Abstract
Background/purpose
Hirschsprung-associated enterocolitis (HAEC) is a life-threatening complication of Hirschsprung’s disease. HAEC is reported to occur in 6–50% of patients preoperatively and in 2–35% postoperatively. The exact cause of HAEC is not fully understood, but disturbances of intestinal microbiota have recently been reported in patients with HAEC. In recent years, the administration of probiotics has been proposed to reduce the incidence of HAEC. We conducted a systematic review and meta-analysis to determine the effect of probiotics on postoperative HAEC.
Methods
A systematic literature search for relevant articles was performed in four databases using the combinations of following terms “probiotics”, “microbiota”, “enterocolitis”, “Lactobacillus”, “Bifidobacterium”, “Saccharomyces”, “Streptococcus”, and “Hirschsprung disease/Hirschsprung’s disease” for studies published between 2002 and 2017. The relevant cohorts of the effect of probiotics in postoperative patients were systematically searched for clinical outcomes. Odds ratio (OR) or standard mean difference (SMD) with 95% confidence intervals (CI) were calculated using standardized statistical methodology.
Results
The search strategy identified 1274 reports. Overall, five studies met defined inclusion criteria, reporting a total of 198 patients. Two studies were prospective multicenter randomized control trials. Lactobacillus, Bifidobacterium, Streptococcus, and Enterococcus were used as probiotics. The incidence of HAEC with/without probiotics was 22.6 and 30.5%, respectively, but this was not statistically different (OR 0.72; 95% CI 0.37–1.39; P = 0.33).
Conclusion
This study shows that the administration of probiotics was not associated with a significant reduction in the risk of HAEC. Additional studies are required to understand more fully the role of microbiota and complex interactions that cause HAEC. With increasing knowledge of the role of microbiota in HAEC, we are likely to understand better the potential benefits of probiotics in this disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Probiotics are defined as live microorganisms, which when consumed in adequate amounts, which confer a health benefit on the host. In vitro studies suggest that probiotics potentially act favorably in the host through several different mechanisms. They have an antimicrobial effect through modifying the microflora, secreting antibacterial substances, competing with pathogens to prevent their adhesion to the intestinal epithelium, competing for nutrients necessary for pathogen survival, producing an antitoxin effect, and reversing some of the consequences of infection on the intestinal epithelium, such as secretory changes and neutrophil migration [1, 2]. Probiotics have been suggested to be efficacious in the prevention and treatment of viral diarrhea [3], necrotizing enterocolitis (NEC) [4], and pouchitis [5]. As well, probiotics can promote mucin production, which is deranged in Hirschsprung-associated enterocolitis (HAEC) [6]. It is well known that mucin layer blocks the direct attachment of commensal bacteria to the epithelial layer [6].
HAEC is a life-threatening complication of Hirschsprung’s disease (HSCR). HAEC is reported to occur in 6–50% of patients preoperatively and in 2–35% postoperatively. The exact cause of HAEC is not fully understood. However, based on the previous knowledge of the beneficial effects of probiotics in inflammatory conditions of the gastrointestinal tract, some centres have advocated the prophylactic administration of probiotics after a pull-through procedure in patient with HSCR in anticipation that this will decrease the incidence of HAEC [7, 8]. We conducted a systematic review and meta-analysis to determine the effect of probiotics on postoperative HAEC.
Materials and methods
A systematic review and meta-analysis were conducted based on Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. A systematic search of the literature was performed in the PubMed, Embase, Medline, and Cochrane Library electronic database for the keywords “probiotics”, “microbiota”, “enterocolitis”, “Lactobacillus”, “Bifidobacterium”, “Saccharomyces”, “Streptococcus”, and “Hirschsprung disease/Hirschsprung’s disease” for studies published between 2002 and 2017. Reference lists of relevant articles were manually searched for further cohorts. Duplicates were deleted. Resulting publications were reviewed in detail for epidemiology, operative treatment, morbidity, and clinical outcome. The relevant articles were reviewed by title, keywords, and abstract by the authors (H.N., T.L., and P.P.) and a full-text assessment of selected articles was performed.
Odds ratio (OR) or standard mean difference (SMD) with 95% confidence intervals (CI) was calculated using standardized statistical methodology.
Results
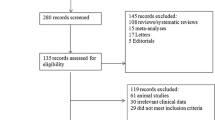
The initial search yielded a total of 1279 publications, of which 1274 were identified by electronic database searching and five from cross-referencing (Fig. 1). After removal of 512 duplicate listed articles, 767 titles, keywords, and abstracts were screened. Of these, 751 non-relevant studies were excluded. The remaining 16 publications were assessed in full text for eligibility and 11 articles were excluded, because they did not address any of the selection criteria. In total, data from five studies (published between 2002 and 2017) met defined inclusion criteria and were included in the cumulative analysis.
Five studies during the period 2011–2016 which met defined inclusion criteria reported a total of 198 patients [7,8,9,10,11] (Table 1). Two studies reported the type of HSCR; the incidence of long segment aganglionosis was not significantly different between probiotics group and placebo group. Two studies were prospective multicenter randomized control trials. Lactobacillus, Bifidobacterium, Streptococcus, and Enterococcus were used as probiotics. The incidence of HAEC with/without probiotics was 22.6 and 30.5%, respectively, but this was not statistically different (OR 0.72; 95% CI 0.37–1.39; P = 0.33) (Fig. 2).
Discussion
In recent years, a large number of randomized controlled trials have demonstrated a decrease in the incidence of NEC in premature infants following administration of probiotics, and therefore, probiotics have been proposed as an effective prophylaxis for NEC [4]. Based on this knowledge, administration of probiotics has been proposed to reduce the incidence of HAEC. However, the exact mechanism of action of probiotics therapy for HAEC is yet to be fully understood, and the benefits of probiotics for HAEC are not clear. Therefore, we conducted a systematic review and meta-analysis to determine the effect of probiotics prophylaxis on postoperative HAEC. The present meta-analysis revealed that administration of probiotics was not associated with a significant reduction in the risk of HAEC.
As pharmacological effects, it is thought that probiotics can directly supply the body’s normal physiological bacteria, adjust the intestinal flora balance, inhibit and eliminate intestinal pathogens, reduce intestinal toxins, promote the body digesting nutrients, synthesize vitamins that organism required, and stimulate immunity [8]. Khan et al. [12] reported that probiotics potentially play a protective role in maintaining intestinal mucosal integrity through a number of different interaction, including alterations in mucosal cytokine expression, competing with intestinal pathogens for mucosal receptors, thereby increasing transepithelial resistance. Wang et al. [8] investigated the effect of oral probiotics in the patients with HSCR. Their result revealed that probiotics not only significantly decreased the incidence of HAEC but also decreased pro-inflammatory cytokines and increased anti-inflammatory cytokines. Shen et al. [13] reported markedly decreased Lactobacilli and Bifidobacteria in HAEC patients and speculated that this may result in a decrease in epithelial barrier function and be a predisposing factor in the development of HAEC. They suggested that treatment with probiotics may be beneficial in the prevention of HAEC [13].
Some case studies have reported sepsis associated with the use of probiotics [14]. In the present study, no studies reported side effects of probiotics. A recent review article summarized the side effects of probiotics [14]. There were some case reports that clearly documented occurrences of sepsis caused by the administered probiotic microbe. This risk is particularly relevant to premature infants given the immaturity of their intestinal barrier and increased risk of translocation of intestinal microbes into the lymphatic and/or systemic circulation. Similarly, in the patients with HSCR, intestinal barrier dysfunction has been proposed as one of the causes of HAEC [15, 16]. Among the probiotic products commonly administered to premature infants, there are reports of sepsis from Saccharomyces boulardii in one preterm infant and three term infants [17, 18], Lactobacillus rhamnosus GG in several premature infants and term infants many of whom had either congenital heart disease, gastroschisis, or short gut syndrome [19,20,21,22,23,24], and in a few infants receiving probiotic Bifidobacteria [25,26,27].
The limitations of this study include small number of articles, only two randomized control trials, and ununified antibiotics. Accordingly, additional randomized control studies are needed using unified antibiotics including comparing probiotic strains, doses, duration of administration, and the incidence of sepsis.
In conclusion, this study showed that administration of probiotics was ineffective in a significant reduction in the risk of HAEC. Additional studies are required to understand more fully the role of microbiota and complex interactions that cause HAEC. With increasing knowledge of the role of microbiota in HAEC, we are likely to understand better the potential benefits of probiotics in this disease.
References
Michail S, Abernathy F (2003) Lactobacillus plantarum inhibits the intestinal epithelial migration of neutrophils induced by enteropathogenic Escherichia coli. J Pediatr Gastroenterol Nutr 36:385–391
Michail S, Abernathy F (2002) Lactobacillus plantarum reduces the in vitro secretory response of intestinal epithelial cells to enteropathogenic Escherichia coli infection. J Pediatr Gastroenterol Nutr 35:350–355
Salari P, Nikfar S, Abdollahi M (2012) A meta-analysis and systematic review on the effect of probiotics in acute diarrhea. Inflamm Allergy Drug Targets 11:3–14
Deshpande G, Rao S, Patole S, Bulsara M (2010) Updated meta-analysis of probiotics for preventing necrotizing enterocolitis in preterm neonates. Pediatrics 125:921–930
Holubar SD, Cima RR, Sandborn WJ, Pardi DS (2010) Treatment and prevention of pouchitis after ileal pouch-anal anastomosis for chronic ulcerative colitis. Cochrane Database Syst Rev 6:CD001176
Mattar AF, Coran AG, Teitelbaum DH (2003) MUC-2 mucin production in Hirschsprung’s disease: possible association with enterocolitis development. J Pediatr Surg 38:417–421 (discussion 417–421)
El-Sawaf M, Siddiqui S, Mahmoud M, Drongowski R, Teitelbaum DH (2013) Probiotic prophylaxis after pullthrough for Hirschsprung disease to reduce incidence of enterocolitis: a prospective, randomized, double-blind, placebo-controlled, multicenter trial. J Pediatr Surg 48:111–117
Wang X, Li Z, Xu Z, Wang Z, Feng J (2015) Probiotics prevent Hirschsprung’s disease-associated enterocolitis: a prospective multicenter randomized controlled trial. Int J Colorectal Dis 30:105–110
Frykman PK, Nordenskjold A, Kawaguchi A, Hui TT, Granstrom AL, Cheng Z et al (2015) Characterization of bacterial and fungal microbiome in children with Hirschsprung disease with and without a history of enterocolitis: a multicenter study. PLoS One 10:e0124172
Demehri FR, Frykman PK, Cheng Z, Ruan C, Wester T, Nordenskjold A et al (2016) Altered fecal short chain fatty acid composition in children with a history of Hirschsprung-associated enterocolitis. J Pediatr Surg 51:81–86
Yin Y, Wang Z, He X (2011) Clinical study on probiotics in the prevention of hirschsprung’s enterocolitis after surgery. Anhui Med J 6:5
Khan MW, Kale AA, Bere P, Vajjala S, Gounaris E, Pakanati KC (2012) Microbes, intestinal inflammation and probiotics. Expert Rev Gastroenterol Hepatol 6:81–94
Shen DH, Shi CR, Chen JJ, Yu SY, Wu Y, Yan WB (2009) Detection of intestinal bifidobacteria and lactobacilli in patients with Hirschsprung’s disease associated enterocolitis. World J Pediatr 5:201–205
Underwood MA (2017) Impact of probiotics on necrotizing enterocolitis. Semin Perinatol 41:41–51
Keita AV, Soderholm JD (2010) The intestinal barrier and its regulation by neuroimmune factors. Neurogastroenterol Motil 22:718–733
Snoek SA, Verstege MI, Boeckxstaens GE, van den Wijngaard RM, de Jonge WJ (2010) The enteric nervous system as a regulator of intestinal epithelial barrier function in health and disease. Expert Rev Gastroenterol Hepatol 4:637–651
Belet N, Dalgic N, Oncel S, Ciftci E, Ince E, Guriz H et al (2005) Catheter-related fungemia caused by Saccharomyces cerevisiae in a newborn. Pediatr Infect Dis J 24:1125
Perapoch J, Planes AM, Querol A, Lopez V, Martinez-Bendayan I, Tormo R et al (2000) Fungemia with Saccharomyces cerevisiae in two newborns, only one of whom had been treated with ultra-levura. Eur J Clin Microbiol Infect Dis 19:468–470
Dani C, Coviello CC, Corsini II, Arena F, Antonelli A, Rossolini GM (2016) Lactobacillus sepsis and probiotic therapy in newborns: two new cases and literature review. AJP Rep 6:e25-29
Brecht M, Garg A, Longstaff K, Cooper C, Andersen C (2016) Lactobacillus sepsis following a laparotomy in a preterm infant: a note of caution. Neonatology 109:186–189
Kunz AN, Noel JM, Fairchok MP (2004) Two cases of Lactobacillus bacteremia during probiotic treatment of short gut syndrome. J Pediatr Gastroenterol Nutr 38:457–458
Land MH, Rouster-Stevens K, Woods CR, Cannon ML, Cnota J, Shetty AK (2005) Lactobacillus sepsis associated with probiotic therapy. Pediatrics 115:178–181
De Groote MA, Frank DN, Dowell E, Glode MP, Pace NR (2005) Lactobacillus rhamnosus GG bacteremia associated with probiotic use in a child with short gut syndrome. Pediatr Infect Dis J 24:278–280
Sadowska-Krawczenko I, Paprzycka M, Korbal P, Wiatrzyk A, Krysztopa-Grzybowska K, Polak M et al (2014) Lactobacillus rhamnosus GG suspected infection in a newborn with intrauterine growth restriction. Benef Microbes 5:397–402
Bertelli C, Pillonel T, Torregrossa A, Prod’hom G, Fischer CJ, Greub G et al (2015) Bifidobacterium longum bacteremia in preterm infants receiving probiotics. Clin Infect Dis 60:924–927
Zbinden A, Zbinden R, Berger C, Arlettaz R (2015) Case series of Bifidobacterium longum bacteremia in three preterm infants on probiotic therapy. Neonatology 107:56–59
Ohishi A, Takahashi S, Ito Y, Ohishi Y, Tsukamoto K, Nanba Y et al (2010) Bifidobacterium septicemia associated with postoperative probiotic therapy in a neonate with omphalocele. J Pediatr 156:679–681
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Nakamura, H., Lim, T. & Puri, P. Probiotics for the prevention of Hirschsprung-associated enterocolitis: a systematic review and meta-analysis. Pediatr Surg Int 34, 189–193 (2018). https://doi.org/10.1007/s00383-017-4188-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-017-4188-y