Abstract
Objective
In this prospective, controlled, monocentric study, we described the clinical and neuroimaging 12-month follow-up of two parallel cohorts of subjects with idiopathic normal pressure hydrocephalus (iNPH), who did or did not undergo lumboperitoneal shunt (LPS).
Methods
We recruited 78 iNPH patients. At baseline, subjects underwent clinical and neuropsychological assessments, 3 T magnetic resonance imaging (MRI), and tap test. After baseline, 44 patients (LPS group) opted for LPS implantation, whereas 34 subjects (control group) declined surgery. Both cohorts were then followed up for 12 months through scheduled clinical and neuropsychological evaluations every 6 months. 3 T MRI was repeated at 12-month follow-up.
Results
Gait, balance, and urinary continence improved in the LPS group, without significant influence on cognitive functions. Conversely, gait and urinary continence worsened in the control group. No preoperative MRI parameter was significant outcome predictor after LPS. Of relevance, in responders to LPS, we found postoperative reduction of periventricular white matter (PWM) hyperintensities, which were instead increased in the control group.
Conclusions
LPS is safe and effective in iNPH. An early surgical treatment is desirable to prevent clinical worsening. Post-surgery decrease of PWM hyperintensities may be a useful MRI marker surrogate for clinical effectiveness of LPS.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Idiopathic normal pressure hydrocephalus (iNPH) is a syndrome affecting the elderly population and characterized by gait and balance disturbances, urinary incontinence, and cognitive impairment in the setting of ventriculomegaly and cerebrospinal fluid (CSF) opening pressure within the normal range [1, 2].
Ventriculoperitoneal shunt (VPS) is the current standard treatment for iNPH patients [3], leading to long-lasting clinical improvement [4]. While VPS is the most common surgical approach in North America and Europe [3], lumboperitoneal shunt (LPS) is increasingly performed in Japan [5,6,7]. Despite shorter surgery time and lower risks of intracranial complications (e.g. hemorrhages, seizures, and infections) compared to VPS [8], LPS is not widely performed and is not considered as the first-choice treatment for iNPH [5,6,7, 9, 10]. This may be due to associated higher failure rates and the possibility of symptomatic overdrainage, especially in the past [8].
Several studies reported the safety and non-inferiority of LPS compared to VPS in improving symptoms in iNPH patients [5,6,7, 9,10,11]. However, prospective, controlled trials are few and incomplete, as—for instance—they lack a standardized outcome assessment. Furthermore, to date, there are no studies evaluating preoperative magnetic resonance imaging (MRI) predictors of outcome after LPS as well as neuroimaging correlates of clinical improvement.
In this prospective, controlled, monocentric study, we first aimed to describe the clinical and neuroimaging 12-month follow-up of two parallel cohorts of iNPH patients, who did or did not undergo LPS as first-line surgical treatment. Second, we investigated preoperative MRI predictors of outcome after LPS.
Methods
We consecutively recruited 78 patients with probable iNPH, diagnosed according to the iNPH International Guidelines [1]. Subjects were enrolled at the “Parkinson's Disease and Movement Disorders Unit” of the Mondino Foundation in Pavia (Italy) from January 2016 to June 2018. At baseline, all patients underwent clinical and neuropsychological assessments, 3 T MRI scan, and tap test (i.e. large volume lumbar puncture). Gait, cognitive functions, balance, and urinary continence were evaluated with the iNPH Rating Scale (iNPHRS), where a value of 100 represents normal function and 0 the most severe level of dysfunction [12]. Gait was also assessed measuring the number of seconds and steps needed for ten-meter walking at free pace along a straight line, turning around, and walking back to the starting position, with a walking aid if usually adopted. The mean values of two consecutive video-recorded trials were used for the ten-meter seconds and steps. The neuropsychological evaluation also included the education-adjusted scores of the Mini Mental State Examination (MMSE) [13] and the Montreal Cognitive Assessment (MoCA) [14].
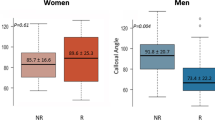
MRI scans (3 T Skyra, Siemens, Erlangen, Germany) were assessed for calculating the Evans’ index [15] on axial T1-weighted images and for investigating the following supportive features [1, 2]: small callosal angle [16] on coronal T1-weighted images at the level of the posterior commissure; aqueductal or fourth ventricular flow void [17] on sagittal T2-weighted images; disproportionately enlarged subarachnoid space hydrocephalus (DESH) [18], i.e. obliteration of the high-convexity sulci on axial T1-weighted images and dilation of the Sylvian fissures on coronal T1-weighted images. Periventricular (PWM) and deep white matter (DWM) hyperintensities on axial FLAIR images were assessed with the Fazekas scale [19].
A positive response to tap test [20], defined as at least 10% improvement of ten-meter seconds or steps, was evaluated in all subjects.
A surgical CSF diversion was offered to all patients but 34 subjects declined in favor of a “wait and see” approach given only mild impairment of activities of daily living and less often fear of surgery. These patients could reconsider shunt anytime but nobody opted for surgery throughout the follow-up period. Among the remaining 44 subjects, all patients with a negative response to tap test had a positive response to external lumbar drainage [21]. The 44 subjects accepting surgery underwent lumboperitoneal shunt (LPS) within 1 month after baseline at the “Neurosurgery Unit” of the San Matteo Foundation in Pavia, after exclusion of spinal canal stenosis or previous lumbar spine arthrodesis. In all cases, an adjustable pressure valve (Medtronic PS Medical® Strata® NSC LPS) was used. A one-step procedure was employed by placing the patient in the lateral position with valve housing in a lumbar pocket.
All patients who did (LPS group) or did not (control group) undergo surgery were then followed up for 12 months through scheduled clinical and neuropsychological evaluations every 6 months. 3 T MRI scan was repeated at 12-month follow-up.
Statistical analyses
Between-group comparisons were performed with Student’s t test or Wilcoxon rank-sum test for continuous variables when appropriate, while Pearson’s chi-square test was used for categorical variables. Spearman’s rank correlation coefficient was applied for bivariate analyses. Within-group follow-up differences were obtained with Wilcoxon signed-rank test for continuous variables and McNemar’s test for categorical variables. We also calculated the mean percentage rates of change at follow-up evaluations compared to the baseline as follows: [(b − a)/a] x 100, where ‘a’ is the mean baseline score and ‘b’ is the mean follow-up score of a clinical rating scale. In particular, responders to LPS experienced at least 10% improvement at follow-up evaluations compared to the baseline. Nominal univariate logistic regression analyses were performed to identify preoperative MRI parameters predicting outcome after LPS through log-likelihood chi-square test. With this regard, a positive outcome was defined as at least 10% improvement of iNPHRS total score after LPS at 12-month follow-up. A value of p < 0.05 was considered significant. Statistical analyses were performed using JMP Pro 14.0 (SAS Institute Inc., USA).
Results
Demographic, clinical, and instrumental findings of iNPH patients at baseline are listed in Table 1. Compared to control subjects, before surgery the LPS cohort showed more severe gait and balance impairment, more frequent and more pronounced urinary urgency/incontinence. Furthermore, LPS patients more often had a positive response to tap test and higher Fazekas scores (PWM and total).
Significant correlations between clinical and MRI features of iNPH patients at baseline are shown in Table 2. Of relevance, gait and balance impairment positively correlated with PWM hyperintensities. Urinary incontinence was instead positively associated with DWM hyperintensities. Conversely, no relationship between cognitive dysfunction and MRI findings was detected.
In the immediate post-surgery phase, four (9.1%) subjects complained of headache, which disappeared increasing the valve opening pressure. At the hospital discharge, the valve opening pressure was set to 1.5 (90–110 mmH2O) in 39 and 2.0 (145–165 mmH2O) in five patients. As a result of poor or absent improvement of motor performances within 1 month after surgery, the valve opening pressure was reduced in eight subjects, i.e. from 1.5 to 1.0 (35–55 mmH2O) in six and from 2.0 to 1.5 in two patients.
We reported no case of subdural hygroma or hematoma, shunt tube migration or obstruction, bowel perforation, meningitis or other infectious complications.
Within-group and between-group clinical follow-up differences are shown in Fig. 1 and Table 3, respectively. Responders to LPS are indicated in Table 3.
6-Month clinical follow-up
Improvement of gait and balance was showed in the LPS group. To a lesser extent, the urinary continence also improved in the LPS cohort. An increase of iNPHRS total score with LPS was thus noticed. Instead, no significant cognitive changes were recognized in the LPS group. None of the patients developed shunt-related complications.
No significant variations were noted in the control group.
12-Month clinical follow-up
Improvement of gait and balance persisted in the LPS group. However, a trend to slightly decreased effectiveness was noted in this cohort if compared to the 6-month follow-up. No significant cognitive changes with LPS were recognized similarly to the previous evaluation. Change of urinary continence was no longer significant in the within-group analysis of the LPS cohort. However, improvement of urinary continence with LPS persisted in the between-group comparison. Accordingly, a reduction of iNPHRS total score was noted in the LPS cohort compared to the 6-month follow-up (p = 0.0047). Nevertheless, an increase of this score persisted in LPS patients both at within-group and between-group analyses compared to the baseline. None of the subjects had shunt-related complications.
On the other hand, control patients showed worsening of gait and urinary continence. In particular, the decrease of iNPHRS continence score in the control cohort was also significant if compared to the 6-month follow-up (p = 0.0120). In contrast, balance and cognitive functions were not significantly different in the untreated cohort. Hence, a decrease of iNPHRS total score was showed in the control group compared to both the baseline and the 6-month follow-up (p = 0.0230).
12-Month MRI follow-up
Compared to the baseline, small callosal angle and DESH were less frequently detected in the LPS group, whereas they did not significantly change in control patients (Fig. 2). Of note, Fazekas score for PWM and total score were reduced in the LPS group and increased in control subjects (Fig. 2). Conversely, Evans’ index, flow void sign, and Fazekas score for DWM did not significantly change at follow-up in any of the cohorts (Fig. 2).
MRI parameters in iNPH patients who did or did not undergo LPS. Mean scores for continuous variables and frequency distribution for categorical variables are shown. Within-group follow-up differences compared to the baseline were calculated (*p < 0.05, **p < 0.01, ***p < 0.001). Vertical error bars represent standard errors
Interestingly, a decrease of Fazekas score for PWM and total score was identified in responders to LPS (Fig. 3). No other MRI parameters could significantly distinguish responders from not responders to LPS (Fig. 3).
MRI parameters in iNPH patients who did (Resp) or did not (No resp) respond to LPS. Mean scores for continuous variables and frequency distribution for categorical variables are shown. Within-group follow-up differences compared to the baseline were calculated (*p < 0.05, **p < 0.01, ***p < 0.001). Vertical error bars represent standard errors
Outcome prediction after lumboperitoneal shunt
No preoperative MRI parameter was significant outcome predictor after LPS.
Discussion
In our study, we showed the safety and effectiveness of LPS in improving gait, balance, and to a lesser extent urinary continence without any significant influence on cognitive functions in iNPH patients. The efficacy of LPS occurred within 6 months after surgery and persisted with a slight reduction at 12-month follow-up.
In keeping with previous studies [3, 11], gait and balance demonstrated the highest mean improvement rates. However, we noted a lower average improvement for urinary continence and cognitive functions. In this regard, controversy still exists on whether urinary and neuropsychological effectiveness can be achieved after surgery, especially in terms of reversibility of cognitive impairment [1, 3, 22].
A direct comparison between our findings and few previous reports concerning the efficacy of LPS in iNPH is not reliable given the different methods used to determine and quantify the outcome. To date, three retrospective, uncontrolled studies [5, 9, 10] and two prospective, controlled trials [6, 7] reported the effectiveness of LPS in iNPH. Among the retrospective investigations, only one study assessed each typical clinical domain through rating scales [5]. Both prospective trials instead evaluated iNPH patients with objective outcome measures [6, 7]. However, several studies used the modified Rankin Scale, which was developed for subjects with stroke providing a general disability measure, and ordinal or nominal rating scales for assessment of symptoms severity in iNPH [5,6,7, 10]. Quantitative outcome measures, such as the iNPHRS, should be employed to maximize accuracy and validity of results [12]. Unfortunately, there is still no agreement on a standardized, unbiased, and practical system of measuring outcome for research or clinical use in iNPH [2, 23]. Of note, we standardized mean rates of change and provided a consistent definition of responsivity or positive outcome for each clinical rating scale.
Among previous prospective trials, one study was controlled for the first three months (after which also the control group underwent LPS) [6], whereas a multicentric investigation used a previously conducted VPS cohort study as historical control [7]. In our study, we considered iNPH patients who declined surgery as control cohort, further describing their natural history of progressive worsening of gait and urinary continence at 12-month follow-up. This partially agrees with a Swedish study, in which shunt was inadvertently delayed in a cohort of iNPH subjects, who showed worsening of gait, balance, and cognitive functions in the meantime [24]. Other authors also stated the progression of symptoms in iNPH patients while waiting for surgery [25].
Regardless of the subsequent response, among patients who underwent LPS, we observed a decreased frequency of small callosal angle and DESH at 12-month follow-up compared to the baseline, whereas Evans’ index and flow void sign did not significantly change. Our results are in keeping with reports showing trend to normalization of callosal angle [26, 27], disappearance of DESH [18, 28], and no remarkable reduction of Evans’ index [29] after shunt. Conversely, few studies noticed a significant postoperative decrease of Evans’ index [26, 27], which is, however, commonly deemed as sign of overdrainage.
In our study, no baseline MRI parameter was significant outcome predictor. The lack of correlation between preoperative morphologic MRI markers and post-surgery outcome corroborates some studies [26, 29,30,31] and argues against other investigations that reported associations between postoperative improvement and baseline presence of small callosal angle [32], DESH [33], flow void sign [17], and white matter hyperintensities [28, 34].
Of interest, in responders to LPS, we found postoperative reduction of PWM hyperintensities without significant changes of DWM hyperintensities. This may be related to post-shunt improvement of gait and balance disturbances, which were positively associated with PWM hyperintensities at baseline. These findings may also partially explain the less noticeable improvement of continence and cognitive functions, which demonstrated no correlation with PWM hyperintensities at baseline. Control subjects instead showed an increase of PWM hyperintensities at 12-month follow-up, further corroborating the relationship between motor impairment and PWM involvement. In agreement with our results, several studies in iNPH reported reversible white matter lesions, mostly in periventricular regions, and their possible relationship with positive outcome after surgery [30, 34, 35]. Interestingly, acetazolamide was also able to reduce PWM hyperintensities and improve gait in iNPH, with a mechanism probably similar to the effect of shunt [36]. PWM hyperintensities can reflect transependymal resorption secondary to reverse flow of CSF or compression resulting from raised intraventricular pressure [37, 38]. Both these mechanisms can lead to increased diffusivity [38] and decreased perfusion in periventricular regions [39, 40]. Accordingly, reduced diffusivity [38, 40], increased perfusion [39, 40], and improvement of the axonal and myelin damage [41] may play a role in PWM changes after surgery. In particular, it is considered that the decreased compression of periventricular regions may be associated with gait improvement after shunt [42].
In our study, we acknowledge several limitations. First, our trial was not randomized. However, randomized controlled trials and sham surgery may be unethical in iNPH as a delayed surgery has significant long-term effects on functional outcome [24]. On the other hand, adopting patients unwilling to undergo shunt as control cohort is also biased as these subjects had milder impairment compared to LPS patients. Still, this notion strengthens our findings as we observed an improvement despite more severe impairment of gait, balance, and urinary continence in the LPS cohort at baseline. In this regard, a greater impairment might lead iNPH patients to choose surgery. Second, this study was not blinded, thus meaning that part of the reported outcome may be due to a placebo effect. Interestingly, however, the reversibility of an MRI parameter (i.e. PWM hyperintensities) and its correlation with the clinical outcome substantiate a real effect beyond placebo. Finally, our study has a rather short follow-up, especially given the recent controversy around the real nosological value of iNPH [23]. Nevertheless, our study duration is in keeping with most trials published so far [5,6,7, 9, 10].
In conclusion, our study suggests that LPS is safe and effective in improving symptoms, mainly gait disturbances and balance impairment in iNPH. Hence, this technique would deserve to be considered among the first-line treatments for iNPH, particularly for fragile patients unable to undergo intracranial procedures. An early shunt is desirable to prevent clinical worsening. Of note, post-surgery decrease of PWM hyperintensities may be a useful MRI marker surrogate for clinical effectiveness of LPS. Future studies with blinded evaluators, longer follow-ups, and possible direct comparisons with VPS are certainly warranted.
Data availability
Anonymized data will be shared upon appropriate request from any qualified investigator.
References
Relkin N, Marmarou A, Klinge P, Bergsneider M, Black PM (2005) Diagnosing idiopathic normal-pressure hydrocephalus. Neurosurgery 57:S4–16 discussion ii–v
Mori E, Ishikawa M, Kato T, Kazui H, Miyake H, Miyajima M, Nakajima M, Hashimoto M, Kuriyama N, Tokuda T, Ishii K, Kaijima M, Hirata Y, Saito M, Arai H, Japanese Society of Normal Pressure Hydrocephalus (2012) Guidelines for management of idiopathic normal pressure hydrocephalus: second edition. Neurol Med Chir 52:775–809
McGirt MJ, Woodworth G, Coon AL, Thomas G, Williams MA, Rigamonti D (2005) Diagnosis, treatment, and analysis of long-term outcomes in idiopathic normal-pressure hydrocephalus. Neurosurgery 57:699–705
Toma AK, Papadopoulos MC, Stapleton S, Kitchen ND, Watkins LD (2013) Systematic review of the outcome of shunt surgery in idiopathic normal-pressure hydrocephalus. Acta Neurochir 155:1977–1980
Nakajima M, Miyajima M, Ogino I, Sugano H, Akiba C, Domon N, Karagiozov KL, Arai H (2015) Use of external lumbar cerebrospinal fluid drainage and lumboperitoneal shunts with Strata NSC valves in idiopathic normal pressure hydrocephalus: a single-center experience. World Neurosurg 83:387–393
Kazui H, Miyajima M, Mori E, Ishikawa M, SINPHONI-2 Investigators (2015) Lumboperitoneal shunt surgery for idiopathic normal pressure hydrocephalus (SINPHONI-2): an open-label randomised trial. Lancet Neurol 14:585–594
Miyajima M, Kazui H, Mori E, Ishikawa M, on behalf of the SINPHONI-2 Investigators (2016) One-year outcome in patients with idiopathic normal-pressure hydrocephalus: comparison of lumboperitoneal shunt to ventriculoperitoneal shunt. J Neurosurg 125:1483–1492
Karabatsou K, Quigley G, Buxton N, Foy P, Mallucci C (2004) Lumboperitoneal shunts: are the complications acceptable? Acta Neurochir 146:1193–1197
Bloch O, McDermott MW (2012) Lumboperitoneal shunts for the treatment of normal pressure hydrocephalus. J Clin Neurosci 19:1107–1111
Bayar MA, Tekiner A, Celik H, Yilmaz A, Menekse G, Yildirim T, Alagoz F, Guvenc Y, Erdem Y (2018) Efficacy of lumboperitoneal shunting in patients with normal pressure hydrocephalus. Turk Neurosurg 28:62–66
Giordan E, Palandri G, Lanzino G, Murad MH, Elder BD (2018) Outcomes and complications of different surgical treatments for idiopathic normal pressure hydrocephalus: a systematic review and meta-analysis. J Neurosurg 131:1–13
Hellström P, Klinge P, Tans J, Wikkelsø C (2012) A new scale for assessment of severity and outcome in iNPH. Acta Neurol Scand 126:229–237
Folstein MF, Folstein SE, McHugh PR (1975) "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Nasreddine ZS, Phillips NA, Bédirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H (2005) The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53:695–699
Evans WA Jr (1942) An encephalographic ratio for estimating the size of the cerebral ventricles: further experience with serial observations. Am J Dis Child 64:820–830
Ishii K, Kanda T, Harada A, Miyamoto N, Kawaguchi T, Shimada K, Ohkawa S, Uemura T, Yoshikawa T, Mori E (2008) Clinical impact of the callosal angle in the diagnosis of idiopathic normal pressure hydrocephalus. Eur Radiol 18:2678–2683
Bradley WG Jr, Whittemore AR, Kortman KE, Watanabe AS, Homyak M, Teresi LM, Davis SJ (1991) Marked cerebrospinal fluid void: indicator of successful shunt in patients with suspected normal-pressure hydrocephalus. Radiology 178:459–466
Kitagaki H, Mori E, Ishii K, Yamaji S, Hirono N, Imamura T (1998) CSF spaces in idiopathic normal pressure hydrocephalus: morphology and volumetry. AJNR Am J Neuroradiol 19:1277–1284
Fazekas F, Chawluk JB, Alavi A, Hurtig HI, Zimmerman RA (1987) MR signal abnormalities at 1.5 T in Alzheimer's dementia and normal aging. AJR Am J Roentgenol 149:351–356
Virhammar J, Cesarini KG, Laurell K (2012) The CSF tap test in normal pressure hydrocephalus: evaluation time, reliability and the influence of pain. Eur J Neurol 19:271–276
Walchenbach R, Geiger E, Thomeer RT, Vanneste JA (2002) The value of temporary external lumbar CSF drainage in predicting the outcome of shunting on normal pressure hydrocephalus. J Neurol Neurosurg Psychiatry 72:503–506
Chaudhry P, Kharkar S, Heidler-Gary J, Hillis AE, Newhart M, Kleinman JT, Davis C, Rigamonti D, Wang P, Irani DN, Williams MA (2007) Characteristics and reversibility of dementia in normal pressure hydrocephalus. Behav Neurol 18:149–158
Espay AJ, Da Prat GA, Dwivedi AK, Rodriguez-Porcel F, Vaughan JE, Rosso M, Devoto JL, Duker AP, Masellis M, Smith CD, Mandybur GT, Merola A, Lang AE (2017) Deconstructing normal pressure hydrocephalus: ventriculomegaly as early sign of neurodegeneration. Ann Neurol 82:503–513
Andrén K, Wikkelsø C, Tisell M, Hellström P (2014) Natural course of idiopathic normal pressure hydrocephalus. J Neurol Neurosurg Psychiatry 85:806–810
Kameda M, Yamada S, Atsuchi M, Kimura T, Kazui H, Miyajima M, Mori E, Ishikawa M, Date I, SINPHONI, and SINPHONI-2 Investigators (2017) Cost-effectiveness analysis of shunt surgery for idiopathic normal pressure hydrocephalus based on the SINPHONI and SINPHONI-2 trials. Acta Neurochir 159:995–1003
Virhammar J, Laurell K, Cesarini KG, Larsson EM (2018) Increase in callosal angle and decrease in ventricular volume after shunt surgery in patients with idiopathic normal pressure hydrocephalus. J Neurosurg 130:130–135
Azuma S, Kazui H, Kanemoto H, Suzuki Y, Sato S, Suehiro T, Matsumoto T, Yoshiyama K, Kishima H, Shimosegawa E, Tanaka T, Ikeda M (2019) Cerebral blood flow and Alzheimer's disease-related biomarkers in cerebrospinal fluid in idiopathic normal pressure hydrocephalus. Psychogeriatrics 19:527–538
Wada T, Kazui H, Yamamoto D, Nomura K, Sugiyama H, Shimizu Y, Yoshida T, Yoshiyama K, Yamashita F, Kishima H, Yoshimine T, Takeda M (2013) Reversibility of brain morphology after shunt operations and preoperative clinical symptoms in patients with idiopathic normal pressure hydrocephalus. Psychogeriatrics 13:41–48
Meier U, Mutze S (2004) Correlation between decreased ventricular size and positive clinical outcome following shunt placement in patients with normal-pressure hydrocephalus. J Neurosurg 100:1036–1040
Tullberg M, Jensen C, Ekholm S, Wikkelsø C (2001) Normal pressure hydrocephalus: vascular white matter changes on MR images must not exclude patients from shunt surgery. AJNR Am J Neuroradiol 22:1665–1673
Agerskov S, Wallin M, Hellström P, Ziegelitz D, Wikkelsö C, Tullberg M (2019) Absence of disproportionately enlarged subarachnoid space hydrocephalus, a sharp callosal angle, or other morphologic MRI markers should not be used to exclude patients with idiopathic normal pressure hydrocephalus from shunt surgery. AJNR Am J Neuroradiol 40:74–79
Virhammar J, Laurell K, Cesarini KG, Larsson EM (2014) The callosal angle measured on MRI as a predictor of outcome in idiopathic normal-pressure hydrocephalus. J Neurosurg 120:178–184
Shinoda N, Hirai O, Hori S, Mikami K, Bando T, Shimo D, Kuroyama T, Kuramoto Y, Matsumoto M, Ueno Y (2017) Utility of MRI-based disproportionately enlarged subarachnoid space hydrocephalus scoring for predicting prognosis after surgery for idiopathic normal pressure hydrocephalus: clinical research. J Neurosurg 127:1436–1442
Tullberg M, Hultin L, Ekholm S, Månsson JE, Fredman P, Wikkelsø C (2002) White matter changes in normal pressure hydrocephalus and Binswanger disease: specificity, predictive value and correlations to axonal degeneration and demyelination. Acta Neurol Scand 105:417–426
Akiguchi I, Ishii M, Watanabe Y, Watanabe T, Kawasaki T, Yagi H, Shiino A, Shirakashi Y, Kawamoto Y (2008) Shunt-responsive parkinsonism and reversible white matter lesions in patients with idiopathic NPH. J Neurol 255:1392–1399
Alperin N, Oliu CJ, Bagci AM, Lee SH, Kovanlikaya I, Adams D, Katzen H, Ivkovic M, Heier L, Relkin N (2014) Low-dose acetazolamide reverses periventricular white matter hyperintensities in iNPH. Neurology 82:1347–1351
Ringstad G, Vatnehol SAS, Eide PK (2017) Glymphatic MRI in idiopathic normal pressure hydrocephalus. Brain 140:2691–2705
Keong NC, Pena A, Price SJ, Czosnyka M, Czosnyka Z, DeVito EE, Housden CR, Sahakian BJ, Pickard JD (2017) Diffusion tensor imaging profiles reveal specific neural tract distortion in normal pressure hydrocephalus. PLoS ONE 12:e0181624
Klinge PM, Brooks DJ, Samii A, Weckesser E, van den Hoff J, Fricke H, Brinker T, Knapp WH, Berding G (2008) Correlates of local cerebral blood flow (CBF) in normal pressure hydrocephalus patients before and after shunting–A retrospective analysis of [15O]H2O PET-CBF studies in 65 patients. Clin Neurol Neurosurg 110:369–375
Tuniz F, Vescovi MC, Bagatto D, Drigo D, De Colle MC, Maieron M, Skrap M (2017) The role of perfusion and diffusion MRI in the assessment of patients affected by probable idiopathic normal pressure hydrocephalus A cohort-prospective preliminary study. Fluids Barriers CNS 14:24
del Mar MM, Pueyo R, Poca MA, Falcón C, Mataró M, Bargalló N, Sahuquillo J, Junquéet C (2007) Post-surgical changes in brain metabolism detected by magnetic resonance spectroscopy in normal pressure hydrocephalus: results of a pilot study. J Neurol Neurosurg Psychiatry 78:760–763
Kanno S, Abe N, Saito M, Takagi M, Nishio Y, Hayashi A, Uchiyama M, Hanaki R, Kikuchi H, Hiraoka K, Yamasaki H, Iizuka O, Takeda A, Itoyama Y, Takahashi S, Mori E (2011) White matter involvement in idiopathic normal pressure hydrocephalus: a voxel-based diffusion tensor imaging study. J Neurol 258:1949–1957
Funding
This study was approved and funded by Italian Ministry of Health and co-funded by Health-Service of Lombardy (project code number RF-2013-02355908: “Idiopathic Normal Pressure Hydrocephalus (iNPH), parkinsonism and dementia: improving the accuracy of diagnosis and the patient care to reverse the symptomatology. Neurodegeneration, phenotypes and outcome measures”).
Author information
Authors and Affiliations
Contributions
Conception and design of the study: MT, CP. Data collection: MT, MP, PP, RZ, BM, PV, ER. Data analysis: MT. Interpretation of results: MT, CP. Writing the first draft of the manuscript: MT. Manuscript review and critique: AP, RC, NV, AF, CP.
Corresponding author
Ethics declarations
Conflicts of interest
AF provided consultancies to AbbVie, Medtronic, Boston Scientific, Sunovion, Chiesi Farmaceutici, UCB, and Ipsen; he is part of Advisory Boards for AbbVie, Boston Scientific, and Ipsen; he received honoraria from AbbVie, Medtronic, Boston Scientific, Sunovion, Chiesi Farmaceutici, UCB, and Ipsen as well as grants from University of Toronto, Weston Foundation, AbbVie, Medtronic, and Boston Scientific. Other authors declare that they have no conflict of interest.
Ethical approval
The study has been approved by the Ethics Committee of the IRCCS Mondino Foundation in Pavia (Italy) and has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
All patients gave written informed consent to all study procedures and to personal data processing for research purposes.
Rights and permissions
About this article
Cite this article
Todisco, M., Picascia, M., Pisano, P. et al. Lumboperitoneal shunt in idiopathic normal pressure hydrocephalus: a prospective controlled study. J Neurol 267, 2556–2566 (2020). https://doi.org/10.1007/s00415-020-09844-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-020-09844-x