Abstract
Paraneoplastic cerebellar degeneration (PCD) can occur severely and appear as subacute cerebellar syndrome. PCD may be associated with small cell lung cancer, adenocarcinoma, breast cancer, ovarian carcinoma, and Hodgkin’s lymphoma. An 11-year-old male was admitted with acute cerebellar ataxia, dysarthria, and diplopia. Mediastinal conglomerated lymph nodes were depicted in a chest computed tomography (CT) examination, and diagnosis of stage IV Hodgkin’s lymphoma was obtained after a lymph node biopsy. The antibodies against Purkinje cells (anti-Tr antibody) were positive immunohistochemically. Thus, paraneoplastic cerebellar degeneration depending on Hodgkin’s disease was diagnosed. Despite the completion of chemotherapy, neurological recovery was not observed in the patient and plasmapheresis with immunoadsorption, and intravenous immunoglobulin (IVIG) was performed. Truncal ataxia, gait disturbance, and tremors decreased. Consequently, we thought that plasmapheresis with the immunoadsorption method and IVIG therapy might be a treatment option for cerebellar ataxia caused by a mechanism of immune ancestry.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Paraneoplastic cerebellar degeneration (PCD) occurs due to extensive cerebellar Purkinje cell death with acute or subacute onset and rapidly progressive pancerebellar syndrome. Although metastasis or direct invasion, indirect toxicity of tumor cells, and ectopic hormones are among the underlying theories to explain pathophysiological mechanisms, it is considered to occur with the neurological autoimmune mechanism of a substantial portion [1]. It is typically presented with the findings of subacute cerebellar ataxia [2]. Sometimes, paraneoplastic symptoms appear before the detection of the tumor; the diagnosis of the tumor can even take months or years. Detection of antineuronal antibodies against onconeuronal antigens is important to determine the tumor type and diagnose paraneoplastic syndrome [3]. This case report presents a child with acute cerebellar syndrome who was diagnosed with Hodgkin’s lymphoma during clinical follow-up.
Case
An 11-year-old male patient presented with a one-week history of unstable walking, slow speech, and double vision complaints. There were no accompanying complaints of fever or any systemic findings. There was no lymphadenopathy nor hepatosplenomegaly. His speech was slurred. Muscle strength and deep tendon reflexes were normal. There was bilateral dysmetria and diadochokinesis, inability of tandem walking, and ataxic walking upon examination.
Routine hematological and biochemical investigations were within normal limits. Contrast-enhanced cranial magnetic resonance imaging (MRI) and cerebrospinal fluid (CSF) analysis were normal. Due to the suggestion of paraneoplastic syndrome in differential diagnosis, thoracoabdominal CT was performed. There was no lymphadenopathy in the mediastinum and abdomen. Paraneoplastic antibodies including anti-Hu, anti-Yo, anti-Ri, anti-Ma2/Ta, anti-CV2, anti-amphiphysin, anti-recoverin, SOX1 antibodies, and thyroid autoantibodies were negative. Urine valine mandelic acid and homovalinic acid levels were detected within normal limits. During follow-up, the patient was referred to another hospital at the parents’ request.
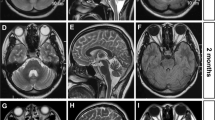
Six months after the beginning of the complaints, he was admitted again with symptoms of cough, fever, and weight loss. The patient’s physical examination revealed cervical-, axillary-, and inguinal-generalized lymph nodes in addition to cerebellar symptoms and truncal ataxia. Conglomerated lymph nodes were detected in the upper mediastinum, both hilar and subcarinal regions in the chest CT examination (Fig. 1). A lymph node biopsy was obtained from the cervical region, and nodular sclerosing type of non-Hodgkin’s lymphoma was diagnosed and classified as stage IVB to Ann Arbor. He was treated with the COEP (vincristine, cyclophosphamide, etoposide, prednisolone) protocol 4 and the OCEP (vincristine, adriamycin, etoposide, prednisolone) protocol 2 times. After completion of the chemotherapy, positron emission tomography (F-18 FDG PET/CT) revealed no activity suggesting residual lymphoma disease but a significant decrease in the cerebellar uptake (Fig. 2). Cerebral and cerebellar atrophy was indicated when compared with initial cranial MRI findings (Fig. 3). Paraneoplastic cerebellar degeneration was suspected, and anti-Tr antibodies were detected on paraneoplastic antibody screening of blood and CSF. Plasmapheresis with the immunoadsorption method (using IMMUSORBA TR 350 selective column) was repeated six times, and then IVIG was given at 400 mg/kg/day for 5 days. During the following period, IVIG was additionally given four more times every 3 weeks. At the sixth month of the follow-up, dysarthria disappeared. Truncal ataxia and tremors did not completely disappear, but he became able to walk unassisted. Long-term immunosuppressive treatment was not required.
Discussion
PCD pathogenesis is still unclear, although antibodies of intrathecal synthesis and autoimmune mechanism in the cerebellum with the presence of inflammatory infiltration against Purkinje cell antigens are held responsible [4]. The most recent studies, especially high levels of anti-Tr and anti-mGluR1 antibodies, were classified as predominant antibodies in Hodgkin’s lymphoma [1]. Anti-Tr antibodies are responsible for PCD by a specific immunohistochemical mechanism against cerebellum in Hodgkin’s lymphoma. The target antigen of this antibody expresses in the dendrites of Purkinje cells. This antigen is likely delta protein which has a role in the formation of transmembrane proteins or notch-like epidermal growth factor-related receptor (DNER) [5]. Anti-Tr antibodies can be detected in serum and CSF [1]. In our patient, anti-Tr antibodies were positive in serum and CSF, and DNER was also found reactive for the antibodies.
In PCD, as mentioned earlier, pathogenesis of neuronal damage is the result of autoimmunity. According to this hypothesis, antineuronal antibodies normally found in neuronal tissues, although responsibility for the pathogenesis is expressed by overcoming available limits in the mutagen or abnormal tissue [6]. Polyclonal B-cell activation shown in Hodgkin’s lymphoma may be responsible for autoimmune pathology without the Tr antigen expression released from malignant tissue in the cerebellum. Also, it is considered that PCD can occur as a result of the T-cell-mediated cytotoxic response in the central nervous system in addition to the effects of antineuronal antibodies [7].
Treatment options are limited, and a decline in antibody titers is possible with treatment of malignancy [3]. In addition to chemotherapy, intravenous immunoglobulin, plasmapheresis, immunosuppressive therapy (prednisolone, cyclophosphamide), as well as other therapeutic interventions had been tried, and variable results had been obtained [7]. Irreversible loss of Purkinje cells and cerebellar atrophy determines the effectiveness of the treatment [8]. Although the target is surface antigen of the DNER in anti-Tr-associated PCD, it does not respond well to the treatment when compared with the diseases characterized by the antibodies when compared to the other surface antigens [9]. Plasmapheresis is also an important treatment method in the presence of autoimmune cerebellar ataxia. By this method, antineuronal antibodies, such as medium- and large-sized molecules can be eliminated by plasma protein fraction [8]. In our case, anti-Tr antibodies against Purkinje cells were detected in the serum and CSF. After plasmapheresis with the immunoadsorption method and IVIG treatment, cerebellar ataxia declined, and body control and unassisted walking were regained.
In conclusion, PCD is characterized by panserebellar syndrome with acute/subacute onset. It is a very rare condition in childhood. The diagnosis and treatment of underlying malignancy determine the prognosis before widespread and irreversible neuronal loss. In cases with Hodgkin’s lymphoma related to paraneoplastic syndrome, the presence of ongoing cerebellar symptoms despite treatment of primary malignancy should suggest the diagnosis of PCD. In this condition, the immune elimination method can be used to eliminate the antigenic stimulus. Immunoadsorption with plasma exchange and IVIG combination can be used as a reliable treatment method in children with PCD.
References
Graus F, Arino H, Dalmau J (2014) Paraneoplastic neurological syndromes in Hodgkin and non-Hodgkin lymphomas. Blood 123:3230–3238
Darnell RB, Posner JB (2011) Paraneoplastic syndromes. Contemporary neurology series. Oxford University Press 79:1–482
Shams’ili S, Grefkens J, de Leeuw B, van den Bent M, Hooijkaas H, van der Holt B et al (2003) Paraneoplastic cerebellar degeneration associated with antineuronal antibodies: analysis of 50 patients. Brain 126:1409–1418
Dalmau J, Rosenfeld MR (2008) Paraneoplastic syndromes of the CNS. Lancet Neurol 7:327–340
Bernal F, Shams’ili S, Rojas I, Sanchez-Valle R, Saiz A, Dalmau J et al (2003) Anti-Tr antibodies as markers of paraneoplastic cerebellar degeneration and Hodgkin’s disease. Neurology 60:230–234
Posner JB (1995) Paraneoplastic syndromes. In: Posner JB (ed) Neurologic complications of cancer. F.A. Davis, Philadelphia, pp. 353–385
Rewcastle NB (1963) Subacute cerebellar degeneration with Hodgkin’s disease. Arch Neurol 9:407–413
Trotter JL, Hendin BA, Osterland CK (1976) Cerebellar degeneration with Hodgkin’s disease. An immunological study. Arch Neurol 33:660–661
Graus F, Dalmau J (2007) Paraneoplastic neurological syndromes: diagnosis and treatment. Curr Opin Neurol 20:732–737
Acknowledgments
None
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare they have no conflict of interest.
Funding
None
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s00381-017-3396-z.
Rights and permissions
About this article
Cite this article
Gungor, S., Kilic, B., Arslan, M. et al. Hodgkin’s lymphoma associated with paraneoplastic cerebellar degeneration in children: a case report and review of the literature. Childs Nerv Syst 33, 509–512 (2017). https://doi.org/10.1007/s00381-016-3284-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-016-3284-y