Abstract
To assess spontaneous blinking and anomalous eyelid movements in patients with hemifacial spasm with an emphasis on interocular differences. Spontaneous eyelid movements were registered bilaterally for 3 min using a high-speed video camera in 28 patients with hemifacial spasm (HFS) who had not been treated with botulinum toxin injections for at least 5 months. The degree of blink conjugacy, maximum velocity, and amplitude of the closing phase of the blinks were determined for the affected and non-affected sides. Out of the 28 subjects, 23 (82%) presented with abnormal nonconjugate spasms that were similar to blinks, and in 17 (61%), high-frequency eyelid twitches were detected between blinks on the affected eye. The rate of nonconjugate blink-like spasms ranged from 0.3 to 24.7 movements/min. With regard for conjugate blinks, there was no significant interocular difference in amplitudes or eyelid closure velocities. The amplitude and velocity were significantly lower for nonconjugate movements than for spontaneous blinks. HFS is a unique condition in which complex patterns of eyelid movements, including both conjugate and nonconjugate movements, are present. Conjugate movements correspond to spontaneous blinking, and the same metrics were observed in affected and non-affected eyes. Nonconjugate movements correspond to anomalous nonconjugate blink-like spasms and high-frequency eyelid twitches in the affected eye, both of which were characterized by lower amplitudes and velocities than were observed in conjugate movements.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hemifacial spasm (HFS) is a disorder characterized by unilateral, involuntary, tonic, or clonic contractions of the mimetic muscles [1,2,3,4].
Spontaneous human blinks are unconscious and conjugate lid movements characterized by the fast and transient closure of both upper lids and occur in the absence of any evident stimulus [5,6,7]. Blinks result from reciprocal action between two antagonistic muscles acting in a conjugate fashion in both upper eyelids: the levator palpebrae muscle, an eyelid retractor, and the orbicularis oculi muscle, an eyelid protractor muscle. The first event that occurs during spontaneous blinking is the inhibition of the levator palpebrae muscle, which results in a down-phase; this is followed by the contraction of the orbicularis oculi muscle, which brings the lid down at high velocity. Levator palpebrae muscle contraction then results in an up-phase [7, 8]. Proper eyelid blinking is imperative for visual function, tear film distribution over the ocular surface and tear drainage [7, 9].
Although blinking metrics have been quantified in several conditions [6, 10,11,12,13], little is known about spontaneous blinking activity and the features of anomalous eyelid motion in HFS. Using a high-speed video camera, we analyzed eyelid movement features in both affected and non-affected eyes in patients with HFS.
Materials and methods
This research adhered to the tenets of the Declaration of Helsinki. The Federal University of S. Paulo Ethics Committee approved the study, and all subjects provided written informed consent.
Twenty-eight HFS patients (19 females) with moderate to severe HFS (very noticeable or incapacitating spasms) according to the Jankovic rating scale (JRS) [14] who had not been treated with botulinum toxin injections in the previous 5 months were recruited for this study. All patients underwent a complete ophthalmological evaluation. The mean age of the patients was 64.3 ± 10.6 years old (range: 42–84 years old).
The exclusion criteria included HFS patients who had undergone prior refractive, eyelid or intraocular surgeries or had any abnormal corneal, lid, or systemic conditions, including dry eye, lid malpositioning, and contact lens wear, which could affect eyelid movements. Patients who had been treated with botulinum toxin in the previous 5 months and those with essential blepharospam or other neurological disorders were excluded.
Eyelid movements were registered bilaterally in a standard manner using a high-speed video system for 3 min while the subjects watched a commercial movie. A small blue light-emitting diode (LED) measuring 2 × 1 × 1 mm with a brightness of 100 mcd was placed on the pretarsal region of both upper eyelids (Fig. 1). The LED was coupled to an electronic circuit that permitted its brightness to be adjusted. A commercially available high-speed camera (Point Grey Research® Inc., model FL3-U3-13S2C-CS) was aligned with the small LED in the patient’s primary position of gaze. Patients were seated at a slit lamp under uniform lighting conditions. All recordings took place in the same exam room to standardize environmental conditions. A camera was coupled to a laptop computer and used to register upper eyelid motion in both eyes based on red, green, and blue (RGB) color images (800 × 350 pixels, 150 dpi resolution) at 120 frames/s. This method was developed by one of the authors (D.M.G.) and was previously demonstrated to be equivalent to the magnetic search coil methodology used to assess spontaneous blinking [15].
Eyelid kinematics analysis
Custom software was developed in MATLAB R2015a (MathWorks, Natick, MA, USA) to analyze the data recorded in this study. An algorithm based on sudden variations of velocity of the eyelid movements (derivatives of the lid movement trace) automatically selected spontaneous blink movements. In addition, the data retrieved were manually examined to avoid any inconsistency in the temporal conjugacy of records. Blink conjugacy was defined as two blink movements occurring at the same time between the affected and non-affected side. The software registered the number of eyelid movements as well as the amplitude and peak velocity of the descending phase of these lid movements.
Statistical analyses were performed using JMP SAS 10.0 (SAS Institute, Inc., Cary, NC). Data are reported as the mean ± standard error (SE). A paired Student’s t test was applied to test the difference between affected and non-affected eyes regarding the number of movements, amplitude, and peak velocity. A p value of less than 0.05 was considered statistically significant.
Results
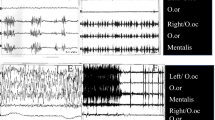
The number of eyelid movements was significantly higher on the affected side (affected: 1884 movements; non-affected: 1491 movements; p = 0.0006). An absence of abnormal movements was observed in five patients who displayed perfect conjugacy of lid movements between the eyes (Fig. 2a). Of the remaining patients (82%), all showed nonconjugate blink-like spasms (Fig. 2b), and in 17 (61%), high-frequency eyelid twitches were detected between blinks (Fig. 2c) on the affected side.
Conjugate and nonconjugate eyelid movements in patients with hemifacial spasm. a Normal conjugacy of spontaneous blinks between affected (red line) and non-affected (blue line) eyes. b Nonconjugate blink-like spasms in the affected eye. c Conjugate blinks admixed with high-frequency eyelid twitches on the affected side
The rate of nonconjugate blink-like spasms ranged from 0.3 to 24.7 movements/min (1 to 75 movements). The temporal rate of conjugacy between the affected and non-affected eyes was 80.2 ± 4.3% (range: 16.7–100%, Fig. 3).
Table 1 shows the eyelid movement metrics of both sides. In conjugate eyelid movements, which correspond to spontaneous blinking, there was no significant difference in amplitude (p = 0.37) or eyelid peak closure velocity (p = 0.08) between the affected and non-affected sides. However, our analysis of nonconjugate eyelid movements, which correspond to anomalous eyelid spasms and eyelid twitches occurring on the affected side, showed that velocity (p < 0.001) and amplitude (p < 0.001) were significantly lower than the values obtained for conjugate movements.
Figure 4 a shows the relationship between amplitudes (mm) obtained for the affected and non-affected eyes in a patient with severe HFS. Figure 4 b shows the peak eyelid closure velocities (mm/s) of eyelid movements in the affected and non-affected eyes of the same patient.
Discussion
Hemifacial spasm (HFS) is a unilateral condition in the vast majority of cases, and therefore, there is an affected side and a contralateral non-affected side (bilateral cases are exceedingly rare, and none of the patients included in this study presented bilateral HFS). In this condition, both conjugate and nonconjugate eyelid movements are present. Conjugate movements, which correspond to spontaneous blinking, have identical metrics between the affected and non-affected eye. In addition to the presence of spontaneous blinking, the eyelid motion traces obtained in the present study demonstrate that HFS also presents complex and heterogeneous patterns: the affected eyes displayed nonconjugate spasms characterized by reduced amplitudes and velocities, and 61% of the patients also presented high-frequency eyelid twitches (Fig. 2). No previous studies have assessed and compared this complex array of eyelid kinematics in both affected and non-affected sides in patients with this condition.
Few studies have assessed blink dynamics in movement disorders. In 1990, Manning et al. [16] evaluated upper lid movement in four HFS patients using the magnetic search coil method. They observed that blink closing velocity and amplitude were lower on the affected side than in normal subjects; however, the affected and non-affected sides of the patients were not compared. Their hypothesis was that the fast-twitch orbicularis oculi muscle fibers had potentially converted into slow-twitch fibers after years of tonic contractions, and this conversion could be associated with the reduced blink closing velocity observed on the affected side. Because approximately 10% of normal orbicularis oculi fibers are slow [17], an increase in their number could lead to a marked reduction in blink closing velocity. Another hypothesis they proposed was related to the possibility of subclinical facial palsy on the affected side in HFS because there is evidence that this condition arises from compression of the facial nerve root at the entry zone [16]. Nevertheless, HFS is a unique condition in which both conjugate and nonconjugate eyelid movements are present. In contrast to Manning et al., we studied both affected and non-affected eyelid movements, and this permitted us to analyze these two types of movements separately. We observed that during spontaneous blinking (conjugate movements), the closure of both upper eyelids occurred in a symmetric and coordinated fashion, with no significant interocular differences in velocity or amplitude, as has been observed in normal individuals [5]; this suggests that the interocular coordination of spontaneous blinking is maintained in HFS. Because there was no significant difference in velocity or amplitude between affected and non-affected eyelids during spontaneous blinking, it is unclear whether our results could be explained by the conversion of orbicularis oculi fiber type or the compression of the facial nerve root on the affected side. Our group recently demonstrated that orbicularis oculi samples obtained from HFS patients presented chronic alterations that led to muscle degeneration on the affected side and that morphological changes were found on even the non-affected side [18]. All these data suggest that HFS is a complex condition, and further research is needed to better understand this disorder.
Hasan et al. [19] investigated eyelid movements in five patients with BEB and observed that in these patients, eyelid spasms had smaller amplitudes and were slower than blinks. In some BEB patients, the orbicularis oculi spasms were accompanied by the persistence of levator palpebrae function in electromyographic studies [20]. This could partially explain the reduced velocity and amplitude observed during the spasms in that study, and a similar mechanism might explain the reduced amplitude and peak velocity in eyelid spasms observed on the affected side in HFS patients in the present study. As was previously observed in BEB [19], our findings show that HFS also does not disrupt the interocular coordination of blinking.
Bentivoglio et al. [21] evaluated the blink rate in patients with BEB at rest and during conversation, whereas Ferrazzano et al. [22] evaluated the effects of writing, reading, and speaking on orbicularis oculi muscle spasms and the blink rate in patients with blepharospasm. In contrast to our study, in both of those previous studies, the assessments were performed based on direct observation of videotapes: two observers reviewed the videotapes and measured the number of blinks. Considering that blinks have a wide range of amplitudes, reviewing videotapes is not a completely objective method since the observer must decide which upper lid movements should be considered a blink [7]. Because in our study, the assessment of blinking dynamics was performed using high-speed videography and small LED diodes that enabled the camera to more precisely register eyelid motion, we were able to evaluate additional parameters with a greater degree of accuracy for detecting movement. Furthermore, the use of this technology permitted us to observe underlying eyelid movement abnormalities that could go undetected by either the physician or patient.
New technologies have evolved our understanding of spontaneous blinking and permitted the study of the effects of several conditions on blink dynamics [10,11,12,13]. Several methodologies used to evaluate spontaneous blinking have been described; these include direct observation, magnetic-based devices, high-speed, and infrared videography in addition to pupillometry [15, 23,24,25,26,27,28,29,30]. For research purposes, magnetic-based devices are considered the gold standard for evaluating blink dynamics; however, the apparatus required to generate a magnetic field is expensive and not useful in a clinical setting. It has been suggested that high-speed videography combined with the use of small LEDs could provide equivalence at a lower cost and be more convenient for both participants and researchers to use [15]. Moreover, the coil marker has been reported to weigh from 20 to 160 mg and might therefore influence blink kinematics and consequently blink parameters [25], whereas LEDs are smaller than coils and do not cause any positional artifact. In the present study, we employed a methodology that was previously shown to be equivalent to a magnetic-based device [15].
Limitations of this study include the fact that some anomalous eyelid spasms may not have been detected during the recording period. Although we recorded eyelid movements for a longer period (3 min) than was used in several studies that assessed normal blinking, in HFS, spasms do not occur 100% of the time and may not have been registered in all patients. In the present study, video analysis showed that in five patients, no eyelid spasms occurred during the recording period. Consequently, these patients presented a 100% conjugacy rate, and this may have resulted in an overestimation of the average conjugacy rate. A longer recording period would probably permit higher eyelid spasm detection rates.
Conclusions
In conclusion, HFS is a unique condition in which complex patterns of eyelid movements, including both conjugate and nonconjugate movements, are present. Conjugate movements correspond to spontaneous blinking, in which there was no significant difference in blink metrics between affected and non-affected eyes, suggesting that the coordination of normal blinking between both eyelids was maintained in HFS patients. The affected eyes displayed nonconjugate spasms, and in most patients, high-frequency eyelid twitches were also observed; both of these abnormal movements were characterized by reduced amplitudes and velocities.
References
Wang A, Jankovic J (1998) Hemifacial spasm: clinical findings and treatment. Muscle Nerve 21:1740–1747
Chaudhry N, Srivastava A, Joshi L (2015) Hemifacial spasm: the past, present and future. J Neurol Sci 356(1–2):27–313
Abbruzzese G, Berardelli A, Defazio G (2011) Hemifacial spasm. Hyperkinetic Mov Disord 100:675–680
Ross AH, Elston JS, Marion MH, Malhotra R (2011) Review and update of involuntary facial movement disorders presenting in the ophthalmological setting. Surv Ophthalmol 56:54–67
Stava MW, Huffman MD, Baker RS, Epstein AD, Porter JD (1994) Conjugacy of spontaneous blinks in man: eyelid kinematics exhibit bilateral symmetry. Invest Ophthalmol Vis Sci 35:3966–3971
Huffman MD, Baker RS, Stava MW, Chuke JC, Rouholiman BR, Porter JD (1996) Kinematic analysis of eyelid movements in patients recovering from unilateral facial nerve palsy. Neurology 46:1079–1085
Cruz AA, Garcia DM, Pinto CT, Cechetti SP (2011) Spontaneous eyeblink activity. Ocul Surf 9(1):29–41
Evinger C, Ka M, Pa S (1991) Eyelid movements. Mechanisms and normal data. Invest Ophthalmol Vis Sci 32:387–400
Palakuru J, Wang J, Aquavella JV (2007) Effects of blinking on tear dynamics. Invest Ophthalmol Vis Sci 48:3032–3037
Garcia D, Barbosa JC, Pinto CT, Cruz AA (2013) Estimation of spontaneous blinking main sequence in normal subjects and patients with graves’ upper eyelid retraction. Invest Ophthalmol Vis Sci 54:1434–1442
Wambier SP, Garcia DM, Cruz AA, Messias A (2016) Spontaneous blinking kinetics on paralytic Lagophthalmos after lid load with gold weight or autogenous temporalis fascia sling. Curr Eye Res 41(4):433–440
Baccega A, Garcia DM, Cruz AAV (2017) Spontaneous blinking kinematics in patients who have undergone Autogeneous fascia Frontalis suspension. Curr Eye Res 42(9):1248–1253
Kimura N, Watanabe A, Suzuki K, Toyoda H, Hakamata N, Fukuoka H, Washimi Y, Arahata Y, Takeda A, Kondo M, Mizuno T, Kinoshita S (2017) Measurement of spontaneous blinks in patients with Parkinson's disease using a new high-speed blink analysis system. J Neurol Sci 380:200–204
Jankovic J, Kenney C, Grafe S, Goertelmeyer R, Comes G (2009) Relationship between various clinical outcome assessments in patients with blepharospasm. Mov Disord 24:407–413
Wambier S, Ribeiro SF, Garcia DM et al (2014) Two-dimensional video analysis of the upper eyelid motion during spontaneous blinking. Ophthal Plast Reconstr Surg 30:146–151
Manning KA, Evinger C, Sibony PA (1990) Eyelid movements before and after Botulinum therapy in patients with lid spasm. Ann Neurol 28:653–660
Porter JD, Burns LA, May PJ (1989) Morphological substrate for eyelid movements: innervation and structure of primate levator palpebrae superioris and orbicularis oculi muscles. J Comp Neurol 287:64–81
Osaki MH, Osaki TH, Osaki T, Gameiro GR, Belfort R Jr, Marie SKN (2019) Orbicularis oculi morphological alterations in affected and non-affected sides in hemifacial spasm. J Neuro-ophthalmol. https://doi.org/10.1097/WNO.0000000000000823
Hasan SA, Baker RS, Sun WS, Rouholiman BR, Chuke JC, Cowen DE, Porter JD (1997) The role of blink adaptation in the pathophysiology of benign essential blepharospasm. Arch Ophthalmol 115(5):631–6.27
Aramideh M, Ongerboer de Vissser BW, Devriese PP et al (1994) Electromyographic features of levator palpebrae superioris and orbicularis oculi muscles in blepharospasm. Brain 117:27–38
Bentivoglio AR, Daniele A, Albanese A, Tonali PA, Fasano A (2006) Analysis of blink rate in patients with blepharospasm. Mov Disord 21(8):1225–1229
Ferrazzano G, Conte A, Belvisi D, Fabbrini A, Baione V, Berardelli A, Fabbrini G (2019) Writing, reading, and speaking in blepharospasm. J Neurol 266(5):1136–1140
Lee WH, Seo JM, Hwang JM (2017) The analysis of eye blinking pattern using high-frame-rate camera. Conf Proc IEEE Eng Med Biol Soc 2017:1509–1512
Doane MG (1980) Interactions of eyelids and tears in corneal wetting and the dynamics of the normal human eyeblink. Am J Ophthalmol 89:507–516
Kwon K-A, Shipley RJ, Edirisinghe M, Ezra DG, Rose G, Best SM, Cameron RE (2013) High-speed camera characterization of voluntary eye blinking kinematics. J R Soc Interface 10(85):213–227
Malbouisson J, Messias A, Garcia DM et al (2010) Modeling upper eyelid kinematics during spontaneous and reflex blinks. J Neurosci Methods 191:119–125
Sforza C, Rango M, Galante D et al (2008) Spontaneous blinking in healthy persons: an optoelectronic study of eyelid motion. Ophthalmic Physiol Opt 28:345–353
Frigerio A, Hadlock TA, Murray EH, Heaton JT (2014) Infrared-based blink-detecting glasses for facial pacing: toward a bionic blink. JAMA Facial Plast Surg 16:211–218
Hanuka A, Itzhak M, Berger A et al (2017) A novel eyelid motion monitor. Graefes Arch Clin Exp Ophthalmol 255:1811–1817
Hershman R, Henik A, Cohen N (2018) A novel blink detection method based on pupillometry noise. Behav Res Methods 50:107–114
Author information
Authors and Affiliations
Contributions
Midori H. Osaki: (1) research project: (a) conception, (b) organization, (c) execution; (2) statistical analysis: (a) design, (b) review, (c) and critique; (3) manuscript preparation: (a) writing of the first draft and (b) review and approval of the final version.
Tammy H. Osaki: (1) research project: (a) conception, (b) organization, (c) execution; (2) statistical analysis: (a) review and critique; (3) manuscript preparation: (a) review and critique and (b) approval of the final version.
Denny Garcia: (1) research project: (a) execution; (2) statistical analysis: (a) design and (b) execution; (3) manuscript preparation: (a) review and critique and (b)approval of the final version.
Teissy Osaki: (1) research project: (a) organization and (b) execution; (2) statistical analysis: (a) review and critique; (3) manuscript preparation: (a) review and critique and (b) approval of the final version.
Gustavo Gameiro: (1) research project: (a) execution; (2) statistical analysis: (a) review and critique; (3) manuscript preparation: (a) review and critique and (b) approval of the final version.
Rubens Belfort Jr.: (1) research project: (a) conception; (2) statistical analysis: (a) review and critique; (3) manuscript preparation: (a) review and critique and (b) approval of the final version.
Antonio Augusto Cruz: (1) research project: (a) conception, (b) organization; (2) statistical analysis: (a) review and critique; (3) manuscript preparation: (a) review and critique and (b) approval of the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the UNIFESP IRB (number 1322/2016) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Osaki, M.H., Osaki, T.H., Garcia, D. et al. Analysis of blink activity and anomalous eyelid movements in patients with hemifacial spasm. Graefes Arch Clin Exp Ophthalmol 258, 669–674 (2020). https://doi.org/10.1007/s00417-019-04567-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-019-04567-w