Abstract
Object
Ventriculoperitoneal (VP) shunts in infants with posthemorrhagic hydrocephalus (PHH) are prone to failures, with some patients at risk for multiple revisions. The objective of our study is to observe long-term outcomes and identify factors leading to proximal and distal multiple failures.
Methods
We performed a retrospective review of infants with PHH that required VP shunt placement between 1982 and 2014. These patients were monitored clinically and radiographically after VP shunt placement.
Results
A total of 502 surgical procedures (initial shunt insertion and revisions) were performed, with 380 shunt revisions in 102 (84 %) patients. Median shunt survival time was 54 months (0.03–220 months). Shunt survival was significantly affected by the following factors: intraventricular hemorrhage (IVH, grade II–III, 95 months vs. grade IV, 28 months, p = 0.022), birth weight (<1.5 kg, 59 months vs. >1.5 kg, 22 months, p = 0.005), gestational age (>27 weeks, 90 months vs. <27 weeks, 20 months, p < 0.0001), distal vs. proximal revision (133 months vs. 48 months, p = 0.013), obstruction (yes, 78 months vs. no, 28 months, p = 0.007), and infection (no, 75 months vs. yes, 39 months, p = 0.045). Regression analysis revealed that multiple gestation, head circumference (>27 cm), congenital anomalies, infection, and obstruction increased the proximal and distal shunt malfunction.
Conclusion
Long-term outcome of VP shunt placement in infants revealed a relatively high rate of complications requiring shunt revision as late as 30 years after initial placement. Infants with VP shunts should be monitored lifelong of these patients by neurosurgeons.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Twenty to 25 % of preterm babies with very low birth weight have intraventricular hemorrhage (IVH) [ 1, 2 ]. With advancement in technology and medical care, the mortality rate of extremely low birth weight infants has significantly improved while increasing the incidence of IVH. Fortunately, most of these hemorrhages are mild (grades I and II IVH) and resolved with no or few complications. However, severe IVH (grade III and IV) may result in serious complications including enlarged ventricle and hydrocephalus, cerebral palsy, visual impairment, hearing defects, and learning disabilities. The incidence of short-term complications including hydrocephalus is as high as 25–36 % [ 1 , 3 , 4 ]. Among those infants with posthemorrhagic hydrocephalus requiring surgical cerebrospinal fluid (CSF) diversion, 72.6–90 % eventually need CSF shunts [ 4 , 5 ]. CSF shunts are prone to failures requiring multiple revisions [ 6 , 7 ] and cost up to a billion dollars a year [ 8 , 9 ]. To our knowledge, no study has investigated the long-term outcome (>30 years) and predictors of individual proximal and distal shunt failures in infants with posthemorrhagic hydrocephalus (PHH). We performed a retrospective analysis of patients presenting with PHH that needed CSF diversion between 1982 and 2013. The purpose of the study was to investigate long-term outcome and identify predictors of multiple proximal and distal shunt revisions in patients with PHH.
Methods
The present study was approved by the Institutional Review Board at LSU Health Sciences Center-Shreveport. A surgical database (MD Analyze, Medtech Global Ltd., South Melbourne, Victoria, Australia) and EPIC electronic health record software (Epic) were used to get information about surgical procedures. Once the patients were identified, further data related to clinical history, operative reports, imaging studies, and outcomes of the patients with PHH between 1982 and 2013 were collected retrospectively by review of the patients’ charts and follow-up notes. The details of the patients’ inclusion and exclusion criteria and their selection for the study are summarized in Fig. 1. The inclusion criteria for the study were PHH patients of the Department of Neurosurgery, LSU Health, and who went through CSF diversion.
Patients’ demographics
Of the neurosurgical service’s 122 PHH patients, 67 (54.9 %) were male and 55 (45.1 %) were female; 79 (64.8 %) of the patients were African Americans, and 36 (29.5 %) were Caucasians. Fifty-seven (46.7 %) patients had IVH grade III, and 56 (45.1 %) patients had IVH grade IV. In addition, 93 (76.2 %) patients had associated congenital anomalies. Among congenital anomalies, 50.8 % were central nervous system-related anomalies such as cerebral palsy and spinal dysraphism. However, 25.4 % cases had non-CNS-related congenital anomalies such as cardiac and renal anomalies. Out of 122 patients, 102 patients underwent shunt replacement and revisions (Figs. 2 and 3 , Tables 1 and 2).
a–c Preoperative ultrasonogram of brain in an infant showing intraventricular hemorrhage (a). Preoperative CT scan in an infant showing ventricular dilatation (b). A 2-year follow-up CT scan reveals shunt in situ with reduced ventricular size (c). d–f A preoperative CT scan of an infant showing ventricular dilatation (d). A preoperative MRI scan of an infant showing ventricular dilatation (e). CT scan shows the 1-year follow-up after VP shunt placement showing shunt in situ with reduced ventricular size (f)
Follow-up
Preoperative and follow-up data were collected from the patients’ case reports and follow-up notes. Neuroimaging studies were performed at 3-month intervals in the first year after ventriculoperitoneal (VP) shunt placement, at 6-month intervals for the following 2 years, and annually thereafter. The mean duration of follow-up was 78 (1 month–334 months).
Statistical analysis
The analysis was conducted using Statistical Package for Social Sciences (SPSS) software, version 21.0 (IBM Corp., Armonk, NY). The Kaplan–Meier method of survival analysis was used to estimate the overall and revision-free survival rate. The log-rank (Mantel–Cox) test was used to analyze the survival difference. The Cox proportional hazards regression model was used to determine independent significant factors for shunt survival. A p value <0.05 was considered as significant.
Results
Rate of shunt revision
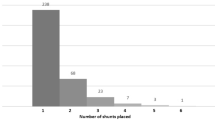
The median follow-up time of our series was 55 (1–334) months. Of the 122 cases with PHH after VP shunt placement, 102 (84 %) patients experienced single or multiple shunt failures requiring shunt revisions. Single shunt revision was required in 30 (24.6 %) patients, and multiple shunt revisions were needed in 72 (59 %) patients. After the initial shunt placement, there were 2–4 revisions in 41(33.6 %) patients, ≥5 revisions in 23 (18.9 %) patients, and ≥10 revisions in 8 (6.5 %) patients (Table 2 , Fig. 2 ). Moreover, after initial shunt placement, 64 (52 %), 69 (57 %), 73 (60 %), and 84 (68.8 %) patients developed shunt failures requiring shunt revisions within 3 months, 6 months, 1 year, and 5 years, respectively.
Survivability of the shunts
The median survival of shunt was 54.3 months (Fig. 4a).
Shunt survival according to different factors
Site of shunt revision (proximal vs. distal)
Overall survival of the shunt varied according to the site of shunt revision. Overall shunt survival for proximal and distal shunt revision was 48 and 133 months, respectively. The difference in the median survival of shunt achieved statistical significance (log-rank p < 0.013) (Fig. 4b).
Grades of IVH
The overall shunt survival for patients with IVH grades II–III and IVH grade IV was 28 and 95 months, respectively. The difference in the median survival achieved statistical significance (log-rank p < 0.022) (Fig. 5a).
Gestational age
The overall shunt survival for patients with gestational age <27 and >27 weeks was 20 and 90 months, respectively. The difference in the median survival achieved statistical significance (log-rank p < 0.0001) (Fig. 5b).
Birth weight
The overall shunt survival for patients with birth weight less than 1.5 kg and greater than 1.5 kg was 22 and 59 months, respectively. The difference in the median survival achieved statistical significance (log-rank p < 0.005) (Fig. 5c).
Causal factors for shunt revision
The most common reasons for shunt revisions in patients with PHH are described in Table 3 and Fig. 2. Neurosurgeons performed 502 surgical procedures (initial shunt insertion and revisions), including 380 shunt revisions in 102 (84 %) patients. The most common indication for revisions was obstruction of the shunt system (156, 41 %), followed by shunt infections (53, 17.2 %) and overdrainage (33, 10.7 %). Among total revision, 147 (39 %) were proximal, 42 (11 %) distal, and 55 (14.5 %) total shunt revisions. In this series, the patients that return with multiple shunt revisions had congenital anomalies leading to multiple shunt failures.
Obstruction
Based on presence or absence of obstruction, shunt survival varied. Shunt survival for patients with and without shunt obstruction was 28 and 78 months, respectively. The difference in the median survival achieved statistical significance (log-rank p = 0.007) (Fig. 6a).
a Survival of the shunt based on patients with or without obstruction; b survival of the shunt based on patients with or without infection; c survival of the shunt based on patients with or without overdrainage. Percentage in the bottom of each graph indicates the percent of patients in follow-up at that particular time. *p < 0.05 is considered as significant
Infection
Shunt survival for patients with and without shunt infection was 39 and 75 months, respectively. The difference in the median survival achieved statistical significance (log-rank p = 0.045) (Fig. 6b).
Overdrainage
Shunt survival for patients with and without shunt overdrainage was 49 and 54 months, respectively. The difference in the median survival did not reach statistical significance (log-rank p = 0.33) (Fig. 6c).
Factors for shunt failure after 10 years of first shunt revision
A total of nine patients were included in this cohort. The shunt survival varied based on different factors.
Birth weight
The median shunt survival for patients with birth weight <1.5 kg and >1.5 kg was 164 and 219 months, respectively. The difference in the median survival achieved statistical significance (log-rank p < 0.021) (Fig. 7a).
Shunt survival analysis in the patients requiring shunt revisions after 10 years. a Survival of the shunt based on birth weight; b survival of the shunt based on gestational age. Percentage in the bottom of each graph indicates the percent of patients in follow-up at that particular time. *p < 0.05 is considered as significant
Gestational age
The median shunt survival for patients with gestational age <27 and >27 weeks was 172 and 219 months, respectively. The difference in the median survival achieved statistical significance (log-rank p = 0.029) (Fig. 7b).
Obstruction
Shunt survival varied based on presence or absence of obstruction. Shunt survival for patients with and without shunt obstruction was 159 and 218 months, respectively. The difference in the median survival achieved statistical significance (log-rank p < 0.045) (Fig. 8a).
Shunt survival analysis in the patients requiring shunt revisions after 10 years. a Survival of the shunt based on patients with or without obstruction; b survival of the shunt based on patients with or without infection; c survival of the shunt based on patients with or without overdrainage. Percentage in the bottom of each graph indicates the percent of patients in follow-up at that particular time. *p < 0.05 is considered as significant
Infection
Shunt survival for patients with and without shunt infection was 162 and 218 months, respectively. The difference in the median survival achieved statistical significance (log-rank p < 0.036) (Fig. 8b).
Overdrainage
The shunt survival for patients with and without shunt overdrainage was 172 and 216 months, respectively. The difference in the median survival did not reach statistical significance (log-rank p < 0.47) (Fig. 8c).
Factors of shunt survival/risk factor for shunt revisions
Multiple revisions
In this series, 102 (83.6 %) patients underwent revisions during the follow-up period. Of the 102 patients, 72 patients had multiple revisions. Among these 72 patients, the total number of revisions was 380 with a maximum of 17 revisions in one patient. In Cox regression analysis, proximal revision (p = 0.018), IVH grade IV, gestational age <27 weeks (p = 0.006), presence of congenital anomalies (p = 0.008), birth weight <1.5 kg (p = 0.038), non-antibiotic-impregnated catheter (p = 0.001), infection (p = 0.047), and obstruction (p = 0.008) were identified as the positive predictors of multiple shunt revisions. However, gender (p = 0.63), ethnicity (p = 0.13), and overdrainage (p = 0.34) did not affect the multiple shunt revision in this series (Table 4).
Proximal revision
Among 380 total revisions, 147 (38.7 %) were proximal and 42 (11.0 %) were distal shunt revisions. In Cox regression analysis, multiple number of gestations (p = <0.0001), head circumference (>27 cm, p = 0.03), presence of congenital anomalies (p < 0.0001), infection (p = 0.048), and obstruction (p = 0.038) of the shunts negatively affected the proximal shunt survival. However, birth weight <1.5 kg (p = 0.95), gestational age (p = 0.95), and overdrainage of shunt (p = 0.83) did not affect the proximal shunt revision rates (Table 4).
Distal revision
Distal shunt survival rates were negatively affected by a number of factors. In Cox regression analysis, multiple number of gestations (p < 0.0001), head circumference (>27 cm, p = 0.04), presence of congenital anomalies (p < 0.0001), and infections (p = 0.001) of the shunts negatively affected the distal shunt survival. Birth weight (p = 0.81), gestational age (p = 0.52), shunt obstruction (p = 0.06), and overdrainage of the shunts (p = 0.67) did not affect the distal shunt survival rates (Table 4).
Discussion
IVH is a quite common and significant complication in premature neonates which may lead to subsequent formation of PHH. Shunt placement is the most widely practiced method for treating PHH [ 10 ]. However, shunt-dependent treatment of PHH is still complicated by high revision rates [ 11 , 12 ]. The patients’ prematurity and incompetent immune systems are conducive to shunt infections. In addition, an excess CSF protein level in the patients with PHH is contributive to shunt obstructions [ 13 ]. Although complications related to CSF shunt placement are known, there needs to be a better understanding of long-term VP shunt durability. To accomplish this, we retrospectively reviewed the shunt database to report our institution’s shunt revision rate and attempt to determine predictors related to proximal or distal multiple shunt failure occurring more than 15 years after shunt placement.
Higher shunt survival reflects the successful shunt placement in PHH. In the present study, overall shunt survival period was 54 months. Findings of this study revealed that shunt survivability depends on the site of shunt revision with distal revision resulting in better shunt survival (p = 0.013). This is the first report in the literature showing shunt survival according to site of shunt revision. A recent report suggested that prognosis after shunt treatment in PHH was better in grade II and III compared to grade IV [ 10 ]. The results of our cohort reflect similar findings (p = 0.022). The present study confirmed that birth weight greater than 1.5 kg (p = 0.005) and gestational age >27 weeks positively affected shunt survival as reported by previous studies [ 5 , 14 ]. In addition, research evidence suggested that multiple other factors including infection, obstruction, and overdrainage or underdrainage are also responsible for VP shunt failure [ 15 ]. Similarly, we have observed that shunt failure in these forms depends on shunt blockade (41 %) more than shunt infection (14 %) or overdrainage (8.7 %) [ 12 ]. This observation of reduced infection rate agrees with a recent publication [ 15 ] and could be explained by evidence-based advancements in aseptic measures and surgical techniques, resulting in reduced surgical site and procedure-related infections [ 16 , 17 ]. Although previous reports suggested that congenital anomalies had a negative effect on shunt survival, our results did not show any significant difference. [ 18 ].
Another goal of our study was to investigate shunt revision rate and risk factors related to shunt failure occurring more than 10 years after first revision. In this series of patients with at least 30 years of follow-up, 84 % required at least one revision, which is in accord with previous reports [ 19 , 20 ]. Shunt revision was required as late as 27 years after initial placement. Although distinguished results are available in the existing literature [ 21 ], this may be explained by the shorter duration of their study period. However, our result is a reminder that neurosurgeons need to educate their patients about the possibility of shunt revision becoming necessary later. In this study, there was a subset of patients who required shunt revision after 10 years. Multiple factors including gestational age, low birth weight (<1.5 kg), obstruction, and infection were responsible for shunt failure in this group of patients. Although Stone et al. [19] attempted to identify the factors for shunt failure in infants who underwent shunt revision after 10 years, they did not observe any. This is the first report showing factors for shunt failure after 10 years. However, only 30 % patients had more than 120 months follow-up and the percentage of patients with >200 months of follow-up period was 8 %.
Predictors for shunt revision
We also examined several variables potentially affecting the overall shunt survival in patients with PHH undergoing VP shunt replacement. In this study, multiple gestation, head circumference (>27 cm), congenital anomalies, infection, and obstruction positively affected the proximal shunt failure. Similarly, multiple gestation, head circumference (>27 cm), congenital anomalies, and infection positively affected the distal shunt failure. Like others, we have also investigated the predictors for multiple shunt revisions [ 1 , 9 ]. This study revealed that proximal shunt revision, IVH grade IV, congenital anomalies, birth weight <1.5 kg, shunt infection, shunt obstruction, and non-antibiotic-impregnated catheter type play significant positive roles on multiple shunt revision. Although a previous study has shown that gender and ethnicity [ 22 ] have significant effects on multiple shunt revisions, these factors trended toward but did not reach significance in our investigation.
Limitation
The present study has inherent limitations due to its retrospective nature. In the cohort of long term follow-up, there were only 8 % cases with >200 months of follow-up which should be further evaluated in large volume of patients. Although uniform technique for VP shunt insertion was practiced, the surgeries were performed by different neurosurgeons.
Conclusion
Although VP shunting is the widely practiced procedure to treat PHH, it is prone to have complications and requires revision. Infants with VP shunts should be monitored lifelong of these patients by neurosurgeons. Proximal shunt revision was identified as the most important factor of shorter shunt survival. Among congenital factors, birth weight (<1.5 kg), presence of congenital anomalies, and gestational age (<27 weeks) had significant effect on shunt failure. Among the causal factors, obstruction was the major cause of shunt failure. In addition, infections had significant effect on shunt failure.
References
Freimann FB, Vajkoczy P, Sprung C (2013) Patients benefit from low-pressure settings enabled by gravitational valves in normal pressure hydrocephalus. Clin Neurol Neurosurg 115:1982–1986
Lemcke J, Meier U (2010) Improved outcome in shunted iNPH with a combination of a Codman Hakim programmable valve and an Aesculap-Miethke ShuntAssistant. Cent Eur Neurosurg 71:113–116
Limbrick DD Jr, Mathur A, Johnston JM, Munro R, Sagar J, Inder T, Park TS, Leonard JL, Smyth MD (2010) Neurosurgical treatment of progressive posthemorrhagic ventricular dilation in preterm infants: a 10-year single-institution study. J Neurosurg Pediatr 6:224–230
Lee IC, Lee HS, Su PH, Liao WJ, Hu JM, Chen JY (2009) Posthemorrhagic hydrocephalus in newborns: clinical characteristics and role of ventriculoperitoneal shunts. Pediatr Neonatol 50:26–32
Willis B, Javalkar V, Vannemreddy P, Caldito G, Matsuyama J, Guthikonda B, Bollam P, Nanda A (2009) Ventricular reservoirs and ventriculoperitoneal shunts for premature infants with posthemorrhagic hydrocephalus: an institutional experience. J Neurosurg Pediatr 3:94–100
Cherian S, Whitelaw A, Thoresen M, Love S (2004) The pathogenesis of neonatal post-hemorrhagic hydrocephalus. Brain Pathol 14:305–311
Mwachaka PM, Obonyo NG, Mutiso BK, Ranketi S, Mwang’ombe N (2010) Ventriculoperitoneal shunt complications: a three-year retrospective study in a Kenyan national teaching and referral hospital. Pediatr Neurosurg 46:1–5
Patwardhan RV, Nanda A (2005) Implanted ventricular shunts in the United States: the billion-dollar-a-year cost of hydrocephalus treatment. Neurosurgery 56:139–144 discussion 144-135
Daszkiewicz P, Barszcz S (2007) Multiple shunt system revisions in patients with hydrocephalus - causes, effects, regularities and prognostic factors. Neurol Neurochir Pol 41:404–410
Koksal V, Oktem S (2010) Ventriculosubgaleal shunt procedure and its long-term outcomes in premature infants with post-hemorrhagic hydrocephalus. Childs Nerv Syst 26:1505–1515
Kadri H, Mawla AA, Kazah J (2006) The incidence, timing, and predisposing factors of germinal matrix and intraventricular hemorrhage (GMH/IVH) in preterm neonates. Childs Nerv Syst 22:1086–1090
Taylor AG, Peter JC (2001) Advantages of delayed VP shunting in post-haemorrhagic hydrocephalus seen in low-birth-weight infants. Childs Nerv Syst 17:328–333
Reinprecht A, Dietrich W, Berger A, Bavinzski G, Weninger M, Czech T (2001) Posthemorrhagic hydrocephalus in preterm infants: long-term follow-up and shunt-related complications. Childs Nerv Syst 17:663–669
Chittiboina P, Pasieka H, Sonig A, Bollam P, Notarianni C, Willis BK, Nanda A (2013) Posthemorrhagic hydrocephalus and shunts: what are the predictors of multiple revision surgeries? J Neurosurg Pediatr 11:37–42
Tuli S, Drake J, Lawless J, Wigg M, Lamberti-Pasculli M (2000) Risk factors for repeated cerebrospinal shunt failures in pediatric patients with hydrocephalus. J Neurosurg 92:31–38
Choksey MS, Malik IA (2004) Zero tolerance to shunt infections: can it be achieved? J Neurol Neurosurg Psychiatry 75:87–91
Gutierrez-Gonzalez R, Boto GR, Perez-Zamarron A (2012) Cerebrospinal fluid diversion devices and infection. A comprehensive review. Eur J Clin Microbiol Infect Dis 31:889–897
Okazaki M, Fukuhara T, Namba Y (2013) Delayed germinal matrix hemorrhage induced by ventriculoperitoneal shunt insertion for congenital hydrocephalus. J Neurosurg Pediatr 12:67–70
Stone JJ, Walker CT, Jacobson M, Phillips V, Silberstein HJ (2013) Revision rate of pediatric ventriculoperitoneal shunts after 15 years. J Neurosurg Pediatr 11:15–19
Wu Y, Green NL, Wrensch MR, Zhao S, Gupta N (2007) Ventriculoperitoneal shunt complications in California: 1990 to 2000. Neurosurgery 61:557–562 discussion 562-553
Simon TD, Hall M, Riva-Cambrin J, Albert JE, Jeffries HE, Lafleur B, Dean JM, Kestle JR, Hydrocephalus Clinical Research N (2009) Infection rates following initial cerebrospinal fluid shunt placement across pediatric hospitals in the United States. Clinical article J Neurosurg Pediatr 4:156–165
Naftel RP, Safiano NA, Falola M, Blount JP, Oakes WJ, Wellons JC 3rd (2013) The association between race and frequent shunt failure: a single-center study. J Neurosurg Pediatr 11:552–557
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Financial disclosure
None
Conflict of interest
None
Rights and permissions
About this article
Cite this article
Bir, S.C., Konar, S., Maiti, T.K. et al. Outcome of ventriculoperitoneal shunt and predictors of shunt revision in infants with posthemorrhagic hydrocephalus. Childs Nerv Syst 32, 1405–1414 (2016). https://doi.org/10.1007/s00381-016-3090-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-016-3090-6