Abstract
Objective
To analyze the interrelation between radiation dose and radiation-induced nasopharyngeal ulcer (RINU) in locoregional recurrent nasopharyngeal carcinoma (NPC) treated with intensity-modulated radiation therapy (IMRT).
Methods
Clinical data were collected from 363 patients with locoregional recurrent NPC who received re-irradiated with definitive IMRT from 2009 to 2017. Twenty-nine patients were diagnosed with RINU. Univariate and multivariate analyses were used to re-evaluate the first and second radiotherapy plans and to identify predictive dosimetric factors.
Results
All dosimetric parameters were notably associated with the progression to RINU (p < 0.01) using paired samples Wilcoxon signed rank tests. Multivariate analysis showed that EQD2_ \(\sum\)D80 (dose for 80 percent volume of the unilateral nasopharynx lesion) was an independent prognostic factor for RINU (p = 0.001). The area under the ROC curve for EQD2_ \(\sum\)D80 was 0.846 (p < 0.001), and the cutoff point of 137.035 Gy could potentially be the dose tolerance of the nasopharyngeal mucosa.
Conclusions
The sum of equivalent dose in 2 Gy fractions (EQD2) in the overlapping volumes between initial and re-irradiated nasopharyngeal mucosal tissue can be effective in predicting the hazard of developing RINU in NPC patients undergoing radical re‑irradiation with IMRT and we propose a EQD2_ \(\sum\)D80 threshold of 137.035 Gy for the nasopharynx.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nasopharyngeal carcinoma (NPC) is one of the high-occurrence cancer among head-and-neck malignancies in Southeast Asia [1]. With the advancement of radiotherapy technology and the renewal of equipment, intensity-modulated radiotherapy (IMRT) has become the main treatment for NPC. Due to the high conformity of tumor coverage as well as the better sparing of normal structure in the IMRT era, the incidence rate of locoregional recurrence and radiotherapy-related toxicities has dropped significantly [2]. However, locoregional relapse rates of NPC patients treated with IMRT were reported, at around 4.8–13% [3,4,5,6]. Considering that patients with locoregional recurrent NPC could still obtain survival benefit by aggressive salvage therapy, high-dose re-irradiation has gained more acceptance and become the major treatment option over the last decade [7,8,9].
The major deficiency of re-irradiation for locoregional recurrent NPC in the IMRT era remains the concern for severe radiation-induced toxicity, including extensive fibrosis, soft tissue necrosis, osteoradionecrosis, etc. [7]. Radiation-induced nasopharyngeal ulcer (RINU) is defined as an ulceration of the surrounding and adjacent structures of the nasopharynx, such as the mucosa, musculus longus capitis, parapharyngeal tissues, and cranial base, which have been irradiated months or years previously [10]. Invasion of carotid sheath might be life-threatening, especially if internal carotid artery is affected. It is essential to obtain more accurate dose constraints for specific structures to avoid RINU in locoregional recurrent NPC patients re-irradiated with IMRT.
The aim of this study is to analyze the clinical characteristics and dose-volume effect of RINU after re-irradiated with IMRT for locoregional recurrent nasopharyngeal carcinoma. This will help to determine how the radiation dosages to the primary and relapse regions affect the occurrence of RINU and to provide a reference for optimizing the re-irradiation treatment plan.
Materials and methods
Patient selection and pretreatment evaluation
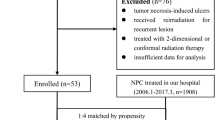
Between February 2009 and December 2017, the medical records of 363 consecutive locoregional recurrent NPC patients receiving IMRT according to our institutional treatment protocol at Fudan University Shanghai Cancer Center were reviewed. Inclusion criteria were (1) aged ≥ 18 years, (2) both primary treatment and retreatment were performed in our cancer center using IMRT with a prescribed dose ≥ 60 Gy, (3) no less than 12 months since the previous treatment, (4) met the diagnosis criteria of RINU after re-irradiation. Exclusion criteria were (1) brachytherapy as part of first or second treatment, (2) developed RINU before re-irradiation. Among the 363 locoregional recurrent NPC patients, 29 patients developed RINU after re-irradiation. However, six patients were excluded due to missing data (n = 3) or receiving intracavitary brachytherapy (n = 3), leaving 23 evaluable patients.
All patients underwent a pretreatment evaluation, including medical history, physical examination, fiberoptic nasopharyngoscopy, complete blood counts, serum biochemistry profile, contrast-enhanced MRI of the head and neck, chest CT scan, abdominal ultrasound. 18F-fluorodeoxyglucose positron emission tomography (18F-FDG-PET) and whole-body bone scan using single-photon emission CT were performed when clinically indicated. For recurrent lesions, thirteen patients were diagnosed by pathological biopsy, and ten patients were diagnosed by at least two types of imaging techniques. Clinical stage was classified in accordance with the eighth edition of the American Joint Committee on Cancer (AJCC) staging system. This retrospective study was approved by the institutional review board of Fudan University Shanghai Cancer Center.
Intensity-modulated radiotherapy
All patients were immobilized in the supine position with thermoplastic masks from head to shoulder. For radiation treatment planning, contrast-enhanced CT was performed at 3–5 mm slice intervals. T1-weighted images from gadolinium-enhanced MRI were fused with the planning CT images for target delineation. All patients underwent radical IMRT with 6 MV photons and completed the prescribed radiation therapy as scheduled.
For the first radiation treatment, the total prescription dose was 66–72.6 Gy to the planning target volume (PTV) of the gross tumor volume of nasopharynx and enlarged retropharyngeal nodes (GTVnx), 66–72.6 Gy to the PTV of the gross tumor volume of the involved positive cervical lymph nodes (GTVnd), 60–63 Gy to the PTV of the clinical target volume 1 (CTV1), and 54–59 Gy to the PTV of the clinical target volume 2 (CTV2). The CTV1 was defined as the high-risk region of microscopic extension which encompassed the GTV plus a margin of 5–10 mm, the entire nasopharynx, retropharyngeal lymph nodal regions, skull base, parapharyngeal space, inferior sphenoid sinus, pterygoid fossae, clivus, the posterior third of the nasal cavity and maxillary sinuses, and any high-risk nodal regions. The CTV2 included the low-risk sites of microscopic extension. The levels of cervical lymphatic drainage that were included were dependent on the tumor sites and nodal staging. The PTV was created by extending the CTV with a margin of 3–5 mm in all orientations. Nevertheless, when the CTV was close to critical structures, the margin of the PTV would be created as small as 1 mm. The dose per fraction was 2–2.2 Gy. Radiotherapy was given over 1 fraction a day, 5 days a week.
Re-irradiation was given to the locoregional recurrent sites, both target volumes and prescription doses were personalized, taking into account the initial radiation dose, interval time between the first and second radiotherapy, normal tissue constraints, and the clinical manifestation of radiation-related toxicities in the previous irradiated tissues. The prescription dosage was 60–70 Gy in 30–35 fractions at 2–2.1 Gy per fraction.
The organs at risk (OAR) included the temporal lobes, brainstem, spinal cord, optic chiasm, pituitary, optic nerves, eyeballs, lens, inner ears, parotid glands, temporomandibular joints, oral cavity, mandible, larynx, thyroid, trachea. The dose constraints for the OAR have been assessed in accordance with the protocols of the Radiation Therapy Oncology Group (RTOG) 0225 and RTOG 0615.
Chemotherapy
During the initial treatment, 95.7% (22/23) patients received chemotherapy: 5 received neoadjuvant chemotherapy, 4 received concurrent chemotherapy, 11 received induction + concurrent chemotherapy, and 2 received induction + adjuvant chemotherapy. 73.9% (17/23) patients were treated with chemotherapy after their recurrence, eight patients underwent neoadjuvant chemotherapy, one patient received adjuvant chemotherapy, seven patients were performed with neoadjuvant + synchronized chemotherapy, and one patient received chemotherapy before and after radiotherapy. The regimens prescribed for induction and adjuvant chemotherapy consisted of PF (cisplatin 25 mg/m2 /day, days 1–3, 5-fluorouracil 0.5 g/m2 /day, days 1–3), TPF (docetaxel 60 mg/m2 /day, day 1, cisplatin 25 mg/m2 /day, days 1–3, and 5-fluorouracil 0.5 g/m2/day, days 1–3), TP (docetaxel 60 mg/m2 /day, day 1, cisplatin 25 mg/m2/day, days 1–3), and GP (gemcitabine 1 g/m2/day, day1, day 8, cisplatin 25 mg/m2/day, days 1–3). The induction and adjuvant chemotherapy regimens were given at 3-week intervals. The regimens for concurrent chemotherapy were as follows: cisplatin alone, nedaplatin alone, or Tegafur, Gimeracil and Oteracil Porassium Capsules. Epidermal growth factor receptor (EGFR: Nimotuzumab or Cetuximab) was given weekly to two patients during the first course of radiotherapy and to three patients during the second course of radiation therapy.
Diagnostic criterion for RINU
RINU was diagnosed in accordance with patients’ clinical symptom, physical signs, and MRI imaging manifestations. MRI imaging features of RINU were reported by our colleagues in the previous study [10], which included inconsecutive nasopharyngeal mucosal line and/or a specific region of low signal intensity on contrast-enhanced T1-weighted images. Figure 1 shows defects in the nasopharyngeal wall on MRI scans of a typical by a patient in our cohort who suffered from the RINU. Two radiologists with expertise in head-and-neck cancers reassessed each MRI scan individually in accordance with the criteria. Any distinction was settled by mutual agreement. 29 patients were diagnosed with nasopharyngeal ulcer and met the diagnostic criteria for RINU. At the time of diagnosis and follow-up MRI, all patients were clinically assessed.
MRI image of a 41-year-old man with rT3N1M0. A Coronal contrast-enhanced, T1-weighted MRI showed a large ulcer on the left posterior wall of the nasopharynx at 2 months after the completion of re-irradiation. B An abnormal nasopharyngeal cavity and soft tissue defects were seen on transverse contrast-enhanced T1-weighted MRI. C Sagittal T1-weighted contrast-enhanced MRI manifested the mucosal ulcer eroding the roof of the nasopharynx
Dose registration
The T1-weighted images of enhanced MRI which including the area and extent of RINU were fused with the planning CT images of the initial and the second course of radiotherapy. Original IMRT plans were reintegrated into the TPS. The volumes of ulcerous side and the corresponding unaffected contralateral nasopharynx were delineated respectively. The sum of the equivalent dose in 2 Gy fractions (EQD2) in the overlapping volumes between initial and re-irradiated nasopharyngeal mucosal tissue of ulcer side and non-ulcer side was regarded as the variable in dosimetric analysis. EQD2 was calculated at α/β = 3 Gy, and the calculation formula of EQD2 was D[d + (α/β)]/[2 + (α/β)], where D = total dose and d = fractional dose. Dose-volume histogram (DVH) curves were exported from the original treatment plans on Pinnacle3 version 8.0 m (Philips Corp, Fitchburg, WI) TPS. Dose parameters encompassing the mean dose (EQD2_ \(\sum\)Dmean), maximum dose (EQD2_ \(\sum\)Dmax), minimum dose (EQD2_ \(\sum\)Dmin), absolute volume irradiated at n Gy (EQD2_ \(\sum\)Vn), dose over n volume (EQD2 _ \(\sum\)Dncc), and dose over n percent volume (EQD2_ \(\sum\)Dn) were calculated from the DVH curves.
Follow-up and statistical analysis
After completion of initial radiotherapy, all patients were required to attend our outpatient clinic for follow-up every 3 months for the first 2 years, every 6 months between years 3 and 5, and annually thereafter. Each follow-up visit included a detailed medical history and examination, indirect nasopharyngoscope, blood routine examination, and blood biochemistry. Contrast-enhanced MRI of the nasopharynx, chest CT, ultrasound of the cervical lymphatic drainage area and abdomen were performed 3 months after the completion of IMRT and every 6–12 months thenceforward. If there was any evidence of locoregional recurrence or distant metastases, further investigations would be ordered.
Follow-up time was counted from the date of completion of the initial IMRT. SPSS software version 21.0 (IBM, Armonk, NY, USA) was used for data analysis. Ulcer-free interval time (UFI) was defined as the time elapsed between the completion of re-irradiation with IMRT and the date of diagnosis of RINU. Time to relapse (TTR) was the interval from the start of initial therapy to the point of locoregional failure. Locoregional relapse-free survival (LRRFS) was defined as the time elapsed between diagnosis of the first locoregional recurrence and the next locoregional relapse. Locoregional recurrence was diagnosed based on contrast-enhanced MRI of the nasopharynx, with or without PET/CT, nasopharyngofiberoscope, and biopsy. Overall survival (OS) was defined as the time from diagnosis of locoregional recurrence to death from any cause or date of last follow-up for surviving patients.
Actuarial rates for OS and LRRFS were generated via Kaplan–Meier method. Paired sample Wilcoxon signed rank tests were used to compare the dosimetric parameters in the paired contralateral nasopharyngeal mucosa (total dose in EQD2), considering the differences between the paired samples by normality tests were failed to obey the normal distribution. Receiver operating characteristics (ROC) curve was performed to figure out the cut-off point for significant dosimetric parameters. Univariate and multivariate analysis (consisting of relevant dosimetric parameters in the paired- sample Wilcoxon signed rank tests) were performed using the Cox proportional hazards model. Two-tailed p values within 0.05 were regarded as statistically significant.
Results
Patient characteristics
Twenty-three patients who met the criteria for unilateral RINU after re-irradiation were enrolled in this study. There were 20 males and 3 females among the 23 patients. All patients had histologically confirmed nasopharyngeal carcinoma (WHO type II/III). The median age was 49 years old, with a range of 38–68 years old. Median TTR was 33 months (range 14–101 months). The recurrent lesion was ipsilateral to the primary lesion in 18 patients. The median ulcer-free interval was 10 months. In conformity with the eighth edition of AJCC staging system, nine patients were rT3, four were rT4. None of these 23 patients received nasopharyngeal boost. At the completion of initial treatment, all patients achieved a complete response (CR). Table 1 summarizes patients’ characteristics.
Dosimetry analysis
Significant association between the dosimetric parameters and RINU manifested itself in the paired samples Wilcoxon signed rank tests (Table 2). Univariate COX regression analysis illustrated that the dosimetric parameters EQD2_ \(\sum\)Dmean, EQD2_ \(\sum\)Dmin, EQD2_ \(\sum\)Vn (n = 20,30,40,50,60,70,75), EQD2_ \(\sum\)Dncc (n = 0.5,1,2,3,4,5), and EQD2_ \(\sum\)Dn (n = 1,5,10,20,30,40,50,60,70,80,95) were remarkably associated with the progression of RINU. By contrast, the EQD2_ \(\sum\)Dmax (p = 0.053) was not pronouncedly correlated with RINU. All the dosimetric parameters that showed significant statistical differences in the single-factor analysis were included in the multivariate analysis (Table 3). The development of RINU was conspicuously related to the EQD2_ \(\sum\) D80 (β = 0.140, SE = 0.042, HR = 1.150, 95% CI = [1.059–1.249], p = 0.001), demonstrating that EQD2_ \(\sum\)D80 is an independent prognostic factor for UFI in patients with locoregional recurrent NPC. The dose tolerance of the overlapping volume between the initial and retreatment target volumes of the nasopharynx was deduced using ROC curve analysis, to be specific, the optimum cut-off points for EQD2_ \(\sum\)D80. The area under the ROC curve for EQD2_ \(\sum\)D80 was 0.846 (p < 0.001). As shown in Fig. 2, the EQD2_ \(\sum\) D80 of 137.035 Gy might be reckoned as the upper limit of the cumulative dose for initial and re-irradiation of the nasopharynx (sensitivity = 0.739, specificity = 0.913). The mean EQD2_ \(\sum\) D80 for ulcer side was 141.46 Gy, and 126.67 Gy for non-ulcerated side.
ROC curve for the EQD2_ \(\sum\) D80 (dose of 80 percentage volume). The cut-off point for EQD2_ \(\sum\) D80 (as the tolerated dose in the nasopharyngeal mucosa) was determined to be 137.035 Gy for recurrent NPC patients treated with IMRT. At an EQD2_ \(\sum\) D80 of 137.035 Gy, the sensitivity and specificity for predicting RINU were 0.739 and 0.913, respectively
Toxicities associated with re-irradiation
Acute adverse events: 7 patients (30.43%) complained of ≥ Grade III mucositis, and grade I or II mucositis was observed in 16 patients (69.57%). Nineteen patients (82.61%) suffered from grade I radiation dermatitis, and 4 patients (17.39%) developed grade II radiodermatitis.
Late adverse events: There was no subcutaneous fibrosis, visual loss or spinal cord or brainstem damage in our series. Two patients (8.7%) had grade II hearing deficit, 5 patients (21.74%) had grade II trismus, 3 patients (13.04%) had grade I or II cranial neuropathy, 3 patients had grade I or II temporal lobe necrosis, 23 patients (100%) had grade IV nasopharyngeal mucositis, and 7 patients (30.43%%) had grade III cephalalgia.
Survival
The median follow-up time was 85 months (range 6–112 months). Twenty-three patients had died when follow-up ceased. Of these, 8 (34.78%) deaths were due to sudden massive nasal bleeding, 12 (52.17%) patients died of cachexia and multiple organ dysfunction, and 3 (13.05%) patients died of tumor progression. The median OS was 29 months, with 1-year, 2-year, 3-year, and 5-year OS rates of 87%, 58.7%, 40.7%, and 16.5%, respectively. The 3-year and 5-year LRRFS were 80.6% and 64.7%, respectively.
Discussion
RINU can occur as a serious complication during or after radiotherapy in NPC patients. In the era of IMRT, how to reduce the incidence of RINU and improve the quality of life of NPC patients remains an important question. The current study provides a unique insight into the direction in which future improvements can be made. In this study, we found that accumulated dose to the nasopharynx was an independent predictor of RINU in patients with locoregional recurrent NPC treated with two courses of IMRT. Therefore, we recommended dose limitation to the nasopharyngeal mucosa to prevent the development of RINU.
The IMRT technique can provide optimized dose delivery to the tumor targets and the surrounding critical organs compared to 3D conformal radiotherapy. Re-irradiation of NPC recurrence using IMRT is clinically profitable and has been performed in several studies [11,12,13,14]. In the study by Hsiung et al. [15], lower doses to normal tissues and more homogeneous target doses can be achieved with IMRT schedules compared to 3D-CRT for the boost or salvage treatment of NPC. Qiu et al. [13] reported their treatment results of high-dose re-irradiation with IMRT for locally recurrent NPC in 70 patients. The 2-year LRRFS, disease-free survival (DFS), and OS rates were 65.8%, 65.8%, and 67.4%, respectively. In the study from Kong et al. [11], the 3-year local recurrence-free survival (LRFS) and OS of recurrent NPC patients re-irradiated with IMRT were 85.1 and 46.0%, respectively. In the current study, the 3-year LRRFS and OS rates were 80.6% and 40.6%, respectively, which were similar to those in other studies.
As local control and survival rates have improved, quality of life (QOL) has increasingly been a priority. Radiation-related late complications are detrimental factors that negatively affect QOL. As reported by Chan et al. [7], even though the use of IMRT, the risk of any grade ≥ 3 complications was exceeding 70% among patients re-irradiated for locally recurrent NPC. These late toxic reactions were inevitable since the tumor was encapsulated, embedded or surrounded by the normal structures. Radiation-induced nasopharyngeal ulcer is one of the serious complications after radical radiotherapy. Based on previous studies, the crude incidence rate of radiation-induced nasopharyngeal necrosis in NPC patients first irradiated with IMRT was 1.9–3.3% [16, 17], while 11–40.5% of patients developed grade 3–5 late adverse events after re-irradiation of recurrent NPC with IMRT, which led to RINU [11, 18, 19]. Nearly half of all patients who suffered from RINU may develop life-threatening massive nasopharyngeal hemorrhage. In the data of Kong et al. [11], a total of 184 recurrent NPC patients who underwent re-irradiation with IMRT died at the time of analysis, and 44 of them died of profuse epistaxis. In the study of Hua and colleagues [20], approximately 30% of patients who received IMRT for recurrent NPC died from severe nasal hemorrhage owing to mucosal necrosis. In our study, 34.78% of the patients died due to massive bleeding. All the above results enlighten us that there is a predominant need to study the mucosal tolerance, and to devise a way to prevent such adverse effects.
Even though the exact mechanism of RINU is unascertained, a conjecture accepted broadly is that high radiation dosage may elicit hypovascular–hypoxic–hypocellular condition which may result in the breakdown of tissues and a chronically non-healing lesion [21]. Moreover, nutritional deficiency and infection have been reported to play an important role in the development of nasopharyngeal ulcer [16, 22]. MRI, nasopharyngoscopy, and pathohistological examination are advantageous for the diagnosis of RINU. Compared with the recurrence of NPC, the MRI images of RINU reveal the defect of soft tissues around the nasopharynx, low signal on T1-weighted images, high or slightly high signal on T2-weighted images, and mild or no enhancement when performing the contrast-enhanced MRI [23]. Nasopharyngoscopy shows that the nasopharynx is covered with a large amount of scab and sticky secretions, mostly yellowish-brown in color. The necrotic mucosa is flocculent with varying degrees of tissue loss, some of the ulcers are deep, and in severe cases, the pulsation of the internal carotid artery may be visible. The ulcer may also destroy the clivus and the basal wall of the sphenoid sinus posteriorly and superiorly, exposing the bone. The biopsy pathology is necrotic tissue without tumor cells, excluding nasopharyngeal ulcers due to tumor recurrence [24].
In the current study, all dosimetric variables were statistically significantly associated with the emergence of RINU, and there was a remarkably high degree of association between these factors. According to the Cox proportional hazards model, the EQD2_ \(\sum\)D80 emerged as the most valuable predictor, suggesting that the accumulated dose to the nasopharynx plays an important role in the RINU occurrence. Some studies also identified dose as an essential hazard factor for the incidence of RINU in re-irradiated patients. Yu et al. [19] observed that the cumulative total prescribed dose to the GTV over 141.5 Gy in two courses of radiation was correlated with lethal nasopharyngeal necrosis. Analogously, Hua et al. [25] found that the accumulated prescription dose over 120 Gy was significantly correlated with post-radiation nasopharyngeal necrosis. The utilization of EQD2_ \(\sum\)D80 cut-off value to avert the emergence of RINU in patients undergoing two courses of IMRT may be clinically necessary, and we recommend an EQD2_ \(\sum\)D80 threshold of 137.035 Gy for the nasopharyngeal mucosa. If such a dose limit is inaccessible, our results still furnish a theoretical basis that can be utilized to predict the occurrence of RINU in patients with recurrent nasopharyngeal carcinoma who have received two courses of IMRT, and thus to identify potential preventive strategies to decrease the possibility of severe bleeding. For patients at high risk of RINU, prophylactic arterial embolization is recommended prior to re-irradiation to decrease the incidence of fatal bleeding during or after radiotherapy.
Currently, immunochemotherapy has become the preferred treatment option for recurrent and/or metastatic NPC. Although most of patients enrolled in the clinical trials were metastatic patients, immunotherapy combined with chemotherapy also demonstrated good efficacy in locoregional recurrent lesions [26,27,28,29]. For patients with non-operable locoregional recurrent disease, the timing of intervention and optimal RT dose of immune-combination chemotherapy followed by recourse radiotherapy deserves further investigation.
Our study has several important limitations. Firstly, all analyses are susceptible to selection bias owing to the limited sample size. Secondly, toxicities were retrospectively identified according to chart reviews, and it is conceivable that not all complications were obtained. Thirdly, due to the lack of a clear formula for the reparation of radiation-induced mucosal damage, this component was ignored in the calculation of EQD2. Finally, our data were merely gathered from a single center, therefore, these results need to be verified in other datasets and further disquisition is expected to reach a statistically convincing conclusion.
Conclusion
Based on this study, we suggest that the EQD2_ \(\sum\)D80 limit for the nasopharyngeal mucosa should be 137.035 Gy, for patients with recurrent NPC who have undergone radical re-irradiation with IMRT. This research presents valuable insight into the prevention of RINU from the standpoint of radiation dose, and physicians should be circumspect about the high nasopharyngeal doses when reviewing and approving second-course IMRT treatment plans. Further explorations are necessitated to identify independent predictors of RINU in recurrent NPC patients, which may suggest strategies to forestall the occurrence of RINU in the IMRT era.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- RINU:
-
Radiation-induced nasopharyngeal ulcer
- NPC:
-
Nasopharyngeal carcinoma
- EQD2 :
-
Equivalent dose in 2 Gy fractions
- ROC:
-
Receiver operating characteristics
- IMRT:
-
Intensity-modulated radiotherapy
- AJCC:
-
American joint committee on cancer
- PTV:
-
Planning target volume
- CTV:
-
Clinical target volume
- GTV:
-
Gross tumor volume
- RTOG:
-
Radiation therapy oncology group
- DVH:
-
Dose and volume histogram
- UFI:
-
Ulcer-free interval time
- TTR:
-
Time to relapse
- LRRFS:
-
Locoregional relapse-free survival
- OS:
-
Overall survival
- CR:
-
Complete response
- QOL:
-
Quality of life
- CT:
-
Computed tomography
- MRI:
-
Magnetic resonance imaging
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65:87–108. https://doi.org/10.3322/caac.21262
Lee AWM, Ng WT, Chan LLK, Hung WM, Chan CCC, Sze HCK et al (2014) Evolution of treatment for nasopharyngeal cancer–success and setback in the intensity-modulated radiotherapy era. Radiother Oncol 110:377–384
Kong F, Ying H, Du C, Huang S, Zhou J, Chen J et al (2014) Patterns of local-regional failure after primary intensity modulated radiotherapy for nasopharyngeal carcinoma. Radiat Oncol 9:60
Ng WT, Lee MCH, Hung WM, Choi CW, Lee KC, Chan OSH et al (2011) Clinical outcomes and patterns of failure after intensity-modulated radiotherapy for nasopharyngeal carcinoma. Int J Radiat Oncol Biol Phys 79:420–428
Lee N, Harris J, Garden AS, Straube W, Glisson B, Xia P et al (2009) Intensity-modulated radiation therapy with or without chemotherapy for nasopharyngeal carcinoma: radiation therapy oncology group phase II trial 0225. J Clin Oncol 27:3684–3690
Liang S-B, Wang Y, Hu X-F, He S-S, Yang X-L, Liu L-Z et al (2017) Survival and Toxicities of IMRT Based on the RTOG Protocols in Patients with Nasopharyngeal Carcinoma from the Endemic Regions of China. J Cancer 8:3718–3724
Chan OSH, Sze HCK, Lee MCH, Chan LLK, Chang ATY, Lee SWM et al (2017) Reirradiation with intensity-modulated radiotherapy for locally recurrent T3–T4 nasopharyngeal carcinoma. Head Neck 39:533–540
Yu KH, Leung SF, Tung SY, Zee B, Chua DTT, Sze WM et al (2005) Survival outcome of patients with nasopharyngeal carcinoma with first local failure: a study by the Hong Kong Nasopharyngeal Carcinoma Study Group. Head Neck 27:397–405
Lee AWM, Fee WE, Ng WT, Chan LK (2012) Nasopharyngeal carcinoma: salvage of local recurrence. Oral Oncol 48:768–774
Li Y, Xu T, Qian W, Lu X, Hu C (2018) Radiation-induced nasopharyngeal ulcers after intensity modulated radiotherapy in primary nasopharyngeal carcinoma patients: a dose-volume-outcome analysis. Oral Oncol 84:1–6
Kong F, Zhou J, Du C, He X, Kong L, Hu C et al (2018) Long-term survival and late complications of intensity-modulated radio therapy for recurrent nasopharyngeal carcinoma. BMC Cancer 18:1139
Roeder F, Zwicker F, Saleh-Ebrahimi L, Timke C, Thieke C, Bischof M et al (2011) Intensity modulated or fractionated stereotactic reirradiation in pati ents with recurrent nasopharyngeal cancer. Radiat Oncol 6:22
Qiu S, Lin S, Tham IWK, Pan J, Lu J, Lu JJ (2012) Intensity-modulated radiation therapy in the salvage of locally recurr ent nasopharyngeal carcinoma. Int J Radiat Oncol Biol Phys 83:676–683
Xiao X-T, Wu Y-S, Chen Y-P, Liu X, Guo R, Tang L-L et al (2023) Patterns and prognosis of regional recurrence in nasopharyngeal carcin oma after intensity-modulated radiotherapy. Cancer Med 12:1399–1408
Hsiung C-Y, Yorke ED, Chui C-S, Hunt MA, Ling CC, Huang E-Y et al (2002) Intensity-modulated radiotherapy versus conventional three-dimensional conformal radiotherapy for boost or salvage treatment of nasopharynge al carcinoma. Int J Radiat Oncol Biol Phys 53:638–647
Xu Y, Liu Y, Wang Z, Wang J, Zhang J, Chen X et al (2022) Irradiation-induced nasopharyngeal necrosis (INN) in newly diagnosed nasopharyngeal carcinoma treated by intensity-modulated radiation therapy: clinical characteristics and the influence of treatment strategies. Radiat Oncol 17:13
Li X-Y, Sun X-S, Liu S-L, Chen Q-Y, Guo S-S, Liu L-T et al (2019) The development of a nomogram to predict post-radiation necro sis in nasopharyngeal carcinoma patients: a large-scale cohort study. CMAR 11:6253–6263. https://doi.org/10.2147/CMAR.S197841
Han F, Zhao C, Huang S-M, Lu L-X, Huang Y, Deng X-W et al (2012) Long-term outcomes and prognostic factors of re-irradiation for locall y recurrent nasopharyngeal carcinoma using intensity-modulated radioth erapy. Clin Oncol (R Coll Radiol) 24:569–576
Yu Y-H, Xia W-X, Shi J-L, Ma W-J, Li Y, Ye Y-F et al (2016) A model to predict the risk of lethal nasopharyngeal necrosis after re -irradiation with intensity-modulated radiotherapy in nasopharyngeal c arcinoma patients. Chin J Cancer 35:59
Hua Y-J, Han F, Lu L-X, Mai H-Q, Guo X, Hong M-H et al (2012) Long-term treatment outcome of recurrent nasopharyngeal carcinoma trea ted with salvage intensity modulated radiotherapy. Eur J Cancer (Oxf Engl 1990) 48:3422–8. https://doi.org/10.1016/j.ejca.2012.06.016
Huang X-M, Zheng Y-Q, Zhang X-M, Mai H-Q, Zeng L, Liu X et al (2006) Diagnosis and management of skull base osteoradionecrosis after radiot herapy for nasopharyngeal carcinoma. Laryngoscope 116:1626–1631
Chen M-Y, Mai H-Q, Sun R, Guo X, Zhao C, Hong M-H et al (2013) Clinical findings and imaging features of 67 nasopharyngeal carcinoma patients with postradiation nasopharyngeal necrosis. Chin J Cancer 32:533–538
Yang B, Chen Y, Xu L, Yao Y, Wu J (2011) Values of diffusion-weighted imaging in differential diagnosing necrosis and recurrence of nasopharyngeal carcinoma after radiotherapy. Chin J Oncol Prev Treat 3:298–302. https://doi.org/10.3969/j.issn.1674-5671.2011.04.08
Yin Z, Gao L, Luo J, Yi J, Huang X, Wang K et al (2016) An analysis of clinical and dosimetric factors for postradiation nasopharyngeal necrosis in intensity-modulated radiotherapy for nasopharyngeal carcinoma. Chin J Radiat Oncol 25:438–42. https://doi.org/10.3760/cma.j.issn.1004-4221.2016.05.004
Hua YJ, Chen MY, Qian CN, Hong MH, Zhao C, Guo L et al (2009) Postradiation nasopharyngeal necrosis in the patients with nasopharyngeal carcinoma. Head Neck 31:807–812
Adkins DR, Haddad RI (2022) Clinical trial data of Anti-PD-1/PD-L1 therapy for recurrent or metastatic nasopharyngeal Carcinoma: a review. Cancer Treat Rev 109:102428
Masterson L, Howard J, Gonzalez-Cruz J, Jackson C, Barnett C, Overton L et al (2020) Immune checkpoint inhibitors in advanced nasopharyngeal carcinoma: beyond an era of chemoradiation? Int J Cancer 146:2305–2314
Yilmaz E, Ismaila N, Bauman JE, Dabney R, Gan G, Jordan R et al (2023) Immunotherapy and biomarker testing in recurrent and metastatic head and neck cancers: ASCO guideline. J Clin Oncol 41:1132–1146
Zhou X, Niu X, Liu P, Ou D, Zhu Y, Wang X (2021) Is immune therapy plus chemotherapy more effective than immune therapy alone for unresectable recurrent nasopharyngeal carcinoma? Front Immunol 12:762663
Acknowledgements
We acknowledge the support of the Department of Radiation Oncology of Fudan University Shanghai Cancer Center.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
MSN and FFK conducted data analysis and drafted the manuscript. GSP, RPZ and CRD participated in data collection. CSH and HMY participated in the design of the study. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no competing interest exists.
Ethics approval
The present study was approved by the Institutional Review Board of Fudan University Shanghai Cancer Center (Approval number 2009224–1).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ni, MS., Kong, FF., Pan, GS. et al. Radiation-induced nasopharyngeal ulcers after re-irradiation with intensity-modulated radiotherapy in locoregional recurrent nasopharyngeal carcinoma patients: a dose–volume–outcome analysis. Eur Arch Otorhinolaryngol 281, 1425–1434 (2024). https://doi.org/10.1007/s00405-023-08330-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-023-08330-0