Abstract
Purpose
This study tried at identifying lifestyle patterns in a sample of Lebanese adults and investigating their association with metabolically healthy overweight and obesity (MHOv/O).
Methods
This study used data from a community-based survey of Lebanese adults living in Greater Beirut (n = 305). Dietary intake was assessed using an 80-item food frequency questionnaire. MHOv/O was defined as having one or none of the following metabolic abnormalities: triglycerides ≥ 150 mg/dL; systolic blood pressure ≥ 130 mmHg; diastolic blood pressure ≥ 85 mmHg; fasting blood glucose ≥ 100 mg/dL; HDL-cholesterol < 40 mg/dL for men and < 50 mg/dL for women. 201 subjects were overweight/obese and 98 had MHOv/O. Factor analysis was implemented to derive lifestyle patterns.
Results
Two lifestyle patterns were identified: the unhealthy lifestyle pattern, which included foods such as fast food and soft drinks and was characterized by alcohol consumption and cigarette smoking. Eating at home loaded negatively on this pattern. The healthy lifestyle pattern was characterized by positive loadings of food groups such as milk and dairy products, fruits, vegetables and legumes. Sleep difficulties loaded negatively on this pattern, while vigorous and moderate physical activity loaded positively. Multiple logistic regression analyses showed that subjects belonging to the 3rd tertile of the healthy pattern scores had higher odds of MHOv/O as compared to those in the 1st tertile (OR 2.33, CI 1.04–4.81).
Conclusions
Findings of this study provided evidence on the combined effect of lifestyle patterns in relation to cardiometabolic abnormalities and highlighted the importance of focusing on ‘holistic’ lifestyle pattern modifications in designing and implementing prevention interventions in overweight and obese subjects.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity is a well-known risk factor for numerous metabolic disorders, including insulin resistance (IR), hyperglycemia [1], hypertension [2, 3] and dyslipidemia, which may contribute to increased morbidity and mortality [4,5,6]. However, a number of studies have shown that a subset of overweight and obese individuals do not develop such metabolic disorders and are thus considered to have a metabolically healthy phenotype (MHP) [7, 8]. There is a lack of consensus on the definition of this phenotype, with several definitions having been proposed in the literature [9]. In some studies, MHP was defined as having one or none of the metabolic abnormalities specified by the adult treatment panel III [9, 10], while in others, C-reactive protein (CRP) was included among the criteria identifying this phenotype [11, 12]. Other definitions were based on the absence of insulin resistance as assessed by HOMA [13] or on oral glucose tolerance based on the Matsuda index [13, 14]. Despite the lack of consensus on the definition of MHP, this phenotype is usually characterized by favorable metabolic profiles such as insulin sensitivity, normal blood pressure and lipid profiles [15]. Other protective factors that were identified among individuals with MHP comprised lower circulating levels of CRP [16] and higher levels of adiponectin compared to their metabolically unhealthy counterparts [17]. Furthermore, it has been suggested that metabolically healthy obese and overweight individuals may have a lower risk of mortality and a better prognosis than their metabolically unhealthy counterparts [18, 19]. These differences were noted across different definitions of MHP [5]. The prevalence of MHP was reported to vary between 7 and 74%, depending on the definition used to identify this phenotype, the age of the population and the inclusion of both overweight and obese or strictly obese subjects [9, 11, 20,21,22].
Metabolically heathy overweight and obesity may have a genetic basis, but recent evidence suggested that lifestyle factors may also play an important role in the etiology of this phenotype [20, 23, 24]. More specifically, dietary intake, physical activity, sleep, smoking and alcohol drinking were found to be associated with cardiometabolic health in overweight and obese subjects [25, 26]. However, the evidence regarding these associations remains inconclusive [25,26,27]. It is worth noting that dietary and lifestyle factors often exert their effects on cardiometabolic health in a synergistic or even cumulative manner, which would not be reflected when examining each factor individually [28, 29]. In many studies that investigated single lifestyle factors, sophisticated statistical techniques such as multiple linear and logistic regression models were adopted to account for the interaction and synergistic effects between these factors [20, 22, 23, 25, 26, 30,31,32]. However, an alternative approach in nutritional epidemiology may allow for a more holistic investigation of MHP determinants, whereby “lifestyle patterns” may be examined in relation to metabolic health. This approach has been adopted by some studies that investigated adiposity [33, 34], metabolic syndrome [28], diabetes [35] and hypertension [29, 36], but none of these studies investigated lifestyle patterns and their association with MHP in overweight and obese subjects.
In Lebanon, similar to other countries of the Eastern Mediterranean region, the rates of adult overweight and obesity were found to follow an increasing secular trend, escalating from 54.4 to 65.0% during the past decade [37]. Previous research conducted by our group has shown that more than one-third of Lebanese overweight and obese adults were metabolically healthy and that this phenotype was positively associated with the Traditional-Lebanese dietary pattern [20]. However, to better capture “real life” associations and implications, it is crucial to investigate combined “lifestyle patterns” in relation to metabolic health in overweight and obese subjects. The adoption of such an integrated holistic approach was highlighted as a priority research area by a recent scoping review on nutrition and non-communicable diseases in the Arab world [38]. In this context, this study aims to (1) identify and characterize lifestyle patterns in a sample of Lebanese adults, including normal, overweight and obese adults; (2) investigate the association of these patterns with metabolic health among overweight and obese adults; and (3) examine the sociodemographic correlates of the identified patterns in the overall population. Findings of this study can be used for the development of holistic, culturally sensitive, evidence-based intervention strategies that contribute to better cardiometabolic health in overweight and obese subjects and hence may help reduce their morbidity and mortality risks [30].
Methodology
This study used data from a community-based survey of Lebanese adults living in Beirut and its suburbs (also known as the Greater Beirut area). Data collection spanned over 3 months extending from March until May of 2014. The Institutional Review Board (IRB) of the American University of Beirut reviewed the survey protocol and granted it approval under the following protocol number (IM.HT.03). All subjects agreeing to take part in the study provided a signed written consent form.
Study participants
Study participants were selected to constitute a representative sample of all adults living in the Greater Beirut area. The recruitment of participants was carried out using a door-to-door approach. A multistage stratified probability sampling frame was used to select the households, whereby the strata were districts of the Greater Beirut area, and within each district, neighborhoods and then households were selected based on a systematic random sampling. Further details about the sampling are described in Nasreddine et al. [39]. If more than one adult respondent were present in the selected household, the youngest eligible adult was invited to participate. To be eligible to take part in the survey, the subject had to be 18 years of age or older with primary residence located in Greater Beirut Area. Exclusion criteria included adults with kidney disease and undergoing dialysis, mentally ill or pregnant. Individuals working in a plastic or any other chemical company were also excluded, given that the original survey aimed to investigate exposure to Bisphenol A.
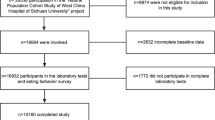
To be included in the present study, subjects had to have complete anthropometric, biochemical and dietary data. Subjects who have reported the diagnosis of one or more chronic disease or metabolic abnormalities that may have affected their dietary or lifestyle habits were excluded: diabetes (n = 64), dyslipidemia (n = 120) or hypertension (n = 119). Therefore, of the original 501 subjects participating in the survey, a total of 305 were included in this study, of whom 201 subjects were either overweight (25 kg/m2 < BMI ≤ 29.9 kg/m2; n = 108) or obese (BMI ≥ 30 kg/m2; n = 91).
Study protocol and data collection
Following the household visits, subjects consenting to take part in the study were asked to visit the Department of Nutrition and Food Sciences, American University of Beirut, where data collection took place. For this visit, subjects were instructed to bring any medication they were taking and to come following a 10-h fast. Data collection included (1) sociodemographic questionnaire, (2) dietary intake assessment, (3) anthropometric measurements as well as (4) biochemical assessments. All aspects of data collection were conducted by trained field workers.
Sociodemographic and lifestyle questionnaire
During a one-to-one interview and using the sociodemographic and lifestyle questionnaire, information was obtained regarding age (in years), sex (male, female), education level (up to intermediate, secondary or technical and university), crowding index (calculated as the ratio of individuals living in the house over the number of rooms in the house-excluding kitchen and bathrooms), marital status (single-including not married, widowed, separated and divorced- and married), family history of cardiovascular diseases (CVD). As for the lifestyle factors, information was collected regarding eating at home, current smoking status, physical activity, sleep difficulties, drinking water and alcohol consumption. For ‘eating at home’, participants were asked to indicate the frequency of consuming the main meal of the day at home, using five choices: never, rarely (1 time/week–2 times per month); few times (2–3 times per week); most of the times (4–5 times per week); and always (6–7 times per week). For smoking, subjects were grouped as either nonsmokers or current smokers. In addition, among smokers, information was collected with regard to the number of cigarettes smoked per day. Physical activity was assessed using the Arabic short version of the International Physical Activity Questionnaire (IPAQ) [40], whereby physical activity was calculated by weighting each type of activity by its energy requirements defined in MET-minutes (multiples of the resting metabolic rate for an activity multiplied by the minutes performed). Based on METS-min per week, three variables of physical activity were derived, including light, moderate and vigorous intensity [40]. Sleep difficulties were examined using the following question: ‘Do you wake up during the night and have difficulty resuming sleep?’. Subjects were given 6 frequency options: never, rarely, once per month, 2–4 times per month, frequently 5–15 per month and almost always 16–30/month. Water intake was assessed by asking the participants ‘how many times per day do you drink one cup of water’ and was shown a standard cup (250 mL) as reference. Alcohol consumption was reported as either ‘consumes alcohol’ or ‘does not consume alcohol’.
Dietary intake assessment
During the same interview, participants completed a culture-specific 80-item semi quantitative Food Frequency Questionnaire (FFQ), referring to the subject’s dietary intake during the year preceding the interview. These 80 items were chosen as a comprehensive representation of the Lebanese diet. Further details about the development of the original questionnaire were described elsewhere [41]. The FFQ used in the study collected information about the amount of food consumed as well as the frequency of consumption. To assist participants in the estimation of the amount of food consumed, the two-dimensional food portion visual (Millen and Morgan, Nutrition Consulting Enterprises, Framingham, MA, USA) was used [42]. Whenever applicable, common household measures were also used in the estimation of the portion size. As for the frequency of consumption of each food item, five options were included in the FFQ: per day, per week, per month, per year or never. Participants had the choice to report their intake in terms of a reference portion size or in grams. The daily intake of each food items was then derived from its reported frequency of consumption. Total energy intake was computed using the food composition database of the Nutritionist ProTM software (Axxya Systems LLC, Stafford, TX, USA). In order to prepare the dietary intake data for the derivation of lifestyle patterns, the 80 food items of the FFQ were grouped into 13 food groups. The grouping of the food items was carried out taking into account the nutrient profile of the food items and/or their culinary usages (“Appendix 1”). For each food group, the total consumption was calculated by adding the daily intake of servings from each item in this group.
Anthropometric measurements
Anthropometric measurements including weight, height, and waist circumference were obtained using standard techniques and as described in Nasreddine et al. [39]. The body mass index (BMI) was calculated as the ratio of weight (kg) to square of height (m2). Systolic and diastolic blood pressure measurements were taken two times, while the participants were seated, using a digital sphygmomanometer, at 10-min intervals. The mean of the two measurements was used in this study.
Biochemical assessments
During the visit and at the Nutrition and Food Sciences department, a 15-mL sample of blood was collected by a trained phlebotomist. The biochemical assessments conducted were: fasting glucose levels using the enzymatic method and levels of triglycerides (TG) and HDL-C using the enzymatic spectrophotometric technique. Further details about the equipment used in the assays are described elsewhere [39].
MHOv/O classification
Although MHOv/O referred typically to the phenotype of overweight and obese individuals who do not present metabolic abnormalities, no agreement exists on the definition of MHOv/O [43]. In this study, the adult treatment panel criteria for the metabolic syndrome (ATP-III) were used [10]. Accordingly, an overweight or obese subject was classified as MHOv/O if he/she had one or none of the following metabolic abnormalities [9, 11]: TG ≥ 150 mg/dL; systolic blood pressure (BP) ≥ 130 mmHg; diastolic blood pressure ≥ 85 mmHg; fasting blood glucose ≥ 100 mg/dL; and HDL- cholesterol < 40 mg/dL for men and < 50 mg/dL for women. Furthermore, in order to be classified as MHOv/O, subjects must not be on any relevant therapy for the metabolic syndrome-associated morbidities of established diabetes, hypertension, hyperlipidemia, CVD, or cerebrovascular disease, liver or renal disease and obstructive sleep apnea. The latter criterion, however, was of little relevance given that participation in this study was restricted to subjects with no known diagnosis of disease. Overweight and obese subjects presenting more than one of the aforementioned metabolic conditions were classified as metabolically unhealthy overweight and obese (MUHOv/O).
Derivation of lifestyle patterns
In order to examine the lifestyle patterns in the study population, variables related to dietary intake, eating at home, alcohol consumption, drinking water, sleep difficulties, current smoking status and physical activity were entered into the factor analysis (FA). Physical activity was used as a continuous variable, expressed in MET-minutes. The FA was carried out with a Varimax rotation of patterns, where the axes are maintained at 90°. A few diagnostics of the data were conducted prior to running the FA and included examination of the correlation matrix between all the variables: the Kaiser–Meyer–Olkin test (> 0.7) and the Chi square for Bartlett test of sphericity (p < 0.05). The number of patterns to be retained depended on (1) the Kaiser criterion (eigenvalue greater than 1), (2) the scree plot (inflection point) and (3) relevance and interpretability of the patterns. For all participants and for each pattern, scores were calculated using the least squares regression method where for each pattern the mean was equal to zero. Each participant had a score on each of the two patterns. For a participant, the higher his/her pattern score indicated higher adherence to this particular pattern. Factor loadings indicated the strength and direction of the association between the lifestyle variables and the identified patterns. For each pattern, participants were grouped into tertiles of pattern scores. For the naming of the patterns, lifestyle variables with a factor score greater than 0.3 were considered [44]. In case a variable had factor loadings > 0.3 on more than one of the identified patterns, the pattern with the highest loading was considered for labeling. The dietary and lifestyle variables were standardized before entry into the FA.
Statistical analyses
For the purpose of evaluating the associations of lifestyle patterns and odds of metabolic health, the derivation of the patterns was conducted among overweight and obese subjects only. The availability of 201 overweight and obese participants allowed for the detection of an OR 2.362 (effect size) with a power of 80% and a 5% margin of errors [45].
Participants’ characteristics were presented through mean ± SD and proportions for continuous and categorical variables, respectively (n = 305). Of these characteristics, none of the continuous variables used was found to have a normal distribution (as per the Shapiro test, skewness and Q–Q plots). Therefore, Kruskal–Wallis tests were used to compare these variables among study participants. Chi square test was used to compare categorical variables. In order to examine the association among the identified lifestyle patterns with MHOv/O, logistic regression was conducted, with the MHOv/O as outcome variable and the tertiles of the patterns as independent variables. In the multiple logistic regression models, variables found to be significantly associated with MHOv/O in the univariate analyses were included in the model, in addition to the pattern’s tertiles. The association of each of the identified patterns with MHOv/O was examined using separate logistic regressions models. For the associations among the patterns and MHOv/O, only overweight and obese subjects were included (n = 201). Multiple logistic regression analyses were used to investigate correlates of the identified lifestyle patterns. For each pattern, the regression model included adherence to the pattern score as outcome variable (belonging to the 3rd tertile of the pattern score) and the sociodemographic characteristics as the independent variables. A p value < 0.05 was used to indicate statistical significance. The Statistical Package for Social Sciences (SPSS) version 22 (IBM Corp. Released 2013. IBM SPSS Statistics for Windows, Version 22.0. IBM Corp: Armonk, NY, USA) was used for data cleaning, management and analyses. Data cleaning referred to identifying and removing (or correcting) inaccurate entries from the data set before conducting the statistical analyses.
Results
In this study, among overweight and obese subjects (n = 201), 103 subjects (51.24%) were MUHOv/O. Subjects’ characteristics including sociodemographic and lifestyle characteristics, anthropometric measurements as well as prevalence of metabolic abnormalities are presented in Table 1. Mean age was 39.35 ± 13.84 years, with normal weight subjects significantly younger than both MHOv/O and MUHOv/O subjects. In the study population, 38.7% and 61.3% were males and females, respectively. The proportion of females, as compared to males, within the MHOv/O group was higher than normal weight and MUHOv/O groups. A considerable proportion of the study population (57.4%) had an education level ‘up to intermediate’ with only 14.9% attaining the university level. There was no significant difference in the education level between the three groups. On the other hand, crowding index, another indicator of socioeconomic status (SES), showed significant difference between the three groups studied, whereby among MHOv/O subjects, the proportion with a crowding index < 2 was lower as compared to normal weight and MUHO/O groups. MHOv/O subjects had also significantly lower energy intake as compared to the MHUOv/O and normal weight subjects. Normal weight subjects had significantly lower waist circumference compared to both MHOv/O and MUHOv/O groups. The levels of blood glucose, TG and HDL-C did not differ significantly between normal weight and MHOv/O groups, but subjects with MUHOv/O had significantly higher levels of blood glucose and TG and lower levels of HDL-C. There were statistically significant differences between the groups in the proportions of subjects with elevated blood glucose, TG and HDL-C (Table 1).
Table 2 displayed the results of the FA and showed the factor loadings of the various lifestyle variables on the patterns identified. In this study, two lifestyle patterns emerged: the unhealthy lifestyle pattern and the healthy lifestyle pattern. The main dietary food groups that characterized the unhealthy lifestyle pattern and their factor loadings were fast food (0.75), sugar-sweetened beverages (SSB) (0.73), condiments (0.63) and meat, poultry and sausages (0.56). Eating at home loaded negatively on this pattern (− 0.66), while alcohol consumption (0.50) and cigarette smoking (0.25) both loaded positively. The healthy lifestyle pattern was characterized by positive loadings of the following food groups: milk and dairy products (0.58), fruits, vegetables and legumes (0.56), fish (0.53) and vegetable oils (0.41). Sleep difficulties loaded negatively on the healthy lifestyle pattern (− 0.40), while vigorous and moderate physical activity had a positive loadings (0.45 and 0.42, respectively) (Table 2).
The associations among the lifestyle patterns identified and the odds of metabolic health were examined among the overweight and obese subjects, using logistic regression models (n = 201) (Table 3). In the age- and sex-adjusted model, subjects who belonged to the 3rd tertile of the scores for the healthy lifestyle pattern had significantly higher odds (OR 2.32, 95% CI 1.11–4.86) of belonging to the MHOv/O group as compared to those who belonged to the 1st tertile. This association was maintained after adjustment for the variables that showed significant associations with metabolic health in the univariate analysis of Table 1 and which included age, sex, crowding index, marital status and caloric intake. In this multivariate model, the odds of metabolic health was 2.23 times higher among overweight and obese subjects who belonged to the 3rd tertile of the healthy pattern as compared to those in the 1st tertile (OR 2.23, 95% CI 1.04–4.81). No associations were observed between adherence to the unhealthy lifestyle pattern and the odds of metabolic health in either of the logistic regression models (Table 3).
The associations of the sociodemographic characteristics with the identified lifestyle patterns were examined among study participants (n = 305), using logistic regression models. For the unhealthy lifestyle pattern, older subjects had significantly lower odds of adherence (OR 0.93, 95% CI 0.89–0.96), so did females as compared to males (OR 0.24, 95% CI 0.10–0.61). As for the healthy pattern, a higher crowding index was associated with lower adherence to this pattern (OR 0.48, 95% CI 0.24–0.93) (Table 4).
Discussion
To our knowledge, this study is the first to examine the association between lifestyle patterns and the MHOv/O phenotype. Two major lifestyle patterns were identified in our study population: (1) the unhealthy and (2) the healthy patterns, with only the latter being associated with MHOv/O. Unlike the traditional methods in nutritional epidemiology, the lifestyle pattern approach transcends reductionism in nutrition research and confers a holistic lens in the investigation of metabolic abnormalities that are multifactorial in nature [46]. Instead of examining one lifestyle factor alone (diet, physical activity, smoking, alcohol drinking and sleep habits) and its association with metabolic health, this holistic approach investigates the whole lifestyle pattern and the circular causalities that may exist between the various risk factors [28]. Accordingly, a lifestyle pattern is not to be viewed as the sum of each risk factor alone but is rather perceived as the dynamic interaction between them [28]. This indicates that the effect of a lifestyle pattern on cardiometabolic health would exceed that of any of its parts (dietary, physical activity, alcohol drinking, smoking and sleep) and could thus capture more ‘real life’ associations and implications [28]. Therefore, the significance of this study is in combining dietary, physical activity, alcohol drinking, current smoking status and sleep characteristics into lifestyle patterns and studying the association of these patterns with metabolic health in overweight and obese subjects.
In this study, around half of the study participants had the MHO/Ov phenotype, a prevalence estimate that is within the range reported in the literature [20, 27, 47, 48]. In agreement with other studies, levels of blood glucose, TG and HDL-C did not differ significantly between normal weight and MHOv/O subjects, but there were significant differences with the MUHOv/O group [49]. In addition, and in line with findings reported by other studies, subjects with MHOv/O were significantly younger, had a lower BMI and were more likely to be females, in comparison with MUHOv/O [20, 50]. The healthy lifestyle pattern identified in our study, and which was characterized by the consumption of fruits, vegetables, dairy products, fish, olive oil, physical activity and less sleep difficulties, was associated with higher odds of MHOv/O in our study sample. The identified healthy pattern confirms the previously described clustering of favorable lifestyle factors, such as fruit and vegetable consumption and physical activity [51,52,53,54] and shares many similarities with the “Prudent” or the “Mixed diet-physically active” lifestyle pattern described in other investigations [28, 33, 36]. However, our study is the first to have considered sleep quality, particularly sleep difficulties, as part of the lifestyle factors included in pattern analyses. Although the evidence is still conflicting, recent studies are indicating that sleep characteristics may be associated with cardiometabolic health [30]. In agreement with our findings, Kanagasabai et al. [30] showed, based on data from the US National Health and Nutritional Examination Survey (NHANES) 2005–2008, that the odds of having a metabolically healthy phenotype were the lowest in subjects who reported “almost always” having difficulties getting back to sleep after waking up during the night. Sleep disorders may increase preference for foods that are rich in fats and carbohydrates [55] and which may exert an adverse impact on insulin sensitivity and the lipid profile. Poor sleep may also be linked with elevated sympathetic nervous system activity, higher inflammation [56], decreased nocturnal glucose and insulin resistance [57, 58].
The healthy lifestyle pattern identified in this study was also characterized by physical activity, a factor that was previously reported to be associated with favorable metabolic health among adults [59, 60]. A growing body of evidence suggests that obese individuals who are physically active have a similar cardiovascular risk as healthy non-obese individuals, since physical activity leads to positive changes in body composition, stimulates fatty acid uptake and oxidation, enhances insulin sensitivity and reduces cardiometabolic risk [61,62,63]. In addition, the diet profile of the healthy lifestyle pattern, which included fruits, vegetables, dairy products, fish and olive oil, shares considerable similarities with the traditional Lebanese diet, a variant of the Mediterranean dietary pattern [54]. As such, our findings of the positive association between MHOv/O and the healthy lifestyle pattern are similar to those reported by Matta et al. [20], where adherence to the traditional Lebanese dietary pattern was associated with higher odds of MHOv/O among Lebanese adults. Adherence to the Mediterranean diet, as assessed by the Mediterranean dietary score (MDS), was also found to be associated with a metabolically healthy phenotype among adults aged less than 45 years in the US [64]. Fruits and vegetables contain dietary fibre, micronutrients, phytochemicals and other bioactive compounds that have antioxidant and anti-inflammatory effects, enhance insulin sensitivity and contribute to a healthier metabolic profile [27, 65,66,67,68]. Olive oil and fish may bring a rich array of monounsaturated and polyunsaturated fatty acids, which may also buffer lipid and insulin fluctuations and peaks, increase insulin sensitivity, enhance fat oxidation and decrease cardio-metabolic risk [69, 70]. The consumption of dairy products, another hallmark of the healthy lifestyle pattern and of the traditional Lebanese diet, has been linked to reduced visceral fat, an effect that may be mediated by independent or synergistic actions of calcium and dairy protein, on lipolysis, lipogenesis and thermogenesis [71, 72].
In our study, we did not observe an association between metabolic health and the unhealthy lifestyle pattern, which was characterized by smoking, alcohol consumption, high intake of fast food and sugar-sweetened beverages, as well as a lower frequency of eating at home. In contrast to our findings, Al-Thani et al. [28, 36] showed that a “High Risk” lifestyle pattern, characterized by an unhealthy diet, low physical activity and smoking, was associated with higher odds of hypertension and metabolic syndrome among Qatari women. The diet profile of the unhealthy lifestyle pattern shares considerable similarities with the Western dietary pattern, which was also shown to increase the risk for cardiometabolic abnormalities among Lebanese adults [73]. The fact that the unhealthy lifestyle pattern identified in our study was characterized by alcohol and coffee consumption may have attenuated its association with metabolic health. In fact, in our study, the assessment of alcohol consumption was based on a yes/no response and thus did not allow us to differentiate between moderate and excessive alcohol consumption. Available evidence suggests that moderate alcohol consumption may have a positive impact on cardiometabolic health and may decrease the prevalence of metabolic syndrome and some of its components among adults [74]. Similarly, coffee is a complex beverage that contains numerous biologically active compounds, some of which may exert cardio-protective effects on glycaemia and blood pressure [75].
This study has also investigated the sociodemographic determinants of the identified lifestyle patterns in our study population. It showed that older individuals were more likely to adhere to the healthy lifestyle pattern, while a higher crowding index was associated with lower adherence to this pattern. The observed direct association between age and adherence to the healthy lifestyle pattern is in agreement with findings reported by Steele et al. [29] among Brazilian adults, where adherence to the “prudent” lifestyle pattern was higher among older subjects. Similar findings were reported by studies investigating dietary patterns. For instance, older age was associated with higher adherence to the Mediterranean diet among Lebanese as well as Spanish adults [54, 76]. A possible explanation may be that older individuals are more likely to uphold traditional dietary and lifestyle habits as compared to younger generations who have greater exposure to “fashionable” foods and emerging marketing trends [77]. The inverse association between age and adherence to the healthy pattern may also reflect a state of lifestyle and nutrition transition, from traditional to “modern” or “westernized” dietary and lifestyle habits, a phenomenon that is typically observed in younger age groups, as is currently witnessed by many countries of the Eastern Mediterranean region [54, 73, 78]. The fact that a higher crowding index, indicative of lower SES, was associated with lower adherence to the healthy lifestyle pattern is a manifestation of the possible inverse socioeconomic gradient in healthy lifestyle and dietary habits [79]. In agreement with our findings, a higher SES was found to be associated with a healthier lifestyle in the US, as assessed by the lifestyle index, which incorporated diet, physical activity, smoking and alcohol consumption [79]. In fact, the adoption of healthier lifestyle behaviors involves making decisions and premeditated choices that individuals with higher SES can usually afford, such as purchasing natural instead of processed foods, joining health clubs or buying sports equipment. Even the simple act of walking or running may warrant a safer environment that is more often available to those with a higher SES [79,80,81,82,83]. Interestingly, the results of our study have also highlighted gender differences in adherence to lifestyle patterns, whereby women were less likely to adhere to the unhealthy lifestyle pattern. This finding is in line with other studies describing women as being more health-conscious and followers of dietary recommendations than men [29, 33, 54, 78].
The study has several strengths. It was conducted on a random sample of the adult population living in Beirut and was performed using a well-planned design, protocol and methodology. Its novelty lies in the adoption of the lifestyle pattern approach in investigating the determinants of MHOv/O. However, the findings of this study are to be considered in light of the following limitations, on top of the relatively small sample size. First, given its cross-sectional design, the results of this study could not imply causality, but are rather reflective of an association between exposure and outcome. However, in order to decrease potential reverse causation, subjects who reported having been diagnosed with a chronic disease or metabolic abnormality that may have influenced their dietary and lifestyle habits were excluded from the study [39]. Second, dietary assessment was performed using a FFQ, which may be limited by measurement errors, the number of food items constitutive of the food list and reliance on memory [39, 84]. However, and in order to offset the restriction dictated by the fixed FFQ food list, participants were asked, at the end of the interview, about ‘other foods that are usually consumed’. It is worth noting that although the adopted FFQ was not validated in our study population, it was previously used for the investigation of dietary patterns and their association with obesity and the metabolic syndrome in Lebanese adults and has yielded plausible findings [39, 73, 85]. Despite the possible limitations of the FFQ approach, it has been described as one of the most suitable dietary assessment methods in large epidemiological surveys as it assesses the participant’s habitual diet over longer periods of time [39, 86]. In addition, data collection was performed using a multi-component questionnaire in an interview setting, an approach that may be associated with social desirability bias, whereby participants may respond in a way that they believe is acceptable or favorable to the interviewer [39, 87]. Such a bias could occur when reporting dietary intake, smoking habits and particularly the amount of alcohol consumed, given the cultural context of the study and the religious norms in the country. Therefore, in our study, alcohol consumption was indicated as a dichotomous variable (yes/no), which does not fully capture alcohol intake in terms of frequency and quantities of alcohol consumed. In this study, the field workers who performed data collection underwent extensive training to decrease any judgmental verbal or non-verbal communication and thus to curtail social desirability bias. Another limitation in this study is that former smoking status was not included in the characterization of lifestyle patterns, although previous evidence suggests that former smokers are at a high risk of developing metabolic abnormalities, including the metabolic syndrome [88]. It is also important to note that we have included in our sample both overweight and obese individuals and not only obese. This may have diluted potential associations between diet, lifestyle and metabolic health. However, despite this potential dilution, we have observed significant associations between “lifestyle patterns” and MHOv/O. Finally, this study was restricted to the urban setting of the Greater Beirut area, and hence, findings related to food consumption and lifestyle characteristics may not be representative of less urban areas in the country, and future nationally representative studies are warranted.
Conclusion
The present study identified two main lifestyle patterns among Lebanese adults, the healthy and the unhealthy patterns, and showed that adherence to the healthy lifestyle pattern was associated with higher odds of MHOv/O. These findings provide evidence on the combined effect of lifestyle patterns in relation to cardiometabolic abnormalities that are complex and multifactorial in nature [28]. The fact that, in our study, individuals with lower SES were less likely to adhere to the healthy lifestyle pattern emphasizes the need for policies that address health inequities and ensure the accessibility of lifestyle-based interventions to socially disadvantaged groups [33]. Public health policy may promote the integration of lifestyle interventions in the healthcare system, interventions that are anchored in the community, its culture, norms and values [36].
References
Eckel RH, Kahn R, Robertson RM, Rizza RA (2006) Preventing cardiovascular disease and diabetes: a call to action from the American Diabetes Association and the American Heart Association. Diabetes Care 29(7):1697–1699. https://doi.org/10.1161/CIRCULATIONAHA.106.176583
Correia ML, Haynes WG, Mark AL (2005) Obesity-associated hypertension. Hypertension. https://doi.org/10.1161/01.hyp.0000151325.83008.b4
Blood Pressure Lowering Treatment Trialists’ Collaboration (2015) Effects of blood pressure lowering on cardiovascular risk according to baseline body-mass index: a meta-analysis of randomised trials. Lancet 385(9971):867–874. https://doi.org/10.1016/S0140-6736(14)61171-5
Yusuf S, Hawken S, Ôunpuu S, Dans T, Avezum A, Lanas F, McQueen M, Budaj A, Pais P, Varigos J (2004) Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case–control study. Lancet 364(9438):937–952. https://doi.org/10.1016/S0140-6736(04)17018-9
Eckel N, Li Y, Kuxhaus O, Stefan N, Hu FB, Schulze MB (2018) Transition from metabolic healthy to unhealthy phenotypes and association with cardiovascular disease risk across BMI categories in 90257 women (the Nurses’ Health Study): 30 year follow-up from a prospective cohort study. Lancet Diabetes Endocrinol. https://doi.org/10.1016/s2213-8587(18)30137-2
Adams KF, Schatzkin A, Harris TB, Kipnis V, Mouw T, Ballard-Barbash R, Hollenbeck A, Leitzmann MF (2006) Overweight, obesity, and mortality in a large prospective cohort of persons 50 to 71 years old. N Engl J Med 355(8):763–778. https://doi.org/10.1056/NEJMoa055643
Stefan N, Häring H-U, Hu FB, Schulze MB (2013) Metabolically healthy obesity: epidemiology, mechanisms, and clinical implications. Lancet Diabetes Endocrinol 1(2):152–162. https://doi.org/10.1016/S2213-8587(13)70062-7
Stefan N, Häring H-U, Schulze MB (2017) Metabolically healthy obesity: the low-hanging fruit in obesity treatment? Lancet Diabetes Endocrinol. https://doi.org/10.1016/s2213-8587(17)30292-9
Hinnouho G-M, Czernichow S, Dugravot A, Batty GD, Kivimaki M, Singh-Manoux A (2013) Metabolically healthy obesity and risk of mortality: does the definition of metabolic health matter? Diabetes Care. https://doi.org/10.2337/dc12-1654
Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, Fruchart JC, James WP, Loria CM, Smith SC Jr (2009) Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 120(16):1640–1645. https://doi.org/10.1161/circulationaha.109.192644
Wildman RP, Muntner P, Reynolds K, McGinn AP, Rajpathak S, Wylie-Rosett J, Sowers MR (2008) The obese without cardiometabolic risk factor clustering and the normal weight with cardiometabolic risk factor clustering: prevalence and correlates of 2 phenotypes among the US population (NHANES 1999–2004). Arch Intern Med 168(15):1617–1624
Karelis A, Rabasa-Lhoret R (2008) Inclusion of C-reactive protein in the identification of metabolically healthy but obese (MHO) individuals. Diabetes Metab 2(34):183–184
Stefan N, Kantartzis K, Machann J, Schick F, Thamer C, Rittig K, Balletshofer B, Machicao F, Fritsche A, Häring H-U (2008) Identification and characterization of metabolically benign obesity in humans. Arch Intern Med 168(15):1609–1616
Matsuda M, DeFronzo RA (1999) Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care 22(9):1462–1470
Karelis AD, Faraj M, Bastard J-P, St-Pierre DH, Brochu M, Prud’homme D, Rabasa-Lhoret R (2005) The metabolically healthy but obese individual presents a favorable inflammation profile. J Clin Endocrinol Metab 90(7):4145–4150. https://doi.org/10.1210/jc.2005-0482
Semple RK, Savage DB, Cochran EK, Gorden P, O’rahilly S (2011) Genetic syndromes of severe insulin resistance. Endocr Rev 32(4):498–514. https://doi.org/10.1210/er.2010-0020
Ahl S, Guenther M, Zhao S, James R, Marks J, Szabo A, Kidambi S (2015) Adiponectin levels differentiate metabolically healthy vs unhealthy among obese and nonobese white individuals. J Clin Endocrinol Metab 100(11):4172–4180
Calori G, Lattuada G, Piemonti L, Garancini MP, Ragogna F, Villa M, Mannino S, Crosignani P, Bosi E, Luzi L (2010) Prevalence, metabolic features and prognosis of metabolically healthy obese Italian individuals: the Cremona Study. Diabetes Care. https://doi.org/10.2337/dc10-0665
Hamer M, Stamatakis E (2012) Metabolically healthy obesity and risk of all-cause and cardiovascular disease mortality. J Clin Endocrinol Metab 97(7):2482–2488. https://doi.org/10.1210/jc.2011-3475
Matta J, Nasreddine L, Jomaa L, Hwalla N, Mehio Sibai A, Czernichow S, Itani L, Naja F (2016) Metabolically healthy overweight and obesity is associated with higher adherence to a traditional dietary pattern: a cross-sectional study among adults in Lebanon. Nutrients 8(7):432. https://doi.org/10.3390/nu8070432
Heinzle S, Ball G, Kuk J (2016) Variations in the prevalence and predictors of prevalent metabolically healthy obesity in adolescents. Pediatr Obes 11(5):425–433. https://doi.org/10.1111/ijpo.12083
Messier V, Karelis AD, Prud’homme D, Primeau V, Brochu M, Rabasa-Lhoret R (2010) Identifying metabolically healthy but obese individuals in sedentary postmenopausal women. Obesity 18(5):911–917. https://doi.org/10.1038/oby.2009.364
de Winter M, Rioux BV, Boudreau JG, Bouchard DR, Sénéchal M (2018) Physical activity and sedentary patterns among metabolically healthy individuals living with obesity. J Diabetes Res. https://doi.org/10.1155/2018/7496768
Fung MD, Canning KL, Mirdamadi P, Ardern CI, Kuk JL (2015) Lifestyle and weight predictors of a healthy overweight profile over a 20-year follow-up. Obesity 23(6):1320–1325. https://doi.org/10.1002/oby.21087
Phillips CM, Dillon C, Harrington JM, McCarthy VJ, Kearney PM, Fitzgerald AP, Perry IJ (2013) Defining metabolically healthy obesity: role of dietary and lifestyle factors. PLoS One 8(10):e76188. https://doi.org/10.1371/journal.pone.0076188
Hankinson AL, Daviglus ML, Horn LV, Chan Q, Brown I, Holmes E, Elliott P, Stamler J (2013) Diet composition and activity level of at risk and metabolically healthy obese American adults. Obesity 21(3):637–643. https://doi.org/10.1002/oby.20257
Kimokoti RW, Judd SE, Shikany JM, Newby P (2015) Metabolically healthy obesity is not associated with food intake in white or black men. J Nutr 145(11):2551–2561. https://doi.org/10.3945/jn.115.221283
Al Thani M, Al Thani AA, Al-Chetachi W, Al Malki B, Khalifa SA, Haj Bakri A, Hwalla N, Nasreddine L, Naja F (2016) A ‘high risk’ lifestyle pattern is associated with metabolic syndrome among Qatari women of reproductive age: a cross-sectional national study. Int J Mol Sci 17(6):698. https://doi.org/10.3390/ijms17060698
Steele EM, Claro RM, Monteiro CA (2014) Behavioural patterns of protective and risk factors for non-communicable diseases in Brazil. Public Health Nutr 17(2):369–375. https://doi.org/10.1017/S1368980012005472
Kanagasabai T, Dhanoa R, Kuk JL, Ardern CI (2017) Association between sleep habits and metabolically healthy obesity in adults: a cross-sectional study. J Obes. https://doi.org/10.1155/2017/5272984
Bell JA, Hamer M, van Hees VT, Singh-Manoux A, Kivimäki M, Sabia S (2015) Healthy obesity and objective physical activity. Am J Clin Nutr 102(2):268–275. https://doi.org/10.3945/ajcn.115.110924
Camhi SM, Crouter SE, Hayman LL, Must A, Lichtenstein AH (2015) Lifestyle behaviors in metabolically healthy and unhealthy overweight and obese women: a preliminary study. PLoS One 10(9):e0138548. https://doi.org/10.1371/journal.pone.0138548
Pérez-Rodrigo C, Gianzo-Citores M, Gil Á, González-Gross M, Ortega RM, Serra-Majem L, Varela-Moreiras G, Aranceta-Bartrina J (2017) Lifestyle patterns and weight status in spanish adults: the ANIBES study. Nutrients 9(6):606. https://doi.org/10.3390/nu9060606
Moschonis G, Kalliora AC, Costarelli V, Papandreou C, Koutoukidis D, Lionis C, Chrousos GP, Manios Y (2014) Identification of lifestyle patterns associated with obesity and fat mass in children: the Healthy Growth Study. Public Health Nutr 17(3):614–624. https://doi.org/10.1017/S1368980013000323
Waidyatilaka I, de Silva A, de Lanerolle-Dias M, Wickremasinghe R, Atukorala S, Somasundaram N, Lanerolle P (2014) Lifestyle patterns and dysglycaemic risk in urban Sri Lankan women. Br J Nutr 112(6):952–957. https://doi.org/10.1017/S0007114514001676
Al Thani M, Al Thani AA, Al-Chetachi W, Al Malki B, Khalifa SA, Bakri AH, Hwalla N, Nasreddine L, Naja F (2015) Lifestyle patterns are associated with elevated blood pressure among qatari women of reproductive age: a cross-sectional national study. Nutrients 7(9):7593–7615. https://doi.org/10.3390/nu7095355
Nasreddine L, Naja F, Sibai A-M, Helou K, Adra N, Hwalla N (2014) Trends in nutritional intakes and nutrition-related cardiovascular disease risk factors in Lebanon: the need for immediate action. J Med Liban 103(1151):1–9. https://doi.org/10.12816/0004102
Naja F, Shatila H, Meho L, Alameddine M, Haber S, Nasreddine L, Sibai AM, Hwalla N (2017) Gaps and opportunities for nutrition research in relation to non-communicable diseases in Arab countries: call for an informed research agenda. Nutr Res. https://doi.org/10.1016/j.nutres.2017.07.011
Nasreddine L, Tamim H, Itani L, Nasrallah MP, Isma’eel H, Nakhoul NF, Abou-Rizk J, Naja F (2018) A minimally processed dietary pattern is associated with lower odds of metabolic syndrome among Lebanese adults. Public Health Nutr 21(1):160–171. https://doi.org/10.1017/S1368980017002130
Committee IR (2005) Guidelines for data processing and analysis of the International Physical Activity Questionnaire (IPAQ)-short and long forms. http://www.sdp.univ.fvg.it/sites/default/files/IPAQ_English_self-admin_short.pdf. Accessed 20 June 2018
Pellet P, Shadarevian S (1970) Food composition: tables for use in the Middle East, 2nd edn. American University of Beirut, Beirut, Lebanon
Millen BE, Morgan JL (1996) The 2D food portion visual. Nutrition and Consulting Enterprises, Framingham, MA
Blüher M (2012) Are there still healthy obese patients? Curr Opin Endocrinol Diabetes Obes 19(5):341–346. https://doi.org/10.1097/MED.0b013e328357f0a3
McCann SE, Marshall JR, Brasure JR et al (2001) Analysis of patterns of food intake in nutritional epidemiology: food classification in principal components analysis and the subsequent impact on estimates for endometrial cancer. Public Health Nutr 4:989–997. https://doi.org/10.1079/PHN2001168
Fleiss J et al (2003) Statistical methods for rates and proportions. http://www.sample-size.net/proportions-effect-size/. Accessed 30 June 2018
Hoffmann I (2003) Transcending reductionism in nutrition research. Am J Clin Nutr 78(3):514S–516S. https://doi.org/10.1093/ajcn/78.3.514S
Kim S, Lee H, Lee D-C, Lee H-S, Lee J-W (2014) Predominance of small dense LDL differentiates metabolically unhealthy from metabolically healthy overweight adults in Korea. Metabolism 63(3):415–421. https://doi.org/10.1016/j.metabol.2013.11.015
Hong HC, Lee J-S, Choi HY, Yang SJ, Yoo HJ, Seo JA, Kim SG, Kim NH, Baik SH, Choi DS (2013) Liver enzymes and vitamin D levels in metabolically healthy but obese individuals: Korean National Health and Nutrition Examination Survey. Metabolism 62(9):1305–1312. https://doi.org/10.1016/j.metabol.2013.04.002
Mathew H, Farr OM, Mantzoros CS (2016) Metabolic health and weight: understanding metabolically unhealthy normal weight or metabolically healthy obese patients. Metab Clin Exp 65(1):73–80
Gonçalves CG, Glade MJ, Meguid MM (2016) Metabolically healthy obese individuals: key protective factors. Nutrition 32(1):14–20. https://doi.org/10.1016/j.nut.2015.07.010
Steffen LM, Jacobs DR Jr, Stevens J, Shahar E, Carithers T, Folsom AR (2003) Associations of whole-grain, refined-grain, and fruit and vegetable consumption with risks of all-cause mortality and incident coronary artery disease and ischemic stroke: the Atherosclerosis Risk in Communities (ARIC) Study. Am J Clin Nutr 78(3):383–390. https://doi.org/10.1093/ajcn/78.3.383
Liu S, Manson JE, Lee I-M, Cole SR, Hennekens CH, Willett WC, Buring JE (2000) Fruit and vegetable intake and risk of cardiovascular disease: the Women’s Health Study. Am J Clin Nutr 72(4):922–928. https://doi.org/10.1093/ajcn/72.4.922
McCullough ML, Bandera EV, Patel R, Patel AV, Gansler T, Kushi LH, Thun MJ, Calle EE (2007) A prospective study of fruits, vegetables, and risk of endometrial cancer. Am J Epidemiol 166(8):902–911. https://doi.org/10.1093/aje/kwm156
Naja F, Hwalla N, Itani L, Baalbaki S, Sibai A, Nasreddine L (2015) A novel Mediterranean diet index from Lebanon: comparison with Europe. Eur J Nutr 54(8):1229–1243. https://doi.org/10.1007/s00394-014-0801-1
St-Onge M, Wolfe S, Sy M, Shechter A, Hirsch J (2014) Sleep restriction increases the neuronal response to unhealthy food in normal-weight individuals. Int J Obes 38(3):411. https://doi.org/10.1038/ijo.2013.114
Spiegel K, Tasali E, Leproult R, Van Cauter E (2009) Effects of poor and short sleep on glucose metabolism and obesity risk. Nat Rev Endocrinol 5(5):253. https://doi.org/10.1038/nrendo.2009.23
Spiegel K, Knutson K, Leproult R, Tasali E, Cauter EV (2005) Sleep loss: a novel risk factor for insulin resistance and type 2 diabetes. J Appl Physiol 99(5):2008–2019. https://doi.org/10.1152/japplphysiol.00660.2005
Spiegel K, Leproult R, Van Cauter E (1999) Impact of sleep debt on metabolic and endocrine function. Lancet 354(9188):1435–1439. https://doi.org/10.1016/S0140-6736(99)01376-8
de Rooij BH, van der Berg JD, van der Kallen CJ, Schram MT, Savelberg HH, Schaper NC, Dagnelie PC, Henry RM, Kroon AA, Stehouwer CD (2016) Physical activity and sedentary behavior in metabolically healthy versus unhealthy obese and non-obese individuals—the Maastricht study. PLoS One 11(5):e0154358. https://doi.org/10.1371/journal.pone.0154358
Poelkens F, Eijsvogels TM, Brussee P, Verheggen RJ, Tack CJ, Hopman MT (2014) Physical fitness can partly explain the metabolically healthy obese phenotype in women. Exp Clin Endocrinol Diabetes 122(2):87–91. https://doi.org/10.1055/s-0033-1363686
Velho S, Paccaud F, Waeber G, Vollenweider P, Marques-Vidal P (2010) Metabolically healthy obesity: different prevalences using different criteria. Eur J Clin Nutr 64(10):1043. https://doi.org/10.1038/ejcn.2010.114
Brown R, Kuk J (2015) Consequences of obesity and weight loss: a devil’s advocate position. Obes Rev 16(1):77–87. https://doi.org/10.1111/obr.12232
van der Heijden G-J, Toffolo G, Manesso E, Sauer PJ, Sunehag AL (2009) Aerobic exercise increases peripheral and hepatic insulin sensitivity in sedentary adolescents. J Clin Endocrinol Metab 94(11):4292–4299. https://doi.org/10.1210/jc.2009-1379
Park Y-MM, Steck SE, Fung TT, Zhang J, Hazlett LJ, Han K, Lee S-H, Kwon H-S, Merchant AT (2017) Mediterranean diet, dietary approaches to stop hypertension (DASH) style diet, and metabolic health in US adults. Clin Nutr 36(5):1301–1309. https://doi.org/10.1016/j.clnu.2016.08.018
Jonnalagadda SS, Harnack L, Hai Liu R, McKeown N, Seal C, Liu S, Fahey GC (2011) Putting the whole grain puzzle together: health benefits associated with whole grains—summary of American Society for Nutrition 2010 satellite symposium. J Nutr 141(5):1011S–1022S. https://doi.org/10.3945/jn.110.132944
Slavin J (2013) Fiber and prebiotics: mechanisms and health benefits. Nutrients 5(4):1417–1435. https://doi.org/10.3390/nu5041417
Liu RH (2013) Health-promoting components of fruits and vegetables in the diet. Adv Nutr 4(3):384S–392S. https://doi.org/10.3945/an.112.003517
Esmaillzadeh A, Kimiagar M, Mehrabi Y, Azadbakht L, Hu FB, Willett WC (2006) Fruit and vegetable intakes, C-reactive protein, and the metabolic syndrome. Am J Clin Nutr 84(6):1489–1497. https://doi.org/10.1093/ajcn/84.6.1489
Fernandez ML, West KL (2005) Mechanisms by which dietary fatty acids modulate plasma lipids. J Nutr 135(9):2075–2078. https://doi.org/10.1093/jn/135.9.2075
Lopez S, Bermudez B, Ortega A, Varela LM, Pacheco YM, Villar J, Abia R, Muriana FJ (2011) Effects of meals rich in either monounsaturated or saturated fat on lipid concentrations and on insulin secretion and action in subjects with high fasting triglyceride concentrations. Am J Clin Nutr 93(3):494–499. https://doi.org/10.3945/ajcn.110.003251
Zemel MB (2003) Mechanisms of dairy modulation of adiposity. J Nutr 133(1):252S–256S. https://doi.org/10.1093/jn/133.1.252S
Zemel MB (2003) Role of dietary calcium and dairy products in modulating adiposity. Lipids 38(2):139–146. https://doi.org/10.1007/s11745-003-1044-6
Naja F, Nasreddine L, Itani L, Chamieh MC, Adra N, Sibai AM, Hwalla N (2011) Dietary patterns and their association with obesity and sociodemographic factors in a national sample of Lebanese adults. Public Health Nutr 14(9):1570–1578. https://doi.org/10.1017/S136898001100070X
Du D, Bruno R, Dwyer T, Venn A, Gall S (2017) Associations between alcohol consumption and cardio-metabolic risk factors in young adults. Eur J Prev Cardiol 24(18):1967–1978. https://doi.org/10.1177/2047487317724008
O’Keefe JH, Bhatti SK, Patil HR, DiNicolantonio JJ, Lucan SC, Lavie CJ (2013) Effects of habitual coffee consumption on cardiometabolic disease, cardiovascular health, and all-cause mortality. J Am Coll Cardiol 62(12):1043–1051. https://doi.org/10.1016/j.jacc.2013.06.035
Patino-Alonso MC, Recio-Rodríguez JI, Belio JFM, Colominas-Garrido R, Lema-Bartolomé J, Arranz AG, Agudo-Conde C, Gomez-Marcos MA, García-Ortiz L, Group E (2014) Factors associated with adherence to the Mediterranean diet in the adult population. J Acad Nutr Diet 114(4):583–589. https://doi.org/10.1016/j.jand.2013.07.038
Hu EA, Toledo E, Diez-Espino J, Estruch R, Corella D, Salas-Salvado J, Vinyoles E, Gomez-Gracia E, Aros F, Fiol M (2013) Lifestyles and risk factors associated with adherence to the Mediterranean diet: a baseline assessment of the PREDIMED trial. PLoS One 8(4):e60166. https://doi.org/10.1371/journal.pone.0060166
Nasreddine L, Hwalla N, Sibai A, Hamzé M, Parent-Massin D (2006) Food consumption patterns in an adult urban population in Beirut, Lebanon. Public Health Nutr 9(2):194–203. https://doi.org/10.1079/PHN2005855
Kim S, Symons M, Popkin BM (2004) Contrasting socioeconomic profiles related to healthier lifestyles in China and the United States. Am J Epidemiol 159(2):184–191. https://doi.org/10.1093/aje/kwh006
Appelhans BM, Milliron B-J, Woolf K, Johnson TJ, Pagoto SL, Schneider KL, Whited MC, Ventrelle JC (2012) Socioeconomic status, energy cost, and nutrient content of supermarket food purchases. Am J Prev Med 42(4):398–402. https://doi.org/10.1016/j.amepre.2011.12.007
Darmon N, Drewnowski A (2008) Does social class predict diet quality? Am J Clin Nutr 87(5):1107–1117. https://doi.org/10.1093/ajcn/87.5.1107
Giskes K, Avendaňo M, Brug J, Kunst A (2010) A systematic review of studies on socioeconomic inequalities in dietary intakes associated with weight gain and overweight/obesity conducted among European adults. Obes Rev 11(6):413–429. https://doi.org/10.1111/j.1467-789X.2009.00658.x
Pechey R, Jebb SA, Kelly MP, Almiron-Roig E, Conde S, Nakamura R, Shemilt I, Suhrcke M, Marteau TM (2013) Socioeconomic differences in purchases of more vs. less healthy foods and beverages: analysis of over 25,000 British households in 2010. Soc Sci Med 92:22–26. https://doi.org/10.1016/j.socscimed.2013.05.012
Willett W (1998) Nutritional epidemiology, 2nd edn. Oxford University Press, New York
Naja F, Nasreddine L, Itani L, Adra N, Sibai A, Hwalla N (2013) Association between dietary patterns and the risk of metabolic syndrome among Lebanese adults. Eur J Nutr 52(1):97–105. https://doi.org/10.1007/s00394-011-0291-3
Caan BJ, Lanza E, Schatzkin A, Coates AO, Brewer BK, Slattery ML, Marshall JR, Bloch A (1999) Does nutritionist review of a self-administered food frequency questionnaire improve data quality? Public Health Nutr 2(4):565–569. https://doi.org/10.1017/S1368980099000750
Okamoto K, Ohsuka K, Shiraishi T, Hukazawa E, Wakasugi S, Furuta K (2002) Comparability of epidemiological information between self-and interviewer-administered questionnaires. J Clin Epidemiol 55(5):505–511. https://doi.org/10.1016/S0895-4356(01)00515-7
Kolovou GD, Kolovou V, Mavrogeni S (2016) Cigarette smoking/cessation and metabolic syndrome. Clin Lipidol 11(1):6–14
Acknowledgements
We would like to acknowledge the contribution of Mrs. Hibeh Shatila for coordinating data analysis and editing the manuscript.
Funding
The original survey was supported by a Grant from the National Council for Scientific Research (LNCSR) (L. N., award number 02-06-14); the American University of Beirut Research Board (L. N. award number 103006/22691); the Medical Practice Plan at the American University of Beirut (H. T., award number 100410, project number 20061); and the Novo Nordisk development programme. The funders had no role in the design, analysis or writing of this article.
Author information
Authors and Affiliations
Contributions
LN and FN formulated the research question. LN drafted the manuscript and contributed to the interpretation of the data. HT led the parent study. MN and LN provided support in conceiving and designing the parent study. LI contributed to data analysis and interpretation. LN, HT and MN acquired the data. FN conducted data analysis and contributed to data interpretation and write up of the manuscript. All authors critically reviewed the manuscript and guarantee the accuracy of their input and the integrity of this work.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Appendix 1
Appendix 1
See Table 5.
Rights and permissions
About this article
Cite this article
Naja, F., Itani, L., Nasrallah, M.P. et al. A healthy lifestyle pattern is associated with a metabolically healthy phenotype in overweight and obese adults: a cross-sectional study. Eur J Nutr 59, 2145–2158 (2020). https://doi.org/10.1007/s00394-019-02063-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-019-02063-9