Abstract
Purpose
The aim of this study was to investigate if postoperative mortality after acute surgical treatment of colorectal cancer has decreased in Denmark during this period and to investigate risk factors associated with early death.
Methods
This is a nationwide and population-based cohort study. From the Danish Colorectal Cancer Group database and National Patient Registry, we collected data on all patients operated with bowel resection, diverting stoma only, or placement of an endoscopic stent from 2005 to 2015. Year of surgery was the main exposure variable and 90-day postoperative mortality the primary outcome.
Results
We included 6147 patients. The incidence of patients per year was stable during 2005–2015. The 90-day mortality decreased from 31% in 2005 to 24% in 2015 with a significant time trend (p < 0.0001). Other factors associated with postoperative mortality were increasing age, presence of comorbidity (measured as Charlson comorbidity index score ≥ 1), and stage IV disease. Insertion of self-expanding metallic stent was protective for 90-day postoperative mortality compared with other surgical procedures.
Conclusion
Ninety-day postoperative mortality from acute colorectal surgery has improved in Denmark from 2005 to 2015. Nevertheless, almost one out of four patients undergoing acute surgery for colorectal cancer dies within 90 days.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Worldwide, colorectal cancer is the third most common cancer with an incidence of around 1.4 million per year and an estimated 700,000 deaths annually [1]. In Denmark, the incidence is 5000 of which approximately 10% undergo acute therapy [2], due to complete obstruction, perforation, abscess formation, or diffuse peritonitis. Acute onset of colorectal cancer is associated with a high short-term mortality risk [3, 4]. In Denmark, postoperative 90-day mortality from acute colorectal cancer surgery is 21% versus only 3% after elective surgery [1]. Besides surgical intervention and diverting stoma, insertion of self-expandable metallic stents (SEMS) can be performed in chosen cases to recreate intestinal continuity as the only treatment or as bridge to surgery.
During the past 10 years, both short- and long-term survival after elective colorectal cancer surgery has improved in Denmark [5, 6]. Changes in short-term mortality after surgery depend on patient-related factors, clinical factors, and structural factors. For instance, changes in mortality can be a result of patient selection and perioperative treatment. The Danish Colorectal Cancer Group (DCCG.dk) and the Danish health authorities have since the 1990s taken several national initiatives to improve survival in patients with colorectal cancer [6]. Initiatives such as evidence-based guidelines, fast-track cancer pathways, cancer plans, and centralization of colorectal cancer surgery in fewer high-volume units [6].
The aim of this study was to evaluate 90-day mortality rate after acute treatment of colorectal cancer within a period of 11 years in Denmark and associated risk factors. Resection, SEMS, and diverting stoma as bridge to surgery, and palliative surgery are all included as treatment strategies.
Method
Study design and setting
This study is a nationwide and population-based historical cohort study. Data are collected prospectively in the Danish national registries. We report the results according to the STROBE statement for cohort studies [7]. The study is based on data from the DCCG.dk database and the National Patient Registry (NPR) [8, 9]. All patients included in the study were operated at a surgical department in a public hospital in Denmark between 1 January 2005 and 31 December 2015.
All Danish residents have access to tax financed, free of charges hospital care. Information on services are registered for all residents and linked with a unique identification number in national registries, e.g., the NPR and population-based clinical quality databases, e.g., the DCCG.dk database. All patients were followed up for 90 days after acute surgery or stent placement. Information about postoperative mortality within this period was collected from the Danish Civil Registration Registry [10].
Registries
Data were collected from the DCCG.dk database, a national population-based, clinical quality database with a completeness proportion of 95–99% of all incident colorectal cancer patients in Denmark [9]. Patients included in the database were treated or diagnosed with colorectal cancer in a surgical department in Denmark. Patients with metachronous cancer, recurrence, and tumors of other histological origin than primary adenocarcinoma, mucinous adenocarcinoma, signet ring cell carcinoma, medullary carcinoma, or undifferentiated carcinoma are not registered in the DCCG.dk database. All clinical and pathological data are registered prospectively.
Only the definitive surgical procedure is registered in DCCG.dk [9]. Thus, patients who are acutely relieved with a diverting stoma or SEMS prior to an elective surgical resection, will only have a registration of the elective procedure in DCCG.dk, and data on these patients were extracted from the Danish NPR. The Danish NPR is the most comprehensive nationwide register worldwide [8]. From the Danish NPR, we use date of emergency admission, stoma as only procedure (KJFF10, KJFF11, KJFF20, KJFF21, KJFF23, KJFF24, KJFF26, KJFF27, KJFF30, and KJFF31), and insertion of endoscopic stent in colon or rectum (KJFA68 and KJGA58A) to include patients with diverting stoma or SEMS prior to elective resection. None of the codes for these variables changed within the study period. Using the unique identification number we obtained information on vital status by linking to the Danish Civil Registration System that holds continuously updated information on name, sex, date of birth, vital status, etc. on all Danish residents alive [10]. All patients included via the Danish NPR were also diagnosed with colorectal cancer in the DCCG.dk within the study period.
Participants
All patients registered in the DCCG.dk database with an acute surgical procedure between 2005 and 2015 were included. Procedures included right hemicolectomy, transverse colectomy, left hemicolectomy, sigmoid colectomy, Hartmann’s procedure, other colectomy ± stoma, diverting stoma, intestinal bypass, subtotal/total colectomy, explorative laparotomy/diagnostic laparoscopy only, anterior resection ± stoma, proctocolectomy with ileostomy, or insertion of SEMS in colon or rectum. Furthermore, we included all patients registered in the Danish NPR with an acute insertion of SEMS or diverting stoma within 72 h after acute admission at any department from 2005 to 2015. If a patient was registered with more than one acute procedure in, e.g., one in the NPR and one in DCCG.dk, the first procedure was used for this analysis. All patients registered in the Danish NPR also needed to have a date of diagnosis in DCCG.dk within the study period. Patients were excluded if they emigrated within 90 days after the surgery, or had an elective surgical procedure for colorectal cancer before the acute procedure. Furthermore, we excluded all patients registered in the DCCG.dk database with acute surgery and one of the following procedures: abdominoperineal excision of rectum, transanal endoscopic microsurgery (TEM), and other local procedures including polypectomy.
Variables
The primary outcome was 90-day mortality after acute colorectal cancer surgery including insertion of SEMS. The secondary outcome was 30-day mortality. Acute surgery is defined in the DCCG.dk as indication of surgery due to suspected ileus, perforation, bleeding, or other acute reasons. Year of surgery was the primary exposure variable. We consider the following to be confounders: age, sex, comorbidity estimated with Charlson comorbidity index (CCI), UICC stage, and tumor localization (right, transverse or left colon, and rectum). Additionally, we adjusted for primary surgical procedure (SEMS insertion, acute surgery with resection, or acute surgery with only diverting stoma).
Subgroup analysis
Patients that directly undergo surgery, and not SEMS, represent a certain risk group and we therefore performed a subgroup analysis of these patients. We did a stepwise analysis, first including surgical specialization, then operative approach and finally hospital volume to investigate if the effect of these variables on 90-day postoperative mortality. Surgery was performed by a specialist if either the surgeon, or the assistant, was specialized in surgical gastroenterology or was a certified colorectal cancer surgeon. The operative approach was either laparotomy or minimal invasive (laparoscopic or robot-assisted). Hospital volume was calculated as the total numbers of surgical colorectal cancer procedures per year, both acute and elective. Volume intervals were based on quartiles of the procedures with 25%, 50%, and 75%, which gave the cut points: < 150 per year, 150–220 per year, 220–280 per year, and > 280 per year.
Additionally, we described the subgroup of patients who had an acute stent or diverting stoma as either bridge to elective surgery with curative attend or compromised resection, or as a palliative procedure without bridge to elective surgery.
Statistical analysis
Association between year of surgery and 90-day postoperative mortality was analyzed using a logistic regression model. We reported the p value of the trend in year of surgery with the chi square test. P values < 0.05 were considered significant. We adjusted analyses for sex, age, CCI score, primary procedure, tumor localization, and UICC stage. Missing data were categorized within each variable. Quantitative variables were tested for linearity by inserting a squared term in the model. Age was found non-linear and was included as a linear spline with cut points at the tertiles. Kaplan-Meier curves were used to illustrate the survival over the calendar periods, after diverting the study period in to four groups.
Interaction was tested in the full model between age and comorbidity (CCI), and between year of surgery and comorbidity (CCI).
A subgroup analysis was performed to investigate the association between 90-day mortality and hospital volume, surgical specialization, and surgical approach with a multiple regression model adjusting for year of surgery, sex, age, CCI, year of surgery, primary procedure, tumor localization, and UICC stage.
All analyses were performed using SAS software, version 9.3. This study was reported to the Danish Data Protection Agency with registration number 2015-41-3726. Under Danish law, registration with the Danish Ethical Committee was not required.
Results
Participants
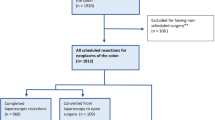
We included 6147 patients admitted and treated with acute surgery for colorectal cancer in Denmark in 2005 through 2015. We excluded 245 patients, Fig. Fig. 1. No difference was found in the gender distribution, and the mean age was 72 years (SD ± 12). Nineteen percent had a CCI score of three or more, while 52% of the participants had no comorbidity. The number of patients operated each year varied from 501 to 631 with a median of 560. Surgical resection was the most common surgical procedure, and most tumors were localized in the left side. Metastatic disease was found in 42% of the patients and local disease (stage I–II) in only 26%. Of the right-sided tumors, 86% were treated with surgery as primary procedure, 3% with SEMS and 11% with diverting stoma. Among the left-sided tumors, 47% were treated with surgery, 44% with SEMS and 9% with diverting stoma. The tumors localized in transverse colon and rectum were mainly treated with surgery (64%) and diverting stoma (45%), respectively. As expected, few variables included in the analysis had missing values. The variables sex, age, CCI, year of surgery, and 90-day mortality had no missing values, while stage had most missing values (8%).
Mortality
The 90-day overall mortality was 25% after acute surgery, insertion of SEMS, or diverting stoma, whereas 30-day mortality was 16%, Table 1. Of the patients, aged 85 years or more, 46% died within 90 days after the acute procedure, whereas this rate was 14% for patients less than 70 years old.
The Kaplan-Meier plot illustrates the unadjusted 90-day survival in the four different periods, Fig. Fig. 2.
Risk factors for postoperative mortality after acute colorectal cancer surgery
The year of surgery was associated with 90-day postoperative mortality also when adjusting for confounders, Table 2. From 2012 to 2015, the mortality was reduced significantly compared with 2005 and the trend in the total period was significantly reduced with an OR of 0.94 per year (95% confidence interval, 0.92–0.96, p < 0.0001).
Increasing age, comorbidity, and UICC stage IV were all significant independent risk factors for postoperative 90-day mortality, Table 2. SEMS, as the primary treatment of acute colorectal cancer, was protective for postoperative mortality compared with other surgical interventions, Table 2.
No interaction was found between age and comorbidity nor year of surgery and comorbidity.
Subgroup analysis
We included 4354 patients in the subgroup analysis not including SEMS. The 90-day mortality was 28% whereof 18% died within the first 30 days after surgery. In adjusted analysis, specialization of the surgeon was associated with 90-day mortality, OR = 1.16 (95% CI, 0.99–1.35), however not significant. When adjusting for specialization of the surgeon, open surgery was associated with an increased risk of 90-day postoperative mortality, OR = 1.66 (95% CI, 1.23–2.23). There was a decreased 90-day mortality in hospitals with a low annual volume of colorectal cancer surgery; however, only significant in hospitals with 150–220 procedures per year, Table 3.
We described the demography of 854 patients in the second subgroup of patients undergoing acute stent or diverting stoma prior to elective resection with a curative intent or compromised resection, and the 1777 patients with stent or stoma as palliative procedure, Table 4. The 90-day mortality in patients with further bridge to surgery 2% compared with 38% in the palliative procedure group. In the patients having bridge to surgery, mean age was 69 years (SD ± 11), 64% had no comorbidity (CCI = 0), 76% of the patients had stage II or III disease, 75% of the tumors were localized in left colon, and the majority had an SEMS procedure (98%). In comparison, patients having a stent procedure or diverting stoma as a palliative procedure mean age was 73 (SD ± 12), 48% had no comorbidity (CCI = 0), 89% had stage IV disease or missing stage, 53%of the tumors were localized in the rectum, and 58% had SEMS as primary procedure.
Discussion
In this present Danish population-based study, 6147 patients operated in an acute setting for colorectal cancer from 2005 to 2015 and the overall postoperative 90-day mortality decreased from 31 to 24%. The 90-day mortality decreased significantly in the latest years from 2012 to 2015 and mortality was reduced with an OR of 0.94 per year (95% CI, 0.92–0.95). Other risk factors for postoperative mortality after acute colorectal cancer surgery were high age, comorbidity, stage IV disease, and diverting stoma. SEMS was associated with reduced risk of mortality.
The primary limitation in this study is the observational design enabling a description of correlation between the year of surgery and postoperative mortality without proving causality. Several individual factors could determine how the patients were treated which can introduce confounding by indication. One example of this is that our results indicate that patients treated with SEMS have a reduced risk of postoperative mortality than the patients treated with surgical resection or diverting stoma. This result could reflect confounding by indication meaning that the patients in the less acute clinical condition like bowel obstruction only were chosen for SEMS and the high-risk patients with, for instance, perforation and peritonitis needed surgical resection or a diverting stoma only. However, there could also be an actual benefit of SEMS in short-term survival, which is a less invasive procedure that prolong the period for medical optimization. Surprisingly, diverting stoma did not have the same protective effect on mortality even though it is an alternative procedure to SEMS and have the same benefits of postponing the final procedure. This could reflect confounding by indication between patients who get SEMS and diverting stoma. For instance, diverting stoma may be chosen in more severe cases if SEMS was not possible and/or in case of an advanced tumor or massive carcinomatosis. Two previous meta-analysis of 334 and 382 patients, and overlapping studies, showed that treatment of malignant, left-sided colon obstruction did not find any advantages of SEMS compared with emergency surgery [11, 12]. A Dutch cohort study with 1860 treated for obstruction of the proximal colon showed that stent was possible in 2.4% as bridge to surgery and resulted in improved in the unadjusted 30-day mortality and morbidity [13]. In the long-term perspective, some studies have, however, reported an increased risk of recurrence and decreased disease-free survival after SEMS [14,15,16].
Lack of information about the clinical condition at the time of surgery, e.g., sepsis or perforation of the tumor associated with a high postoperative mortality [17], can introduce residual confounding in our results. Furthermore, information about life style factors such as smoking, alcohol intake, and BMI could also affect postoperative mortality. Sufficient data on these variables were not available in our study. However, through adjusting for comorbidity, we indirectly account for some of the effect of lifestyle factors.
Previous studies have focused on acute colorectal cancer surgery as a risk factor for early postoperative death and analyzed differences in patient and clinical related characteristics in acute versus elective surgery [18,19,20]. This study is, to our knowledge, the largest study investigating if postoperative mortality has improved over the past decade, and which factors are associated with 90-day mortality. It is a major strength of this study that it is nationwide and population-based using a high-quality database with a completeness of 95–99% of all patients treated for colorectal cancer in Denmark. Data are collected prospectively, eliminating the risk of recall bias. Follow-up is almost complete (97%) and validity is high on primary outcome and exposure variables. All clinical data have an almost complete coverage with missing values on tumor localization or stage in only 532 patients (9%) and the validity on all variables included in the model is considered very high [9, 10].
The European Society of Gastrointestinal Endoscopy recommends SEMS as an alternative to emergency surgery only in patients at high operative risk, e.g., ASA ≥ III and/or age > 70 years, and not as standard treatment in left-sided obstructive tumors [21]. In England, 24% of the all patients with colorectal cancer from 2006 to 2013 presented as an emergency [22]. The relative 90-day survival was 65% after acute diagnosed colorectal cancer. In the English national guidelines, it is recommended to use SEMS in left-sided colorectal cancers with complete or nearly complete bowel obstruction [23]. However, guidelines are not clear in defining the indication for emergency surgery versus SEMS [23]. In Sweden, handling of acute colorectal cancer patients is more precise [24]. Treatment with SEMS is not recommended in patients with potential curable resection unless surgical intervention is considered a high risk. They argue that the risk of stent-related perforation increases the risk of postoperative mortality and of tumor spread. If curative surgery cannot be guaranteed, it is recommended to perform diverting stoma and no resection [24].
In the subgroup analysis based on 4354 patients undergoing surgical resection, there was a tendency to decreased 90-day mortality with low hospital volume of yearly surgical colorectal cancer procedures. This result deviate from both previous Danish and American studies on acute colorectal cancer surgery [18, 25], and a meta-analysis with both acute and elective surgical mode [26]. In a Danish study from 2011, 4.3% of the variation in 30-day postoperative mortality after acute colorectal cancer surgery was explained by the hospital volume level [18]. They did not adjust for operative approach or specialization of the surgeon [18]. This could explain differences in the results from the same population. In the American study, they evaluated the load of operation for the individual surgeon with the maximum level of > 10 annually resections [25]. In a Danish context, ten resections per year are low for a specialized surgeon. Furthermore, it is plausible that some severe cases have been removed to more specialized, high-volume departments for surgical intervention. In our data, we cannot adjust for this type of confounding by indication due to lack of clinical data.
We saw a positive effect on minimal invasive surgery versus open. Again, this could be a real effect due to reduced stress response in minimal invasive surgery [27, 28]. Alternatively, the result is caused by confounding by indication, if the more complex tumors, e.g., local advanced tumors were operated with an open approach, and the less complicated tumors where operated with minimal invasive surgery. However, we cannot explore this further in the current data.
In conclusion, the 90-day postoperative mortality after acute colorectal cancer surgery, including SEMS, has improved significantly in Denmark from 2005 to 2015. However, postoperative mortality remains high at 24% in 2015 and further studies should investigate how the perioperative period could be optimized to improve survival even more.
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65:87–108. https://doi.org/10.3322/caac.21262
Annual report of Danish Colorectal Cancer Group (DCCG.dk) 2016. http://dccg.dk/pdf/2017/Aarsrapport_2016.pdf. Accessed Sep 24th 2018
Kobayashi H, Miyata H, Gotoh M, Baba H, Kimura W, Kitagawa Y, Nakagoe T, Shimada M, Tomita N, Sugihara K, Mori M (2014) Risk model for right hemicolectomy based on 19,070 Japanese patients in the national clinical database. J Gastroenterol 49:1047–1055. https://doi.org/10.1007/s00535-013-0860-8
Sjo OH, Larsen S, Lunde OC, Nesbakken A (2009) Short term outcome after emergency and elective surgery for colon cancer. Color Dis 11:733–739. https://doi.org/10.1111/j.1463-1318.2008.01613.x
Iversen LH, Ingeholm P, Gogenur I, Laurberg S (2014) Major reduction in 30-day mortality after elective colorectal cancer surgery: a nationwide population-based study in Denmark 2001-2011. Ann Surg Oncol 21:2267–2273. https://doi.org/10.1245/s10434-014-3596-7
Iversen LH, Green A, Ingeholm P, Osterlind K, Gogenur I (2016) Improved survival of colorectal cancer in Denmark during 2001-2012—the efforts of several national initiatives. Acta Oncol 55(Suppl 2):10–23. https://doi.org/10.3109/0284186x.2015.1131331
University of Bern https://strobe-statement.org/fileadmin/Strobe/uploads/checklists/STROBE_checklist_v4_cohort.pdf Accessed Sep 24th 2018
Lynge E, Sandegaard JL, Rebolj M (2011) The Danish National Patient Register. Scand J Public Health 39:30–33. https://doi.org/10.1177/1403494811401482
Ingeholm P, Gogenur I, Iversen LH (2016) Danish colorectal cancer group database. Clin Epidemiol 8:465–468. https://doi.org/10.2147/clep.s99481
Pedersen CB (2011) The Danish civil registration system. Scand J Public Health 39:22–25. https://doi.org/10.1177/1403494810387965
Liu Z, Kang L, Li C, Huang M, Zhang X, Wang J (2014) Meta-analysis of complications of colonic stenting versus emergency surgery for acute left-sided malignant colonic obstruction. Surg Laparosc Endosc Percutan Tech 24:73–79. https://doi.org/10.1097/sle.0000000000000030
Huang X, Lv B, Zhang S, Meng L (2014) Preoperative colonic stents versus emergency surgery for acute left-sided malignant colonic obstruction: a meta-analysis. J Gastrointest Surg 18:584–591. https://doi.org/10.1007/s11605-013-2344-9
Amelung FJ, Consten ECJ, Siersema PD, Tanis PJ (2016) A population-based analysis of three treatment modalities for malignant obstruction of the proximal colon: acute resection versus stent or stoma as a bridge to surgery. Ann Surg Oncol 23:3660–3668. https://doi.org/10.1245/s10434-016-5247-7
Avlund TH, Erichsen R, Ravn S, Ciplys Z, Andersen JC, Laurberg S, Iversen LH (2017) The prognostic impact of bowel perforation following self-expanding metal stent as a bridge to surgery in colorectal cancer obstruction. Surg Endosc 32:328–336. https://doi.org/10.1007/s00464-017-5680-0
Gorissen KJ, Tuynman JB, Fryer E, Wang L, Uberoi R, Jones OM, Cunningham C, Lindsey I (2013) Local recurrence after stenting for obstructing left-sided colonic cancer. Br J Surg 100:1805–1809. https://doi.org/10.1002/bjs.9297
Sabbagh C, Browet F, Diouf M, Cosse C, Brehant O, Bartoli E, Mauvais F, Chauffert B, Dupas JL, Nguyen-Khac E, Regimbeau JM (2013) Is stenting as “a bridge to surgery” an oncologically safe strategy for the management of acute, left-sided, malignant, colonic obstruction? A comparative study with a propensity score analysis. Ann Surg 258:107–115. https://doi.org/10.1097/SLA.0b013e31827e30ce
Al-Temimi MH, Griffee M, Enniss TM, Preston R, Vargo D, Overton S, Kimball E, Barton R, Nirula R (2012) When is death inevitable after emergency laparotomy? Analysis of the American College of Surgeons National Surgical Quality Improvement Program database. J Am Coll Surg 215:503–511. https://doi.org/10.1016/j.jamcollsurg.2012.06.004
Osler M, Iversen LH, Borglykke A, Martensson S, Daugbjerg S, Harling H, Jorgensen T, Frederiksen B (2011) Hospital variation in 30-day mortality after colorectal cancer surgery in Denmark: the contribution of hospital volume and patient characteristics. Ann Surg 253:733–738. https://doi.org/10.1097/SLA.0b013e318207556f
Gooiker GA, Dekker JW, Bastiaannet E, van der Geest LG, Merkus JW, van de Velde CJ, Tollenaar RA, Liefers GJ (2012) Risk factors for excess mortality in the first year after curative surgery for colorectal cancer. Ann Surg Oncol 19:2428–2434. https://doi.org/10.1245/s10434-012-2294-6
Jestin P, Nilsson J, Heurgren M, Pahlman L, Glimelius B, Gunnarsson U (2005) Emergency surgery for colonic cancer in a defined population. Br J Surg 92:94–100. https://doi.org/10.1002/bjs.4780
van Hooft JE, van Halsema EE, Vanbiervliet G, Beets-Tan RG, DeWitt JM, Donnellan F, Dumonceau JM, Glynne-Jones RG, Hassan C, Jimenez-Perez J, Meisner S, Muthusamy VR, Parker MC, Regimbeau JM, Sabbagh C, Sagar J, Tanis PJ, Vandervoort J, Webster GJ, Manes G, Barthet MA, Repici A (2014) Self-expandable metal stents for obstructing colonic and extracolonic cancer: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy 46:990–1053. https://doi.org/10.1055/s-0034-1390700
National Cancer Registration and Analysis Service (NCRAS) http://www.ncin.org.uk/publications/routes_to_diagnosis# Accessed Sep 24th 2018
National guidelines from National Institute for Health and Care Excellence (NICE) https://www.nice.org.uk/guidance/cg131/evidence/full-guideline-pdf-183509677 Accessed Sep 24th 2018
National guidelines, Sweeden http://www.cancercentrum.se/globalassets/cancerdiagnoser/tjock%2D%2Doch-andtarm-anal/vardprogram/nvpkolorektalcancer_2016-03-15.pdf. Accessed Sep 24th 2018
Aquina CT, Becerra AZ, Xu Z, Boscoe FP, Schymura MJ, Noyes K, Monson JRT, Fleming FJ (2017) Nonelective colon cancer resection: a continued public health concern. Surgery 161:1609–1618. https://doi.org/10.1016/j.surg.2017.01.001
Archampong D, Borowski D, Wille-Jorgensen P, Iversen LH (2012) Workload and surgeon's specialty for outcome after colorectal cancer surgery. Cochrane Database Syst Rev 14:CD005391. https://doi.org/10.1002/14651858.CD005391.pub3
Kehlet H, Nielsen HJ (1998) Impact of laparoscopic surgery on stress responses, immunofunction, and risk of infectious complications. New Horiz 6:S80–S88
Wichmann MW, Huttl TP, Winter H, Spelsberg F, Angele MK, Heiss MM, Jauch KW (2005) Immunological effects of laparoscopic vs open colorectal surgery: a prospective clinical study. Arch Surg 140:692–697. https://doi.org/10.1001/archsurg.140.7.692
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Degett, T.H., Dalton, S.O., Christensen, J. et al. Mortality after emergency treatment of colorectal cancer and associated risk factors—a nationwide cohort study. Int J Colorectal Dis 34, 85–95 (2019). https://doi.org/10.1007/s00384-018-3172-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-018-3172-x