Abstract
Background
To evaluate the predictors of visual improvement using spectral-domain optical coherence tomography (SD-OCT) in eyes with resistant diabetic macular edema (DME) treated with pars plana vitrectomy.
Methods
Thirty-four eyes with resistant DME were evaluated in a retrospective manner. Several SD-OCT variables including photoreceptor inner segment/outer segment (IS/OS) junction, external limiting membrane (ELM) integrity, and central macular thickness (CMT) before and after the surgery, were evaluated by two experienced observers, masked to visual acuity. The visual improvement was used as the outcome measure for a stepwise regression, while the OCT factors were used as predictors.
Results
The strongest predictor of vision improvement was pre-operative damage to the ELM (p = 0.0277) compared to IS/OS junction (p = 0.03). Pretreatment central macular thickness was a very weak predictor (p = 0.18) of visual improvement. For each percentage increase of ELM integrity there was a 0.13 letter gain in vision. ELM integrity explained 16% of the visual acuity improvement after treatment, which rose to 21% with the addition of CMT. The addition of IS/OS junction in the model did not add predictive information.
Conclusions
Evaluation of ELM preoperatively predicts the vision improvement more accurately than the IS/OS junction and CMT in eyes with DME.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Diabetic macular edema (DME) is the most common cause of moderate vision loss in diabetics [1]. Various treatment modalities including laser photocoagulation [1], intravitreal steroids [2], and intravitreal anti-vascular endothelial growth factor (VEGF) drugs [3] have been reported to be useful in management of DME. Various reports have shown favorable effects of pars plana vitrectomy (PPV) for treating DME with or without obvious abnormalities of the vitreoretinal interface [4]. Even with seemingly successful treatment, the visual outcome may occasionally remain poor despite complete resolution of DME by PPV. Causes for this limited visual improvement include macular ischemia [5], photoreceptor dysfunction [6], and accumulated sub-foveal hard exudates [7].
Increased retinal thickness usually correlates well with vision decrease in eyes with DME. In our previous publication, we investigated spectral-domain optical coherence tomography (SD-OCT) in eyes with DME, and found a better correlation of the photoreceptor inner segment/outer segment (IS/OS) junction integrity with visual acuity than with central macular thickness (CMT) [8]. In the present study, our interest was to determine structural predictors of visual improvement in DME eyes, as seen on SD-OCT. We investigated this in a series of eyes undergoing vitrectomy for resistant DME. We performed a retrospective analysis of SD-OCT scans taken before and after surgery. Factors including IS/OS junction integrity, external limiting membrane (ELM) integrity, and CMT were investigated as potential predictive factors for vision improvement in eyes that underwent vitrectomy for non-responsive DME.
Patients and methods
For this retrospective study, we reviewed the medical records of 34 eyes of 34 consecutive patients with DME that were treated with pars plana vitrectomy at Jacobs Retina Center at Shiley Eye Center at the University of California, San Diego (UCSD) between July 2008 and December 2009. DME was diagnosed by fundus examination, and was confirmed by fluorescein angiography and spectral-domain optical coherence tomography (SD-OCT). All eyes had a reduction of central vision despite medical management including focal/grid laser (15 eyes) or intravitreal triamcinolone (four eyes), or had the primary traction pathology (15 eyes) as a cause for macular edema. Laser photocoagulation for DME was not performed within 500 microns from the fovea. All eyes had a follow-up of over 6 months, and good quality pre- and post-operative SD-OCT images, which permitted visualization of all structures of interest. Eyes with dense macular lipid deposits (seven eyes) were excluded due to poor visualization of the structures at the fovea. Eyes with macular ischemia on fluorescein angiography were also excluded. Informed consent was obtained from all participants. All procedures conformed to the Declaration of Helsinki for research involving human subjects, and were approved by the Institutional Review Board of the University of California, San Diego.
All the patients had a complete ophthalmologic examination including best-corrected Early Treatment Diabetic Retinopathy Study (ETDRS) visual acuity measurement, slit-lamp biomicroscopy, indirect fundus ophthalmoscopy, fluorescein angiography, and SD-OCT examination. All the patients had standard three-port 25G PPV by a single surgeon (WRF).
Imaging
DME was evaluated with scanning laser SD-OCT using a Heidelberg Spectralis instrument (Heidelberg Engineering, Vista, CA, USA). For each patient, horizontal and vertical SD OCT images through the fovea were obtained for evaluation. Two experienced observers (JC and JK) masked to visual acuities measured several variables. First, foveal thickness was calculated by using the calipers feature on the SD OCT instrument. An average thickness value was obtained after reviewing both scans.
Image grading
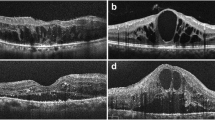
A disruption in IS/OS or ELM was defined as the loss of the back-reflection line. Analysis of the retinal structures was done as per our previous publication [8]. The IS/OS junction and ELM damage were measured as the percentage of disruption along the IS/OS and ELM layers, measured 500 microns in either direction from the foveal center. The percentage disruption was averaged to generate a number between 0% (no disruption) and 100% (total loss of the layer in both horizontal and vertical scans (Fig. 1). Reduced back-scattering from the ELM, IS/OS, and the other outer retinal layers, which included the outer plexiform layer, the outer nuclear layer, the retinal pigment epithelium, and the choroid, was regarded to be the result of a shadowing effect, not disruption.
Evaluation of external limiting membrane (ELM) (arrowhead) and inner segment/outer segment (IS/OS) (arrow) junction integrity in the central 1000 microns in eyes with resistant diabetic macular edema using spectral-domain optical coherence tomography (SD-OCT) a Both IS/OS junction (arrow) and ELM (arrowhead) are preserved with increase in retinal thickness and presence of epiretinal membrane (ERM). b IS/OS (arrow) junction and ELM (arrowhead) are disrupted (approximately 50%) with distorted foveal contour, intraretinal cyst and ERM. c ELM (arrowhead) appears intact in the central 1000 microns; however, IS/OS damage (arrow) (approximately 40%) is noted. Foveal contour is distorted with vitreomacular traction, and intraretinal cysts are seen. d Gross cystic edema with disruption of both IS/OS junction (arrow) and ELM (arrowhead), noted in the central 1000 microns
Surgical procedure
Standard three-port 25-gauge pars plana vitrectomy was performed using either Accurus Surgical Systems (Alcon Laboratories, Inc., Fort Worth, TX, USA) vitrectomy machines. The posterior vitreous adherent to the macula was removed using aspiration. In cases where visualization of the vitreous remnants was difficult, we used triamcinolone to stain the cortical vitreous. All triamcinolone particles were aspirated after complete removal of the posterior hyaloid. In addition to vitrectomy, removal of epiretinal membrane or vitreo-macular traction without internal limiting membrane peel was performed.
Statistical analysis
Visual acuity was recorded as the number of ETDRS letters correctly read by patient. The visual improvement, measured as the difference between pre-treatment and post-treatment, was used as the response variable for a stepwise regression. The pre-treatment central macular thickness and percentage of IS-OS and ELM disruption were used as predictors of post-operative visual acuity. The final model was selected based on the maximum R2 improvement and the lowest Mallow’s Cp value. All statistical analysis was performed using SAS software version 9.2 (SAS Institute, Cary, NC, USA).
Results
This case series included 34 eyes of 34 patients who underwent PPV for non-resolving DME with high quality SD-OCT images before and after the surgery. Twenty-six were men and eight were women, with a mean age of 76 years (range 51–84 years). Twenty eyes were pseudophakic and 14 were phakic with a cataract range of nuclear sclerosis (NS1-NS2) according to the Lens Opacities Classification System III (LOCS III) [9]. OCT findings included epiretinal membrane (27 eyes), vitreo-macular traction (five eyes), large intraretinal cysts (four eyes) and neurosensory detachment (three eyes) (Figs. 2 and 3). Mean follow-up period was 391 days (range 180–595 days). Comparison of anatomical and visual outcome in groups classified on the basis of integrity of IS/OS and ELM are shown in Table 1.
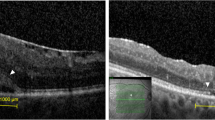
Case 1. Intact external limiting membrane (ELM) (arrowhead) and inner segment / outer segment (IS/OS) (arrow) junction. Pre-operative spectral-domain optical coherence tomography (SD-OCT) of a 68-year-old diabetic woman, with best-corrected visual acuity (BCVA) of 40 ETDRS letters (20/80), showing increased retinal thickness with the presence of vitreomacular traction and intraretinal cysts (a). IS/OS junction (arrow) and ELM (arrowhead) appears to be intact. This patient underwent vitrectomy for persistent diabetic macular edema and recovered normal foveal contour (b), and BCVA improved to 67 letters (20/25+2) at 6 months of follow-up
Case 2. Disrupted external limiting membrane (ELM) (arrowhead) and inner segment / outer segment (IS/OS) (arrow) junction at baseline. Pre-operative spectral-domain optical coherence tomography (SD-OCT) of a 72-year-old diabetic man, with BCVA of 20 ETDRS letters (20/200), SD-OCT showing increased retinal thickness with the presence of epiretinal membrane (ERM). IS/OS junction (arrow) and ELM (arrowhead) damage (100%) is seen in central 1,000 microns (a). This patient underwent vitrectomy for persistent diabetic macular edema and recovered normal foveal contour but with damaged outer retina (b), with BCVA of 23 letters (20/160-2) at 8 months of follow-up
A model incorporating CMT, IS/OS integrity, and ELM integrity showed that knowledge of ELM integrity explains the 16% (r 2 = 0.16, Cp = 1.82) of variability of visual improvement. This increases to 21% (r 2 =0.21, Cp = 2.03) (Fig. 4) if CMT is added, but adding IS/OS makes this model less efficient (r 2 = 0.21, Cp = 4.00) due to the co-linearity between ELM and IS/OS (r = 0.9, p < 0.0001). The analysis revealed that the percentage of ELM damage was a significant predictor of visual improvement (p = 0.0277) compared to IS/OS junction (p = 0.03). Pretreatment central macular thickness was a very weak predictor (p = 0.18) of visual improvement. Each percentage increase of ELM damage was associated with reduction of 0.13 letter improvement (Fig. 4), while pretreatment central macular thickness was only weakly associated with the visual improvement.
Visual acuity change in letters after {(+) = numbers of letters gained} versus external limiting membrane (ELM) disruption in percentage. Note that ELM disruption is associated with less vision improvement. Each percentage increase of ELM damage is associated with reduction of 0.13 letter improvement
Discussion
Studies have shown that macular edema and thickness is the only one factor among many to affect visual acuity in patients with diabetic macular edema [5, 10, 11]. Macular ischemia and foveal exudates also contribute to the poor prognosis [5]. Several prior studies using OCT had differing conclusions when correlating retinal thickness and visual acuity. The correlation coefficient between the retinal thickness and visual acuity was in a range of 0.28 to 0.73 [10, 11].Our current study showed a low correlation coefficient of 0.18 between retinal thickness and vision improvement. The reason for this poor correlation is likely to be the damage to the photoreceptor layer.
We have previously showed that IS/OS junction integrity is better correlated with visual acuity than CMT in a cross-sectional study [8]. It has been suggested that the integrity of the IS/OS layer could be an important predictor of vision in patients with retinitis pigmentosa, central serous chorioretinopathy, acute zonal occult outer retinopathy, branch retinal vein occlusion, and macular hole treated with vitrectomy [12–15]. Sakamoto et al. also showed a good correlation of photoreceptor layer integrity with visual acuity in a cross-sectional study of eyes with DME [16].
In our study, pre-operative ELM integrity was a better predictor for vision improvement than CMT or IS/OS junction integrity. The ELM is not a true membrane, but is formed by a tangentially oriented series of adhesions, zonulae adherens, connecting apical processes of the Muller cells with inner segments of the photoreceptors. This layer may help to maintain the alignment and orientation of the photoreceptors. The ELM also acts as a diffusion barrier for the passage of proteins from the subretinal space to inner retina. Thus, the ELM is an OCT landmark of integrity of inner segments of the photoreceptors. Our study has shown that the integrity of this structure is the best predictor of vision improvement in eyes undergoing surgery for non-responsive DME [17].
Evaluation of ELM is very difficult on time-domain OCT, and can be difficult on some spectral-domain OCT images. Wakabayashi et al. demonstrated the significance of the ELM by showing the better correlation of the ELM with visual acuity in eyes that were repaired for retinal detachment [18].Recently, Otani et al. showed a close relationship of the photoreceptor IS/OS junction and ELM integrity with the visual acuity in eyes with DME [7]. However, these studies did not evaluate these structures as predictive factors for visual improvement after treatment.
Severe retinal edema or large cystic changes may reduce the visibility of outer retinal structures, including the outer plexiform layer or retinal pigmented epithelium (RPE). These false negatives should not be considered disruptions, but rather a shadowing effect.
It is unlikely that the ERM peeling maneuver directly affects outer retinal structures, and in our surgical experience we do not see any direct damage to ELM and IS/OS junction. Wakayabashi et al. showed that the eyes with damage to both IS/OS and ELM preoperatively did not achieve restoration of these structures after macular hole repair [19]. We also believe that the restoration of outer retinal structures may not be possible due to permanent damage to ELM caused by chronic edema. However, we certainly believe that vitrectomy for causative factors for DME may prevent further damage to the outer retinal structures; however, preoperative status of the outer retinal structures would predict the visual outcome after surgery.
ELM integrity has been shown to have a better correlation with visual acuity than retinal thickness in eyes with cystoid macular edema secondary to venous occlusion, and in eyes with age-related macular degeneration [20, 21]. To the best of our knowledge, our study is the first to evaluate multiple OCT factors including CMT, IS/OS integrity, and ELM integrity as predictors of visual improvement after PPV in eyes with resistant DME. Knowledge of which DME eyes are most likely to respond to treatment may be helpful for clinicians to plan therapy and for clinical trials design.
In conclusion, preoperative status of ELM on SD-OCT predicts vision improvement more accurately than the IS/OS junction and CMT in eyes with DME. We certainly believe that vitrectomy would prevent further damage to the outer retinal structures; however, preoperative status of the outer retinal structures would predict the visual outcome after surgery. High-resolution OCT should be the integral part of DME management for the better assessment of the outer retinal structures, which are important prognostic markers.
References
Early Treatment Diabetic Retinopathy Study Research Group (1985) Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study Report number 1. Arch Ophthalmol 103(12):1796–1806
Martidis A, Duker JS, Greenberg PB, Rogers AH, Puliafito CA, Reichel E, Baumal C (2002) Intravitreal triamcinolone for refractory diabetic macular edema. Ophthalmology 109(5):920–927
Nicholson BP, Schachat AP (2010) A review of clinical trials of anti-VEGF agents for diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 248(7):915–930
Capone A Jr, Panozzo G (2000) Vitrectomy for refractory diabetic macular edema. Semin Ophthalmol 15(2):78–80
Shah SP, Patel M, Thomas D, Aldington S, Laidlaw DA (2006) Factors predicting outcome of vitrectomy for diabetic macular oedema: results of a prospective study. Br J Ophthalmol 90(1):33–36
Otani T, Kishi S (2002) A controlled study of vitrectomy for diabetic macular edema. Am J Ophthalmol 134(2):214–219
Otani T, Yamaguchi Y, Kishi S (2010) Correlation between visual acuity and foveal microstructural changes in diabetic macular edema. Retina 30(5):774–780
Maheshwary AS, Oster SF, Yuson RM, Cheng L, Mojana F, Freeman WR (2010) The association between percent disruption of the photoreceptor inner segment-outer segment junction and visual acuity in diabetic macular edema. Am J Ophthalmol 150(1):63–67, e61
Chylack LT Jr, Wolfe JK, Singer DM, Leske MC, Bullimore MA, Bailey IL, Friend J, McCarthy D, Wu SY (1993) The lens opacities classification system III. The Longitudinal Study of Cataract Study Group. Arch Ophthalmol 111(6):831–836
Blumenkranz MS, Haller JA, Kuppermann BD, Williams GA, Ip M, Davis M, Weinberg DV, Chou C, Whitcup SM (2010) Correlation of visual acuity and macular thickness measured by optical coherence tomography in patients with persistent macular edema. Retina 30(7):1090–1094
Browning DJ, Glassman AR, Aiello LP, Beck RW, Brown DM, Fong DS, Bressler NM, Danis RP, Kinyoun JL, Nguyen QD, Bhavsar AR, Gottlieb J, Pieramici DJ, Rauser ME, Apte RS, Lim JI, Miskala PH (2007) Relationship between optical coherence tomography-measured central retinal thickness and visual acuity in diabetic macular edema. Ophthalmology 114(3):525–536
Eandi CM, Chung JE, Cardillo-Piccolino F, Spaide RF (2005) Optical coherence tomography in unilateral resolved central serous chorioretinopathy. Retina 25(4):417–421
Murakami T, Tsujikawa A, Ohta M, Miyamoto K, Kita M, Watanabe D, Takagi H, Yoshimura N (2007) Photoreceptor status after resolved macular edema in branch retinal vein occlusion treated with tissue plasminogen activator. Am J Ophthalmol 143(1):171–173
Piccolino FC, de la Longrais RR, Ravera G, Eandi CM, Ventre L, Abdollahi A, Manea M (2005) The foveal photoreceptor layer and visual acuity loss in central serous chorioretinopathy. Am J Ophthalmol 139(1):87–99
Schocket LS, Witkin AJ, Fujimoto JG, Ko TH, Schuman JS, Rogers AH, Baumal C, Reichel E, Duker JS (2006) Ultrahigh-resolution optical coherence tomography in patients with decreased visual acuity after retinal detachment repair. Ophthalmology 113(4):666–672
Sakamoto A, Nishijima K, Kita M, Oh H, Tsujikawa A, Yoshimura N (2009) Association between foveal photoreceptor status and visual acuity after resolution of diabetic macular edema by pars plana vitrectomy. Graefes Arch Clin Exp Ophthalmol 247(10):1325–1330
Bunt-Milam AH, Saari JC, Klock IB, Garwin GG (1985) Zonulae adherentes pore size in the external limiting membrane of the rabbit retina. Invest Ophthalmol Vis Sci 26(10):1377–1380
Wakabayashi T, Oshima Y, Fujimoto H, Murakami Y, Sakaguchi H, Kusaka S, Tano Y (2009) Foveal microstructure and visual acuity after retinal detachment repair: imaging analysis by Fourier-domain optical coherence tomography. Ophthalmology 116(3):519–528
Wakabayashi T, Fujiwara M, Sakaguchi H, Kusaka S, Oshima Y (2010) Foveal microstructure and visual acuity in surgically closed macular holes: spectral-domain optical coherence tomographic analysis. Ophthalmology 117(9):1815–1824
Oishi A, Hata M, Shimozono M, Mandai M, Nishida A, Kurimoto Y (2010) The significance of external limiting membrane status for visual acuity in age-related macular degeneration. Am J Ophthalmol 150(1):27–32, e21
Yamaike N, Tsujikawa A, Ota M, Sakamoto A, Kotera Y, Kita M, Miyamoto K, Yoshimura N, Hangai M (2008) Three-dimensional imaging of cystoid macular edema in retinal vein occlusion. Ophthalmology 115(2):355–362, e352
Acknowledgement
This study was supported by an Unrestricted Research Fund to Jacobs Retina Center at Shiley Eye Center, University of California, San Diego (LC) and NIH EYO 7366 (WRF).
Financial disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors have full control of all primary data, and we agree to allow Graefe's Archive for Clinical and Experimental Ophthalmology to review their data upon request.
Rights and permissions
About this article
Cite this article
Chhablani, J.K., Kim, J.S., Cheng, L. et al. External limiting membrane as a predictor of visual improvement in diabetic macular edema after pars plana vitrectomy. Graefes Arch Clin Exp Ophthalmol 250, 1415–1420 (2012). https://doi.org/10.1007/s00417-012-1968-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-012-1968-x