Abstract
Purpose
To compare the 2-year results after photodynamic therapy (PDT) alone and PDT combined with intravitreal injections of bevacizumab and triamcinolone acetonide (triple therapy) for polypoidal choroidal vasculopathy (PCV).
Methods
We retrospectively reviewed the medical records of 40 consecutive patients (40 eyes) with subfoveal PCV. Of these 40 eyes, 16 were treated with PDT alone and 24 were treated with triple therapy.
Results
The change in visual acuity in the triple therapy group was significantly better than that in the PDT group (P < 0.001). At 24 months, improvement in visual acuity was seen in only two eyes (12.5 %) of the PDT group, while it was seen in ten eyes (41.7 %) of the triple therapy group. Retreatment was given to 12 eyes (75.0 %) in the PDT group and to nine eyes (37.5 %) in the triple therapy group, although the retreatment-free period was significantly longer in the triple therapy group than in the PDT group (P < 0.001). Post-treatment vitreous hemorrhage was seen in only two eyes (12.5 %), all of which were in the PDT group.
Conclusion
Compared with PDT alone, triple therapy appears to reduce the postoperative hemorrhagic complications and recurrences of PCV and to improve the 2-year visual outcomes of PCV.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Previously, photodynamic therapy (PDT) with verteporfin was used primarily for the treatment of classic choroidal neovascularization (CNV) associated with age-related macular degeneration (AMD) [1], while today, intravitreal injections of anti-vascular endothelial growth factor (VEGF) agents (bevacizumab or ranibizumab) have become the principal treatment for exudative AMD [2–4]. Three monthly injections of anti-VEGF agents often lead to visual acuity (VA) improvement, but after these initial injections, repeated injections are usually required in order to maintain initial visual recovery. However, repeated injections increase the risk of ophthalmic and systemic adverse events, such as endophthalmitis and stroke [5]. To achieve better visual outcomes with fewer treatments, several reports have shown promising short-term effects of PDT combined with intravitreal injection of bevacizumab and a steroid (triamcinolone acetonide [TA] or dexamethasone) for exudative AMD [6, 7].
Polypoidal choroidal vasculopathy (PCV) is now recognized as a distinct clinical entity, differing in many ways from exudative AMD [8]. PCV is characterized by a branching vascular network that terminates in polypoidal lesions seen on indocyanine green angiography (IA), and is more common in Asians than in Caucasians [9, 10]. It has been reported that the treatment effects of anti-VEGF agents on the vascular lesions of PCV are limited in short-term follow-up [11, 12]. In contrast, a number of studies have shown encouraging results of PDT for the treatment of PCV. A small number of sessions of PDT causes regression of the polypoidal lesions, and often results in stable, or even improved VA [13–15]. Subsequent reports, however, have shown extensive hemorrhagic complications and recurrences of the polypoidal lesions after the initially successful treatment with PDT [16, 17].
Recently, PDT combined with anti-VEGF agents has been reported to improve the short-term visual outcome in PCV, compared with PDT alone [18–20]. In addition, it has been suggested that this combination therapy may reduce the risk of postoperative hemorrhagic complications [18]. Furthermore, it has been reported that TA suppresses the early proangiogenic response of retinal pigment epithelium (RPE) cells after PDT treatment [21], and that the intravitreal injection of TA per se has a suppressive effect on CNV [22]. For the treatment of PCV, when PDT is combined with an anti-VEGF agent and also with TA, this triple therapy might reduce postoperative complications and the recurrence rate, and lead to a better visual prognosis. To date, however, no information is available on the effects of this triple therapy for PCV. Accordingly, the study described herein aimed to evaluate the long-term results of this triple therapy on symptomatic subfoveal PCV by comparing it with PDT alone.
Patients and methods
For this retrospective study, we reviewed the medical records of 40 consecutive eyes (40 patients) with treatment-naïve subfoveal PCV, who were treated with PDT alone or with PDT combined with an intravitreal injection of bevacizumab and TA (triple therapy) at Kyoto University Hospital between September 2004 and December 2008. PDT alone was performed on 16 patients (PDT group) between September 2004 and December 2007 and PDT combined with bevacizumab and TA was performed on 24 patients (triple therapy group) between January 2008 and December 2008. Inclusion criteria of the study were: (1) symptomatic PCV in patients older than 50 years, (2) the presence of subfoveal vascular lesions, (3) best-corrected VA of 20/25 or worse, and (4) a minimum follow-up of 24 months after initial treatment. Exclusion criteria were: (1) eyes with other macular abnormalities (i.e., AMD, pathologic myopia, idiopathic CNV, presumed ocular histoplasmosis, angioid streaks, and other secondary CNV), (2) any contraindications for fluorescein angiography (FA), IA, or verteporfin, (3) the presence of an RPE tear, (4) any previous treatment for subfoveal PCV, (5) a history of previous vitrectomy, or (6) any other additional therapy during the study period (i.e., anti-VEGF therapy). This study was approved by the Institutional Review Board at Kyoto University Graduate School of Medicine, and adhered to the tenets of the Declaration of Helsinki.
The diagnosis of PCV was based on IA, which shows a branching vascular network terminating in polypoidal dilation. In the present study, pseudophakic eyes were included. When both eyes with PCV that were treated with PDT or triple therapy met the inclusion criteria, only the eye which was treated initially was included in the current study. Some patients in the PDT group were included in a previous study [17].
At the initial visit, each patient underwent a comprehensive ophthalmologic examination, including measurement of best-corrected VA with a Landolt chart, determination of intraocular pressure, indirect ophthalmoscopy, slit-lamp biomicroscopy with a contact lens, optical coherence tomography examinations, and FA and IA (HRA-2, Heidelberg Engineering, Dossenheim, Germany).
In eyes of the PDT group, standard-fluence PDT was performed using a 689 nm diode laser unit (Visulas PDT system 690S; Carl Zeiss, Dublin, CA, USA) after an injection of verteporfin (Visudyne; Novartis Pharma AG, Basel, Switzerland), according to PDT guidelines for AMD [23]. In eyes that received triple therapy, injection of bevacizumab (1.25 mg) and TA (2 mg) was performed in a sterile manner, and prophylactic topical antibiotics were applied for 1 week after the injection. At 3–4 days after the intravitreal injection, standard-fluence PDT was performed according to PDT guidelines for AMD. The greatest linear dimension was calculated based on FA and IA, as described in detail previously [24]. All polypoidal lesions, the entire branching vascular network, and type 2 CNV detected by FA and IA were included. Serous pigment epithelial detachment was not included in the lesion area when the absence of underlying CNV was confirmed by IA.
After the initial treatment, each patient was scheduled to be seen at 3 months, at which time they again underwent a comprehensive ophthalmologic examination. When IA showed recurrent or residual polypoidal lesions and exudative change was seen on ocular coherence tomography (OCT), retreatment with PDT or the triple therapy was given according to the initial treatment. When residual polypoidal lesions were detected on IA but no exudative change was seen on OCT, no retreatment was given and the patient was reevaluated at the next visit.
All values are presented as mean ± standard deviation. For statistical analysis, best-corrected VA as measured with a Landolt chart was converted to a logarithm of the minimum angle of resolution (logMAR). VA was considered to be improved or deteriorated when the logMAR change was greater than 0.2. On OCT scans, foveal thickness was defined as the distance between the inner surface of the neurosensory retina and the RPE beneath the fovea. In each group, VA or foveal thickness after treatment was studied by one-way repeated measures analysis of variance with the Dunnett test. To compare VA and foveal thickness between the PDT group and the triple therapy group, two-factor repeated measures analysis of variance was used. The retreatment-free period was calculated from the date of the initial therapy to the date when the treating physician determined the necessity of retreatment by PDT or by triple therapy. Survival analysis using Kaplan–Meier methods was used to compare the difference in the retreatment-free period after initial treatment between the PDT and the triple therapy groups. In patients who underwent no retreatment by either PDT or triple therapy, the retreatment-free period was established at 2 years of follow-up. Descriptive statistics for all demographic and clinical variables were calculated, and comparisons made using the unpaired t-test for means with continuous data (e.g., age) and the Chi-squared test for categorical data (e.g., gender). Statview version 5.0 software (SAS Institute, Inc., Cary, NC, USA) was used for statistical analyses. A P value < 0.05 was considered to be statistically significant.
Results
In the study described herein, a total of 40 patients with treatment-naïve PCV were evaluated; 16 eyes which received only treatment with PDT (PDT group) and 24 eyes which received only triple therapy (triple therapy group) during the 24-month study period. Table 1 shows baseline characteristics of each group. There were no significant differences in age, gender, or baseline VA between the two groups. However, baseline foveal thickness of the triple therapy group was significantly larger than that of the PDT group, and the rate of serous retinal detachment was also higher in the triple therapy group.
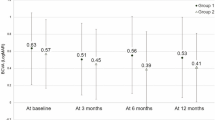
Figure 1a shows the time-course in mean foveal thickness of each group. In the triple therapy group, the mean foveal thickness decreased immediately after initiation of treatment and remained throughout the 2-year follow-up period. Figure 1b shows the time-course of mean VA in each group. In the PDT group, there was no significant improvement of VA after initiation of treatment; in fact, mean VA was significantly decreased (+0.25 ± 0.45) at 24 months (P = 0.041). In contrast, VA somewhat improved after initiation of treatment in the triple therapy group, while the improvement was not statistically significant. Some improvement was maintained throughout the 2-year follow-up period.
Mean foveal thickness (a) and mean visual acuity (b) in eyes with polypoidal choroidal vasculopathy treated with photodynamic therapy (PDT group) or with PDT combined with intravitreal injections of bevacizumab and triamcinolone acetonide (triple therapy group). Visual acuity is shown in logMAR fashion. *P < 0.05, compared with pretreatment values. Error bars represent the standard error
Figure 2 shows the change in mean VA from baseline in each group. The mean change in VA in the triple therapy group was significantly better than that in the PDT group (P < 0.001). At 24 months after initial treatment, mean change in VA in the triple therapy group (−0.10 ± 0.32) was significantly better than that in the PDT group (+0.25 ± 0.45, P = 0.007). Figure 3 shows the percentage of eyes with improved or decreased VA at 24 months; improvement in VA was seen in two eyes (12.5 %) of the PDT group and in ten eyes (41.7 %) of the triple therapy group, and reduction in VA was seen in nine eyes (56.3 %) of the PDT group and in five eyes (20.1 %) of the triple therapy group. Thus, improvement in VA was seen more frequently in the triple therapy group (P = 0.044).
Change in visual acuity in eyes with polypoidal choroidal vasculopathy treated with photodynamic therapy (PDT group) or with a combination of PDT and intravitreal injection of bevacizumab and triamcinolone acetonide (triple therapy group). Change in visual acuity in eyes treated with triple therapy was significantly better than that in eyes treated with PDT alone (P < 0.001). Visual acuity is shown in logMAR fashion. *P < 0.05, compared with values in the PDT group. Error bars represent the standard error
Percentages of eyes with improved and deteriorated visual acuity at 24 months after treatment. All eyes with polypoidal choroidal vasculopathy were treated with photodynamic therapy (PDT group) or PDT combined with intravitreal injection of bevacizumab and triamcinolone acetonide (triple therapy group). Visual acuity was considered to be improved or deteriorated when the change in logMAR units was greater than 0.2. Improvement of visual acuity was seen more frequently in the triple therapy group (P = 0.044)
Table 2 summarizes final characteristics and complications during the study period. In both groups, the polypoidal lesions disappeared after treatment (Fig. 4). At 24 months, complete disappearance of the polypoidal lesions was confirmed in 13 eyes (81.3 %) in the PDT group and in 22 eyes (91.7 %) of the triple therapy group. Fifteen eyes (62.5 %) of the triple therapy group and four eyes (25.0 %) of PDT group underwent a single session of PDT during the 24-month study period (P = 0.020). Retreatment by PDT was done in 12 eyes (75.0 %) in that group, and nine eyes (37.5 %) in the triple therapy group received retreatment by triple therapy (Fig. 5). The mean numbers of treatment were 2.19 ± 0.91 in the PDT group and 1.50 ± 0.78 in the triple therapy group (P = 0.015). Figure 6 shows the overall survival analysis curve for the retreatment-free periods in each group, which was significantly longer in the triple therapy group (20.6 ± 6.8 months) than in the PDT group (11.7 ± 8.6 months, P < 0.001).
Polypoidal choroidal vasculopathy successfully treated with photodynamic therapy combined with intravitreal injection of bevacizumab and triamcinolone acetonide (triple therapy). a Initial fundus photograph (left) shows reddish-orange nodules and fibrin exudate (20/50 OS). Indocyanine green angiography (middle) shows a branching vascular network that terminates in polypoidal lesions. A vertical optical coherence tomography section through the fovea (right) reveals sharp protrusions of the retinal pigment epithelium due to polypoidal lesions (arrows). A branching vascular network is seen as flat protrusions (arrowheads). b Three months after triple therapy. No polypoidal lesions are seen on indocyanine green angiogram. A branching vascular network is still seen. Protrusions due to polypoidal lesions have become flattened (20/60 OS). c 12 months after treatment, no recurrence is seen (20/50 OS)
Recurrence of polypoidal lesions after successful treatment with photodynamic therapy of polypoidal choroidal vasculopathy. a Initial fundus photograph (left) shows a reddish-orange nodule with a hard exudate and with a subretinal hemorrhage (20/40 OD). Indocyanine green angiography (right) shows a branching vascular network that terminates in polypoidal lesions. b Three months after photodynamic therapy. Reddish-orange nodules and polypoidal lesions have regressed (20/30 OD). c At 12 months after treatment, recurrence has occurred. Fundus photograph shows pigment epithelial detachments with surrounding subretinal hemorrhage and hard exudate (20/100 OD). Polypoidal lesions have recurred at the terminus of the remaining branching vascular network. d At 3 months after retreatment with photodynamic therapy, pigment epithelial detachments and polypoidal lesions have completely regressed (20/70 OD)
Overall survival curve for the retreatment-free period in both treatment groups. Eyes with polypoidal choroidal vasculopathy were treated with either photodynamic therapy (PDT group) or PDT combined with intravitreal injection of bevacizumab and triamcinolone acetonide (triple therapy group). The period until the treating physician opted to perform additional treatment was significantly longer in the triple therapy group than in the PDT group (P < 0.001)
In the current study, two eyes (12.5 %) in the PDT group developed cataract and underwent surgery during the study period; additionally, one eye (4.2 %) in the triple therapy group (P = 0.327) underwent similar surgery. In the PDT group, two eyes (12.5 %) developed a vitreous hemorrhage and one eye developed an RPE tear during the study period, while no eye in the triple therapy group developed a vitreous hemorrhage. No eye underwent glaucoma surgery for ocular hypertension after intravitreal injections of TA.
Discussion
Several investigators have reported the short-term outcomes of anti-VEGF agents for the treatment of PCV, and have shown the limited effect of these agents on the vascular lesions of PCV, even though the anti-VEGF agents reduced the exudative change that was due to PCV. It has been reported that complete resolution of the polypoidal lesions was achieved in only 16.1 % of eyes, with a mean of 3.3 injections of bevacizumab over a 12 month period [25]. In an earlier report, although monthly injections of ranibizumab successfully reduced the exudative manifestations of PCV, a reduction in the polypoidal lesions was seen in only 33 % of eyes [11].
In contrast, a number of studies have shown encouraging results of PDT for the vascular lesions of PCV, with complete regression of the polypoidal lesions achieved in many cases with fewer sessions. In a report by Chan et al. [26], PDT led to complete regression of the polypoidal lesions in 95 % of eyes with PCV, and resulted in either stable or improved VA 1 year after treatment in 95 % of eyes. However, a year or more after successful treatment with PDT, recurrences of the polypoidal lesions sometimes cause a substantial decrease in VA. Using Kaplan–Meier methods, Yamashiro et al. reported that the recurrence of polypoidal lesions after successful PDT treatment was estimated to be 11.5 % at 15 months, 20.4 % at 18 months, and 38.8 % at 21 months [16]. Thus, the recurrence of polypoidal lesions after PDT is a major problem in the treatment of PCV.
In the combination therapy, anti-VEGF agents, which can cause rapid reduction of the exudative change, are thought to contribute to the visual recovery that is associated with regression of the polypoidal lesions induced by PDT. Previous experimental studies have shown increased expression of VEGF shortly after PDT treatment [27, 28], which suggests that an intravitreal injection of bevacizumab before PDT may well exert a protective effect. With regard to the injection of TA, Okubo and colleagues reported a case of PCV successfully treated with trans-Tenons retrobulbar injection of TA [29], and Mukai and colleagues reported the protective effects of TA against occlusion of the choriocapillaris which was induced by PDT [30]. However, Lai et al. reported that the adjunctive use of TA during PDT did not appear to result in additional benefit for the treatment of PCV [31], so the effect of TA on PCV remains controversial.
In the current study, the PDT group showed no significant improvement in VA after initiation of treatment; mean VA was significantly decreased at 24 months. In the triple therapy group, however, VA was improved at 3 months after treatment and mean VA was improved by −0.11 (logMAR) at 6 months and by −0.09 (logMAR) at 12 months. In this triple therapy group, although improvement was not statistically significant, at least some improvement in VA was maintained throughout the 2-year follow-up period. At 24 months, VA improvement was achieved in only 12.5 % of eyes in the PDT group and in 41.7 % in the triple therapy group (P = 0.044), so, based on our findings, triple therapy for PCV, compared to PDT alone, results in more rapid visual recovery and improved visual outcome at 24 months.
In our case series, both PDT alone and the triple therapy successfully reduced polypoidal lesions and exudative change, with complete disappearance of the polypoidal lesions confirmed at 24 months in 81.3 % of cases in the PDT group and in 91.7 % of cases in the triple therapy group. There were significant differences in the number of eyes with a recurrence and in the number of PDT treatments between the two groups. Furthermore, the retreatment-free period was significantly longer in the triple therapy group (20.6 ± 6.8 months) than in the PDT group (11.7 ± 8.6 months).
Following treatment for PCV, one of the most vision-threatening complications of PDT is extensive hemorrhage. A previous report of PCV treated with PDT indicated that postoperative subretinal hemorrhage was seen in 28 of 91 eyes, and that bleeding resulted in a vitreous hemorrhage in six eyes [32]. In the current study, no eye in the triple therapy group developed a vitreous hemorrhage, although two eyes in the PDT group developed a vitreous hemorrhage. Recent reports by Gomi et al. [18] and by Sato et al. [19] suggested a lower incidence of subretinal hemorrhage after PDT when it was combined with bevacizumab, and it has been reported that the vasoconstrictive effect of bevacizumab may contribute to the suppression of postoperative hemorrhages [33].
Major limitations of the current study are its retrospective nature and its relatively small sample size. In addition, there were some statistical differences between the two groups, including baseline foveal thickness and the rate of serous retinal detachment, which may affect the response to treatment. Furthermore, this study was not a randomized, comparative trial. However, selection bias is small as both groups consisted of consecutive eyes that were treated at different time periods. Our findings suggest that intravitreal injection of bevacizumab and TA combined with PDT improves the 2-year visual outcome of PCV and may reduce postoperative hemorrhagic complications and the recurrence rate. However, because our findings are based on an observation period of only 24 months, it remains unclear whether triple therapy has a long-term effect.
Another limitation is that the safety and efficacy of the triple therapy were not compared with PDT combined with anti-VEGF therapy. Recently, the EVEREST study has shown the 6-month effects of PDT in combination with ranibizumab for PCV [34], in which the eyes treated with PDT combined with ranibizumab achieved the highest gains at 6 months. However, it remains unclear whether this combination therapy reduces the recurrence of polypoidal lesions after successful initial treatment. Further prospective, randomized, long-term studies are necessary to determine the efficacy and safety of triple therapy for PCV.
References
Bressler NM, Arnold J, Benchaboune M, Blumenkranz MS, Fish GE, Gragoudas ES, Lewis H, Schmidt-Erfurth U, Slakter JS, Bressler SB, Manos K, Hao Y, Hayes L, Koester J, Reaves A, Strong HA (2002) Verteporfin therapy of subfoveal choroidal neovascularization in patients with age-related macular degeneration: additional information regarding baseline lesion composition’s impact on vision outcomes—TAP report No. 3. Arch Ophthalmol 120:1443–1454
Avery RL, Pieramici DJ, Rabena MD, Castellarin AA, Nasir MA, Giust MJ (2006) Intravitreal bevacizumab (Avastin) for neovascular age-related macular degeneration. Ophthalmology 113:363–372, e365
Heier JS, Antoszyk AN, Pavan PR, Leff SR, Rosenfeld PJ, Ciulla TA, Dreyer RF, Gentile RC, Sy JP, Hantsbarger G, Shams N (2006) Ranibizumab for treatment of neovascular age-related macular degeneration: a phase I/II multicenter, controlled, multidose study. Ophthalmology 113(633):e631–e634
Michels S, Rosenfeld PJ, Puliafito CA, Marcus EN, Venkatraman AS (2005) Systemic bevacizumab (Avastin) therapy for neovascular age-related macular degeneration twelve-week results of an uncontrolled open-label clinical study. Ophthalmology 112:1035–1047
Martin DF, Maguire MG, Ying GS, Grunwald JE, Fine SL, Jaffe GJ (2011) Ranibizumab and bevacizumab for neovascular age-related macular degeneration. N Engl J Med 364:1897–1908
Ahmadieh H, Taei R, Soheilian M, Riazi-Esfahani M, Karkhaneh R, Lashay A, Azarmina M, Dehghan MH, Moradian S (2007) Single-session photodynamic therapy combined with intravitreal bevacizumab and triamcinolone for neovascular age-related macular degeneration. BMC Ophthalmol 7:10
Yip PP, Woo CF, Tang HH, Ho CK (2009) Triple therapy for neovascular age-related macular degeneration using single-session photodynamic therapy combined with intravitreal bevacizumab and triamcinolone. Br J Ophthalmol 93:754–758
Ciardella AP, Donsoff IM, Huang SJ, Costa DL, Yannuzzi LA (2004) Polypoidal choroidal vasculopathy. Surv Ophthalmol 49:25–37
Sho K, Takahashi K, Yamada H, Wada M, Nagai Y, Otsuji T, Nishikawa M, Mitsuma Y, Yamazaki Y, Matsumura M, Uyama M (2003) Polypoidal choroidal vasculopathy: incidence, demographic features, and clinical characteristics. Arch Ophthalmol 121:1392–1396
Maruko I, Iida T, Saito M, Nagayama D, Saito K (2007) Clinical characteristics of exudative age-related macular degeneration in Japanese patients. Am J Ophthalmol 144:15–22
Kokame GT, Yeung L, Lai JC (2010) Continuous anti-VEGF treatment with ranibizumab for polypoidal choroidal vasculopathy: 6-month results. Br J Ophthalmol 94:297–301
Hikichi T, Ohtsuka H, Higuchi M, Matsushita T, Ariga H, Kosaka S, Matsushita R, Takami K (2010) Improvement of angiographic findings of polypoidal choroidal vasculopathy after intravitreal injection of ranibizumab monthly for 3 months. Am J Ophthalmol 150:674–682, e671
Spaide RF, Donsoff I, Lam DL, Yannuzzi LA, Jampol LM, Slakter J, Sorenson J, Freund KB (2002) Treatment of polypoidal choroidal vasculopathy with photodynamic therapy. Retina 22:529–535
Gomi F, Ohji M, Sayanagi K, Sawa M, Sakaguchi H, Oshima Y, Ikuno Y, Tano Y (2008) One-year outcomes of photodynamic therapy in age-related macular degeneration and polypoidal choroidal vasculopathy in Japanese patients. Ophthalmology 115:141–146
Tsuchiya D, Yamamoto T, Kawasaki R, Yamashita H (2009) Two-year visual outcomes after photodynamic therapy in age-related macular degeneration patients with or without polypoidal choroidal vasculopathy lesions. Retina 29:960–965
Yamashiro K, Tsujikawa A, Nishida A, Mandai M, Kurimoto Y (2008) Recurrence of polypoidal choroidal vasculopathy after photodynamic therapy. Jpn J Ophthalmol 52:457–462
Kurashige Y, Otani A, Sasahara M, Yodoi Y, Tamura H, Tsujikawa A, Yoshimura N (2008) Two-year results of photodynamic therapy for polypoidal choroidal vasculopathy. Am J Ophthalmol 146:513–519
Gomi F, Sawa M, Wakabayashi T, Sasamoto Y, Suzuki M, Tsujikawa M (2010) Efficacy of intravitreal bevacizumab combined with photodynamic therapy for polypoidal choroidal vasculopathy. Am J Ophthalmol 150:48–54
Sato T, Kishi S, Matsumoto H, Mukai R (2010) Combined photodynamic therapy with verteporfin and intravitreal bevacizumab for polypoidal choroidal vasculopathy. Am J Ophthalmol 149:947–995
Ruamviboonsuk P, Tadarati M, Vanichvaranont S, Hanutsaha P, Pokawattana N (2010) Photodynamic therapy combined with ranibizumab for polypoidal choroidal vasculopathy: results of a 1-year preliminary study. Br J Ophthalmol 94:1045–1051
Obata R, Iriyama A, Inoue Y, Takahashi H, Tamaki Y, Yanagi Y (2007) Triamcinolone acetonide suppresses early proangiogenic response in retinal pigment epithelial cells after photodynamic therapy in vitro. Br J Ophthalmol 91:100–104
Wang YS, Friedrichs U, Eichler W, Hoffmann S, Wiedemann P (2002) Inhibitory effects of triamcinolone acetonide on bFGF-induced migration and tube formation in choroidal microvascular endothelial cells. Graefes Arch Clin Exp Ophthalmol 240:42–48
Treatment of age-related macular degeneration with photodynamic therapy (TAP) Study Group (1999) Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin: one-year results of 2 randomized clinical trials—TAP report. Arch Ophthalmol 117:1329–1345
Otani A, Sasahara M, Yodoi Y, Aikawa H, Tamura H, Tsujikawa A, Yoshimura N (2007) Indocyanine green angiography: guided photodynamic therapy for polypoidal choroidal vasculopathy. Am J Ophthalmol 144:7–14
Cheng CK, Peng CH, Chang CK, Hu CC, Chen LJ (2011) One-year outcomes of intravitreal bevacizumab (Avastin) therapy for polypoidal choroidal vasculopathy. Retina 31:846–856
Chan WM, Lam DS, Lai TY, Liu DT, Li KK, Yao Y, Wong TH (2004) Photodynamic therapy with verteporfin for symptomatic polypoidal choroidal vasculopathy: one-year results of a prospective case series. Ophthalmology 111:1576–1584
Schmidt-Erfurth U, Schlotzer-Schrehard U, Cursiefen C, Michels S, Beckendorf A, Naumann GO (2003) Influence of photodynamic therapy on expression of vascular endothelial growth factor (VEGF), VEGF receptor 3, and pigment epithelium-derived factor. Invest Ophthalmol Vis Sci 44:4473–4480
Tatar O, Adam A, Shinoda K, Stalmans P, Eckardt C, Luke M, Bartz-Schmidt KU, Grisanti S (2006) Expression of VEGF and PEDF in choroidal neovascular membranes following verteporfin photodynamic therapy. Am J Ophthalmol 142:95–104
Okubo A, Ito M, Kamisasanuki T, Sakamoto T (2005) Visual improvement following trans-Tenon’s retrobulbar triamcinolone acetonide infusion for polypoidal choroidal vasculopathy. Graefes Arch Clin Exp Ophthalmol 243:837–839
Mukai R, Kishi S, Sato T, Watanabe G, Matsumoto H (2010) Protective effect of intravitreal bevacizumab and sub-tenon triamcinolone acetonide against occlusion of choriocapillaris induced by photodynamic therapy. Ophthalmologica 224:267–273
Lai TY, Lam CP, Luk FO, Chan RP, Chan WM, Liu DT, Lam DS (2010) Photodynamic therapy with or without intravitreal triamcinolone acetonide for symptomatic polypoidal choroidal vasculopathy. J Ocul Pharmacol Ther 26:91–95
Hirami Y, Tsujikawa A, Otani A, Yodoi Y, Aikawa H, Mandai M, Yoshimura N (2007) Hemorrhagic complications after photodynamic therapy for polypoidal choroidal vasculopathy. Retina 27:335–341
Papadopoulou DN, Mendrinos E, Mangioris G, Donati G, Pournaras CJ (2009) Intravitreal ranibizumab may induce retinal arteriolar vasoconstriction in patients with neovascular age-related macular degeneration. Ophthalmology 116:1755–1761
Koh A, Lee WK, Chen LJ, Chen SJ, Hashad Y, Kim H, Lai TY, Pilz S, Ruamviboonsuk P, Tokaji E, Weisberger A, Lim TH (2012) EVEREST STUDY: Efficacy and safety of verteporfin photodynamic therapy in combination with ranibizumab or alone versus ranibizumab monotherapy in patients with symptomatic macular polypoidal choroidal vasculopathy. Retina Mar 21 [Epub ahead of print]
Acknowledgments
This study was supported in part by the Japan Society for the Promotion of Science (JSPS), Tokyo, Japan (Grant-in-Aid for Scientific Research, no. 21592256), and by the Japan National Society for the Prevention of Blindness, Tokyo, Japan. We thank the following clinicians at Kyoto University Hospital for their assistance in gathering the treatment histories for our study: Hiroshi Tamura, MD, Hideo Nakanishi, MD, Hisako Hayashi, MD, Satoko Nakagawa, MD, Kohei Takayama, MD, Yumiko Ojima, MD, and Takahiro Horii, MD.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakata, I., Tsujikawa, A., Yamashiro, K. et al. Two-year outcome of photodynamic therapy combined with intravitreal injection of bevacizumab and triamcinolone acetonide for polypoidal choroidal vasculopathy. Graefes Arch Clin Exp Ophthalmol 251, 1073–1080 (2013). https://doi.org/10.1007/s00417-012-2137-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-012-2137-y