Abstract
We present a unique case of a prolactin (PRL)-producing pituitary adenoma showing incomplete neuronal differentiation without ganglion cells. A 27-year-old man presented with nausea, headaches, and instability over the last 2 months. Clinical examination revealed obesity with no other endocrinological signs. His serum PRL levels were slightly elevated (38 ng/ml), whereas concentrations of the other adenohypophysial hormones were within normal range. Histology revealed an unusual pituitary adenoma containing many hypocellular areas with fibrillar appearance. The sizable tumor cells were polyhedral or elongated harboring an ovoid, vesicular nucleus with prominent nucleolus, lacking, however, the typical features of ganglion cells. By immunohistochemistry, many adenoma cells were positive for PRL. Immunostain for neurofilament protein revealed variable amounts of fibrils dispersed throughout the stroma, mostly in the hypocellular areas. In addition, neurofilament protein and chromogranin were strongly reactive in approximately 15% of the tumor cell population, whereas reactivity for synaptophysin was uniform throughout the tumor. These findings led to the conclusion that part of the tumor-cell population expressed a hybrid immunoprofile of adenoma–neuronal cell. Our case is the first PRL-producing pituitary adenoma showing incomplete neuronal differentiation lacking mature ganglion cells.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coexistence of neuronal tissue and pituitary adenoma elements has been reported as a separate entity by many authors since its first description in 1926 [9]. The histogenesis and terminology of these unexpected intriguing tumors has caused much debate among pathologists during the last decade. The currently used term to describe such sellar tumors is pituitary adenoma with neuronal choristoma (PANCH), which was introduced in 1994 [8]. The neuronal compartment of PANCH tumors, composed of ganglion cells within a variably fibrillar substrate of neuropil, is admixed with adenomatous pituitary cells. The latter may express various adenohypophysial hormones, mostly growth hormone (GH) [2, 3, 6, 8, 24, 25]. Prolactin (PRL) or adrenocorticotropic hormone (ACTH) producing a number of clinically nonfunctioning PANCH tumors have also been infrequently described [10, 11, 14, 23, 24, 25, 26, 29, 31]. Only a single case of a pituitary corticotroph adenoma associated with Cushing’s disease containing neuropil without ganglion cells was recently reported [32].
We report a unique case of PRL-producing pituitary adenoma showing neurofilament accumulation in the cytoplasm of many adenoma cells and in the extracellular matrix as well. This type of adenoma with neuronal component may represent a new entity, possibly a variant of PANCH tumor, with incomplete neuronal differentiation.
Case report
A 27-year-old man presented with nausea, headaches and walking instability during the last 2 months. Clinical examination revealed obesity with no other endocrinological signs. Basal serum levels of his adenohypophysial hormones were found within the normal range, except for a slightly elevated PRL (38 ng/ml, normal <5 ng/ml). A CT scan showed an intrasellar tumor measuring 1.3 cm in its largest diameter extending up to the optic chiasm with no alterations of compression (Fig. 1). The patient underwent transsphenoidal surgery. A year after the operation, he was in good health and free of symptoms.
Morphology methods
For light microscopy, 4–6 μm sections of formalin-fixed and paraffin-embedded tissue were used. Slides of paraffin sections were stained with H&E and the PAS method. For immunohistochemistry, the standard avidin-biotin-peroxidase complex (ABC) technique was utilized using the elite Vectastain detection system (Vector Laboratories, Inc., Burlingame, CA, USA). Antibodies directed against adenohypophysial hormones were used: GH (1:4000), PRL (1:3000), ACTH (1:2500), β-thyroid stimulating hormone (β-TSH, 1:2000), β-follicle stimulating hormone (β-FSH, 1:1500), β-luteinizing hormone (β-LH, 1:2000), and α-subunit of glycoprotein hormones (α-SU, 1:1500). Pretreatment of sections with 1 mg/100 ml pronase (Sigma, St. Louis, MO, USA) was performed in room temperature before the application of primary antibodies. Tissue sections were also immunostained for S-100 protein (1:2000, Dakopatts Co., Carpinteria, CA), glial fibrillary acidic protein (GFAP; dilution 1:1000, Dakopatts), neurofilament protein (NF, 1:100, clone NR4, Dakopatts, and 1:100, clone RT 97, Novocastra Labs. Newcastle upon Tyne, UK), chromogranin (CGR, 1:1100, Dakopatts), synaptophysin (SYN, 1:1000, Dakopatts), and Ki-67 (clone MIB-1, 1:50, Novacastra). The two different NF protein antibodies used react with the 68-kDa and the 200-kDa component of the three major polypeptide subunits of neurofilaments, respectively. Before immunostaining for NF protein and Ki-67, sections were incubated in sodium citrate buffer, pH 6.0 in high-pressure cooking for 2 min. Incubations with all primary antibodies were carried out overnight at 4°C. Additional sections were immunostained for NF protein without microwaving. Forty-six additional unselected PRL-producing tumors were also immunostained for NF protein. No tissue appropriately fixed for electron microscopic studies was available, and samples retrieved from the formalin-fixed, paraffin-embedded material were suboptimal.
Results
Histology
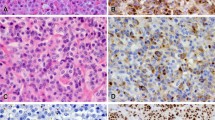
By histology, the tumor was diagnosed as a chromophobic, slightly acidophilic, pituitary adenoma. It was cellular with a diffuse pattern of growth showing small hypocellular foci with fibrillar appearance (Figs. 2, 3). The tumor cells were uniformly large polyhedral often unipolar with elongated cytoplasmic processes. They harbored sizable ovoid, vesicular nuclei with thick nuclear membrane and prominent nucleolus. A few tumor cells contained lobulated or double nuclei. No Nissl substance was noted in their cytoplasm. Few tumor cells showed signs of apoptosis, exhibiting pyknotic nuclei and dark cytoplasm. The majority of tumor cells showed conclusive immunoreactivity for PRL (Fig. 4), often with a typical paranuclear distribution (Golgi pattern). Immunohistochemistry yielded negative results for all the remaining adenohypophysial hormones. Immunostains for NF protein revealed variable amounts of fibrils dispersed throughout the stroma with focal accumulation, particularly in the hypocellular areas of the tumor. The distribution of neurofilaments was identical for both NF protein antibodies used (68-kDa and 200-kDa subunits). In addition, approximately 15% of the tumor cell population, particularly in the fibrillary areas, showed strong cytoplasmic immunoreactivity for NF protein, highlighting the fine cytoplasmic processes of the elongated unipolar cells (Fig. 5). By examining a number of consecutive sections, immunostain for CGR showed similar strong cytoplasmic distribution (Fig. 6) with that of NF protein in the same tumor cells, whereas immunoreactivity for SYN was uniform in the tumor cells, highlighting their cytoplasmic membranes (Fig. 7). The immunocytochemical results for PRL, NF protein, CGH, and SYN are shown in Table 1. Sections immunostained for NF protein with and without microwaving showed the same distribution of neurofilaments. However, after microwaving, immunoreactivity was more intense. The tumor was negative for S-100 protein and GFAP. The Ki-67 labeling index was estimated at 5%. Forty-six additional PRL-producing adenomas immunostained for NF protein in both collaborating institutions showed no positivity in either the cytoplasm of adenoma cells or the stroma. NF protein antibodies were localized only in the neurohypophysial fragments included in five of these specimens, which served as internal controls.
Discussion
The histogenesis of tumors composed of both pituitary adenoma cells and neural tissue is an intriguing enigma for pathologists regarding the different embryonal origin of adenohypophysis and neuronal elements. Three theories have been proposed to explain this association. The first one accepts the existence of neuronal elements possibly as embryonic remnants in the adenohypophysis; the development of the two lesions being incidental with no histogenetic relation and interaction. Alternatively, these tumors may originate in remnants, showing intermediate features of neuronal-adenohypophysial cells [10, 31]. The second is a cause and effect theory proposing that heterotopic hypothalamic neurons within the adenohypophysis can produce stimulatory peptides (for example GRH) leading to hyperplasia and subsequent neoplastic transformation of adenohypophysial cells [2, 3]. The paracrine effect of ectopic hypothalamic factors on pituitary cells has been proposed to act mainly as the promoter signal during tumorigenesis. The third hypothesis suggests the pituitary adenoma as the initial lesion. Some adenoma cells under unknown stimulatory mechanisms can be transformed into neuronal elements [7]. More studies are needed to reveal which theory is valid.
Regarding the established embryological concepts, it is difficult to accept neuronal metaplasia of pituitary adenoma cells. However, Horvath et al. [7], in a detailed immunohistochemical and ultrastructural study of 15 GH-producing PANCH tumors, provided morphological evidence for this contemporary and attributable hypothesis. They noted the presence of many intermediate cells bearing features of both somatotroph adenoma and ganglion cell in at least one case. One of these tumors gradually progressed towards a neuronal phenotype as documented in the tissue obtained at subsequent operations. In the same study, retrospective examination of several cases of sparsely granulated somatotroph adenomas disclosed scattered cells with similar intermediate features.
Furthermore, one experimental study indicated the potential of spontaneous neural transformation of adenohypophysial cells in culture [12]. This potential has also been observed in vitro in other endocrine neoplasms (pheochromocytoma, medullary thyroid carcinoma, carcinoid tumor, insulin-producing pancreatic tumors, and small-cell carcinoma of the lung) and may represent a metaplastic process [1, 5, 13, 21, 30]. In addition, gangliocytic and ganglion-like differentiation has recently been observed in a bronchial carcinoid tumor [18]. Lastly, the presence of neuronal cells in the posterior lobe in a case report raises the possibility of transdifferentiation of adenohypophysial cells [8].
The mechanisms of neural transformation of pituitary adenoma cells are still obscure. Previous studies proposed that nerve growth factor (NGF) may be involved in this process, because its receptors have been detected in all adenohypophysial cell types [15, 17, 20] and in both adenoma and neuronal cells of PRL-producing PANCH tumors [29]. In addition, in vitro studies also proved that NGF has a mitogenic effect on human and rat pituitary cells and can modulate the secretion of adenohypophysial hormones [16, 22]. However, whether NGF plays a role in neural transformation of adenohypophysial cells has not been proven so far [29].
Vidal et al. [32], in a recent report, presented a case of a corticotroph adenoma with neural differentiation. The tumor stroma contained neurofilaments, but no neurons were identified. Our case is unique because it is the first PRL-producing pituitary adenoma showing incomplete neuronal transformation. By histology, the adenoma cells resembled small ganglion cells but lacked the typical features; they were unipolar with cytoplasmic process and large vesicular nuclei with prominent nucleolus. Approximately 15% of adenoma cells, in addition to NF protein, showed strong immunopositivity for CGR, whereas SYN was extensive and uniform. Careful examination of parallel sections showed immunoreactivity for PRL in the substantial majority of these cells. The same was true for CGR, which also represents a marker for neuronal differentiation. This observation represents additional evidence that at least part of the adenoma cells expressed a dual adenoma–neuronal immunoprofile, but they were not fully differentiated towards neurons. Hypocellular fibrillar areas corresponding to accumulated neurofilament-rich cytoplasmic processes of tumor cells, identified by histology and immunohistochemistry for NF protein, is a unique novel finding not previously reported in PRL-producing adenomas. In a single previous report immunoreactivity for NF protein was observed in five PRL-secreting adenomas among 75 formalin-fixed pituitary tumors [19]. Immunoreactivity was more intense for the 68-kDa as compared to the 160-kDa subunit and negative for the 200-kDa subunit. In our studies, we also detected no reactivity for the 200-kDa subunit in an additional 46 PRL-producing pituitary adenomas examined, but we failed to reveal NF protein positivity using the 68-kDa subunit. These adverse results might be attributed to technical differences. Nevertheless, in the presented case, immunoreactivity for NF protein was restricted to the areas of hypocellular fibrillar stroma and the cytoplasmic distribution of NF protein in the adjacent tumor cells was not patchy as it was observed by these authors, but strong and diffuse highlighting the elongated cytoplasmic processes. In addition, selective localization of NF protein exclusively in the neurohypophysial fragments of five PRL-producing adenomas examined excludes the possibility of false positivity induced by microwaving.
By description, our case does not fulfill the criteria of a PANCH tumor. The presence of neural elements and cells with intermediate adenoma–neuronal phenotype can be explained by the neural metaplasia theory. The NF protein may represent a product of adenoma cells being stimulated towards neuronal differentiation, and this might be the first step in the metaplastic process preceding the full development of ganglion cells. The ultrastructural findings in the case described by Vidal et al [32] are consistent with our suggestion. It is uncertain whether these tumors would proceed towards a neuronal phenotype if left unresected, because neural transformation mechanisms are complex, regulated by many signals and different pathways [12, 16, 28]. The presence of neuronal features in PANCH tumors containing somatotrophs might be attributed to the ability of somatotrophs to act as adult stem cells that under certain circumstances might transform and exhibit a different cell phenotype. However, our case further suggests the view that PRL-producing cells have also the capacity for neuronal transformation [29]. This evidence may lead to reconsideration of the established embryologic concept that the adenohypophysis derives from Rathke’s pouch an ectodermal structure unrelated to the nervous system [5]. The question remains whether this tumor represents a PANCH variant or a new entity of an intermediate adenoma–neuronal tumor.
References
Ahiman H, Wigander AL, Nilsson O (1989) Presence of nerve growth factor like immunoreactivity in carcinoid tumour cells and induction of a neuronal phenotype in long term culture. Int J Cancer 43:949–955
Asa SL, Scheithauer BW, Bilbao JM, Horvath E, Ryan N, Kovacs K, Randall RV, Laws ER Jr, Singer W, Linfoot JA, Thorner MO, Vale W (1984) A case for hypothalamic acromegaly: a clinicopathological study of six patients with hypothalamic gangliocytomas producing growth hormone-releasing factor. J Clin Endocrinol Metab 58:796–780
Bevan JS, Asa SL, Rossi ML, Esiri MM, Adams CB, Burke CW (1989) Intrasellar gangliocytoma containing gastrin and growth hormone-releasing hormone associated with a growth hormone-secreting pituitary adenoma. Clin Endocrionol (Oxf) 30:213–224
Dubois PM, El Anraoui A, Heretier AG (1997) Development and differentiation of pituitary cells. Microsc Res Tech 39:98–113
Franke WW, Grund C, Archatatter T (1986) Co-expression of cytokines and neurofilament proteins in a permanent cell line: Cultured rat PC12 cells combine neuronal and epithelial features. J Cell Biol 103:1933–1943
Geddes JF, Jansen GH, Robinson SFD, Gömöri E, Holton JL, Monson JP, Besser GM, Révész T (2000) Gangliocytomas of the pituitary. A heterogeneous group of lesions with differing histogenesis. Am J Surg Pathol 24:607–613
Horvath E, Kovacs K, Scheithauer BW, Lloyd RV, Smyth HS (1994) Pituitary adenoma with neuronal choristoma (PANCH): Composite lesion or lineage infidelity? Ultrastruct Pathol 18:565–574
Horvath E, Kovacs K, Tran E (2000) Ganglion cells in the posterior pituitary: result of ectopia or transdifferentiation? Acta Neuropathol 100:106–110
Kiyono H (1926) Die Histopathology der hypophyse. Virchows Arch A Pathol Anat Histopathol. 259:388–465
Lach B, Rippstein P, Benoit BG, Staines W (1996) Differentiating neuroblastoma of pituitary gland: neuroblastic transformation of epithelial adenoma cells. J Neurosurg 85:953–960
Li JY, Racadot O, Kujas M, Koyadri M, Peillon F, Racadot J (1989) Immunocytochemistry of four mixed pituitary adenomas and intrasellar gangliocytomas associated with different clinical syndromes: acromegaly, amenorrhea-galactorrhea, Cushing’s disease and isolated tumoral syndrome. Acta Neuropathol 77:320–328
Martinez-Campos A, Dannies PS (1986) A possible differentiation of anterior pituitary cells in collagen gels into neurons. Cell Tissue Res 244:21–26
McCann FV, Pettengill OS, Cole JJ, Russell JAG, Sorenson GD (1981) Calcium spike electrogenesis and other electrical activity in continuously cultured small cell carcinoma of the lung. Science 212:1155–1157
McCowen KC, Glickman JN, Black PMcL, Zervas NT, Lidov HGW, Garber JR (1999) Gangliocytoma masquerating as prolactinoma. J Neurosurg 91:490–495
Missale C, Boroni F, Sigala S, Buriani A, Fabris M, Leon A, Dal Toso R, Spano P (1996a) Nerve growth factor in the anterior pituitary: localization in mammotroph cells and cosecretion with prolactin by a dopamine-regulated mechanism. Proc Natl Acad Sci USA 93:4240–4245
Missale C, Losa M, Sigala S, Balsari A, Giovanelli M, Spano PF (1996b) Nerve growth factor controls proliferation and progression of human prolactinoma cell lines through an autocrine mechanism. Mol Endocrinol 10:272–285
Missale C, Fiorentini C, Finardi A, Spano P (1999) Growth factors in pituitary tumors. Pituitary 153–158
Nakamura M, Abe Y, Hatanaka H, Tsutsumi Y, Kijima H, Ueyama Y, Inoue H, Shimosato Y, Osamura RY (2003) Bronchial carcinoid tumor with gangliocytic- and paraganglionlike differentiation. Virchows Arch 442:183–185
Ogawa A, Sugihara S, Hasegawa M, Sasaki A, Nakazato Y, Kawada T, Ishiuchi S, Tamura M (1990) Intermediate filament expression in pituitary adenomas Virchows Arch B 58:341–349
Patterson JC, Childs GW (1994) Nerve growth factor and its receptor in the anterior pituitary. Endocrionol 135:1689–1696
Polak M, Scharfmann R, Seilheimer B, Eisenbarth G, Dressler D, Verma IM, Porter H (1993) Nerve growth factor induces neuron- like differentiation of an insulin- secreting pancreatic beta cell line. Proc Natl Acad Sci USA 90:5781–5785
Proesmans M, van Bael A, Andries M, Denef C (1997) Mitogenic effects of nerve growth on different cell types in reaggregate cell cultures of immature rat pituitary. Mol Cell Endocrinol 134:119–127
Puchner MJA, Lüdecke DK, Valdueza JM, Saeger W, Willig RP, Stalla GK, Odink RJ (1993) Cushing’s disease in a child caused by a corticotropin-releasing hormone-secreting intrasellar gangliocytoma associated with an adrenocorticotropic hormone-secreting pituitary adenoma. Neurosurgery 33:920–925
Puchner MJA, Lüdecke DK, Saeger W, Riedel M, Asa SL (1995) Gangliocytomas of the sellar region-a review. Exp Clin Endocrinol 103:129–149
Puchner MJΑ, Herrmann HD (1997) Intrasellar pituitary gangliocytoma-adenoma presenting with acromegaly: case report. Neurosurgery 40:611–614
Saeger W, Puchner MJA, Lüdecke DK (1994) combined sellar gangliocytoma and pituitary adenoma in acromegaly or Cushing’s disease. A report of three cases Virchows Arch 425:93–99
Schechter J, Windle JJ, Stauber C, Mellon PL (1992) Neural tissue within anterior pituitary tumors generated by oncogene expression in transgenic mice. Neuroendocrinol 56:300–311
Schechter J, Stauber C, Windle JJ, Mellon PL (1995) Basic fibroblastic growth factor: the neurotrophic factor influencing the ingrowth of neural tissue into the anterior pituitary of alpha-T7 transgenic mice? Neuroendocrinol 61:622–627
Scheithauer BW, Horvath E, Kovacs K, Lloyd RV, Stefaneanu L, Buchfelder M, Fahlbusch R, Werder K von, Lyons DF (1999) Prolactin-producing pituitary adenoma and carcinoma with neuronal components-a metaplastic lesion. Pituitary 1:197–205
Tamir H, Liu KP, Payette RF (1989) Human medullary thyroid carcinoma: characterization of the serotoninergic and neuronal properties of a neuroectodermally derived cell line. J Neurosci 9:1199–1212
Towfighi J, Salam MM, McLendon RE, Powers S, Page RB (1996) Ganglion-cell-containing tumors of the Pituitary Gland. Arch Pathol Lab Med 120:369–377
Vidal S, Horvath E, Bonert V, Shaninian H K, Kovacs K (2002) Neural transformation in pituitary corticotroph adenoma Acta Neuropathol 104:435–440
Acknowledgements
The authors are indebted to the National Hormone and Pituitary Program (NHPP), Torrance, California, USA, for providing the pituitary hormone antibodies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thodou, E., Kontogeorgos, G., Horvath, E. et al. Prolactin-producing pituitary adenoma with incomplete neuronal transformation: an intermediate adenoma–neuronal tumor. Acta Neuropathol 108, 115–120 (2004). https://doi.org/10.1007/s00401-004-0862-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-004-0862-y