Abstract
The neural correlates of stuttering are to date incompletely understood. Although the possible involvement of the basal ganglia, the cerebellum and certain parts of the cerebral cortex in this speech disorder has previously been reported, there are still not many studies investigating the role of white matter fibers in stuttering. Axonal stimulation during awake surgery provides a unique opportunity to study the functional role of structural connectivity. Here, our goal was to investigate the white matter tracts implicated in stuttering, by combining direct electrostimulation mapping and postoperative tractography imaging, with a special focus on the left frontal aslant tract. Eight patients with no preoperative stuttering underwent awake surgery for a left frontal low-grade glioma. Intraoperative cortical and axonal electrical mapping was used to interfere in speech processing and subsequently provoke stuttering. We further assessed the relationship between the subcortical sites leading to stuttering and the spatial course of the frontal aslant tract. All patients experienced intraoperative stuttering during axonal electrostimulation. On postsurgical tractographies, the subcortical distribution of stimulated sites matched the topographical position of the left frontal aslant tract. This white matter pathway was preserved during surgery, and no patients had postoperative stuttering. For the first time to our knowledge, by using direct axonal stimulation combined with postoperative tractography, we provide original data supporting a pivotal role of the left frontal aslant tract in stuttering. We propose that this speech disorder could be the result of a disconnection within a large-scale cortico-subcortical circuit subserving speech motor control.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stuttering is a speech disorder characterized by disruptions in speech motor behavior (blocks, repeated or prolonged articulatory and phonatory actions) that result in sound and syllable repetitions, sound prolongations and broken words [1–3]. Stuttering can be considered as a dynamic disturbance of motor control, even if there may be cases where stuttering is explained by purely cognitive/linguistic or emotional/psychological factors [4, 5]. Developmental stuttering starts in childhood without any neurological impairment, but with possible genetic derangements [5–7]. It persists in only 1 % of cases. Spontaneous recovery before adulthood is proposed to reveal the maturation of speech motor control mechanisms [8]. Acquired forms due to a neurological disease or as a result of emotional trauma may also occur in adults [9–12]. Neurogenic acquired stuttering may be distinguished from other types of dysfluencies, including developmental stuttering by a number of features, and notably the absence of secondary symptoms (such as eye blinking or face grimacing) and signs of anxiety, the absence of adaptation effect and the fact that repetitions, prolongations and blocks may occur in all kinds and positions of words, whatever the articulatory features, number and complexity of syllable are [13].
Neuroimaging and lesion studies on the neural correlates of stuttering have suggested the role of several brain areas, including the supplementary motor area (SMA), the bilateral sensorimotor cortices and auditory areas, the anterior cingulate cortex, the bilateral frontal operculae and the cerebellar vermis [4, 14–18]. In addition, many authors considered the main problem to be in the defective production of timing cues provided by the basal ganglia–thalamocortical circuit [5, 19, 20]. Indeed, lesions in thalamus and/or striatum may cause acquired stuttering [21]. Moreover, in persistent developmental stuttering, a correlation between severity of stuttering and activity in the basal ganglia has been demonstrated. It was also shown that this activity was modified by fluency shaping therapy through long-term therapy effects that reflect speech production improvement [22, 23].
For a long time, the white matter connectivity mediating this large-scale network has received less attention. Recent but still scattered evidence derived from diffusion imaging yet suggests a decreased connectivity in the speech network of stutterers, as testified by a reduced fractional anisotropy within the white matter tracts underlying the left sensorimotor cortex, the superior longitudinal fascicle, the left corticospinal tract and the cerebellar peduncles [18, 24–27]. However, diffusion imaging is only an indirect reflect of the actual functional connectivity, justifying the need of more direct evidence.
Here, we used intraoperative direct electrical stimulation (DES) during awake surgery for brain gliomas to investigate the anatomo-functional connectivity subserving stuttering. DES mimics a transient virtual lesion of the brain. If the patient produces incorrect responses during DES, both at cortical and subcortical levels, the surgeon leaves the region intact to preserve the patient’s cognitive and sensorimotor functions. The location of electrically inactivated subcortical structures can be precisely identified using postoperative diffusion tensor imaging. Therefore, DES enables to map the white matter tracts sustaining brain functions [28]. By this means, we were able to demonstrate that stimulation-induced disruption of the left frontal aslant tract (FAT) induced transitory stuttering. On the basis of this original finding, we suggest that this speech disorder could be the result of a disconnection within the large-scale cortico-subcortical circuit underlying speech motor control.
Patients and methods
Patients
Eight patients (5 females; age 34.7 ± 7.9 years) diagnosed with a diffuse low-grade glioma within the left frontal lobe were selected from September 2012 to September 2014. In all cases, they underwent maximal tumor resection under local anesthesia with an intraoperative functional mapping via electrostimulation. Sensorimotor and speech functions were systematically assessed during the procedure.
In accordance with our care protocol, all patients received a neurological examination before surgery, including the Karnofsky Performance Status (KPS) score. A language assessment was also performed by a trained speech therapist the day before, immediately after (between 3 and 5 days) and 3 months after surgery.
All patients gave informed consent to participating in the study.
Intraoperative functional mapping
The goal of WHO grade II glioma surgery is to maximize the extent of resection while preserving the integrity of eloquent networks by performing tumor removal according to the individual functional limits [29, 30]. In accordance with this approach, resections were performed under local anesthesia so that a cortical and subcortical mapping could be carried out using DES, as precisely described in previous works [31, 32]. A bipolar electrode with 5 mm spaced tips delivering a biphasic current (pulse frequency of 60 Hz, single pulse phase duration of 1 ms, amplitude from 1 to 4 mA—Nimbus*, Newmedic) was applied on the brain. DES has the transient effect (about 4 s) of a virtual lesion. Therefore, if the region stimulated is critical within a given functional network, the function sub-served by this cortical area or this white matter tract will be disturbed during a few seconds [28].
In practice, a wide craniotomy was performed under sedation. The tumor margins were verified in relation to the sulcal and gyral brain surface anatomy with ultrasonography. Letters tags marked the cortical boundaries of the glioma. Prior to tumor resection, a cortical mapping was achieved in awake condition. This electrical mapping was performed over the primary sensory-motor area and the ventral premotor cortex (during a combined counting and movement task) by progressively increasing the level of stimulation of 0.5 mA (from a baseline of 1 mA) until a functional response (movement disruption, dysesthesia, articulatory disturbances or speech arrest) was elicited—indicating the optimal threshold of stimulation [33].
Language mapping was performed using the DO naming test task [34], a French standardized naming test constituted of 80 black and white drawn pictures presented on a computer screen, and a semantic association task [35], a standardized visual non-verbal semantic test constituted of 52 black and white drawn pictures. A speech therapist and a neuropsychologist analyzed speech and language disorders induced during electrostimulation, i.e. speech arrest, anomia, articulatory troubles, phonemic paraphasia, semantic paraphasia, speech slowness, initiation troubles, perseveration or other speech disturbances, especially stuttering.
The patient and the speech therapist/neuropsychologist were unaware of the timing of electrostimulation application. A cortical site was considered positive for speech and/or language when any interference was met at three non-sequential stimulations followed by a return to a normal brain function after the end of stimulation. The same site was never stimulated twice successively to avoid seizures. All positive stimulation sites were marked with a tag number and a photograph before resection was taken capturing the cortical map.
After completion of cortical mapping, the glioma resection was started during which the subcortical structures, especially the white matter tracts, were systematically stimulated while the patient performed a dual task combining continuous picture naming with upper limb movement. Pyramid and palm tree test (PPTT) was also administrated to identify non-verbal semantic pathways [36]. The same electrical parameters were used at the subcortical level as the cortical one. The resection cavity was extended up to the connecting fascicles, with no margin, so that a maximal glioma resection was obtained, while preserving critical subcortical structures. After completion of the resection, a second photograph was taken with numbered tags marking subcortical positive sites [28].
Imaging acquisition and analysis
MRI scanning protocol
All patients underwent a brain MRI acquisition 3 months after surgery, including a 3D post-contrast T1-GRE sequence and a 3D FLAIR sequence using a 3.0 T magnet (Skyra, Siemens, Erlangen, Germany). The parameters were set as follows: TR = 5000 ms, TE = 384 ms, TI (inversion time) = 1800 ms, slice thickness = 0.898 mm, matrix = 256 × 256. A diffusion tensor imaging (DTI) was also acquired, consisting of diffusion-weighted echo planar imaging sequences (TR = 6700 ms, TE = 82 ms, FOV = 240 × 240 mm, matrix = 96 × 96, slice thickness = 2.5 mm with no gap, effective resolution; 2.5 mm3 isotropic voxels). Diffusion gradients were applied in 30 directions with b values of 0 and 1000 s/mm2.
Probabilistic tractography
For each subject, diffusion-weighted data were processed using FSL (University of Oxford’s Center for Functional Magnetic Resonance Imaging; Brain Software Library release 5.0) and its FMRIB’s diffusion toolbox (FDT v2.0, http://fsl.fmrib.ox.ac.uk/fsl/fslwiki/FDT). The processing included BET, DTIFIT and Bedpostx [37, 38]. Tracking was performed in subject space using FDT probtrackx with the default setting (5000 streamlines, step length = 0.5 mm, curvature threshold = 0.2) [37]. Region of interests (ROI) were defined to identify the frontal aslant tract: pars opercularis of the inferior frontal gyrus was selected as the seed region and pre-SMA as the target region on high-resolution 3D T1-weighted images using MRIcron software (http://www.mccauslandcenter.sc.edu/mricro/mricron). ROIs were manually delineated by following gyral and sulcal landmarks for each patient, according to coordinates mentioned in previous reports [39, 40]. The pre-SMA was manually defined as the area of the medial frontal cortex in the superior frontal gyrus lying dorsal to the cingulate sulcus and rostral to the VCA line [41, 42]. ROIs were adapted manually according to the shifts of brain parenchyma and surgical margins on the postoperative MRIs. We did not perform any control on the quality of these ROIs.
The voxels with the maximum connectivity value within the resulting connectivity distribution map of each participant were identified using MATLAB (the MathWorks, Natick, MA). The masks of these maps were then generated using a threshold of 5 % of the maximum connectivity value [43, 44].
Finally, T1 and resulting tracts were normalized in the MNI space according to the MNI-152 1 mm3 standard brain template given in FSL using FLIRT (12 parameters).
Volume of interests and the measurements associated with tracts
To determine the localizations of stimulation points according to surgical cavities, volume of interests (VOI) were created manually on 3D-T1 images for each patient. The intraoperative stimulation points related to stuttering were established on the basis of their spatial association with well-known functional and anatomical landmarks, viewable on the intraoperative pictures as previously reported [44, 45]. The stimulation points were plotted in each postoperative b = 0 diffusion-weighted images used in drawing the resection cavity. They were all overlaid with the VOIs of thresholded tracts and resection cavities on MNI-152 1 mm3 brain.
In summary, T1 was coregistered to the MNI-152 1 mm template using FLIRT while diffusion images were coregistered to the T1 using the b0 image. This processing allows to switch from T1 or diffusion space to MNI space.
As a next step, we measured the minimum and mean distances between the tracts and the stimulation points and also the surgical cavities to evaluate the spatial relationships of VOIs and the white matter fibers. We used an in-house software to measure the distances between cavities, stimulation points and thresholded tracts.
Anatomo-functional correlations
The exact location of the white matter fasciculi was determined using both the types of dysfunction during intraoperative electrostimulation mapping and anatomic correlation with postoperative MRI and tractography. Indeed, the control MRI examination carried out 3 months after surgery allowed us to analyze accurately the anatomical location of the eloquent pathways, i.e., by definition at the periphery of the cavity, where the resection was stopped according to the functional responses elicited by intraoperative subcortical stimulation (with no margin). It is worth noting that we have extensively used and validated this reliable and reproducible method in many previous studies—for a recent review, see [28].
Results
Patients
Patient’s sociodemographic and clinical characteristics are summarized in Table 1.
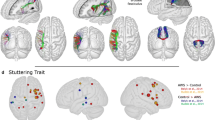
Seizure was the initial clinical symptom in all patients but one (incidental discovery). All patients had a Karnofsky Performance Status score of 90 or 100, with no functional deficit on the preoperative neurological examination. No patients had preoperative stuttering. The preoperative MRI localized the tumor within the SMA in four patients, the pre-SMA in three patients and in the posterior part of the middle frontal gyrus in one patient. A probability map of the gliomas before surgery is shown in Fig. 1.
On postoperative MRI, the median extent of resection was 96 %, with subtotal tumor removal in five cases, total removal in two cases, and a supratotal resection in one case (i.e. the postoperative resection volume was higher than preoperative tumor volume, see [46]). According to the intraoperative photos and postoperative MRIs, it was understood that the resections were extended posteromesially to SMA in five patients and up to precentral sulcus in three patients. Anterior part of the cingulate gyrus was resected in four cases. The posterolateral limit was the dorsolateral prefrontal cortex (DLPFC) in five patients and the inferior frontal sulcus in two cases.
In the immediate postoperative period, six patients experienced transient self-initiated difficulties of voluntary actions, namely “initiation disorders”, affecting speech with complete mutism. The eight patients resumed a normal life (KPS score of 90 or 100) within 3 months after surgery, with a complete recovery of speech in all cases but two, who still had slight speech initiation disorders. No patients experienced postoperative stuttering. Note that all patients had benefited from speech rehabilitation immediately after surgery.
Intraoperative findings
At the cortical level, DES over the ventral premotor cortex (vPMC), i.e. the lateral part of the precentral gyrus, elicited speech arrest in the eight patients. Primary motor area of face and hand situated mesially to vPMC were evoked in all cases, resulting in involuntary movement of face and right upper extremity, respectively. During dual task of counting and moving the upper extremity, complete arrest of movement and speech was noted on the posterior and mesial part of superior frontal gyrus in the eight patients, corresponding to cortical negative motor area. Anomia was generated in three patients by stimulating the DLPFC on the posterior part of the middle frontal gyrus and in one patient by stimulating the pars opercularis of the inferior frontal gyrus. Finally, a semantic paraphasia was observed in one patient during DES of the mid-part of the middle frontal gyrus. In addition, stimulation of DLPFC induced disturbances during the PPTT in one patient. No stuttering has been evoked cortically.
At the subcortical level, resection was pursued posteriorly until negative motor responses were found in all cases. DES elicited interruption of movement of the upper limb in the eight patients, associated with speech arrest in three cases. Moreover, semantic paraphasia and/or anomia and/or PPTT disorders have been generated in seven patients by stimulating the lateral part of the resection cavity, corresponding to the inferior fronto-occipital fascicle (IFOF)—as we previously reported [31, 36, 47]. In addition, stimulation of the head of the caudate nucleus provoked verbal perseverations in four patients.
The main result is that subcortical DES of the fibers running in the posterior wall of the resection cavity (and running in the medial and inferior part of the cavity in one case) elicited part-word stuttering during the naming test in all patients. From a behavioral standpoint, this stuttering was characterized by repetitions, prolongations and/or blocks during the production of the target word (picture name). Stuttering moments were neither related to articulatory features (i.e. stuttering could occur as well in “peacock” as in “snake” or “lemon”) nor related to number or complexity level of syllables. There were no secondary symptoms such as eye blinking or facial grimacing, but the production was visibly effortful. Each of these neuropsychological manifestations was analyzed and recorded by an experienced appraiser (i.e., the speech therapist or the neuropsychologist). The white matter tract generating stuttering when stimulated has been surgically preserved in the eight patients.
Tractography
On the postoperative tractographies, the left FAT was observed as a white matter tract connecting pars opercularis to pre-SMA in five, and to anterior SMA proper in three cases (cases 3, 4, 7). Tract continuity was preserved in all cases, thanks to subcortical functional mapping during surgical resection. Figure 2 shows the tract reconstruction of all the subjects.
The stimulation points, which caused stuttering during surgery, overlapped with FAT in all patients (Fig. 3). All the tracts were located posterior to the surgical cavities except one tract that runs medially and inferiorly to the resection cavity. The tracts coincided with the stimulation points in seven cases and had a close relationship (min. distance 1.3 mm.) in one case. The mean value of minimal distances between the left FAT and the surgical cavity was 0.96 mm. (range 0–4.9 mm). The minimal distances from left FAT to stimulation points had a mean value of 0.1 (range 0–1.3 mm).
Illustrative cases
Case 1 (patient 7) (Fig. 4)
A 48-year-old right-handed man without any neurological deficits was evaluated for epileptic seizures. The MRI revealed a diffuse tumor involving the left mesial premotor area. The patient underwent awake surgery with intraoperative electrical cortical DES over the vPMC elicited speech arrest. The primary motor cortex of hand and face was also detected by stimulating the precentral gyrus more medially. The tumor was removed up to the precentral sulcus posteriorly and up to the inferior frontal sulcus laterally. Verbal perseveration was elicited by electrostimulation of the deep part of the cavity, near the head of the caudate. Anomia during DO80 test was induced by DES of the lateral part of the surgical cavity, corresponding to the IFOF. The patient also interrupted movement of his right arm during stimulation of the posterolateral edge of the cavity, corresponding to the “negative motor network” [45]. In addition, stuttering (repetition and prolongation of the first syllable of words during picture naming) was elicited by subcortical electrostimulation of the fibers coming from anterior SMA (according to the postoperative tractography, see Fig. 3), on the posterior edge of the cavity, close to the sites generating arrest of upper limb movement. The surgical resection was stopped according to these functional boundaries, by preserving all fascicles. The patient experienced a complete mutism in the immediate postoperative period. He was discharged without any sensory or motor deficits on the fifth day following surgery, he benefited from speech rehabilitation, and he completely recovered within the 3 months after resection. He never had postoperative stuttering.
Illustrative case 1. The preoperative FLAIR and T1-weighted MR images demonstrating a left frontal diffuse lesion invading SMA and pre-SMA (a, b). The intraoperative picture of brain mapping (c). Tags 2 and 3: vPMC (arrest of speech on the lateral side of premotor cortex), tags 4 and 1: primary motor areas of hand and face (involuntary movements of hand and face). Tag 47: head of the caudate nucleus (perseveration), tag 48: IFOF (anomia), tag 49: negative motor network (interruption of the movement of right arm during dual task), tag 50: FAT (repetition of the first syllable of the words during picture naming)
Case 2 (patient 6) (Fig. 5)
A 34-year-old right-handed female patient experienced epileptic seizure leading to the discovery of a left frontal glioma on MRI. During the awake surgery, DES allowed the detection of cortical eloquent areas, i.e. the vPMC (speech arrest), the primary motor cortex of face and upper limb in the central gyrus, and a site within the DLPFC which generated PPTT disturbances during stimulation. Subcortically, DES of the IFOF (running in the posterolateral wall of the surgical cavity up to the DLPFC) elicited semantic disorders, during both D080 test and PPTT. Posteriorly, subcortical stimulation induced negative motor responses of the right upper limb. Posteromedially, stuttering (either blocks or syllable repetition) was evoked by stimulation of fibers connecting the frontomesial cortex to the inferior frontal gyrus, thus corresponding to the FAT, as shown by postoperative tractography (see Fig. 2). The patient had mutism immediately after surgery, with no sensorimotor deficit. She completely recovered within the next weeks, thanks to speech rehabilitation. She never experienced postoperative stuttering.
Illustrative case 2. The preoperative cranial MRI revealing a diffuse glioma in the premotor area (a, b). Intraoperative image after resection of the tumor showing the cortical and subcortical stimulation areas (c). Cortical eloquent areas which were detected by direct electrical stimulation—tag 6: vPMC (arrest of speech fluency), tag 1: negative motor area (arrest of facial movement), tag 2: primary motor area of face (involuntary contraction on right side of the face), tags 3, 4, 5: primary motor areas of hand and right upper extremity (involuntary movements of right hand and upper limb), tag 7: dorsolateral prefrontal cortex (perturbation during PPTT). Subcortical stimulation findings—tags 48 and 49: pyramidal fibers (involuntary dystonic movements of right upper extremity), tag 40: IFOF (comprehension problems during PPTT), tags 42 and 43: negative motor response, tag 50: FAT (stuttering during picture naming test)
Discussion
To the best of our knowledge, it is the first study to use intraoperative axonal mapping combined to postoperative tractography to investigate the white matter correlates of stuttering. Interestingly, Penfield and Welch have previously observed stuttering during cortical stimulation of the SMA [48]. Later, Ojemann and Ward reported part word stuttering by stimulating the thalamus [49]. More recently, cases of stuttering have been described following implantation of stimulating electrodes into basal ganglia nuclei for other indications [50, 51]. These preliminary electrophysiological data already pled in favor of a wide cortico-subcortical circuit underpinning stuttering. However, to date, no electrostimulation mapping of white matter tracts has been performed to study the subcortical pathways implicated in stuttering.
Our findings provide direct support for a key role of the left frontal aslant tract in stuttering. The characteristics of the dysfluencies observed in our study are those of acquired neurogenic stuttering (versus developmental stuttering) as underlined in recent studies (for a review, see [13]), namely: dysfluencies consist in either repetitions, prolongations or blocks and do not depend on articulatory features, the patient does not appear anxious about the stuttering behavior, but his production seems to be effortful; there are no secondary symptoms such as eye blinking or facial grimacing.
Using postmortem dissections and tractography, the FAT has been described as a frontal white matter bundle connecting the SMA proper and the pre-SMA to the posterior inferior frontal gyrus, in particular pars opercularis [52–55]. While its functional role is still poorly known, it was recently proposed that degeneration of the FAT in primary progressive aphasia might account for impairment in verbal fluency [56]. Furthermore, Kronfeld-Duenias et al. has shown diffusion properties of FAT in people with persistent developmental stuttering in a recent DTI study [57]. More recently, our team as well as Vassal et al. demonstrated the implication of the left FAT on speech control using intraoperative axonal mapping [44, 58]. However, the language deficit which was elicited by DES of FAT in these studies was speech arrest, that is, a complete blockage of articulation (anarthria, i.e. the loss of the motor ability to speak, with no sound, no movement of the face) and not a stuttering (syllable repetition which do not depend on articulatory features, as defined above) as observed in our present study.
It is already known that lesions of SMA proper and pre-SMA may lead to speech disturbances affecting both fluency and intonation [59]. Indeed, besides the role of SMA proper in the execution of speech, pre-SMA is involved in supramotor actions such as word selection, planning and coordination of movement sequences [60–63]. SMA is part of the basal ganglia–thalamocortical circuit, which is presumed to be responsible for the temporal configuration of internally driven sequential movements [64]. In a recent investigation, Lu et al. hypothesized that uncontrolled hyperactivity in basal ganglia–thalamus–pre-SMA circuit could partly explain for difficulties in selection and sequencing of speech movements in stutterers [20]. This is in agreement with previous electrophysiological studies which showed that SMA stimulation may elicit part word repetition [48] and that stimulation of the left ventrolateral thalamus may generate syllable repetition [49]. In addition, the left ventral premotor cortex and posterior inferior frontal gyrus contain cells encoding for learned speech sounds [65]. Interestingly, it seems that SMA operates initiation of planned speech actions [66]. Therefore, it has been suggested that such a role in speech control was made possible owing to the connections between SMA/pre-SMA and pars opercularis/pars triangularis [59, 67].
In the current study, stuttering was elicited during DES of the left FAT, as confirmed by probabilistic tractography. Indeed, we evidenced close spatial relationships between the intraoperative stimulation points and the FAT reconstructed on postsurgical diffusion tensor imaging maps. In addition, six patients experienced transient speech initiation disturbances, eventually complete mutism, immediately following surgery, supporting that the resection came into the contact of the FAT—as already demonstrated [44]. On the basis of these data, we hypothesize that this speech disorder could be explained by a disconnection between SMA/pre-SMA and the posterior part of the inferior frontal gyrus due to a transitory dysfunction of the FAT. This would be in agreement with previous research suggesting that stuttering may be a problem of disruption between articulation and movement control [20]. Therefore, we could propose that FAT plays a role in control of timing and planning of utterances as well as phonological sequencing.
Our study suffers from certain limitations. Besides the small number of patients, our series concerned only the left hemisphere. Therefore, we cannot rule out a possible role of the right FAT in stuttering. However, it is worth noting that this pathway is left lateralized in most right-handed healthy subjects [54, 68] and that we did not elicit speech disorders during DES of the right FAT in our previous stimulation study which included tumors in both hemispheres [44]. Furthermore, previous neuroimaging investigation revealed that hyper-activation of the right hemisphere in stuttering was probably due to compensatory changes and that primary dysfunction was circumscribed to the left hemisphere [69].
Another issue that should be addressed is the lack of postoperative stuttering in our series of patients in spite of the onset of other kind of speech disorders in the immediate postsurgical period, related to a transient SMA syndrome. Interestingly, the pre-SMA and SMA proper sub-loops interconnected with different areas of globus pallidus and thalamus have already been proposed for basal ganglia–thalamocortical pathway [70]. In the same vein, we could suggest the existence of different fiber layers in the FAT, with distinct effects on speech production immediately following resective surgery. Further dissection, imaging and stimulation studies are needed to validate this hypothesis.
Conclusion
Despite the small number of patients, this is the first study combining intraoperative subcortical DES and postoperative tractography that supports the involvement of the left FAT in stuttering. We might suggest that the left FAT could convey signals for timing and motor control of fluent speech, and that its disruption may generate stuttering. Therefore, we propose that this speech disorder could be the result of a disconnection within a large-scale cortico-subcortical network subserving speech motor control, including the left FAT, in addition to the previously reported basal ganglia–thalamocortical circuit.
References
Andrews G, Craig A, Feyer AM, Hoddinott S, Howie P, Neilson M (1983) Stuttering: a review of research findings and theories circa 1982. J Speech Hear Disord 48:226–246
Espir MLE, Rose FC (1970) The basic neurology of speech. Blackwell Scientific Publications, Oxford
Max L, Guenther FH, Gracco VL, Ghosh SS, Wallace ME (2004) Unstable or insufficiently activated internal models and feedback-biased motor control as sources of dysfluency: a theoretical model of stuttering. Contemp Issues Commun Sci Disord 31:105–122
Craig-McQuaide A, Akram H, Zrinzo L, Tripoliti E (2014) A review of brain circuitries involved in stuttering. Front Hum Neurosci 884:1–20
Ludlow CL, Loucks T (2003) Stuttering: a dynamic motor control disorder. J Fluency Disord 28:273–295
Bloodstein O (1995) A handbook on stuttering, 5th edn. Singular, San Diego
Riaz N, Steinberg S, Ahmad J, Pluzhnikov A, Riazuddin S, Cox NJ, Drayna D (2005) Genome wide significant linkage to stuttering on chromosome 12. Am J Hum Genet 76:647–651
Forster DC, Webster WG (2001) Speech-motor control and interhemispheric relations in recovered and persistent stuttering. Dev Neuropsychol 19:125–145
Doi M, Nakayasu H, Soda T, Shimoda K, Ito A, Nakashima K (2003) Brainstem infarction presenting with neurogenic stuttering. Int Med 42:884–887
Dworkin JP, Culatta RA, Abkarian GG, Meleca RJ (2002) Laryngeal anesthetization for the treatment of acquired disfluency: a case study. J Fluency Disord 27:215–225
Iverach L, Rapee RM (2014) Social anxiety disorder and stuttering: current status and future directions. J Fluency Disord 40:69–82
Lim EC, Wilder-Smith E, Ong BK, Seet RC (2005) Adult-onset re-emergent stuttering as a presentation of Parkinson’s disease. Ann Acad Med Singapore 34:579–581
Lundgren K, Helm-Estabrooks N, Klein R (2010) Stuttering following acquired brain damage: a review of the literature. J Neurolinguistics 23:447–454
Brown S, Ingham RJ, Ingham JC, Laird AR, Fox PT (2005) Stuttered and fluent speech production: an ALE meta-analysis of functional neuroimaging studies. Hum Brain Mapp 25:105–117
Cykowski MD, Kochunov PV, Ingham RJ, Ingham JC, Mangin JF, Rivière D, Lancaster JL, Fox PT (2008) Perisylvian sulcal morphology and cerebral asymmetry patterns in adults who stutter. Cereb Cortex 18:571–583
Foundas AL, Bollich AM, Corey DM, Hurley M, Heilman KM (2001) Anomalous anatomy of speech-language areas in adults with persistent developmental stuttering. Neurology 57:207–215
Jäncke L, Hänggi J, Steinmetz H (2004) Morphological brain differences between adult stutterers and non-stutterers. BMC Neurol 4:23
Watkins KE, Smith SM, Davis S, Howell P (2008) Structural and functional abnormalities of the motor system in developmental stuttering. Brain 131:50–59
Alm P (2004) Stuttering and the basal ganglia circuits: a critical review of possible relations. J Commun Disord 37:325–369
Lu C, Peng D, Chen C, Ning N, Ding G, Li K, Yang Y, Lin C (2010) Altered effective connectivity and anomalous anatomy in the basal ganglia-thalamocortical circuit of stuttering speakers. Cortex 46:49–67
Carluer L, Marié RM, Lambert J, Defer GL, Coskun O, Rossa Y (2000) Acquired and persistent stuttering as the main symptom of striatal infarction. Mov Disord 15:343–346
Giraud AL, Neumann K, Bachoud-Levi AC, von Gudenberg AW, Euler HA, Lanfermann H, Preibisch C (2008) Severity of dysfluency correlates with basal ganglia activity in persistent developmental stuttering. Brain Lang 104:190–199
Neumann K, Euler HA, von Gudenberg AW, Giraud AL, Lanfermann H, Gall V, Preibisch C (2003) The nature and treatment of stuttering as revealed by fMRI A within- and between-group comparison. J Fluency Disord 28:381–409
Chang SE, Erickson KI, Ambrose NG, Hasegawa-Johnson MA, Ludlow CL (2008) Brain anatomy differences in childhood stuttering. Neuroimage 39:1333–1344
Chang SE, Zhu DC, Choo AL, Angstadt M (2015) White matter neuroanatomical differences in young children who stutter. Brain 138:694–711
Connally EL, Ward D, Howell P, Watkins KE (2014) Disrupted white matter in language and motor tracts in developmental stuttering. Brain Lang 131:25–35
Sommer M, Koch MA, Paulus W, Weiller C, Büchel C (2002) Disconnection of speech-relevant brain areas in persistent developmental stuttering. Lancet 360:380–383
Duffau H (2015) Stimulation mapping of white matter tracts to study brain functional connectivity. Nat Rev Neurol 11:255–265
Duffau H (2012) The challenge to remove diffuse low-grade gliomas while preserving brain functions. Acta Neurochir 154:569–574
Duffau H, Taillandier L (2015) New concepts in the management of diffuse low-grade glioma: proposal of a multistage and individualized therapeutic approach. Neuro-Oncology 17:332–342
Duffau H, Gatignol P, Mandonnet E, Peruzzi P, Tzourio-Mazoyer N, Capelle L (2005) New insights into the anatomo-functional connectivity of the semantic system: a study using cortico-subcortical stimulations. Brain 128:797–810
Duffau H, Gatignol P, Mandonnet E, Capelle L, Taillandier L (2008) Intraoperative subcortical stimulation mapping of language pathways in a consecutive series of 115 patients with Grade II glioma in the left dominant hemisphere. J Neurosurg 109:461–471
Fernández Coello A, Moritz-Gasser S, Martino J, Martinoni M, Matsuda R, Duffau H (2013) Selection of intraoperative tasks for awake mapping based on relationships between tumor location and functional networks. J Neurosurg 119:1380–1394
Metz-Lutz MN, Kremin H, Deloche G (1991) Standardisation d’un test de dénomination orale: contrôle des effets de l’âge, du sexe et du niveau de scolarité chez les sujets adultes normaux. Rev Neuropsychol 1:73–95
Howard D, Patterson K (1992) The pyramid and palm trees test. Thames Valley Test Company, Bury StEdmunds
Moritz-Gasser S, Herbet G, Duffau H (2013) Mapping the connectivity underlying multimodal (verbal and non-verbal) semantic processing: a brain electrostimulation study. Neuropsychologia 51:1814–1822
Behrens TE, Berg HJ, Jbabdi S, Rushworth MF, Woolrich MW (2007) Probabilistic diffusion tractography with multiple fiber orientations: what can we gain? Neuroimage 34:144–155
Smith SM (2002) Fast robust automated brain extraction. Hum Brain Mapp 17:143–155
Amunts K, Weiss PH, Mohlberg H, Pieperhoff P, Eickhoff S, Gurd JM, Marshall JC, Shah NJ, Fink GR, Zilles K (2004) Analysis of neural mechanisms underlying verbal fluency in cytoarchitectonically defined stereotaxic space–the roles of Brodmann areas 44 and 45. Neuroimage 22:42–56
Kaplan E, Naeser MA, Martin PI, Ho M, Wang Y, Baker E, Pascual-Leone A (2010) Horizontal portion of arcuate fasciculus fibers track to pars opercularis, not pars triangularis, in right and left hemispheres: a DTI study. Neuroimage 52:436–444
Picard N, Strick PL (2001) Imaging the premotor areas. Curr Opin Neurobiol 11:663–672
Rizzolatti G, Luppino G, Matelli M (1996) The classic supplementary motor area is formed by two independent areas. Adv Neurol 70:45–56
Bennett IJ, Motes MA, Rao NK, Rypma B (2012) White matter tract integrity predicts visual search performance in young and older adults. Neurobiol Aging 33:433.e21–e31
Kinoshita M, de Champfleur NM, Deverdun J, Moritz-Gasser S, Herbet G, Duffau H (2015) Role of fronto-striatal tract and frontal aslant tract in movement and speech: an axonal mapping study. Brain Struct Funct 220:3399–3412
Schucht P, Moritz-Gasser S, Herbet G, Raabe A, Duffau H (2013) Subcortical electrostimulation to identify network subserving motor control. Hum Brain Mapp 4:3023–3030
Yordanova Y, Moritz-Gasser S, Duffau H (2011) Awake surgery for WHO Grade II gliomas within “noneloquent” areas in the left dominant hemisphere: toward a “supratotal” resection. J Neurosurg 115:232–239
Duffau H (2014) The huge plastic potential of adult brain and the role of connectomics: new insights provided by serial mappings in glioma surgery. Cortex 58:325–337
Penfield W, Welch K (1951) The supplementary motor area of the cerebral cortex: a clinical and experimental study. Arch Neurol Psychiatry 66:289–317
Ojemann GA, Ward AA Jr (1971) Speech representation in ventrolateral thalamus. Brain 94:669–680
Moretti R, Torre P, Antonello RM, Capus L, Gioulis M, Zambito Marsala S, Cazzato G, Bava A (2003) Speech initiation hesitation’ following subthalamic nucleus stimulation in a patient with Parkinson’s disease. Eur Neurol 49:251–253
Nebel A, Reese R, Deuschl G, Mehdorn HM, Volkmann J (2009) Acquired stuttering after pallidal deep brain stimulation for dystonia. J Neural Transm 116:167–169
Lawes IN, Barrick TR, Murugam V, Spierings N, Evans DR, Song M, Clark CA (2008) Atlas-based segmentation of white matter tracts of the human brain using diffusion tensor tractography and comparison with classical dissection. Neuroimage 39:62–79
Ford A, McGregor KM, Case K, Crosson B, White KD (2010) Structural connectivity of Broca’s area and medial frontal cortex. Neuroimage 52:1230–1237
Catani M, Dell’acqua F, Vergani F, Malik F, Hodge H, Roy P, Valabregue R, Thiebaut de Schotten M (2012) Short frontal lobe connections of the human brain. Cortex 48:273–291
Oishi K, Zilles K, Amunts K, Faria A, Jiang H, Li X, Akhter K, Hua K, Woods R, Toga AW, Pike GB, Rosa-Neto P, Evans A, Zhang J, Huang H, Miller MI, van Zijl PC, Mazziotta J, Mori S (2008) Human brain white matter atlas: identification and assignment of common anatomical structures in superficial white matter. NeuroImage 43:447–457
Catani M, Mesulam MM, Jakobsen E, Malik F, Martersteck A, Wieneke C, Thompson CK, Thiebaut de Schotten M, Dell’Acqua F, Weintraub S, Rogalski E (2013) A novel frontal pathway underlies verbal fluency in primary progressive aphasia. Brain 136:2619–2628
Kronfeld-Duenias V, Amir O, Ezrati-Vinacour R, Civier O, Ben-Shachar M (2014) The frontal aslant tract underlies speech fluency in persistent developmental stuttering. Brain Struct Funct. doi:10.1007/s00429-014-0912-8
Vassal F, Boutet C, Lemaire JJ, Nuti C (2014) New insights into the functional significance of the frontal aslant tract—an anatomo-functional study using intraoperative electrical stimulations combined with diffusion tensor imaging-based fiber tracking. Br J Neurosurg 28:685–687
Ackermann H, Riecker A (2011) The contribution(s) of the insula to speech production: a review of the clinical and functional imaging literature. Brain Struct Funct 214:419e433
Fridriksson J, Moser D, Ryalls J, Bonilha L, Rorden C, Baylis G (2009) Modulation of frontal lobe speech areas associated with the production and perception of speech movements. J Speech Lang Hear Res 52:812–819
Ikeda A, Yazawa S, Kunieda T, Ohara S, Terada K, Mikuni N, Nagamine T, Taki W, Kimura J, Shibasaki H (1999) Cognitive motor control in human pre-supplementary motor area studied by subdural recording of discrimination/selection-related potentials. Brain 122:915–931
Ridderinkhof KR, Ullsperger M, Crone EA, Nieuwenhuis S (2004) The role of the medial frontal cortex in cognitive control. Science 306:443–447
Zentgraf K, Stark R, Reiser M, Kunzell S, Schienle A, Kirsch P, Walter B, Vaitl D, Munzert J (2005) Differential activation of pre-SMA and SMA proper during action observation: effects of instructions. Neuroimage 26:662–672
Cunnington R, Bradshaw JL, Iansek R (1996) The role of the supplementary motor area in the control of voluntary movement. Hum Mov Sci 15:627–647
Guenther FH, Ghosh SS, Tourville JA (2006) Neural modeling and imaging of the cortical interactions underlying syllable production. Brain Lang 96:280–301
Bohland JW, Bullock D, Guenther FH (2010) Neural representations and mechanisms for the performance of simple speech sequences. J Cogn Neurosci 22:1504–1529
Papoutsi M, de Zwart JA, Jansma JM, Pickering MJ, Bednar JA, Horwitz B (2009) From phonemes to articulatory codes: an fMRI study of the role of Broca’s area in speech production. Cereb Cortex 19:2156–2165
Kinoshita M, Shinohara H, Hori O, Ozaki N, Ueda F, Nakada M, Hamada J, Hayashi Y (2012) Association fibers connecting the Broca center and the lateral superior frontal gyrus: a microsurgical and tractographic anatomy. J Neurosurg 116:323–330
Braun AR, Varga M, Stager S, Schulz G, Selbie S, Maisog JM, Carson RE, Ludlow CL (1997) Altered patterns of cerebral activity during speech and language production in developmental stuttering. An H2(15)O positron emission tomography study. Brain 120:761–784
Nakano K, Kayahara T, Tsutsumi T, Ushiro H (2000) Neural circuits and functional organization of the striatum. J Neurol 247(Suppl 5):V1–V15
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical standards
This study has been approved by the appropriate ethics committee and has therefore been performed in the accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Rights and permissions
About this article
Cite this article
Kemerdere, R., de Champfleur, N.M., Deverdun, J. et al. Role of the left frontal aslant tract in stuttering: a brain stimulation and tractographic study. J Neurol 263, 157–167 (2016). https://doi.org/10.1007/s00415-015-7949-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-015-7949-3