Abstract
Stomatogastric musculature from crabs in the genus Cancer provides a system in which modulatory roles of peptides from the FLRFamide family can be compared. The anterior cardiac plexus (ACP) is a neuroendocrine release site within the Cancer stomatogastric nervous system that is structurally identical in C. borealis, C. productus, and C. magister but that appears to contain FLRFamide-like peptide(s) only in C. productus. We measured the effect of TNRNFLRFamide on nerve-evoked contractions of muscles that were nearby, an intermediate distance, or far from the ACP. We found the spatial pattern of FLRFamidergic modulation of muscles in C. productus to be qualitatively different than in C. borealis or C. magister. In C. productus, muscles proximal to the ACP were more responsive than distal muscles. In C. borealis, FLRFamidergic response was less dependent on muscle location. These results suggest that functionally different roles of FLRFamides in modulating stomatogastric muscle movements may have evolved in different Cancer species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Peptidergic modulation in the stomatogastric nervous system (STNS) has been studied in a fair amount of detail in creatures from three infraorders within the decapod suborder Reptantia: Palinura (spiny lobsters), Astacidea (lobsters and crayfish) and Brachyura (true crabs) (Katz and Tazaki 1992; Skiebe 2001). While many aspects of structure and function in the STNS are conserved across species, staining patterns in peptide immunolabeling assays have shown considerable variability, even for two species in the same genus (Cancer) (Mortin and Marder 1991). Investigating the physiological and functional significance corresponding to one of these anatomical differences is a challenging but rewarding task, as it can provide insight about neuromodulatory function and its evolution in the STNS.
The center of the STNS is the stomatogastric ganglion (STG), which consists of 25–30 mostly motor neurons that control muscle movements that produce the gastric mill (grinding) and pyloric (filtering) rhythmic motor patterns (Harris-Warrick et al. 1992). In Cancer crabs, the muscles themselves provide an opportunity to compare the effects of peptides from the FLRFamide family. In Cancer borealis, the best studied species in the genus, FLRFamide potentiates nerve-evoked contractions of the stomach muscles (Jorge-Rivera and Marder 1996; Jorge-Rivera et al. 1998) through modulatory effects both at the neuromuscular junction and on the contractile machinery (Jorge-Rivera and Marder 1996). Previous immunohistochemical investigations (Christie et al. 2004b; Marder et al. 1987) have revealed no staining in the vicinity of the musculature, so the prevailing view (Jorge-Rivera and Marder 1996) has been that the FLRFamide that acts on C. borealis muscles is secreted by neuroendocrine organs extrinsic to the STNS (Fu et al. 2005; Li et al. 2003).
Recently, however, a neuroendocrine system intrinsic to the STNS was identified that may release FLRFamide directly onto stomatogastric muscles, but in only one of three Cancer species studied (Christie et al. 2004a; Savage et al. 2006). This system is comprised of anterior commissural neurons 1 and 2 (ACN1/2), which are located and arborize in each commissural ganglion (CoG), and the anterior cardiac plexus (ACP), an endocrine release site the neurons form in each anterior cardiac nerve (acn). While the structure of the ACN1/2-ACP system is identical in C. productus, C. borealis and C. magister (Christie et al. 2004a; Savage et al. 2006), FLRFamide-like immunoreactivity was found in the ACP of C. productus (Christie et al. 2004a) but not in the other two species (Cruz-Bermudez et al. 2006; Savage et al. 2006).
To better understand the functional significance of FLRFamide release by the C. productus ACP, we measured the effects of peptide application on nerve-evoked contractions of six stomatogastric muscles in each of the three species. We report that the spatial pattern of FLRFamidergic modulation of stomatogastric musculature in C. productus is qualitatively different than in C. borealis or C. magister. In C. productus, gastric muscles that are proximal to the ACP are more responsive than distal pyloric muscles, while in C. borealis the response is less dependent on muscle location. These results suggest that the role of the ACP in modulating stomatogastric motor patterns may have evolved differently in the three species. Some of these data previously appeared in abstract form (Savage et al. 2006).
Materials and methods
Animals and solutions
Jonah crabs Cancer borealis Stimpson were purchased from the Marine Biological Laboratory (Woods Hole, Massachusetts, USA). Dungeness crabs Cancer magister Dana and red rock crabs Cancer productus Randall were collected by hand, trap or trawl at multiple locations throughout the San Juan Archipelago and greater Puget Sound areas of Washington State, USA. Regardless of species, animals were maintained in aerated seawater aquaria chilled to 10°C until used. Physiological saline had the following composition: 440 mM NaCl; 11 mM KCl; 13 mM CaCl2; 26 mM MgCl2; 11.2 mM Trisma base; 5.1 mM maleic acid; pH 7.4. The peptide TNRNFLRFamide was purchased from American Peptide (Sunnyvale, CA, USA), dissolved in distilled water at 10−3 M and stored at −20°C. Aliquots were diluted in saline to desired concentrations immediately before use.
Contraction measurements
Measurements of nerve-evoked contractions were made from the cardiac 6 (c6), cardiopyloric valve 3a (cpv3a), ventral cardiac 2 (cv2), gastric mill 4 (gm4), gastric mill 8b (gm8b), and pyloric 8 (p8) muscles, all of which are intrinsic muscles from the foregut of Cancer crabs. The c6 muscle is innervated by the anterior median (AM) neuron via the acn. The cv2 muscle is innervated by the inferior cardiac (IC) neuron via the medial ventricular nerve (mvn). The gm4 muscle, which consists of distinct gm4b and gm4c bundles of fibers, is innervated by the dorsal gastric (DG) neuron via the dorsal gastric nerve (dgn). The other three muscles are innervated via the lateral ventricular nerve (lvn): cpv3a by the lateral pyloric (LP) neuron, gm8b by the medial gastric (MG) neuron, and p8 by pyloric (PY) neurons. Nerves and muscles were identified using the nomenclature of Maynard and Dando (1974). The AM, DG and MG neurons are part of the gastric network that controls grinding movements of three teeth in the gastric mill. The IC, LP and PY neurons are part of the pyloric network that constricts and dilates valves in the pylorus, a filtering structure (Harris-Warrick et al. 1992; Maynard and Dando 1974; Selverston and Moulins 1987; Weimann et al. 1991).
The basic procedure for making muscle contraction measurements has been described previously (Messinger et al. 2005). Neuromuscular preparations consisting of one of the muscles and the innervating nerve were dissected from the Cancer foregut and pinned flat in 5 ml Petri dishes lined with Sylgard 182 (Dow Corning, Midland, MI, USA). One of the muscle insertions was pinned down in the dish, while the other was tied to a FT03 force displacement transducer (Astro-Med, West Warwick, RI, USA) with a ~3 cm piece of size 6/0 silk suture thread (Fine Science Tools, Foster City, CA, USA).
The nerve was stimulated extracellularly via a stainless steel pin electrode with a train of 1 ms unipolar pulses produced by an isolated pulse stimulator (Model 2100, A-M Systems, Carlsborg, WA, USA). This resulted in muscle shortening, and the force transducer measured the increased tension. The transducer signal was amplified (Model 440, Brownlee Precision, San Jose, DA, USA) by a factor of 10,000, recorded using a Digidata 1322A acquisition system (Axon Instruments, Union City, CA, USA), and analyzed using the Clampfit program (Axon Instruments). During experiments the bath volume was maintained at ~3 ml, and the preparation was continuously superfused (4–5 ml/min) with saline that was cooled with an ice bath and regulated to within half a degree at a temperature between 9 and 11°C. Solutions containing TNRNFLRFamide (10−9–10−7 M) were bath-applied by means of a switching port at the inflow of the superfusion system.
Statistics
A two-way ANOVA was used to analyze and compare the dose–response relationships for the effect of TNRNFLRFamide on gm4 peak contractile force in C. productus and C. borealis. The Holm–Sidak method was then used to make pairwise comparisons. In the experiments in which the effect of only a single concentration (10−7 M) of TNRNFLRFamide was tested, a paired t test was used to test for statistical significance. In instances when the normality condition was not satisfied, the Wilcoxon signed rank test was implemented. All statistical tests were done using SigmaStat (Version 3.1, Systat Software, Point Richmond, CA, USA). Statistical significance was indicated on figures using the following symbols: * P < 0.05, ** P < 0.01, *** P < 0.001. Error bars on all plots correspond to standard errors.
Figure production
Figures were produced using the Canvas (Version 9, ACD Systems, Miami, FL, USA) and SigmaPlot (Version 8.0, Systat Software, Point Richmond, CA, USA) software packages.
Results
Sensitivity of modulation of muscle contraction by TNRNFLRFamide is similar in C. productus and C. borealis
FLRFamides have previously been shown to increase nerve-evoked contraction and/or induce myogenic activity in crustacean stomatogastric muscles (Meyrand and Marder 1991), including those of Cancer borealis (Jorge-Rivera and Marder 1996). In the latter investigation, it was observed that application of either TNRNFLRFamide or SDRNFLRFamide modulated muscle contraction in 15 of 17 muscles tested and that the threshold concentration for an effect was 10−10 M and 10−9–10−8 M, respectively. To determine whether FLRFamides similarly enhance contraction in C. productus, we initially measured the effects of the more potent TNRNFLRFamide peptide on contraction of the gm4 muscle. We chose this muscle because a previous study showed that, in C. borealis, gm4 is modulated by at least ten neuropeptides including both FLRFamides (Jorge-Rivera et al. 1998), and hence we hypothesized that it would be the muscle in C. productus most likely to be modulated.
Figure 1a shows nerve-evoked contractions of gm4 in C. productus that were elicited every minute by 1 s duration, 20 Hz electrical stimulation of the dgn, before and during application of 10−8 M TNRNFLRFamide. The two individual contractions representing the control (C) and maximal (M) modulated states are replotted in Fig. 1b. The peak contractile force in response to a stimulation increased fourfold in the presence of TNRNFLRFamide, but desensitization to the peptide occurred (Fig. 1a), as has been observed elsewhere in the STNS (Birmingham et al. 2003) and in crustacean neuromuscular preparations (Worden et al. 1995). Subsequent applications of TNRNFLRFamide resulted in much smaller effects on contraction, even after an hour of rinse (data not shown), as has been reported previously (Jorge-Rivera and Marder 1996). Figure 1c is a dose–response relationship plotting the ratios of the peak contractile forces under (maximal) modulated and control conditions for gm4 contractions in C. productus elicited by 1 s, 20 Hz stimulation. For comparison, we made dose-response measurements in corresponding preparations from C. borealis (Fig. 1c). Because of desensitization in both species, a particular neuromuscular preparation could be tested at only a single peptide concentration; each bar represents the average of five experiments. For both animals, pronounced effects on contraction were first observed at 10−8 M. Significant differences in the modulatory response were measured between concentrations within each species and between species for a given concentration.
TNRNFLRFamide increases the contractile force produced by a stomatogastric muscle in C. productus. a Contractions of the gastric mill 4 (gm4) muscle before and during application of 10−8 M TNRNFLRFamide in response to 1 s, 20 Hz stimulation of the dgn made once per minute. b Expanded traces for the contractions corresponding to the control (C) and maximal (M) modulated conditions for the experiment shown in a. c Dose–response relationship for the effect of TNRNFLRFamide on gm4 peak contractile force (normalized to control) elicited by 1 s, 20 Hz stimulation in C. productus and C. borealis. Experiments on five preparations were used to generate each bar. Different preparations were required for each concentration because of the desensitization that is illustrated in a. A two-way ANOVA was performed on the data set after applying a logarithmic transform to the ratios (i.e. responses normalized to controls) to satisfy requirements of normality and homogeneity of variances. Statistically significant differences in the mean for the two species (P = 0.0056) and among the different concentrations (P < 0.000001) were found, but there was not a statistically significant interaction between species and concentration (P = 0.089). Pairwise comparisons between concentrations within a species and between species for each concentration were made using the Holm-Sidak method, with significant differences indicated on the figure. For C. borealis, the difference between the means for 10−9 M versus 10−8 M was not quite significant (P = 0.092)
Relative magnitude of modulation of stomatogastric musculature by TNRNFLRFamide varies among species of Cancer crabs
While at least ten substances, including TNRNFLRFamide and SDRNFLRFamide, have been identified as neuromodulators of stomatogastric musculature in C. borealis (Jorge-Rivera and Marder 1996; Jorge-Rivera and Marder 1997; Jorge-Rivera et al. 1998), modulation of stomach muscles in C. productus has been documented only for the peptide CabTRP Ia (Messinger et al. 2005), and in C. magister for the monoamine dopamine (Lingle 1981). In C. borealis, depolarization of the pericardial organs releases TNRNFLRFamide and SDRNFLRFamide into the hemolymph (Li et al. 2003), and we would expect that FLRFamides circulate at similar levels in C. productus and C. magister. As mentioned earlier, anatomical investigations (Christie et al. 2004a; Savage et al. 2006) showed that an additional source of FLRFamide in the STNS of C. productus could be the ACN1/2-ACP system. Local release from the ACP might expose muscles near the release site to higher concentrations of FLRFamides than more distal muscles. Thus we wished to determine if the responses to FLRFamide of muscles proximal and distal to the ACP in C. productus differ from those in C. borealis or C. magister.
We chose three gastric (c6, gm4, gm8b) and three pyloric (cpv3a, cv2, p8) muscles with which to quantify the effect of 10−7 M TNRNFLRFamide on nerve-evoked contraction in each of the three species. We picked these particular muscles because we wanted to study relatively large muscles that would produce easily measured contractions and that were either nearby (c6, gm4), far (cv2, p8), or an intermediate distance (cpv3a, gm8b) from the ACP (Fig. 2). In particular, the c6 muscle is innervated via the acn, the nerve in which the ACP is located. For all muscles, we used a 1 s train of electrical pulses to elicit contractions. As in previous studies of Cancer musculature (Jorge-Rivera and Marder 1996; Jorge-Rivera and Marder 1997; Jorge-Rivera et al. 1998; Le et al. 2006; Messinger et al. 2005) a constant frequency stimulus was used: 20 Hz for muscles (gm4, gm8b, p8) that produced larger, more easily measurable contractions, and 40 Hz for muscles (c6, cpv3a, cv2) that produced smaller contractions.
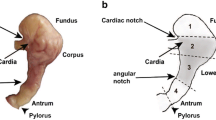
Schematic representation (after Weimann et al. 1991) showing the location of the anterior cardiac plexus (ACP) relative to some of the stomatogastric muscles in Cancer crabs. Dorsal view with only left side is shown. Anterior is up, and medial is to the right. Intrinsic muscles studied in this investigation are shown in light gray, other intrinsic muscles are dark gray, extrinsic muscles are white, and ossicles are black. c6, cardiac 6 muscle; cpv3a, cardiopyloric valve 3a muscle; cv2, ventral cardiac 2 muscle; gm4b,c, gastric mill 4b,c muscle; gm8b, gastric mill 8b muscle; p8, pyloric 8 muscle; acn anterior cardiac nerve; dacn dorsal anterior cardiac nerve; lacn lateral anterior cardiac nerve; on esophageal nerve; son superior esophageal nerve; stn stomatogastric nerve; STG stomatogastric ganglion. The ACP and STG are located within nerves and are not drawn to scale. Muscles are drawn approximately to scale. The gm4 muscles, for example, are approximately 1–2 cm in length, but muscle size varies both across and within species. In general, C. productus is the smallest animal and C. magister the largest
Application of TNRNFLRFamide resulted in enhanced contractions in all three crab species. Representative contractions under control conditions and in the presence of 10−7 M TNRNFLRFamide are shown in Fig. 3a for a muscle from each species. As in Fig. 1a, desensitization was observed in these experiments, and so all results reflect the maximal modulatory effect that occurred 5–10 min after application of the peptide. A summary of results for the six muscles is shown in Fig. 3b. Overall, the effect of TNRNFLRFamide was larger in C. productus than in C. borealis, and the smallest effect was observed in C. magister. Within each species, however, the relative magnitude of the modulatory effect varied greatly and appeared to be correlated with a muscle’s location. In C. productus very large (100–350%) average increases in the contractile forces produced by gastric muscles (c6, gm4, gm8b) were observed, while the effects on the distal pyloric muscles (cpv3a, cv2, p8) were far smaller (≤30% over basal). In C. borealis prominent (>55%) modulatory effects were noted for all muscles except cv2, consistent with an earlier study (Jorge-Rivera and Marder 1996). The effects on the three gastric muscles were smaller than in C. productus, but those for cpv3a and p8 in C. borealis were noticeably larger. In C. magister there was little or no modulatory effect in four muscles (c6, cv2, gm8b, p8) and a smaller enhancement for the other two muscles (cpv3a, gm4) than in C. borealis. The increase in contraction of the C. magister gastric muscle (gm4) was smaller than in C. productus, while the increase for the pyloric muscle (cpv3a) was larger.
Relative magnitude of response of stomatogastric musculature to TNRNFLRFamide varies among species of Cancer crabs. a Representative nerve-evoked contractions elicited by 1 s electrical stimulation under control conditions (solid lines) and in the presence of 10−7 M TNRNFLRFamide (dotted lines) for muscles in each of the three Cancer species. b Summary of effect of 10−7 M TNRNFLRFamide on peak contractile force for six muscles in each of the three species (1 s stimulation for all muscles; c6: stimulus frequency 40 Hz, N = 10 for each species; gm4: 20 Hz, N = 5; gm8b: 20 Hz, N = 5; cpv3a: 40 Hz, N = 6; cv2: 40 Hz, N = 5; p8: 20 Hz, N = 5.) The locations of the groups of bar graphs reflect the approximate relative positions of the muscles. (See Fig. 2.) Paired t tests using the raw data were used to test for statistical significance with respect to control, except when the normality condition was not satisfied (#) and the Wilcoxon signed rank test was used. The P values are shown at the bottom of the figure. We note that in a few cases (e.g. C. productus/gm8b, C. borealis/gm4) very prominent effects were not quite statistically significant due to the relatively small sample size and the large variability in the range of contractile forces measured under control conditions
Discussion
Possible roles of globally- and locally-released FLRFamide in modulating Cancer stomatogastric muscles
At least 20 members of the FLRFamide family have been identified as being present in crabs in the genus Cancer (Cruz-Bermudez et al. 2006; Fu et al. 2005; Fu and Li 2005). TNRNFLRFamide and SDRNFLRFamide, the first two of these to be characterized and isolated (Trimmer et al. 1987), are known to potentiate contractions of stomatogastric muscles in C. borealis (Jorge-Rivera and Marder 1996; Jorge-Rivera et al. 1998). Since no FLRFamide-containing neurosecretory site within the STNS of C. borealis or C. magister has been discovered, nor has direct FLRFamide-like innervation of musculature been observed, FLRFamidergic enhancement of muscle contractions in these two crabs presumably results from release from structures outside of the STNS such as the pericardial organs, which are known to secrete TNRNFLRFamide and SDRNFLRFamide in C. borealis (Li et al. 2003).
Jorge-Rivera and Marder (1996) suggested that simultaneous modulation of all stomatogastric muscles in C. borealis by FLRFamide might allow maintenance of physiologically-relevant muscle movements in the presence of a relatively weak pattern of motor neuron firing. For the five muscles that showed a modulatory response in C. borealis, we measured an average increase in contractile force of 122 ± 66% (SD) and noted that the magnitude of the effect did not vary across the five muscles nearly as much as in C. productus, where the average increase was 161 ± 163%. Thus we predict that, in C. borealis, FLRFamide broadly amplifies but does not qualitatively alter the overall pattern of gastric mill and pyloric muscle movements.
In C. productus, but not the other two Cancer species, the ACP appears to be a possible additional source of FLRFamide. This assertion is based on studies using a commercially-available antibody generated against synthetic FMRFamide that detected immunoreactivity in the ACP of C. productus but not in those of the other two species (Christie et al. 2004a; Savage et al. 2006). This antibody has been used to map the distribution of FLRFamide-related peptides in a number of crustacean species including Cancer crabs (Christie et al. 2004a and references therein) and has been shown to react with both TNRNFLRFamide and SDRNFLRFamide (Christie et al. 2004a), the only two FLRFamides that have been tested in vitro for reactivity with this antibody. However, we cannot rule out the possibility of additional nonreactive members of the peptide family being present in C. borealis or C. magister.
Based on its ultrastructure, the ACP was identified as a neurosecretory site (Christie et al. 2004a); however, it is not yet clear as to whether release of ACP modulators would be more accurately described as global or local. Certain regions of the crustacean STNS are in close contact with circulating hemolymph and would be ideally sited to either release modulators into the circulatory system or be modulated themselves. The STG and portions of its connecting nerves, for example, are located within the lumen of the cor frontale (auxiliary heart) of the anterior aorta (Maynard and Dando 1974; Steinacker 1978). The ACP is located outside this artery, however, so we suspect that FLRFamide released in C. productus by this structure may only reach local targets within the STNS and may not act globally throughout the organism.
If FLRFamide release is local and all muscles have a similar sensitivity (i.e. dose–response relationship) to the peptide, it may be the case that only muscles close to the ACP would be exposed to FLRFamide concentrations sufficient to potentiate contractions. Although Jorge-Rivera and Marder (1996) reported a threshold concentration of 10−10 M TNRNFLRFamide for effects on contractions, our dose–response measurements (Fig. 1c) suggest that the TNRNFLRFamide concentration required to increase contractile force appreciably (~50% or more over basal) is between 10−9 and 10−8 M. We have no way of determining the total amount of FLRFamide released from the ACP, but we can estimate the relative concentrations at different distances if we make simplifying assumptions. If the peptide is released all at once and diffuses freely in two dimensions (being confined by the hyperdermis covering the surface of the stomach), we show in the Appendix that the maximum concentration at a given point varies as the inverse square of the distance to the release site. The concentration at the center of the c6 muscle (seven times closer to the ACP) would thus be roughly 50 times larger than at the center of gm8b. If barriers were to hinder flow to the distal muscles, this ratio could be considerably larger, and proximal and distal muscles might respectively be exposed to saturating and subthreshold FLRFamide concentrations.
We found that the response to 10−7 M TNRNFLRFamide of muscles in C. productus, unlike in C. borealis, strongly depended on the muscle’s proximity to the ACP. Contractile forces produced by the three most proximal muscles, all gastric, increased notably (>100% over basal) in the presence of the peptide, while the distal, pyloric muscles showed little or no response. These results suggest that FLRFamide likely modulates stomatogastric movements in a functionally different fashion in C. productus than in C. borealis (or C. magister), regardless of its origin or the details of its release: FLRFamide released globally by a structure such as the pericardial organs should broadly strengthen the C. productus gastric mill motor program as in C. borealis but leave pyloric movements unaffected. FLRFamide released by the C. productus ACP such that all muscles are exposed to the same concentration (or saturating concentrations) should similarly potentiate gastric but not pyloric contractions. However, FLRFamide released in a local manner by the ACP, producing a concentration gradient across the musculature, should increase the contractions of c6 and perhaps gm4, but not those of more distal gastric muscles. This would result in qualitatively different gastric mill movements.
To confirm that the FLRFamidergic response in C. productus is in general spatially-varying and/or motor program-dependent would require further study of more muscles, with a particular focus on gastric and pyloric muscles like gm8b and cpv3a that are approximately the same distance from the ACP. Additional experiments in which the electrical stimulus parameters (e.g. duration, rate) and/or FLRFamide concentration were varied might generate a family of spatial maps of muscle response. Such results would hint at whether the pattern of FLRFamidergic muscle modulation in vivo depends on the activity of the STG motor neurons and/or of ACN1/2, respectively.
Evolution of modulatory role of FLRFamide in Cancer stomatogastric muscles
Based on outward morphological features such as carapace and claw shape, C. borealis and C. magister were historically grouped in a different Cancer subgenus (Metacarcinus) from C. productus (Nations 1979). More recently, however, phylogenies inferred from DNA sequencing of the mitochondrial cytochrome oxidase I (COI) gene indicate that C. productus and C. borealis are the most closely related of the three species (Harrison and Crespi 1999), suggesting that some or all of the gross morphological similarities or differences in Cancer crabs may have evolved independently and have been driven to convergence or divergence by environmental conditions such as their habitats or diets. It is reasonable to assume that these factors could have similarly shaped development of internal structures such as the stomatogastric system. For this reason we are not terribly surprised that, of the three crab species examined in this study, the two (C. borealis and C. magister) that lack FLRFamide-like peptide in the ACP and have somewhat similar patterns of FLRFamidergic modulation of the musculature are actually the most distantly related with respect to the molecular phylogeny (Harrison and Crespi 1999). Future comparative molecular, anatomical and physiological studies of the Cancer STNS likely will provide a better understanding of the functional role of FLRFamide release by the ACP in C. productus and might shed light on the circumstances driving evolutionary modifications of this structure in particular, and of the STNS in general.
Abbreviations
- acn :
-
Anterior cardiac nerve
- ACN1/2:
-
Anterior commissural neurons 1 and 2
- ACP:
-
Anterior cardiac plexus
- AM:
-
Anterior median neuron
- COI:
-
Cytochrome oxidase I
- cpv3a:
-
Cardiopyloric valve 3a muscle
- c6:
-
Cardiac 6 muscle
- CoG:
-
Commissural ganglion
- cv2:
-
Ventral cardiac 2 muscle
- dacn :
-
Dorsal anterior cardiac nerve
- DG:
-
Dorsal gastric neuron
- dgn :
-
Dorsal gastric nerve
- gm4:
-
Gastric mill 4 muscle
- gm8b:
-
Gastric mill 8b muscle
- IC:
-
Inferior cardiac neuron
- lacn :
-
Lateral anterior cardiac nerve
- p8:
-
Pyloric 8 muscle
- LP:
-
Lateral pyloric neuron
- PY:
-
Pyloric neuron
- lvn :
-
Lateral ventricular nerve
- son :
-
Superior esophageal nerve
- MG:
-
Medial gastric neuron
- STG:
-
Stomatogastric ganglion
- mvn :
-
Medial ventricular nerve
- stn :
-
Stomatogastric nerve
- on :
-
Esophageal nerve
- STNS:
-
Stomatogastric nervous system
References
Birmingham JT, Billimoria CP, DeKlotz TR, Stewart RA, Marder E (2003) Differential and history-dependent modulation of a stretch receptor in the stomatogastric system of the crab, Cancer borealis. J Neurophysiol 90:3608–3616
Christie AE, Cain SD, Edwards JM, Clason TA, Cherny E, Lin M, Manhas AS, Sellereit KL, Cowan NG, Nold KA, Strassburg HP, Graubard K (2004a) The anterior cardiac plexus: an intrinsic neurosecretory site within the stomatogastric nervous system of the crab Cancer productus. J Exp Biol 207:1163–1182
Christie AE, Stein W, Quinlan JE, Beenhakker MP, Marder E, Nusbaum MP (2004b) Actions of a histaminergic/peptidergic projection neuron on rhythmic motor patterns in the stomatogastric nervous system of the crab Cancer borealis. J Comp Neurol 469:153–169
Cruz-Bermúdez ND, Fu Q, Kutz-Naber KK, Christie AE, Li L, Marder E (2006) Mass spectrometric characterization and physiological actions of GAHKNYLRFamide, a novel FMRFamide-like peptide from crabs of the genus Cancer. J Neurochem 97:784–799
Fu Q, Kutz KK, Schmidt JJ, Hsu YW, Messinger DI, Cain SD, de la Iglesia HO, Christie AE, Li L (2005) Hormone complement of the Cancer productus sinus gland and pericardial organ: an anatomical and mass spectrometric investigation. J Comp Neurol 493:607–626
Fu Q, Li L (2005) De novo sequencing of neuropeptides using reductive isotopic methylation and investigation of ESI QTOF MS/MS fragmentation pattern of neuropeptides with N-terminal dimethylation. Anal Chem 77:7783–7795
Harris-Warrick RM, Marder E, Selverston AI, Moulins M (eds) (1992) Dynamic biological networks. The stomatogastric nervous system. MIT Press, Cambridge
Harrison MK, Crespi BJ (1999) Phylogenetics of Cancer crabs (Crustacea:Decapoda:Brachyura). Mol Phylogenet Evol 12:186–199
Jorge-Rivera JC, Marder E (1996) TNRNFLRFamide and SDRNFLRFamide modulate muscles of the stomatogastric system of the crab Cancer borealis. J Comp Physiol A 179:741–751
Jorge-Rivera JC, Marder E (1997) Allatostatin decreases stomatogastric neuromuscular transmission in the crab, Cancer borealis. J Exp Biol 200:2937–2946
Jorge-Rivera JC, Sen K, Birmingham JT, Abbott LF, Marder E (1998) Temporal dynamics of convergent modulation at a crustacean neuromuscular junction. J Neurophysiol 80:2559–2570
Katz PS, Tazaki K (1992) Comparative and evolutionary aspects of the crustacean stomatogastric system. In: Harris-Warrick RM, Marder E, Selverston AI, Moulins M (eds) Dynamic biological networks: the stomatogastric nervous system. MIT Press, Cambridge
Le T, Verley DR, Goaillard JM, Messinger DI, Christie AE, Birmingham JT (2006) Bistable behavior originating in the axon of a crustacean motor neuron. J Neurophysiol 95:1356–1368
Li L, Kelley WP, Billimoria CP, Christie AE, Pulver SR, Sweedler JV, Marder E (2003) Mass spectrometric investigation of the neuropeptide complement and release in the pericardial organs of the crab, Cancer borealis. J Neurochem 87:642–656
Lingle C (1981) The modulatory action of dopamine on crustacean foregut neuromuscular preparations. J Exp Biol 94:285–299
Marder E, Calabrese RL, Nusbaum MP, Trimmer B (1987) Distribution and partial characterization of FMRFamide-like peptides in the stomatogastric nervous systems of the rock crab, Cancer borealis, and the spiny lobster, Panulirus interruptus. J Comp Neurol 259:150–163
Maynard DM, Dando MR (1974) The structure of the stomatogastric neuromuscular system in Callinectes sapidus, Homarus americanus and Panulirus argus (decapoda crustacea). Philos Trans R Soc Lond (Biol) 268:161–220
Messinger DI, Kutz KK, Le T, Verley DR, Hsu YW, Ngo CT, Cain SD, Birmingham JT, Li L, Christie AE (2005) Identification and characterization of a tachykinin-containing neuroendocrine organ in the commissural ganglion of the crab Cancer productus. J Exp Biol 208:3303–3319
Meyrand P, Marder E (1991) Matching neural and muscle oscillators: control by FMRFamide-like peptides. J Neurosci 11:1150–1161
Mortin LI, Marder E (1991) Differential distribution of ß-pigment dispersing hormone (ß-PDH)-like immunoreactivity in the stomatogastric nervous system of five species of decapod crustaceans. Cell Tiss Res 265:19–33
Nations JD (1979) The genus Cancer and its distribution in time and space. Bull Biol Soc Wash 3:153–187
Savage EE, Messinger DI, Trieu Q, Doan V, Verley DR, Birmingham JT, Christie AE (2006) Structural but not neurochemical conservation of the anterior cardiac neuron 1/2-anterior cardiac plexus (ACN1/2-ACP) neuroendocrine system of Cancer crabs: implications for differential sensitivities of the foregut musculature to FLRFamide. 2006 Neuroscience Meeting Planner, Atlanta, GA
Selverston AI, Moulins M (eds) (1987) The Crustacean Stomatogastric System. Springer-Verlag, Berlin
Skiebe P (2001) Neuropeptides are ubiquitous chemical mediators: using the stomatogastric nervous system as a model system. J Exp Biol 204:2035–2048
Steinacker A (1978) The anatomy of the decapod crustacean auxiliary heart. Biol Bull 154:497–507
Trimmer BA, Kobierski LA, Kravitz EA (1987) Purification and characterization of FMRFamidelike immunoreactive substances from the lobster nervous system: isolation and sequence analysis of two closely related peptides. J Comp Neurol 266:16–26
Weimann JM, Meyrand P, Marder E (1991) Neurons that form multiple pattern generators: identification and multiple activity patterns of gastric/pyloric neurons in the crab stomatogastric system. J Neurophysiol 65:111–122
Worden MK, Kravitz EA, Goy MF (1995) Peptide F1, an N-terminally extended analog of FMRFamide, enhances contractile activity in multiple target tissues in lobster. J Exp Biol 198:97–108
Acknowledgments
We would like to thank Y. Hsu and A. Christie for collecting animals and C. Billimoria, M. Marvier and D. Ostrov for valuable conversations. J.T. Birmingham acknowledges support from the Research Corporation, the Grass Foundation, Santa Clara University (SCU), and an award to SCU under the Undergraduate Biological Sciences Education Program of the Howard Hughes Medical Institute. All experiments complied with the “Principles of animal care”, publication No. 86-23, revised 1985, of the National Institute of Health, and also with the current laws of the USA.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Let n(x, y, t) be the time-varying concentration of FLRFamide in saline confined to an infinite plane. Assume its evolution is described by the two-dimensional diffusion equation \( \frac{\partial n}{\partial t} = D\nabla^{2} n \), where D is the diffusion constant, and that n(x, y, 0) = n 0 δ(x)δ(y)—initially all peptide is confined at the origin. The solution to this equation has azimuthal symmetry and is most simply expressed using polar coordinates:
The time at which the concentration at r = R is maximum can be obtained by solving \( \frac{{\partial n\left( {R,t} \right)}}{\partial t} = 0. \) The resulting condition is 4Dt = R 2. This result can be substituted back into Eq. 1 to eliminate t and to yield the maximum concentration at r = R:
which varies as the inverse square of the distance from the origin.
Rights and permissions
About this article
Cite this article
Verley, D.R., Doan, V., Trieu, Q. et al. Characteristic differences in modulation of stomatogastric musculature by a neuropeptide in three species of Cancer crabs. J Comp Physiol A 194, 879–886 (2008). https://doi.org/10.1007/s00359-008-0359-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-008-0359-6