Abstract
Purpose
Stage 4A retinopathy of prematurity (ROP) is a critical phase where retinal detachment develops, but fovea is preserved. The present study aims to evaluate the effect of the first treatment choice (laser photocoagulation (LPC) or intravitreal ranibizumab (IVR)) applied in this critical phase on the prognosis of the disease.
Methods
Records of patients diagnosed with stage 4A ROP and whose first treatment was applied in our clinic were evaluated retrospectively. All patients were referred to our clinic for the treatment of advanced ROP . While group 1 was composed of the patients who were administered LPC as first treatment, group 2 included patients where IVR was applied as first treatment. The patients in both groups were referred to surgical treatment in the presence of progression.
Results
The present study included a total of 31 eyes in 16 patients with stage 4A ROP. Eighteen eyes of nine patients in group 1 were first applied LPC, and 13 eyes of seven patients in group 2 were first applied intravitreal ranibizumab. While anatomic outcomes of ten eyes in both groups were favorable, eight eyes in group 1 and three eyes in group 2 displayed progression and were referred to vitreoretinal surgery.
Conclusions
Laser and/or IVR treatment may be effective as a non-surgical treatment for stage 4A ROP. Especially stage 4A ROP until 6 clock hours can regress without surgical treatment. However, in stage 4A with involvement wider than 6 clock hours, non-surgical regression is difficult. Prospective controlled large series studies are necessary.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
According to the International Classification of Retinopathy of Prematurity (ICROP), the definition of the diagnosis of stage 4 retinopathy of prematurity (ROP) is ROP with extrafoveal (4A) or foveal (4B) partial retinal detachment (RD) [1]. Scleral buckling (SB) or vitrectomy are the surgical treatments for ROP-related RD [2]. Vitreoretinal traction associated with advanced ROP is reported to be relieved effectively with lens-sparing vitrectomy (LSV) [3, 4]. While for some eyes vision can be preserved with early surgical intervention in stage 4 ROP [5], some eyes with stage 4A ROP do not progress after laser treatment [6].
Vitreous vascular endothelial growth factor (VEGF) levels that are significantly higher have been identified in vascular active stage 4 ROP [7, 8]. Recently, the related literature indicates studies that report positive outcomes after intravitreal anti-VEGF agents in stage 4 ROP [9]. A few studies report that ranibizumab (Lucentis; Novartis, Basel, Switzerland), structured with the antibody fragment, is an alternative for intravitreal anti-VEGF therapy in ROP [10, 11].
The purpose of this study is to evaluate the effect of initial treatment choice (diode laser photocoagulation or intravitreal ranibizumab) on the prognosis of 4A ROP in premature infants.
Materials and methods
The present study retrospectively investigated patients who were diagnosed with stage 4A ROP and involved patients whose initial treatment was applied in our clinic. None of the cases received any treatment previously. The study was carried out in the center for the diagnosis and the treatment of retinopathy of prematurity in Adana Numune Training and Research Hospital between October 2013 and December 2015. Before the study was conducted, Institutional Review Board approval was obtained and ethical standards of the Declaration of Helsinki were followed. A written informed consent form was obtained from each patient’s parent before the treatment.
All the examinations were performed using a Heine video omega 2C binocular indirect ophthalmoscope (Heine Optotechnik, Herrsching, Germany) operating with archimed programme and obtaining images. The medical records and retinal images of all patients were obtained. The diagnosis of stage 4 ROP was defined as ROP with extrafoveal (4A) partial retinal detachment according to the international classification of ROP [1]. Gestational age, birth weight, stage and location of ROP, age at treatment, and final outcomes were recorded. The width of partial retinal detachment was recorded as clock hour.
All the patients in the study were born in other hospitals and they were referred to our clinic for treatment of progressive ROP. All the patients were administered the treatment within 24 h after the diagnosis; patients who received any kind of treatment before were excluded from the study. The patients were divided into two groups: group 1, which included patients receiving laser as initial treatment, and group 2, which included patients receiving intravitreal ranibizumab as initial treatment. Treatment choice was done according to statement of pupillary dilatation and clear vitreous. In case of poor pupillary dilatation and absence of clear vitreous, intravitreal ranibizumab was applied as initial treatment. Regression was defined as the decreased partial retinal detachment while progression was defined as increased partial retinal detachment. Persistence of ROP was defined as the lack of adequate regression after treatment, and the retreatment applied within 2 weeks from the previous treatment.
The infants were administered 0.25 mg intravitreal ranibizumab (IVR) in group 2 as initial treatment; those who were detected to have regression in partial retinal detachment were monitored. The patients whose partial retinal detachment was persistent were applied additional diode laser photocoagulation (LPC) within 2 weeks in group 2. Patients from both groups who demonstrated partial retinal detachment progression despite the treatment were referred to vitreoretinal surgery.
Laser ablation technique
Laser ablations were performed with an 810-nm diode laser through an indirect delivery system (IRIDEX; Oculight SL, Mountain View, CA, USA), using a 28-day condensing lens under topical anesthesia 0.5 % proparacaine hydrochloride (Alcaine; SA Alcon-Couvreur NV, Puurs, Belgium) with ketamine sedation. The procedure was carried under supervision of the neonatologist in the neonatal intensive care unit. The treatment of all the eyes was done with a near-confluent pattern of laser ablation with one-half a spot size apart. The laser settings were arranged to a power ranging between 150 and 250 mW and duration of 200 m/s and an interval of 300 so that a moderately white laser burn could be achieved. All the laser treatments were performed by the same specialist who is experienced on this issue (EAS). All the laser parameters, start and finish time of laser, and vital signs of the infant throughout the laser were recorded on nurse observation sheets.
Intravitreal injection technique
Using tropicamide 0.05 % and phenylephrine 2.5 % eye drops, all eyes were dilated 30 min before the IVI. Preparation of the eyes was done using povidone iodine 5 % in a standard fashion. After 3 min of waiting to provide aseptic conditions, 0.25 mg (0.025 ml) IVR was injected under topical anesthesia 0.5 % proparacaine hydrochloride (Alcaine; SA Alcon-Couvreur NV, Puurs, Belgium) 1 mm behind the temporal limbus via pars plicata with a 30-gauge needle. No sedation was used and a nurse held the infants during injections. After the injections, intraocular pressure and retinal artery patency were checked. All patients were given topical antibiotic drops for 1 week after IVR. All patients were monitored in terms of systemic side effects and their signs were recorded.
Data analysis
Analysis of the data was performed using Statistical Package for Social Sciences for Windows software (SPSS version 16.0, SPSS Inc. Chicago, IL, USA). While the descriptive statistics of normally distributed continuous variables were expressed as mean ± standard deviation, abnormally distributed ones were expressed as median (minimum–maximum). Mann–Whitney U test was used for the comparison of the variables. Categorical variables were presented as frequency (%) and compared between the groups using Chi-square test and Fisher’s exact test. Statistically significant differences were taken as a p value of 0.05.
Results
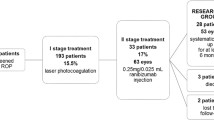
A total of 31 eyes in 16 patients (nine males and seven females) with stage 4A ROP were included in this study. Eighteen eyes of nine patients were initially treated with LPC in group 1; and 13 eyes of seven patients were initially treated with intravitreal ranibizumab in group 2. The mean for gestational age (GA) of the patients was found to be 29.00 ± 2.52 weeks (range, 25–33) in group 1 and 28.07 ± 2.46 weeks (range, 24–33) in group 2. The mean for birth weight (BW) of the patients was found to be 1288.33 ± 402 g (range, 675–1870) in group 1 and 1112.30 ± 331.33 g (range, 680–1720) in group 2. The mean postmenstrual age (PMA) at the time of treatment was 38.72 ± 2.39 weeks (range, 35–43) in group1 and 37.15 ± 1.31 weeks (range, 35–39) in group 2. The width of partial retinal detachment was 6.38 ± 3.43 clock hours (range, 3–12) in group 1 and 6.07 ± 2.06 clock hours (range, 3–9) in group 2. Tables 1 and 2 demonstrate the complete data of the patients.
There were no significant differences in gestational age (p values = 0.291), birth weight (p values = 0.356), postmenstrual age at the time of treatment (p values = 0.080) and width of partial retinal detachment (p value = 0.806) between these two groups.
No apparent ocular complications were detected related to the injection, such as endophthalmitis, ocular inflammation, cataract, or vitreous hemorrhage. Moreover, no major systemic complications occurred.
Retinal reattachment was achieved in ten out of 18 eyes after single LPC treatment in group 1. In spite of LPC treatment, eight eyes required surgery due to progressed ROP in group 1.
In group 2, only four eyes out of 13 attained retinal reattachment without additional LPC or vitreoretinal surgery. Retinal vascularization was completed in the postmenstrual 65th week in the right eye of case 2, postmenstrual 71st week in both eyes of case 3, and postmenstrual 69th week in the right eye of case 4. In group 2, nine eyes received LPC as additional treatment because of the persistence of ROP (see Fig. 1). Retinal reattachment was obtained after LPC in six eyes, but three eyes required vitreoretinal surgery because they progressed ROP.
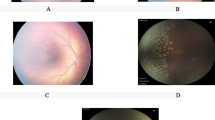
Images of patient where ranibizumab was applied as the initial treatment and then LPC was performed. Left eye of case 5 in group 2. A1 Preoperative poor pupillary dilatation. A2 Plus table is present at posterior pole. A3 Retinal detachment areas. A4 Good pupillary dilatation after IVR. A5 Partial retinal detachment area expands towards vitreous in a more transparent form after IVR. A6 Spots of the laser treatment applied after the IVR treatment on postop first day. A7 One week following application of laser treatment. A8 Two weeks following application of laser treatment. A9 Partial retinal detachment areas that regressed 4 weeks after LFC
Final anatomical outcomes were favorable and the macula was in normal contour in patients who did not make surgery treatment in both groups. Also, retinal reattachment was attained with surgical treatment in patients who had progressive ROP.
The regression without vitreoretinal surgical rates was 55.6 % in group 1; and it was found 76.9 % in group 2. 44.4 % (8/18) of the cases in group 1 and 23 % (3/13) of the cases in group 2 demonstrated progression and were referred to vitreoretinal surgery. No statistically significant differences were found in terms of the effect of first treatment choice on progression between the two groups (p values = 0.227). However, comparison of the laser parameters (see Table 3) showed that laser was completed with less power (p values = 0.001) and in a shorter time (p values = 0.001) in group 2; and this case was found to be statistically significant. In addition, vitreoretinal surgery requirement increased in the presence of younger gestational age (p values = 0.007), smaller birth weight (p values = 0.006), and wider involvement of partial retinal detachment (p values = 0.000), and this case was found to be statistically significant.
Discussion
Stage 4 ROP is an advanced stage of the disease, which is an intermediate phase seen before total retinal detachment in the eyes with progression. Treatment criteria for ROP have been identified by early treatment retinopathy of prematurity cooperative group (ETROP) and they describe these criteria for eyes with threshold or prethreshold ROP [12]. As for the eyes in which partial retinal detachment has started, no standardized criteria have been identified yet; neither for the follow-up nor for the choice of the treatment to be applied. There are quite limited sources about the treatment approach to be applied in particularly stage 4A ROP where macula is not involved. The current approach is to apply surgical treatment, mainly scleral buckling or LSV.
The anatomical success rate of vitrectomy for stage 4 ROP is reported to be between 73.2 and 92.0 %; with the presence of possible adverse effects of endophthalmitis, iatrogenic retinal break, cataract, and delayed-onset intraocular pressure elevation [3, 13, 14]. Also, the success rate of scleral buckling is 75 %; with possible complications of severe myopia, anisometropia, and the need for a secondary procedure for division (or removal) of the encircling element for the growing eye [15, 16]. On the other hand, in the ETROP study, vitrectomy for stage 4A was not as successful as reported in other case series [17]. Recently, anti-VEGF agents have been begun to be used in ROP treatment; and positive outcomes about the ranibizumab chosen have been reported in terms of systemic adverse effects [10, 11]. Anti-VEGF agents are usually used in prethreshold or threshold ROP. However, there are some recent studies showing that anti-VEGF agents have been beneficial in stage 4 ROP both as monotherapy [9] and in therapies combined with surgery [2].
Krohne et al. found that the half–life of ranibizumab was 7.19 days in a non-vitrectomized human eye [18] while this was found to be 9.82 days after bevacizumab [19]. Additionally, it was found that serum VEGF levels were suppressed for 2 months after intravitreal bevacizumab in patients with type 1 ROP [20]. More recurrence could be seen after IVR, which could be explained by the shorter ocular half-life of ranibizumab.
Cheng et al. performed IVR or intravitreal bevacizumab (IVB) in the 13 eyes of 7 patients with stage 4 and found that the disease regressed in all patients without any need for surgery and vascularization was completed [9]. Kusaka et al. [21] and Wu et al. [22] reported that there was retinal reattachment without vitrectomy in stage 4 ROP after IVB. In the present study, only four of 13 eyes that were applied intravitreal ranibizumab as first treatment regressed with IVR monotherapy without any need for additional laser treatment or surgery. After LPC was administered as additional treatment, six eyes were observed to have regression. As for the group applied laser treatment, ten out of 18 eyes regressed with LPC monotherapy. An overall evaluation of the eyes that showed progression in both groups despite the treatment showed that the main factor affecting progression was the width of the partial detachment area (p value = 0.000).
Hartnett et al. reported that in the presence of some clinical findings, threshold or prethreshold ROP progressed to advanced stages despite the treatment. Absence of clear vitreous, plus disease in two or more quadrants, and six or more clock hours of ridge elevation were the features that predicted progressive ROP [5]. The present study also found that the patients who demonstrated progression in stage 4A ROP despite the treatment were the patients who had wider involvement. Especially stage 4A ROP until 4 clock hours can regress with LPC or IVR monotherapy. As for the eyes having involvement in 4–6 clock hours, IVR and then LPC combined treatment might be beneficial. However, in stage 4A, with involvement wider than 6 clock hours, non-surgical regression is difficult despite the combined treatment.
In the series they presented, Futamura et al. reported that the LPC applied in the examinations before scleral buckling was insufficient in the eyes that received laser treatment and had progression to 4A. In the same series, they also found VEGF concentration in the patients’ aqueous to be high [9]. Sometimes, there are some technical difficulties of applying laser in stage 4 ROP due to poor pupillary dilatation, haze vitreous, and hemorrhage. Besides, avascular areas beneath the detachment line do not hold laser spots easily. Due to these technical difficulties, sometimes the laser applied is not sufficient, skip areas remain, and the disease might show progression. However, good pupillary dilatation and clear vitreous are enabled with the IVR applied before LPC in stage 4A ROP, burn is easily obtained in avascular retina. As a result, this study has demonstrated that LPC applied after IVR in stage 4A ROP could be administered more easily and effectively.
Honda et al. reported acute tractional RD after IVB they applied for ROP treatment [23]. Rapid neovascular involution with accelerated fibrosis and posterior hyaloid contraction, as a response to decreased levels of VEGF, is believed to be the cause of the development or progression of tractional RD [24]. No patients in this study had progressive fibrous traction after the injection.
Limitations of the present study include its having small series and short follow-up duration and being retrospective. Retinal reattachment and retinal vascularization were evaluated only clinically because fluorescein angiography was not made. Visual acuity of the patients was not evaluated. Besides, in group 2, the eyes that did not show regression within 2 weeks after IVR were applied LPC as an additional treatment. However, in the series presented by Cheng et al., regression after anti-VEGF was observed after 3 weeks. The eyes that were applied LPC and did not regress in 2 weeks, were referred to surgery. In stage 4A ROP, after IVR and/or LPC, it is not clear how long the patients should be followed before referring to surgery. However, for certain eyes, early surgical intervention in stage 4 ROP can be critical to preserve vision [5].
Conclusions
In some patients, the disease can regress without any need for surgery with IVR and/or LPC applied as monotherapy in early period in stage 4A ROP. The eyes that showed progression despite the treatment were the ones that had wider involvement of partial detachment. Although the effect of the first treatment choice (LPC or IVR) on prognosis was not statistically significant between the groups, applying IVR as first treatment was found to form a basis for an effective laser treatment. As for the eyes having involvement until 6 clock hours, IVR and/or LPC treatment might be beneficial. However, in stage 4A with involvement wider than 6 clock hours, non-surgical regression is difficult, despite the combined treatment. Further randomized controlled studies are necessary to compare the clinical efficacy and safety with other conventional interventions.
References
International Committee for the Classification of Retinopathy of Prematurity (2005) The International Classification of Retinopathy of Prematurity revisited. Arch Ophthalmol 123:991–999
Futamura Y, Asami T, Nonobe N et al (2015) Buckling surgery and supplemental intravitreal bevacizumab or photocoagulation on stage 4 retinopathy of prematurity eyes. Jpn J Ophthalmol 59(6):378–388
El Rayes EN, Vinekar A, Capone A (2008) Three-year anatomic and visual outcomes after vitrectomy for stage 4B retinopathy of prematurity. Retina 28:568–572
Choi J, Kim JH, Kim S-J, Yu YS (2011) Long-term results of lens-sparing vitrectomy for stages 4B and 5 retinopathy of prematurity. Korean J Ophthalmol 25:305–310
Hartnett ME, Mccolm JR (2004) Retinal features predictive of progressive stage 4 retinopathy of prematurity. Retina 24(2):237–241
Hartnett ME, Rodier DW, McColm JR et al (2003) Long-term vision results measured with Teller acuity cards and a new light perception projection scale after management of late stages of retinopathy of prematurity. Arch Ophthalmol 121(7):991–996
Sato T, Kusaka S, Shimojo H, Fujikado T (2009) Vitreous levels of erythropoietin and vascular endothelial growth factor in eyes with retinopathy of prematurity. Ophthalmology 116:1599–1603
Sato T, Kusaka S, Shimojo H, Fujikado T (2009) Simultaneous analyses of vitreous levels of 27 cytokines in eyes with retinopathy of prematurity. Ophthalmology 116:2165–2169
Cheng HC, Lee SM, Hsieh YT, Lin PK (2015) Efficacy of intravitreal injection of anti-vascular endothelial growth factor agents for stage 4 retinopathy of prematurity. Retina 35(4):660–666
Zhou Y, Jiang Y, Bai Y, Wen JCL (2016) Vascular endothelial growth factor plasma levels before and after treatment of retinopathy of prematurity with ranibizumab. Graefes Arch Clin Exp Ophthalmol 254(1):31–36
Menke M, Framme C, Nelle M et al (2015) Intravitreal ranibizumab monotherapy to treat retinopathy of prematurity zone II, stage 3 with plus disease. BMC Ophthalmology;15–20
Early Treatment for Retinopathy of Prematurity Cooperative Group (2003) Revised indications for the treatment of retinopathy of prematurity: results of the Early Treatment for Retinopathy of Prematurity randomized trial. Arch Ophthalmol 121:1684–1694
Kychenthal A, Dorta P (2008) 25-gauge lens-sparing vitrectomy for stage 4A retinopathy of prematurity. Retina 28:S65–S68
Iwahashi-Shima C, Miki A, Hamasaki T et al (2012) Intraocular pressure elevation is a delayed-onset complication after successful vitrectomy for stages 4 and 5 retinopathy of prematurity. Retina 32:1636–1642
Hinz BJ, de Juan E Jr, Repka MX (1998) Scleral buckling surgery for active stage 4A retinopathy of prematurity. Ophthalmology 105:1827–1830
Chow DR, Ferrone PJ, Trese MT (1998) Refractive changes associated with scleral buckling and division in retinopathy of prematurity. Arch Ophthalmol 116:1446–1448
Repka MX, Tung B, Good WV, Capone A Jr, Shapiro MJ (2011) Outcome of eyes developing retinal detachment during the Early Treatment for Retinopathy of Prematurity study. Arch Ophthalmol 129(9):1175–1179
Krohne TU, Liu Z, Holz FG, Meyer CH (2012) Intraocular pharmacokinetics of ranibizumab following a single intravitreal injection in humans. Am J Ophthalmol 154:682–686
Krohne TU, Eter N, Holz FG, Meyer CH (2008) Intraocular pharmacokinetics of bevacizumab after a single intravitreal injection in humans. Am J Ophthalmol 146:508–512
Wu WC, Lien R, Liao PJ, Wang NK, Chen YP, Chao AN, Chen KJ, Chen TL, Hwang YS, Lai CC (2015) Serum levels of vascular endothelial growth factor and related factors after intravitreous bevacizumab injection for retinopathy of prematurity. JAMA Ophthalmol 133:391–397
Kusaka S, Shima C, Wada K et al (2008) Efficacy of intravitreal injection of bevacizumab for severe retinopathy of prematurity: a pilot study. Br J Ophthalmol 92:1450–1455
Wu WC, Yeh PT, Chen SN et al (2011) Effects and complications bevacizumab use in patients with retinopathy of prematurity: a multicenter study in Taiwan. Ophthalmology 118:176–183
Honda S, Hirabayashi H, Tsukahara Y, Negi A (2008) Acute contraction of the proliferative membrane after an intravitreal injection of bevacizumab for advanced retinopathy of prematurity. Graefes Arch Clin Exp Ophthalmol 246:1061–1063
Arevalo JF, Maia M, Flynn HW Jr et al (2008) Tractional retinal detachment following intravitreal bevacizumab (Avastin) in patients with severe proliferative diabetic retinopathy. Br J Ophthalmol 92:213–216
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for this research.
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study. The authors did not receive any financial support from any public or private sources. The authors have no financial or proprietary interest in a product, method, or material described herein.
Rights and permissions
About this article
Cite this article
Sukgen, E.A., Koçluk, Y. Treatment for stage 4A retinopathy of prematurity: laser and/or ranibizumab. Graefes Arch Clin Exp Ophthalmol 255, 263–269 (2017). https://doi.org/10.1007/s00417-016-3443-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-016-3443-6