Abstract
The first survey of sea snakes (Elapidae, Hydrophiinae) at the remote Entrecasteaux atolls, Coral Sea, was conducted using remote unbaited 360° video cameras (RUV360), in 2021. We detected 49 snakes belonging to at least six species (Aipysurus duboisii, A. laevis, Hydrophis coggeri, H. macdowelli, H. major and H. ornatus) at the two largest atolls, Surprise and Huon. Three more are probably present (Emydocephalus annulatus, H. peronii, H. platurus), highlighting the richness of these atolls. No snakes were detected at neighboring smaller atolls. The lack of sea snake observations at the five small atolls may be due to the insufficient area of suitable habitat. Compared to assemblages sampled using the same method in New Caledonia (at Nouméa = A. laevis, E. annulatus, Laticauda sp., H. macdowelli, H. major, H. ornatus; at Touho = A. laevis, H. ornatus), and assemblages sampled with different methods at the Chesterfields (A. duboisii, A. laevis, E. annulatus, H. coggeri, H. laboutei, H. peronii, H. platurus), the Entrecasteaux atolls exhibit a high sea snake species richness. RUV360s appear to sample the diversity of sea snakes more effectively than did baited remote underwater video stations (BRUVS) within the Great Barrier Reef (which detected only A. laevis, H. curtus, H. ocellatus).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sea snakes species are distributed in tropical and subtropical areas of the Indo-Pacific, with the greatest diversity in the Coral Triangle (Elfes et al. 2013). In a recent review of future directions for research on marine snakes, a key question identified was “What are the geographic distributions and boundaries of the species of marine snakes, and where are hotspots of biodiversity, endemism and richness?” (Udyawer et al. 2018). Knowledge about the distribution of sea snake species is useful for several purposes. Most notably, these predators can act as bioindicators to monitor coral reef ecosystems, which face many threats worldwide (Reed et al. 2002; Seret et al. 2008; Rasmussen et al. 2021). Remote atolls might be an important refuge from increasing anthropogenic disturbance, although declines in the abundance of sea snakes have occurred at such sites (Rasmussen et al. 2021). To address this gap in knowledge, we need surveys of a wide array of sites at a range of spatial scales. Unfortunately, little information is available about snake diversity on remote reefs. For example, snakes have not been studied at the Entrecasteaux atolls, although these atolls occur in a region of high sea snake species richness (Brischoux et al. 2012; Elfes et al. 2013). The Entrecasteaux atolls are located in the northern part of the New Caledonia EEZ (Exclusive economic zone), a large (1.3 million-km2) area that became the Coral Sea Natural Park in 2014. This park aims to protect biodiversity while permitting sustainable economic development, a goal that requires knowledge of the biodiversity present. Several animal taxa have been surveyed at the Entrecasteaux atolls (Read et al. 2014; Juhel et al. 2019; Borsa et al. 2021; Wantiez et al. 2022) but sea snakes have been neglected. The only published information was a single photograph of Aipysurus laevis, in a publication about hermit crabs (Evans et al. 2015).
One reason for a scarcity of detailed information on the geographic distribution of sea snake species is that most survey methods are expansive and labor-intensive (e.g., snorkelling; manta-tows). New technologies offer promising ways to overcome these problems (Crowe-Riddell et al. 2019). For example, remote unbaited 360° video sampling (RUV360) enables standardized and efficient census of vertebrates over peri-reefal soft bottoms (Mallet et al. 2021a, b, 2022). This method allowed us to produce the first list of sea snake species at Entrecasteaux atolls. We compare our findings to unpublished research at Touho, on the east coast of New Caledonia, and Nouméa (Mallet, unpublished data, 2021) and to published research from the nearby Chesterfields (Minton and Dunson 1985; Zimmerman et al. 1994). Finally, we compare RUV360 versus baited remote underwater video surveys (BRUVS) (Udyawer et al. 2014) for surveying sea snakes.
Methods
Camera stations
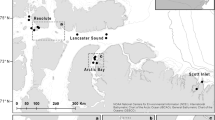
The remote unbaited 360° video sampling method (RUV360) was selected for this study as it provides a less biased representation of vertebrate assemblages than do remote underwater video stations that use bait to attract predators and scavengers to the camera site (BRUVS). Mallet et al. (2021b) demonstrated that RUV360 can provide standardized and efficient censuses of vertebrates over peri-reefal soft bottoms. They estimated the optimal duration of camera deployment at 17 min. We set up unbaited 360° cameras for video census between 30 August 2021 and 10 September 2021 (i.e., at the end of the cold season), over soft bottoms inside Entrecasteaux atolls (see Mallet et al. 2021a, b, 2022). Entrecasteaux atolls comprise two large atolls, bearing coral cays (Huon and Surprise) and five smaller atolls without such cays (Petit Guilbert, Gros Guilbert, Mérite, Pelotas, and Portail) (Fig. 1).
Stations on the same atoll were 1.5 km apart, in order to optimise sampling effectiveness. About 197 stations were deployed successfully (out of 207 attempts): 89 at Huon atoll, 73 at Surprise atoll, 12 at Pelotas atoll, 8 at Portail atoll, 4 at Mérite atoll, 8 at Gros Guilbert atoll, and 3 at Petit Guilbert atoll. Cameras were attached to weighted tripods to keep them upright, and were set up at depths of 2–50 m. Recordings were made from 0700 to 1630 h (i.e., over the diel period with sufficient light). On each deployment, the camera was turned on immediately before immersion and allowed to sink to the bottom with a rope tied to a buoy. It was set to record for 17 min. During that period, the boat went to another one or two stations to deploy additional cameras, before returning to retrieve the first camera (located via the buoy), and moving it to another station. The number of snakes was recorded as MaxN: maximum number of individuals of the same species appearing at the same time throughout the entire video.
Species identification
Videos allow sea snakes to be identified with confidence because there are < 15 species in New Caledonia (Ineich & Laboute 2002; Ineich 2007; Brischoux et al. 2018) and these species differ in body shape (notably, including both microcephalic and macrocephalic taxa: see Sanders et al. 2013) and markings. Species were identified using relevant publications (Ineich & Laboute 2002; Cogger 2018). In the discussion below, we use current generic names, some of which differ from those used in earlier reports.
Results and discussion
Of the 197 stations in which cameras were deployed successfully, 39 (19.8%) recorded at least one snake. This is similar to the proportion of deployments that recorded snakes in a BRUVS study from the Great Barrier Reef (19%: Udyawer et al. 2014). We recorded a total of 49 snakes, of which nine (18.3%) could not be identified to species level. Six species of sea snakes were identified at Huon and Surprise atolls, but no snakes were recorded on the 35 stations at smaller atolls (Fig. 2). The lack of observations of sea snakes at the small atolls may be due to insufficient sampling effort or (more likely) to an inadequate area of suitable habitat to support population persistence. Sea snakes were detected more frequently at the Entrecasteaux atolls that in the other sites sampled to date with the same method. In the New Caledonia lagoon, sea snakes were recorded in 12.5 and 6.5% of the stations off Nouméa and Touho (Fig. 1), respectively (Mallet et al. unpublished data 2020, 2021a, b).
The list of species observed at the Entrecasteaux atolls can be compared with published data from the Chesterfields. These larger systems are similar and geographically close to Entrecasteaux atolls, and are also within the Coral Sea Natural Park (Fig. 2). At the Entrecasteaux atolls and in the Chesterfields, 10 species of sea snakes have been recorded (Table 1), but only four of them are common to both reef systems. These differences may reflect the difference of methods used (videos in our study versus snorkelling, manta-board, scuba diving and netting from a boat in other studies).
The unbaited cameras also gave us the opportunity to record behavior of sea snakes that was not modified by the presence of human beings or predators (i.e., sharks) attracted by a bait, because our cameras were set up to function in the absence of divers or bait. Nevertheless, sea snakes are notoriously curious, often approaching novel objects (Ineich & Laboute 2002) and in the current study, snakes often investigated the camera, facilitating species identification. Observed behaviors are listed in Table 2. Some snakes exhibited more than one behavior during the recording period. 75.5% of the sea snakes that we recorded swam near the bottom, as expected because all species of sea snakes except Hydrophis platurus feed on benthic prey (Sanders et al. 2013). A video of each species is available as Supplementary Information.
Sea Kraits (Laticauda spp.) were not seen in our videos. That result is unsurprising, because these amphibious species were not recorded as part of the terrestrial herpetofauna of Surprise Island (the only nearby land mass, the cay at Huon atoll is only a few meters wide: Bauer et al. 1992; Caut et al. 2008). Sea Kraits are not known from the Chesterfields either.
Turtle-headed Sea Snakes (Emydocephalus annulatus) were not identified with confidence from the videos, but some of the unidentified small dark-colored snakes may be E. annulatus. Indeed, individuals of this species were photographed around coral bommies at Huon atoll in 2019 (A. Aupetit pers. com., see https://www.inaturalist.org/observations/116044037 to 116044043). In the Chesterfields, Minton and Dunson (1985) saw E. annulatus and Zimmerman et al. (1994) described this species as the most commonly encountered. Living mostly on coral reefs (Goiran et al. 2020), E. annulatus can easily be missed by cameras set up on soft bottom substrates such as in the present study.
Reef Shallows Sea Snakes (Aipysurus duboisii) were present at Surprise atoll. We recorded two pairs of A. duboisii, one snake following the other, apparently engaged in courtship. One pair was swimming horizontally mid-water over a 22 m deep sandy bottom, an unusual habitat for this shallow-reef snake (see video in Supplementary Information). Minton and Dunson (1985) saw A. duboisii in the Chesterfields.
Olive Sea Snakes (A. laevis) were the most abundant sea snakes on our videos. Being curious, they frequently investigated the cameras. They also were seen in the Chesterfields by Minton and Dunson (1985), and were reported to be the second most abundant sea snakes there (Zimmerman et al. 1994).
Cogger’s Sea Snake (Hydrophis coggeri) was the second-most-often recorded sea snake species in our surveys of the Entrecasteaux atolls. Minton and Dunson (1985) do not mention this species in the Chesterfields. However, that situation likely reflects identification issues. They mention a species of Hydrophis that resembles H. melanocephalus, a species found only in Vietnam, China, Taiwan and Japan (Sanders et al. 2013). The specimens collected by Minton and his colleagues were deposited in the Field Museum at Chicago, and were later identified as H. coggeri (catalogue #s 224,286–8, 2,053,694–6). Thus, the two unidentified Hydrophis mentioned by Zimmerman et al. (1994) were probably also H. coggeri.
McDowell’s Sea Snakes (Hydrophis macdowelli) were recorded at Huon and Surprise atolls. This species is described as highly wary (Ineich & Laboute 2002), but the specimen recorded at Huon atoll behaved in a relaxed way, showing the advantages of the unbaited video survey (see video in Supplementary Information). It has not been recorded in the Chesterfields.
Greater Sea Snakes (Hydrophis major) were recorded at Huon and Surprise atolls. They were not seen in the Chesterfields by Minton and Dunson (1985) nor by Zimmerman et al. (1994). Feeding exclusively on striped catfish Plotosus lineatus (Shine et al. 2019), these sea snakes are indicators of the presence of P. lineatus at these atolls, although this cryptic species is not included in the published species-list for fishes of the Entrecasteaux atolls (Wantiez et al. 2022).
Ornate Reef Sea Snakes (Hydrophis ornatus) were recorded at Huon atoll. The taxonomy and nomenclature of this species is currently under revision, so the snakes listed here under this name may soon be redescribed. In general appearance, the snakes seen in videos resemble those seen in the waters off Noumea: pale with grey bands extending halfway down the flanks (see https://www.inaturalist.org/observations/105998451).
Horned Sea Snakes (Hydrophis peronii) were not recorded at the Entrecasteaux atolls. Known for foraging on soft bottom habitats (Ineich & Laboute 2002), H. peronii may live in the lagoons that we surveyed but remained unseen. The method we used detects only moving snakes. A pale snake, lying in ambush at the entrance of a goby’s burrow (as is typical for our observations of this species in the Noumea region: R. Shine, pers. obs. and C. Goiran, pers. obs.; see https://www.inaturalist.org/observations/105759087) is likely to remain unnoticed on videos. Therefore, we cannot exclude the presence of this species at the Entrecasteaux atolls. It was seen in the Chesterfields by Minton and Dunson (1985) and by Zimmerman et al. (1994).
Yellow-Bellied Sea Snakes (Hydrophis platurus) were not seen on our videos. Again, observability may be low because of our methodology. This species is pelagic, drifting at the sea surface (Ineich & Laboute 2002) and thus is unlikely to be seen on videos taken near the sea floor. The Entrecasteaux atolls are well within the wide geographic range of this species, so it will almost certainly be found in targeted surveys. It was recorded by Minton and Dunson (1985) and Zimmerman et al. (1994) in the Chesterfields.
More generally, the species detected by surveys in remote sites will depend upon the methods employed. For example, adding bait to underwater stations (baited remote underwater video stations, = BRUVS) may yield results different from those obtained when using unbaited stations. Data from 2,471 BRUVS deployments within the Great Barrier Reef Marine Park revealed the presence of sea snakes in 19% of the videos (Udyawer et al. 2014), but the 572 sea snakes recorded comprised only three identified species: Aipysurus laevis (77%), Hydrophis curtus (9%), and H. ocellatus (8%). The remaining 6% could not be identified. The region in which the cameras were deployed is home to about 16 species (Elfes et al. 2013), suggesting that large teleosts and sharks that were attracted to bait stations may have dissuaded other sea snake species from approaching the BRUVS, with sightings of sea snakes biased towards larger-bodied or generalist species. Therefore, unbaited systems such as our remote unbaited 360°video sampling method (RUV360) may sample the diversity of sea snakes more effectively than is possible with baited systems. Sea snakes are not the only group efficiently surveyed with RUV360. The pilot study demonstrated that this method is efficient for monitoring fish assemblages and soft bottom habitats at the same time (Mallet et al. 2021b).
Conclusion
Sea snakes were abundant on the previously unsurveyed Entrecasteaux atolls, appearing in 19.8% of our videos. We confirmed the presence of six species (Aipysurus duboisii, A. laevis, Hydrophis coggeri, H. macdowelli, H. major, and H. ornatus) and at least three more species are likely to be present (Emydocephalus annulatus, H. peronii, and H. platurus). Therefore, the Entrecasteaux atolls seem to host a rich diversity of sea snakes, consistent with the assemblage identified from the Chesterfields. The lack of records of snakes at five small atolls suggests that these atolls do not offer sufficient area of suitable habitat for sea snakes. Our study also demonstrated the value of the unbaited 360°video sampling method (RUV360) for surveys of the health of soft bottom habitats. We recommend the use of RUV360 for long-term follow-up studies of soft bottom habitats, together with visual census of coral reefs at the Entrecasteaux atolls and other parts of Coral Sea Natural Park, in particular the Chesterfields.
Data availability
All data generated or analyzed during this study are included in this published article [and its Supplementary Information files].
References
Bauer AM, Renevier A, Sadlier RA (1992) Caledoniscincus austrocaledonicus (Reptilia: Scincidae) from lIe Surprise, D’Entrecasteaux Reefs, New Caledonia. Pac Sci 46:86–89
Borsa P, Philippe A, Le Bouteiller A (2021) Oiseaux marins des atolls d’Entrecasteaux (parc naturel de la mer de Corail): bilan des observations des deux dernières décennies. Bull Société Zool Fr 146:175–188
Brischoux F, Tingley R, Shine R, Lillywhite HB (2012) Salinity influences the distribution of marine snakes: implications for evolutionary transitions to marine life. Ecography 35:994–1003
Brischoux F, Bonnet X, Shine R, Goiran C (2018) Les serpents marins des récifs coralliens de Nouvelle-Calédonie. In: Payri C (ed) Nouvelle-Calédonie Archipel de corail. IRD & Solaris Editions, Nouméa, New Caledonia, pp 121–126
Caut S, Angulo E, Courchamp F (2008) Dietary shift of an invasive predator: rats, seabirds and sea turtles. J Appl Ecol 45:428–437
Cogger HG (2018) Reptiles and amphibians of Australia, updated, 7th edn. CSIRO publishing, Clayton, South VIC, Australia
Crowe-Riddell JM, d’Anastasi BR, Nankivell JH, Rasmussen AR, Sanders KL (2019) First records of sea snakes (Elapidae: Hydrophiinae) diving to the mesopelagic zone (>200 m). Austral Ecol 44:752–754
Elfes CT, Livingstone SR, Lane A, Lukoschek V, Sanders KL, Courtney AJ, Gatus JL, Guinea M, Lobo AS, Milton DA (2013) Fascinating and forgotten: the conservation status of the world’s sea snakes. Herpetol Conserv Biol 8:37–52
Evans N, Gilbert A, Andréfouët S, Paulay G (2015) A massive subtidal aggregation of hermit crabs in surprise Atoll lagoon, New Caledonia. Coral Reefs 34:917–917
Goiran C, Brown GP, Shine R (2020) Niche partitioning within a population of sea snakes is constrained by ambient thermal homogeneity and small prey size. Biol J Linn Soc 129(3) 644–651. https://doi.org/10.1093/biolinnean/blz206
Ineich I, Laboute P (2002) Les serpents marins de Nouvelle-Calédonie. Institut de Recherche pour le Développement, Paris
Ineich I (2007) The sea snakes of New Caledonia. In: Payri C., Richer de Forges B. (eds) Doc. Sci. Tech. Institut de Recherche pour le Développement, Nouméa, pp 403–410
Juhel J-B, Vigliola L, Wantiez L, Letessier TB, Meeuwig JJ, Mouillot D (2019) Isolation and no-entry marine reserves mitigate anthropogenic impacts on grey reef shark behavior. Sci Rep 9:2897
Mallet D, Lanos N, Wantiez L (2021a) État des lieux des fonds meubles des atolls d’Entrecasteaux. Note de synthèse.
Mallet D, Olivry M, Ighiouer S, Kulbicki M, Wantiez L (2021b) Nondestructive monitoring of Soft Bottom Fish and Habitats using a Standardized, Remote and Unbaited 360° Video Sampling Method. Fishes 6:50
Mallet D, Lanos N, Goiran C, Wantiez L (2022) Programme HaPéRO. Volume 2 - Communautés biologiques et habitats des fonds meubles de Touho et Poindimié. Etat des lieux 2021. Université de la Nouvelle-Calédonie, VISIOON, 43pp
Minton SA, Dunson WW (1985) Sea snakes collected at the Chesterfield Reefs, Coral Sea. Atoll Res Bull 292:101–108
Rasmussen AR, Hay-Schmidt A, Boneka F, Allentoft M, Sanders KL, Elmberg J (2021) Viviparous sea snakes can be used as bioindicators for diverse marine environments. Philipp J Syst Biol 14:1–16
Read TC, Wantiez L, Werry JM, Farman R, Petro G, Limpus CJ (2014) Migrations of Green Turtles (Chelonia mydas) between Nesting and Foraging Grounds across the Coral Sea. PLoS ONE 9:e100083
Reed RN, Shine R, Shetty S (2002) Sea kraits (Squamata: Laticauda spp.) as useful bioassay for assessing local diversity of eels (Muraenidae, Congridae) in the western Pacific Ocean. Copeia 2002(4):1098–1101
Sanders KL, Rasmussen AR, Mumpuni EJ, de Silva A, Guinea ML, Lee MSY (2013) Recent rapid speciation and ecomorph divergence in Indo-Australian sea snakes. Mol Ecol 22:2742–2759
Séret B, Brischoux F, Bonnet X, Shine R (2008) First record of Cirrimaxilla formosa (Muraenidae) from New Caledonia, found in sea snake stomach contents. Cybium 32:191–192
Shine R, Shine T, Goiran C (2019) Morphology, reproduction and diet of the greater sea snake, Hydrophis major (Elapidae, Hydrophiinae). Coral Reefs 38:1057–1064
Udyawer V, Cappo M, Simpfendorfer CA, Heupel MR, Lukoschek V (2014) Distribution of sea snakes in the Great Barrier Reef Marine Park: observations from 10 yrs of baited remote underwater video station (BRUVS) sampling. Coral Reefs 33:777–791
Udyawer V, Barnes P, Bonnet X, Brischoux F, Crowe-Riddell JM, D’Anastasi B, Fry BG, Gillett A, Goiran C, Guinea ML, Heatwole H, Heupel MR, Hourston M, Kangas M, Kendrick A, Koefoed I, Lillywhite HB, Lobo AS, Lukoschek V, McAuley R, Nitschke C, Rasmussen AR, Sanders KL, Sheehy C, Shine R, Somaweera R, Sweet SS, Voris HK (2018) Future Directions in the Research and Management of Marine Snakes. Front Mar Sci 5:399
Wantiez L, Frolla P, Goroparawa D (2022) Communautés biologiques et habitats coralliens des atolls d’Entrecasteaux. État des lieux 2021. Maintien de l’intégrité. 93
Zimmerman KD, Heatwole H, Menez A (1994) Sea snake in the Coral Sea: an expedition for the collection of animals and venom. Herpetofauna 24(1):25–26
Acknowledgements
The Entrecasteaux Atolls Campaign was part of HaPéRO program, funded by the Government of New Caledonia and the French Government through Fonds Pacifique. The authors thank the crew of the vessel Amborella for their competence and efficiency during the field trip. The survey was authorized by the Government of New Caledonia. The authors thank the editor and reviewers for improving the manuscript.
Funding
Article was funded by Fonds Pacifique, Convention de subvention 2105, Laurent Wantiez, Gouvernement de la Nouvelle-Calédonie, Contrat de collaboration de recherche 05/2021, Laurent Wantiez.
Author information
Authors and Affiliations
Contributions
DM, LW, VU and CG contributed to conceptualization. DM, LW VU and CG contributed to methodology. DM and CG performed formal analysis. DM, NL and LW conducted investigation. DM contributed resources. All authors were involved in writing and original draft preparation.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (MP4 7027 KB)
Supplementary file2 (MP4 7759 KB)
Supplementary file3 (MP4 8568 KB)
Supplementary file4 (MP4 4098 KB)
Supplementary file5 (MP4 9035 KB)
Supplementary file6 (MP4 1732 KB)
Supplementary file7 (MP4 3059 KB)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Goiran, C., Mallet, D., Lanos, N. et al. Sea snake diversity at the Entrecasteaux atolls, Coral Sea, as revealed by video observations at unbaited stations. Coral Reefs 41, 1551–1556 (2022). https://doi.org/10.1007/s00338-022-02307-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-022-02307-x