Abstract
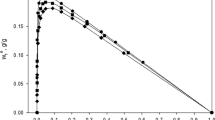
Adsorption of n-butanol and water from butanol–water mixtures is studied on adsorbents of different hydrophobicity (activated carbon and hydrophobized montmorillonites). The shape of the adsorption isotherms of the hydrophobized clay minerals does not indicate preferential adsorption of butanol. Therefore, the adsorption capacity cannot be determined on the basis of the Langmuir isotherm. A new equation for determining the adsorption capacity is derived by combining the free enthalpy of adsorption with the adsorption excess amount. Both collections of data are obtained from the surface excess isotherm. The reliability of the adsorption capacity is checked by X-ray diffraction measurements.
Article PDF
Similar content being viewed by others

Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 1 November 1997 Accepted: 19 February 1998
Rights and permissions
About this article
Cite this article
Regdon, I., Dékány, I. & Lagaly, G. A new way for calculating the adsorption capacity from surface excess isotherms. Colloid Polym Sci 276, 511–517 (1998). https://doi.org/10.1007/s003960050273
Issue Date:
DOI: https://doi.org/10.1007/s003960050273