol Immunopathol 73:15–29
Rich IN (1995) Primordial germ cells are capable of producing cells of the hematopoietic system in vitro. Blood 86:463–472
Saito A, Watanabe K, Kusakabe T, Abe M, Suzuki T (1998) Mediastinal mature teratoma with coexistence of angiosarcoma, granulocytic sarcoma and a hematopoietic region in the tumor: a rare case of association between haematological malignancy and mediastinal germ cell tumor. Pathol Int 48:749–753
Sanchez M-J, Holmes A, Miles C, Dzierzak E (1996) Characterization of the first definitive hematopoietic stem cells in the AGM and liver of the mouse embryo. Immunity 5:513–525
Scaravaglio P, Facta P, Ceresole B, Guglielminetti T, Berruti A, Pautasso M, Mazza U, Saglio G, Rege-Cambrin G (1996) Chromosome abnormalities in a patient with acute myeloid leukemia following mediastinal germ cell tumor. Cytogenetics 91:175
Schachtner SK, Wang Y, Baldwin HS (2000) Qualitative and quantitative analysis of embryonic pulmonary vessel formation. Am J Respir Cell Mol Biol 22:157–165
Sopp P, Kwong LS, Howard CJ (1996) Identification of bovine CD14. Vet Immunol Immunopathol 52:323–328
Tan JC, Nocka KH, Ray P, Traktman P, Besmer P (1990) The dominant W42 spotting phenotype results from a missense mutation in the c-kit receptor kinase. Science 247:209–212
Tavian M, Hallais M-F, Péault B (1999) Emergence of intraembryonic hematopoietic precursors in the pre-liver human embryo. Development 126:793–803
Teyssier-le Discorde M, Prost S, Nandrot E, Kirszenbaum M (1999) Spatial and temporal mapping of c-kit and its ligand, stem cell factor expression during human embryonic haemopoiesis. Br J Haematol 107:247–253
Timens W, Kamps W (1997) Hemopoiesis in human fetal and embryonic liver. Microsc Res Tech 39:387–397
Timens W, Kamps WA, Rozeboom-Uiterwijk T, Poppema S (1990) Haemopoiesis in human fetal and embryonic liver. Virchows Archiv A Pathol Anat 416:429–436
Tisdall DJ, Fidler AE, Smith P, Quirke LD, Stent VC, Heath DA, McNatty KP (1999) Stem cell factor and c-kit gene expression and protein localization in the sheep ovary during fetal development. J Reprod Fertil 116:277–291
Van Kampen C, Mallard BA, Wilkie BN (1999) Adhesion molecules and lymphocyte subsets in milk and blood of periparturient Holstein cows. Vet Immunol Immunopathol 69:23–32
Winquist G (1954) Morphology of the blood and the hemopoietic organs in cattle under normal and some experimental conditions. Acta Anat 22(suppl 21)
Woodruff K, Wang N, May W, Adrone E, Denny C, Feig SA (1995) The clonal nature of mediastinal germ cell tumors and acute myelogenous leukemia. Cancer Genet Cytogenet 79:25–31
Wrobel K-H, Süss F (1998) Identification and temporospatial distribution of bovine primordial germ cells prior to gonadal sexual differentiation. Anat Embryol 197:451–467
Yarden Y, Kuang WJ, Yang-Feng T, Coussens L, Munemitsu S, Dull TJ, Chen E, Schlessinger J, Francke U, Ullrich A (1987) Human proto-oncogene c-kit: a new cell surface receptor tyrosine kinase for an unidentified ligand. EMBO J 6:3341–3351
Yokomizo T, Ogawa M, Osato M, Kanno T, Yoshida H, Fujimoto T, Fraser S, Nishikawa S, Okada H, Satake M, Noda T, Nishikawa SI, Ito Y (2001) Requirement of Runx1/AML1/PEBP2αB for the generation of haematopoietic cells from endothelial cells. Genes to Cells 6:13–23
Yoshida H, Takakura N, Hirashima M, Kataoka H, Tsuchida K, Nishikawa S, Nishikawa SI (1998) Hematopoietic tissues, as a playground of receptor tyrosine kinases of the PDGF-receptor family. Dev Comp Immunol 22:321–332
Yoshinaga K, Nishikawa S, Ogawa M, Hayashi SI, Kunisada T, Fujimoto T, Nishikawa SI (1991) Role of c-kit in mouse spermatogenesis: identification of spermatogonia as a specific site of c-kit expression and function. Development 113:689–699
Young HM (1999) Embryological origin of interstitial cells of Cajal. Microsc Res Tech 47:303–308
Zeng L, Takeya M, Ling X, Nagasaki A, Takahashi K (1996) Interspecies reactivities of anti-human macrophage monoclonal antibodies to various animal species. J Histochem Cytochem 44:845–853
Acknowledgements
The authors greatly appreciate the excellent technical assistance of Mrs. I. Hees, Mrs. M. Schimmel, Mrs. U. Schmitt and Mrs. E. Stauber, and the skilful photographic work of Mrs. A. Zenker and Mr. T. Maurer. We thank Mrs. C. Ross-Cavanna for linguistic help. We are especially grateful to H. Hikono, National Institute of Animal Health, 3-1-1 Kannondai, Tsukuba, Ibaraki, 305, Japan, for his kind donation of anti-c-kit (bk1–6) antibodies. The monoclonal antibody M1/70.15.11.5.2 developed by T.A. Springer was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by the University of Iowa, Department of Biological Sciences, Iowa City, IA 52242, USA. This research was financially supported by DFG grant Wr 7/10-1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kritzenberger, M., Wrobel, KH. Histochemical in situ identification of bovine embryonic blood cells reveals differences to the adult haematopoietic system and suggests a close relationship between haematopoietic stem cells and primordial germ cells. Histochem Cell Biol 121, 273–289 (2004). https://doi.org/10.1007/s00418-004-0629-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-004-0629-5