Abstract
Purpose
Obesity leads to the clustering of cardiovascular (CV) risk factors and the metabolic syndrome (MetS) also in children and is often accompanied by non-alcoholic fatty liver disease. Quality of dietary fat, beyond the quantity, can influence CV risk profile and, in particular, omega-3 fatty acids (FA) have been proposed as beneficial in this setting. The aim of the study was to evaluate the associations of individual CV risk factors, characterizing the MetS, with erythrocyte membrane FA, markers of average intake, in a group of 70 overweight/obese children.
Methods
We conducted an observational study. Erythrocyte membrane FA were measured by gas chromatography. Spearman correlation coefficients (rS) were calculated to evaluate associations between FA and features of the MetS.
Results
Mean content of Omega-3 FA was low (Omega-3 Index = 4.7 ± 0.8%). Not omega-3 FA but some omega-6 FA, especially arachidonic acid (AA), were inversely associated with several features of the MetS: AA resulted inversely correlated with waist circumference (rS = − 0.352), triglycerides (rS = − 0.379), fasting insulin (rS = − 0.337) and 24-h SBP (rS = − 0.313). Total amount of saturated FA (SFA) and specifically palmitic acid, correlated positively with waist circumference (rS = 0.354), triglycerides (rS = 0.400) and fasting insulin (rS = 0.287). Fatty Liver Index (FLI), a predictive score of steatosis based on GGT, triglycerides and anthropometric indexes, was positively correlated to palmitic acid (rS = 0.515) and inversely to AA (rS = − 0.472).
Conclusions
Our data suggest that omega-6 FA, and especially AA, could be protective toward CV risk factors featuring the MetS and also to indexes of hepatic steatosis in obese children, whereas SFA seems to exert opposite effects.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In the last few decades, the lifestyle changes in western countries led to an increase in the prevalence of overweight/obesity and cardiovascular (CV) risk factors, which often accompany the body weight excess, not only in adults but also in children [1]. In particular, a central distribution of obesity is associated with a higher CV risk profile through the clustering of CV risk factors that leads to the so-called metabolic syndrome (MetS). Insulin resistance often accompanies obesity and plays a pivotal pathophysiological role in the development of MetS. Non-alcoholic fatty liver disease (NAFLD) can be considered the hepatic manifestation of MetS because of the common risk factors, like central obesity, insulin resistance and dyslipidemia [2]. NAFLD by itself is associated with an increased risk of cardiovascular disease [3].
An unbalanced diet is a common factor that might promote several of the components of MetS and NAFLD. A hyperlipidemic diet may influence the onset and progression of obesity; however, increasing attention is paid not only to the quantity but also to the quality of fat. Indeed, epidemiological and intervention studies indicate that saturated fatty acids (SFA) might negatively affect insulin sensitivity [4] and plasma lipid profile [5]. However, randomized controlled trials (RCTs) and successive meta-analyses gave controversial results about the link between SFA and cardiovascular risk [6, 7]. Anyhow, there is general agreement that dietary assumption of SFA should be lower than 10% but which is the best macronutrient to replace them remains a matter of debate.
On the other hand, omega-3 polyunsaturated fatty acids (PUFA) are generally recognized as healthy nutrients. They positively affect plasma lipid profile, insulin sensitivity [8], might exert beneficial effect on blood pressure (BP) [9,10,11] and on abdominal obesity [12] and, moreover, can play an important role in prevention and treatment of NAFLD, even in children [2]. Moreover, omega-3 PUFA have anti-inflammatory activity, which may play a role also in the development of CV diseases [13]. The increasing interest in the potential beneficial effect of omega-3 PUFA and the concern that a high omega-6/omega-3 PUFA ratio is associated with an increased cardiovascular risk led to the advisement of reducing the dietary intake of omega-6 PUFA, although insufficient evidence links the omega-6/omega-3 PUFA ratio to the clinical outcomes [14]; anyhow the role of total and single omega-6 PUFA in cardiovascular disease is not clarified yet [15].
Notably, new insights indicate a protective role of omega-6 FA, and in particular linoleic acid (LA), with respect to body fat distribution and insulin resistance [16] and total mortality [17]. A longitudinal study showed an inverse relation between an increase in serum omega-6 PUFA and the prevalence of MetS in a Finnish population [18]. Thus, further investigations are needed to clarify these issues in adults and even more in children. Therefore, we aimed at investigating the associations of individual CV risk factors, characterizing the MetS, with erythrocyte membrane FA, markers of the preceding 2–3 months intake, in a group of overweight and obese children.
Methods
Overweight and obese children were recruited from the “Pediatric Obesity Outpatients Unit” of the University Hospital of Verona and of the “Local Health Unit n. 20” (ASL 20) of Verona. Inclusion criteria were: children and adolescents aged 5–18 years; overweight or obesity (BMI ≥ 90th and 95th percentile for sex and age, respectively). WHO reference for BMI was used for categorizing children into the overweight and obese groups [19]. Exclusion criteria were: hepatic or renal chronic diseases, malignancies, diabetes mellitus, other therapies potentially affecting glucose and/or lipid metabolism; obesity secondary to genetic disorders and/or syndromes.
Study design
The study was conducted according to a cross-sectional observational design. The study was approved by the Ethical Committee of Verona (CE n. 2218), and written informed consent was obtained from each participant’s parents.
Assessments
Each child was evaluated in a single occasion, between 8 and 9 a.m. A questionnaire was administered to the patients and to the parents, dealing with medical history, family history, physiological and pathological information and use of drugs. Then, the participants underwent a physical examination. They were advised not to engage in strenuous exercise and to avoid consuming caffeine-containing beverages within 12 h preceding the vascular studies.
During the visit, blood pressure was measured with a semiautomatic oscillometric device (TM-2551, A&D instruments Ltd, Abingdon Oxford, UK) for 3 times, 3 min apart with the patient lying supine for at least 10 min before the first measurement in a room with controlled temperature (22–24 °C). Ambulatory blood pressure measurement was recorded by an oscillometric device (Spacelabs 90217; Spacelabs Inc., Issaquah, Washington, USA), which measured BP every 15 min during the day and every 30 min during the night. Children and parents recorded physical activities, resting and sleeping time and symptoms on a dedicated diary. After recording, the daytime and nighttime periods (set to default at 0700 and 2200 h, respectively) were adapted to “real” awake and sleep times according to what was declared in the diary of activity, as previously indicated [20].
All of the values derived from BP measurements were transformed in z-score and percentile, according to normative values [21, 22].
Body weight, height, and waist and hip circumferences were measured with the patient wearing light clothes. Body weight was measured by a calibrated balance and height by a calibrated stadiometer [23].
Waist circumference was transformed in z score and percentile according to normative values [24]. Expert pediatricians defined the pubertal status on the basis of Tanner stages; children with a Tanner stage ≥ 3 were classified as pubescent.
Metabolic syndrome definition
In an exploratory analysis, we defined the MetS according to the diagnostic criteria suggested by the International Diabetes Federation [25]. Anyhow we decided to extend the diagnosis to all age categories and replace the suggested cut-off point of BP with the 90th percentile for sex and age of SBP and DBP, according to the normative values [22] and to the definition of pre-hypertension/hypertension indicated by the current guidelines [26].
Laboratory measurements
Blood samples were collected after an overnight fast. Laboratory measurements, including fasting plasma glucose, insulin, total cholesterol, HDL-cholesterol, triglycerides, AST, ALT and GGT were measured using standardized methods. Insulin resistance has been estimated with HOMA Index (HOMA-IR), which was calculated by Matthews formula (fasting insulin (mU/mL) × fasting glucose (mmol/L)/22.5) [27].
Fatty liver index (FLI), derived from an algorithm based on BMI, waist circumference, triglycerides and GGT were calculated as previously described [28].
Red blood cell membrane fatty acid measurement
EDTA–blood tubes were centrifuged, plasma and buffy coat taken off, and erythrocytes frozen at − 80 °C until analysis. Erythrocyte fatty acid composition was analyzed using the HS-Omega-3 Index® methodology as previously described [29]. Fatty acid methyl esters were generated from erythrocytes by acid transesterification and analyzed by gas chromatography using a GC2010 Gas Chromatograph (Shimadzu, Duisburg, Germany) equipped with a SP2560, 100-m column (Supelco, Bellefonte, PA, USA) using hydrogen as carrier gas. Fatty acids were identified by comparison with a standard mixture of fatty acids characteristic of erythrocytes. A total of 26 fatty acids were identified and quantified.
Results are given as percentage of total identified fatty acids after response factor correction. The coefficient of variation for EPA plus DHA and for most other fatty acids was 4%. Analyses were quality controlled according to DIN ISO 15189.
Estimation of Δ9, Δ6 and Δ5 desaturase activity
Δ9, Δ6 and Δ5 desaturase are enzymes responsible for the endogenous formation of monounsaturated and polyunsaturated FA and their activity has been associated with insulin–glucose homeostasis and with central obesity [30].
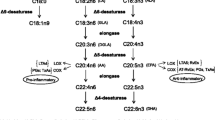
We estimated the desaturase activity as the ratio of product to precursor of individual red blood cell membrane FA as follows: Δ9-desaturase (SCD) = C16:1n − 7/C16:0 and C18:n − 9/C18:0 (they will be referred to as SCD-16 and SCD-18, respectively); Δ6-desaturase (D6D) = C18:3n − 6/C18:2n − 6 and Δ5-desaturase (D5D) = C20:4n − 6/C20:3n − 6.
Hepatic ultrasonography
Children underwent abdomen ultrasonography (US) using a convex probe (ACUSON S2000TM system, Siemens, Erlanger, Germany). The presence of significant liver steatosis was defined by an experience sonographer.
Statistics
Data are presented as median and range unless otherwise stated. The normal distribution of each variable was evaluated by the Kolmogorov–Smirnov test. Differences in the measured parameters between groups (MetS presence, steatosis, gender, pubertal state) were analyzed by T test or Mann–Whitney U test, as appropriate, at univariate analysis, and by unconditional logistic regression models at multivariate analysis. Unless otherwise specified, covariates included in the multivariate models were age, sex, BMI percentile and the features associated, at univariate analysis, with MetS presence, steatosis, gender, and pubertal state, respectively.
Since MetS-associated features were non-normally distributed, bivariate correlations were estimated by the non-parametric Spearman correlation coefficient (rS). After log-transformation of MetS-associated features, general linear models were applied to assess the association of FA with MetS-associated features, after adjustment for age, sex and BMI.
Two-tailed tests with a p < 0.05 were considered statistically significant in the main analysis. To take into account the multiple comparisons, along with original p values, the false discovery rate (FDR) adjusted p values were also calculated and reported in the tables, where appropriate. The analyses were performed with Statistical Analysis System (SAS) Software, version 9.2.
Results
General characteristics
Seventy patients (40 males and 30 females) were included in the study. Median age was 11 years (range 5–17 years); median BMI was 29.0 kg/m2 (range 23.0–42.7 kg/m2), which in all cases was higher than the 90th percentile, as for inclusion criteria. Five children (7.1%) fulfilled the diagnostic criteria of MetS. Sixty-three children underwent abdomen ultrasonography (US) and in 34 (53%) children hepatic steatosis was detected.
The average omega-3 index was 4.7 ± 0.8%; mean value of ALA was 0.08 ± 0.03%, and of LA was 12.0 ± 1.6%. Anthropometric, clinical and biochemical characteristics of the children are listed in Table 1, and the FA contents of erythrocytes in Online Resource 1.
Correlations between red blood cell membrane FA and features of Mets in the whole sample
Omega-3 PUFA, as well as EPA and DHA, correlated positively with waist circumference; moreover, EPA showed positive correlations with insulin, HOMA-IR and with FLI (Table 2).
On the contrary, total omega-6 PUFAs (LA + GLA + DGLA + AA + docosatetraenoic acid (DTA) + eicosadienoic acid + C22:5n6) was inversely correlated with several anthropometric and laboratory measurements related to MetS; within omega-6 PUFA, AA in particular was associated with almost the same characteristics as the class of FA (Table 2; Fig. 1). Conversely, within omega-6 PUFA family, GLA showed opposite correlations with the CV risk factors, as compared to AA, and, in particular, it was positively correlated with some features of Mets, especially with total cholesterol, triglycerides, fasting insulin, HOMA-IR and FLI.
Total content of SFA was positively correlated with several features of the MetS, within SFA, palmitic acid (PA) in particular positively correlated with several individual characteristics (Table 2; Fig. 2).
Omega-9 FAs and trans-FAs, when considered either the single FA or their sum, did not show significant correlations with anthropometric, clinical and laboratory parameters of MetS.
Correlations between red blood cell membrane FA and features of NAFLD in the whole sample
As for the correlation with individual components of the MetS, omega-6 PUFA and in particular AA showed an inverse correlation with FLI and transaminases, whereas SFA, and particularly PA, resulted positively correlated (see Table 2).
Correlations between desaturase activity and features of MetS in the whole sample
D6D activity showed positive correlations with waist circumference (rS = 0.249), triglycerides (rS = 0.370), fasting insulin (rS = 0.404), HOMA-IR (rS = 0.423) and with FLI (rS = 0.402).
D5D activity was inversely correlated to triglycerides (rS = − 0.401), and FLI (rS = − 0.319) (in Online Resource 4).
No significant differences were found in desaturase activity according to gender, pubertal status and presence of hepatic steatosis.
Regressions
After adjustment for sex and age all the correlations shown in Table 2 remained significant. When also BMI was included in the regression most associations remained significant, especially the ones with laboratory parameters (see Table 2 where significant associations even after full adjustment are underlined).
Analysis by subgroups
When comparing children with and without MetS, we observed a significantly higher SBP, triglycerides, PA and D6D and lower HDL-cholesterol, AA and omega-6 in patients with MetS, after adjustment by age and BMI. Otherwise, no feature was associated with liver steatosis at multivariate analysis. The characteristics of the subgroups are detailed in Online Resource 2 and in Online Resource 3. The correlations of FA profile with the clinical parameters found in the whole sample were mainly confirmed in both subgroups of children with and without steatosis, with higher evidence in the steatosis group, especially for the associations with NAFLD characteristics. The correlations of FA with the features of the MetS and of NAFLD, divided according the presence or absence of hepatic steatosis, are detailed in the Online Resource 5.
Discussion
The main result of our study is the evidence that omega-6 FA, and in particular AA, are inversely associated, whereas SFA positively, with many components of MetS in obese children. Even if the observational design of the study does not allow to prove a causal link, our results could suggest, on the one side, a protective role of omega-6 FA and, on the other side, a harmful effect of SFA with respect to cardiovascular risk factors and NAFLD.
Anyhow, these associations of opposite sign could simply reflect a higher concentration of omega-6 PUFA with respect to SFA that, on turn, could reflect a healthier dietary habit. Therefore, the relative increase in omega-6 PUFA could not be, or not only, protective per se but instead be a marker of the reduction of other potentially harmful components of the diet, like saturated FA. Indeed, a diet rich in polyunsaturated FA is often associated with healthier dietary pattern, which may involve also other macronutrients [31].
Despite an increasing expectation from a beneficial effect on cardiovascular risk profile and NAFLD in children by omega-3 PUFA, and in particular EPA and DHA [2], we did not find any significant beneficial association between either EPA or DHA and clinical/laboratory characteristics of our population. On the contrary, we found a positive association of EPA with some characteristics of the MetS, in particular insulin and HOMA-IR. The effect of omega-3 PUFA on glucose metabolism is not clearly defined and trials in humans did not give univocal results [32, 33]. In our sample, that was taking a free diet without any supplement, the level of omega-3 PUFA was very low, far below the 8% threshold suggested for CV protection [34] and this could also be a confounding element.
We choose red blood cell membrane FA as a biomarker of dietary intake of FA because of the stability of their values, especially of essential FA, which reflects the mean dietary intake in the preceding few months [35], and because it is more reliable compared to dietary self-report or questionnaire, especially in children.
Our results confirm that total amount of SFA is associated with an unfavorable cardiovascular risk profile in obese children and, within this family of FA, palmitic acid show to be related to almost all the characteristics of the MetS. It has been already shown that palmitic acid is one of the most abundant circulating FA in obese children [36] and elevated palmitic acid levels can affect insulin homeostasis, which is the principal etiologic driver of the metabolic abnormalities clustering in the MetS. In particular, in vitro studies reported an impairment in insulin secretion in murine [37] and human [38] β-cell lines due to palmitic acid, generally referred to as lipotoxicity. A few in vivo studies support these results: one study in adults reported an altered postprandial insulin secretion and sensitivity in response to a high dietary intake of palmitic acid [39], another study in children and adolescents showed that obese subjects with higher circulating levels of palmitic acid have an increased and delayed insulin secretion [40]. Furthermore, it has been shown that palmitic acid in plasma triglycerides was higher in abdominally obese adults as compared to the controls without central obesity and this FA profile was positively correlated to HOMA-IR [41].
As already stated above, our results might indicate also a protective role of omega-6 PUFA, and in particular of AA, with respect to several cardiovascular risk factors. Previous studies investigating the effect of omega-6 in cardiovascular disease gave conflicting results, some suggesting no association between omega-6 PUFA and the risk of hypertension [42] and other that the replacement of SFA with omega-6 PUFA is not associated with vascular and endothelial function but might improve BP [43]. Even the guidelines for fat intake of the German Society of Nutrition support a possible protective effect on CV disease when SFA are replaced with omega-6 PUFA, especially for primary and secondary prevention of coronary heart disease [14]. Indeed, dietary intake of LA may contribute to prevention and control of elevated blood pressure [44]. Thus, although omega-6 PUFA have been for a long time counter-posed to the beneficial omega-3 PUFA, recent insight supports their potential cardiovascular protective effect [17].
Even when considering specifically AA, the omega-6 which drives the observed associations, available data show contrasting results. A higher amount of AA was found in adipose tissue in metabolically unhealthy obese adults compared to metabolically healthy control [45]. Then, in healthy children, AA, measured in adipose tissue and in skeletal muscle cells, was positively correlated to fasting insulin and HOMA-IR [46]. Nevertheless, some studies indicated a protective effect of AA, especially when measured in red blood cell membranes, on glucose–insulin homeostasis [47, 48], even if some observations suggest a beneficial effect in subject with low–normal insulin sensitivity but not in highly insulin-sensitive individuals [49].
It is not clear why GLA, in contrast to the other omega-6 FA, is positively correlated with a poorer metabolic profile. On the one side, previous studies have indicated that GLA leads to the production of anti-inflammatory compounds, but GLA may also inhibit the metabolism of AA, at least in some types of cells [50].
Only a few studies investigated the role of GLA and DGLA in humans and their biological actions remain still unclear. A recently published nested case-cohort multicentric study showed that a fatty acid pattern, measured on plasma phospholipids, including high concentration of LA and low concentration of GLA and PA, was associated with lower incidence of type 2 diabetes mellitus in a sample of more than 27,000 adults with a median follow-up of 11.9 years [51]. On the contrary, the large observational Ludwigshafen Risk and CV Health Study showed that GLA was inversely correlated to all-cause and CV mortality, even after adjustment for traditional risk factors [52]. The same study showed no significant correlations of AA with the outcomes [52].
Moreover, DGLA, which is yielded from GLA, showed to be positively associated with the risk of type 2 diabetes mellitus, when comparing the highest with the lowest quartile of plasma phospholipids DGLA in a sample of Japanese adults followed up for 5 years [53]. Also in a sample of Chinese children, DGLA was positively associated with HOMA-IR, whereas AA showed an inverse correlation [54]. By contrast, in two studies conducted in patients with coronary artery disease DGLA resulted beneficially associated with CV events and with mortality [55, 56].
Moreover, GLA is yielded from LA by D6D and leads to the production of DGLA, which is the substrate of D5D. Therefore, the association of GLA and DGLA with the clinical parameters could also reflect the activity of the desaturase enzymes.
In fact, also the role of fatty acid desaturase (SCD, D6D and D5D) has been linked to visceral obesity [30] and insulin resistance, suggesting an increased SCD and D6D and a decreased D5D activity in subjects with impaired insulin sensitivity and related disorders [57,58,59]. Our results support the findings of previous studies in adults showing that the estimated D6D activity is positively related to a poorer metabolic profile, whereas D5D activity is inversely related.
In addition, omega-6 PUFA, SFA and D6D showed to be correlated with waist circumference, marker of central adiposity, supporting the association of dietary fat with the body fat distribution and the cardiometabolic profile, even if the association disappears after further adjustment for BMI [60, 61].
Furthermore, the associations of the different FA and FA families with a possible hepatic involvement, configuring the NAFLD, beside the correlation with the individual components of the Mets, support the hypothesis that NAFLD represents a continuum with MetS subtended by insulin resistance as the common pathophysiological background [62].
Beside the possible metabolic effect of the family and/or the individual omega-6 FA, it is worth mentioning their effect on inflammation. The metabolic pathway of AA via cyclooxygenase and lipoxygenase leads to the formation of thromboxanes and leukotrienes with pro-inflammatory actions, whereas the CYP450-derived eicosanoids, especially epoxyeicosatrienoic acids, exert prevalent haemodynamic effects but they can also be anti-inflammatory [63, 64]. On the contrary, the counter-part metabolites derived from EPA and DHA are likely to exert anti-inflammatory actions or less potent pro-inflammatory effect, compared to AA [65]. Moreover, in contrast with AA, in vitro studies and animal models suggest a possible anti-inflammatory effect of GLA [50, 66] but, as above mentioned, studies in human are not unequivocal. Although the evaluation of the effects of omega-6 FA on inflammations go beyond the aim of the present study, this possible role in human health should be taken into account and linked to the metabolic actions.
Finally, our findings underline opposite associations within the individual members of a FA family, i.e., AA and GLA, which raise the question whether the clinical effect should better be considered for the individual FA rather than for the family of FA.
Our study has limitations: the sample size is relatively low which can primarily expose to a problem of statistical power. Nevertheless, we were able to detect some meaningful associations between lipid composition of erythrocyte membrane and many parameters of the MetS. These results need to be confirmed in other studies analyzing also samples of children of different ages and body size, including non-obese children. Moreover, data coming from other ethnic groups, which often have different dietary habits, could help in better understanding of these associations. Then, it remains to be clarified if the putative beneficial effect of omega-6 PUFA is specific for obese children and/or viewable only when omega-3 PUFA are under a certain threshold.
Lastly, we lack data about the total food intake of fatty acids and the amount of fatty acids and the other macronutrients in the diet, even if dietary tools to collect these data are often inaccurate, especially in children. Anyhow, it is possible to speculate that data from dietary assessment, like food frequency questionnaire or food diary, together with the measurement of fatty acids in red blood cell membranes, could have led to discover whether, on the one side, the balance between different fatty acids or with the other macronutrients, and on the other side, if the entire family or a single FA plays a role in metabolic modulation.
Strengths of our study are the exploration of a topic in children, in which only a few studies are available, the in-depth characterization of the children’s clinical and laboratory characteristics of the MetS, finally the use of the gold-standard technique for the assessment of fatty acids in red blood cell membrane.
Further studies, including intervention trials, are needed to better understand the actions of the different FA on the single components of the MetS even in children.
In conclusion, the present study shows an association between individual FA, reflecting their dietary intake, and MetS, which supports the hypothesis that the quality of fat intake, beyond the quantity, can influence the metabolic profile in obese children. Our findings agree with the current dietary recommendation to reduce the intake of SFA and support a possible beneficial effect of polyunsaturated FA intake, especially omega-6 PUFA, even though the actions of individual omega-6 FA and the interplay between metabolic and inflammatory actions should be better understood. The level of omega-3 PUFA in our sample of obese children is extremely low so that its putative beneficial effect could have not been detectable.
Abbreviations
- AA:
-
Arachidonic acid
- ALA:
-
Alpha-linoleic acid
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- BP:
-
Blood pressure
- CV:
-
Cardiovascular
- D5D:
-
Delta-5 desaturase
- D6D:
-
Delta-6 desaturase
- DBP:
-
Diastolic blood pressure
- DGLA:
-
Dihomo-gamma-linolenic acid
- DHA:
-
Docosahexaenoic acid
- EPA:
-
Eicosapentaenoic acid
- FA:
-
Fatty acid
- FLI:
-
Fatty Liver Index
- GGT:
-
Gamma-glutamylatransferase
- GLA:
-
Gamma-linolenic acid
- LA:
-
Linoleic acid
- MetS:
-
Metabolic syndrome
- NAFLD:
-
Non-alcoholic fatty liver disease
- O-DBP:
-
Office diastolic blood pressure
- O-SBP:
-
Office systolic blood pressure
- PA:
-
Palmitic acid
- RCT:
-
Randomized controlled trial
- SBP:
-
Systolic blood pressure
- SCD:
-
Delta-9 desaturase
- SFA:
-
Saturated fatty acid
- TRI:
-
Triglycerides
References
Steinberger J, Daniels SR, Eckel RH et al (2009) Progress and challenges in metabolic syndrome in children and adolescents: a scientific statement from the american heart association atherosclerosis, hypertension, and obesity in the young committee of the council on cardiovascular disease in the young. Circulation 119:628–647. https://doi.org/10.1161/CIRCULATIONAHA.108.191394
Nobili V, Carpino G, Alisi A et al (2014) Role of docosahexaenoic acid treatment in improving liver histology in pediatric nonalcoholic fatty liver disease. PLoS One 9:e88005. https://doi.org/10.1371/journal.pone.0088005
Musso G, Gambino R, Cassader M, Pagano G (2011) Meta-analysis: natural history of non-alcoholic fatty liver disease (NAFLD) and diagnostic accuracy of non-invasive tests for liver disease severity. Ann Med 43:617–649. https://doi.org/10.3109/07853890.2010.518623
Vessby B, Uusitupa M, Hermansen K et al (2001) Substituting dietary saturated for monounsaturated fat impairs insulin sensitivity in healthy men and women: the KANWU Study. Diabetologia 44:312–319
Chiu S, Williams PT, Krauss RM (2017) Effects of a very high saturated fat diet on LDL particles in adults with atherogenic dyslipidemia: a randomized controlled trial. PLoS One 12:e0170664. https://doi.org/10.1371/journal.pone.0170664
Mozaffarian D, Micha R, Wallace S (2010) Effects on coronary heart disease of increasing polyunsaturated fat in place of saturated fat: a systematic review and meta-analysis of randomized controlled trials. PLoS Med 7:e1000252. https://doi.org/10.1371/journal.pmed.1000252
de Souza RJ, Mente A, Maroleanu A et al (2015) Intake of saturated and trans unsaturated fatty acids and risk of all cause mortality, cardiovascular disease, and type 2 diabetes: systematic review and meta-analysis of observational studies. BMJ 351:h3978. https://doi.org/10.1136/bmj.h3978
Derosa G, Cicero AFG, D’Angelo A et al (2016) Effects of n-3 pufas on fasting plasma glucose and insulin resistance in patients with impaired fasting glucose or impaired glucose tolerance. Biofactors 42:316–322. https://doi.org/10.1002/biof.1277
Miller PE, Van Elswyk M, Alexander DD (2014) Long-chain omega-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid and blood pressure: a meta-analysis of randomized controlled trials. Am J Hypertens 27:885–896. https://doi.org/10.1093/ajh/hpu024
Bonafini S, Antoniazzi F, Maffeis C et al (2015) Beneficial effects of ω-3 PUFA in children on cardiovascular risk factors during childhood and adolescence. Prostaglandins Other Lipid Mediat. https://doi.org/10.1016/j.prostaglandins.2015.03.006
Bonafini S, Fava C (2017) Omega-3 fatty acids and cytochrome P450-derived eicosanoids in cardiovascular diseases: which actions and interactions modulate hemodynamics? Prostaglandins Other Lipid Mediat 128–129:34–42. https://doi.org/10.1016/j.prostaglandins.2017.01.004
Zhang YY, Liu W, Zhao TY, Tian HM (2017) Efficacy of omega-3 polyunsaturated fatty acids supplementation in managing overweight and obesity: a meta-analysis of randomized clinical trials. J Nutr Health Aging 21:187–192. https://doi.org/10.1007/s12603-016-0755-5
Calder PC (2013) Omega-3 polyunsaturated fatty acids and inflammatory processes: nutrition or pharmacology? Br J Clin Pharmacol 75:645–662. https://doi.org/10.1111/j.1365-2125.2012.04374.x
Wolfram G, Bechthold A, Boeing H et al (2015) Evidence-based guideline of the german nutrition society: fat intake and prevention of selected nutrition-related diseases. Ann Nutr Metab 67:141–204. https://doi.org/10.1159/000437243
Marventano S, Kolacz P, Castellano S et al (2015) A review of recent evidence in human studies of n-3 and n-6 PUFA intake on cardiovascular disease, cancer, and depressive disorders: does the ratio really matter? Int J Food Sci Nutr 66:611–622. https://doi.org/10.3109/09637486.2015.1077790
Belury MA, Cole RM, Bailey BE et al (2016) Erythrocyte linoleic acid, but not oleic acid, is associated with improvements in body composition in men and women. Mol Nutr Food Res 60:1206–1212. https://doi.org/10.1002/mnfr.201500744
Wang DD, Li Y, Chiuve SE et al (2016) Association of specific dietary fats with total and cause-specific mortality. JAMA Intern Med 176:1134–1145. https://doi.org/10.1001/jamainternmed.2016.2417
Vanhala M, Saltevo J, Soininen P et al (2012) Serum omega-6 polyunsaturated fatty acids and the metabolic syndrome: a longitudinal population-based cohort study. Am J Epidemiol 176:253–260. https://doi.org/10.1093/aje/kwr504
Cole TJ, Bellizzi MC, Flegal KM, Dietz WH (2000) Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ 320:1240–1243
Tagetti A, Bonafini S, Zaffanello M et al (2017) Sleep-disordered breathing is associated with blood pressure and carotid arterial stiffness in obese children. J Hypertens 35:125–131. https://doi.org/10.1097/HJH.0000000000001123
Wühl E, Witte K, Soergel M et al (2002) Distribution of 24-h ambulatory blood pressure in children: normalized reference values and role of body dimensions. J Hypertens 20:1995–2007
National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents (2004) The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. Pediatrics 114:555–576
Maffeis C, Grezzani A, Pietrobelli A et al (2001) Does waist circumference predict fat gain in children? Int J Obes Relat Metab Disord 25:978–983. https://doi.org/10.1038/sj.ijo.0801641
Sharma AK, Metzger DL, Daymont C et al (2015) LMS tables for waist-circumference and waist-height ratio Z-scores in children aged 5–19 y in NHANES III: association with cardio-metabolic risks. Pediatr Res 78:1–7. https://doi.org/10.1038/pr.2015.160
Zimmet P, Alberti KGM, Kaufman F et al (2007) The metabolic syndrome in children and adolescents—an IDF consensus report. Pediatr Diabetes 8:299–306. https://doi.org/10.1111/j.1399-5448.2007.00271.x
Lurbe E, Agabiti-Rosei E, Cruickshank JK et al (2016) 2016 European Society of Hypertension guidelines for the management of high blood pressure in children and adolescents. J Hypertens 34:1887–1920. https://doi.org/10.1097/HJH.0000000000001039
Matthews DR, Hosker JP, Rudenski AS et al (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28:412–419
Bedogni G, Bellentani S, Miglioli L et al (2006) The Fatty Liver Index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol 6:33. https://doi.org/10.1186/1471-230X-6-33
Harris WS, Von Schacky C (2004) The Omega-3 Index: a new risk factor for death from coronary heart disease? Prev Med (Baltim) 39:212–220. https://doi.org/10.1016/j.ypmed.2004.02.030
Alsharari ZD, Risérus U, Leander K et al (2017) Serum fatty acids, desaturase activities and abdominal obesity—a population-based study of 60-year old men and women. PLoS One 12:e0170684. https://doi.org/10.1371/journal.pone.0170684
Heuer T, Krems C, Moon K et al (2015) Food consumption of adults in Germany: results of the German National Nutrition Survey II based on diet history interviews. Br J Nutr 113:1603–1614. https://doi.org/10.1017/S0007114515000744
Ahrén B, Mari A, Fyfe CL et al (2009) Effects of conjugated linoleic acid plus n-3 polyunsaturated fatty acids on insulin secretion and estimated insulin sensitivity in men. Eur J Clin Nutr 63:778–786. https://doi.org/10.1038/ejcn.2008.45
Akinkuolie AO, Ngwa JS, Meigs JB, Djoussé L (2011) Omega-3 polyunsaturated fatty acid and insulin sensitivity: a meta-analysis of randomized controlled trials. Clin Nutr 30:702–707. https://doi.org/10.1016/j.clnu.2011.08.013
Harris WS (2010) The omega-3 index: clinical utility for therapeutic intervention. Curr Cardiol Rep 12:503–508. https://doi.org/10.1007/s11886-010-0141-6
Sun D, Cuevas AJ, Gotlinger K et al (2014) Soluble epoxide hydrolase-dependent regulation of myogenic response and blood pressure. Am J Physiol Heart Circ Physiol 306:H1146–H1153. https://doi.org/10.1152/ajpheart.00920.2013
Ubhayasekera SJKA., Staaf J, Forslund A et al (2013) Free fatty acid determination in plasma by GC-MS after conversion to Weinreb amides. Anal Bioanal Chem 405:1929–1935. https://doi.org/10.1007/s00216-012-6658-3
Sato Y, Fujimoto S, Mukai E et al (2014) Palmitate induces reactive oxygen species production and β-cell dysfunction by activating nicotinamide adenine dinucleotide phosphate oxidase through Src signaling. J Diabetes Investig 5:19–26. https://doi.org/10.1111/jdi.12124
Vasu S, McClenaghan NH, McCluskey JT, Flatt PR (2013) Effects of lipotoxicity on a novel insulin-secreting human pancreatic β-cell line, 1.1B4. Biol Chem. https://doi.org/10.1515/hsz-2013-0115
Bermudez B, Ortega-Gomez A, Varela LM et al (2014) Clustering effects on postprandial insulin secretion and sensitivity in response to meals with different fatty acid compositions. Food Funct 5:1374–1380. https://doi.org/10.1039/c4fo00067f
Staaf J, Ubhayasekera SJKA., Sargsyan E et al (2016) Initial hyperinsulinemia and subsequent β-cell dysfunction is associated with elevated palmitate levels. Pediatr Res 80:267–274. https://doi.org/10.1038/pr.2016.80
Aristizabal JC, Barona J, Gonzalez-Zapata LI et al (2016) Fatty acid content of plasma triglycerides may contribute to the heterogeneity in the relationship between abdominal obesity and the metabolic syndrome. Metab Syndr Relat Disord 14:311–317. https://doi.org/10.1089/met.2015.0168
Wang L, Manson JE, Forman JP et al (2010) Dietary fatty acids and the risk of hypertension in middle-aged and older women. Hypertension 56:598–604. https://doi.org/10.1161/HYPERTENSIONAHA.110.154187
Vafeiadou K, Weech M, Altowaijri H et al (2015) Replacement of saturated with unsaturated fats had no impact on vascular function but beneficial effects on lipid biomarkers, E-selectin, and blood pressure: results from the randomized, controlled Dietary Intervention and VAScular function (DIVAS) study. Am J Clin Nutr 102:40–48. https://doi.org/10.3945/ajcn.114.097089
Miura K, Stamler J, Nakagawa H et al (2008) Relationship of dietary linoleic acid to blood pressure: the international study of macro-micronutrients and blood pressure study. Hypertension 52:408–414. https://doi.org/10.1161/HYPERTENSIONAHA.108.112383
Böhm A, Halama A, Meile T et al (2014) Metabolic signatures of cultured human adipocytes from metabolically healthy versus unhealthy obese individuals. PLoS One 9:e93148. https://doi.org/10.1371/journal.pone.0093148
Aldámiz-Echevarría L, Prieto JA, Andrade F et al (2007) Arachidonic acid content in adipose tissue is associated with insulin resistance in healthy children. J Pediatr Gastroenterol Nutr 44:77–83. https://doi.org/10.1097/01.mpg.0000237931.53470.ba
Das UN (2013) Arachidonic acid and lipoxin A4 as possible endogenous anti-diabetic molecules. Prostaglandins Leukot Essent Fatty Acids 88:201–210. https://doi.org/10.1016/j.plefa.2012.11.009
Clifton PM, Nestel PJ (1998) Relationship between plasma insulin and erythrocyte fatty acid composition. Prostaglandins Leukot Essent Fatty Acids 59:191–194
Felton CV, Stevenson JC, Godsland IF (2004) Erythrocyte-derived measures of membrane lipid composition in healthy men: associations with arachidonic acid at low to moderate but not high insulin sensitivity. Metabolism 53:571–577
Arm JP, Boyce JA, Wang L et al (2013) Impact of botanical oils on polyunsaturated fatty acid metabolism and leukotriene generation in mild asthmatics. Lipids Health Dis 12:141. https://doi.org/10.1186/1476-511X-12-141
Imamura F, Sharp SJ, Koulman A et al (2017) A combination of plasma phospholipid fatty acids and its association with incidence of type 2 diabetes: the EPIC-InterAct case-cohort study. PLoS Med 14:e1002409. https://doi.org/10.1371/journal.pmed.1002409
Delgado GE, März W, Lorkowski S et al (2017) Omega-6 fatty acids: Opposing associations with risk—the Ludwigshafen Risk and Cardiovascular Health Study. J Clin Lipidol 11:1082–1090.e14. https://doi.org/10.1016/j.jacl.2017.05.003
Akter S, Kurotani K, Sato M et al (2017) High serum phospholipid dihomo-γ-linoleic acid concentration and low ∆5-desaturase activity are associated with increased risk of type 2 diabetes among japanese adults in the hitachi health study. J Nutr 147:1558–1566. https://doi.org/10.3945/jn.117.248997
Hua M-C, Su H-M, Yao T-C et al (2017) Alternation of plasma fatty acids composition and desaturase activities in children with liver steatosis. PLoS One 12:e0182277. https://doi.org/10.1371/journal.pone.0182277
Nilsen DWT, Aarsetoey H, Pönitz V et al (2017) The prognostic utility of dihomo-gamma-linolenic acid (DGLA) in patients with acute coronary heart disease. Int J Cardiol 249:12–17. https://doi.org/10.1016/j.ijcard.2017.09.202
Ouchi S, Miyazaki T, Shimada K et al (2017) Decreased circulating dihomo-gamma-linolenic acid levels are associated with total mortality in patients with acute cardiovascular disease and acute decompensated heart failure. Lipids Health Dis 16:150. https://doi.org/10.1186/s12944-017-0542-2
Daneshmand R, Kurl S, Tuomainen T-P, Virtanen JK (2017) Associations of estimated ∆-5-desaturase and ∆-6-desaturase activities with stroke risk factors and risk of stroke: the Kuopio Ischaemic Heart Disease Risk Factor Study. Br J Nutr 117:582–590. https://doi.org/10.1017/S000711451700054X
Warensjö E, Sundström J, Lind L, Vessby B (2006) Factor analysis of fatty acids in serum lipids as a measure of dietary fat quality in relation to the metabolic syndrome in men. Am J Clin Nutr 84:442–448
Vessby B, Gustafsson I-B, Tengblad S et al (2002) Desaturation and elongation of fatty acids and insulin action. Ann N Y Acad Sci 967:183–195
Maffeis C, Banzato C, Talamini G, Obesity Study Group of the Italian Society of Pediatric Endocrinology and Diabetology (2008) Waist-to-height ratio, a useful index to identify high metabolic risk in overweight children. J Pediatr 152:207–213. https://doi.org/10.1016/j.jpeds.2007.09.021
Maffeis C, Corciulo N, Livieri C et al (2003) Waist circumference as a predictor of cardiovascular and metabolic risk factors in obese girls. Eur J Clin Nutr 57:566–572. https://doi.org/10.1038/sj.ejcn.1601573
Reccia I, Kumar J, Akladios C et al (2017) Non-alcoholic fatty liver disease: a sign of systemic disease. Metabolism 72:94–108. https://doi.org/10.1016/j.metabol.2017.04.011
Jiang J, Zhang S, Liu Y et al (2014) EETs alleviate ox-LDL-induced inflammation by inhibiting LOX-1 receptor expression in rat pulmonary arterial endothelial cells. Eur J Pharmacol 727:43–51. https://doi.org/10.1016/j.ejphar.2014.01.045
Shahabi P, Siest G, Meyer UA, Visvikis-Siest S (2014) Human cytochrome P450 epoxygenases: variability in expression and role in inflammation-related disorders. Pharmacol Ther 144:134–161. https://doi.org/10.1016/j.pharmthera.2014.05.011
Arnold C, Konkel A, Fischer R, Schunck W-HH (2010) Cytochrome P450-dependent metabolism of omega-6 and omega-3 long-chain polyunsaturated fatty acids. PharmacolRep 62:536–547
Sergeant S, Rahbar E, Chilton FH (2016) Gamma-linolenic acid, Dihommo-gamma linolenic, eicosanoids and inflammatory processes. Eur J Pharmacol 785:77–86. https://doi.org/10.1016/j.ejphar.2016.04.020
Acknowledgements
Part of this work was performed in the LURM (Laboratorio Universitario di Ricerca Medica) Research Center, University of Verona.
Funding
The study is supported by a Grant of the Italian Ministry of Health (GR-2011-02349630) to CF in agreement with the ‘Regione Veneto’ and the ‘Azienda Ospedaliera Universitaria Integrata di Verona’.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that they have no competing interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bonafini, S., Tagetti, A., Gaudino, R. et al. Individual fatty acids in erythrocyte membranes are associated with several features of the metabolic syndrome in obese children. Eur J Nutr 58, 731–742 (2019). https://doi.org/10.1007/s00394-018-1677-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-018-1677-2