Abstract
Decades of research have revealed relationships between the abundance of coral reef taxa and local conditions, especially at small scales. However, a rigorous test of covariation requires a robust dataset collected across wide environmental or experimental gradients. Here, we surveyed spatial variability in the densities of major coral reef functional groups at 122 sites along a 70 km expanse of the leeward, forereef habitat of Curaçao in the southern Caribbean. These data were used to test the degree to which spatial variability in community composition could be predicted based on assumed functional relationships and site-specific anthropogenic, physical, and ecological conditions. In general, models revealed less power to describe the spatial variability of fish biomass than cover of reef builders (R2 of best-fit models: 0.25 [fish] and 0.64 [reef builders]). The variability in total benthic cover of reef builders was best described by physical (wave exposure and reef relief) and ecological (turf algal height and coral recruit density) predictors. No metric of anthropogenic pressure was related to spatial variation in reef builder cover. In contrast, total fish biomass showed a consistent (albeit weak) association with anthropogenic predictors (fishing and diving pressure). As is typical of most environmental gradients, the spatial patterns of both fish biomass density and reef builder cover were spatially autocorrelated. Residuals from the best-fit model for fish biomass retained a signature of spatial autocorrelation while the best-fit model for reef builder cover removed spatial autocorrelation, thus reinforcing our finding that environmental predictors were better able to describe the spatial variability of reef builders than that of fish biomass. As we seek to understand spatial variability of coral reef communities at the scale of most management units (i.e., at kilometer- to island-scales), distinct and scale-dependent perspectives will be needed when considering different functional groups.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ecological community structure emerges from myriad processes that each act at characteristic temporal and spatial scales. When co-occurring groups are believed to have strong functional linkages, it is tempting to assume that the ecological dynamics of these groups will be associated, resulting in spatial and temporal correlations in abundance. In coastal marine ecosystems, there is often a close coupling between foundational, habitat-building species and the mobile taxa that use this habitat. Examples include mangrove trees creating the habitat for invertebrates and fishes (Nagelkerken 2009; Whitfield 2017), seagrasses creating sediment dynamics exploited by benthic infauna (Nakaoka 2005; Bouma et al. 2009), kelp facilitating growth of invertebrate and fish assemblages (DeMartini and Roberts 1990; Arkema et al. 2009), and scleractinian corals and coralline algae creating shelter, hydrodynamic conditions, and trophic opportunities for fishes and other animals (Harborne et al. 2006).
In coral reef habitats, the linkage between framework builders (e.g., corals) and associated mobile taxa (e.g., fishes) has been explored extensively, generating multiple hypotheses of functional causative associations. For example, fish community composition can be linked to the structure of reef builders through dependence upon shelter and food (Almany 2004; Wilson et al. 2006; Darling et al. 2017). Similarly, fishes can influence the outcome of benthic competitive networks by selectively foraging upon fast-growing seaweeds (Ogden and Lobel 1978) as well as spatially concentrating inorganic nutrients by excreting within the benthos (Meyer et al. 1983; Burkepile et al. 2013). Through such ecological interactions, the spatial variability in the abundance of one group may be assumed to be reflected in the abundance of the other. Hypotheses that link process-based views of dependencies and interactions among key functional groups can be investigated through paired observational surveys. For example, positive correlations between fish biomass and coral cover (Williams and Polunin 2001; Newman et al. 2006) are consistent with the hypothesis of corals creating habitat and shelter for fishes (Harborne et al. 2006).
Process-based hypotheses of interactions among key functional groups within reef communities are often derived from small-scale (10–100 s of m) experimental studies (Vermeij et al. 2010; Zaneveld et al. 2016) or large-scale (100–1000 s of km) observational surveys (Sandin et al. 2008; Lester et al. 2020). In practice, each small- and large-scale approach is designed to test specific hypotheses by targeting extreme variation of an independent variable, for example the abundance of one group (e.g., through experimental manipulation or across a broad geographical range). Such experimental and large-scale designs accentuate the effect size expected from a potential driver, so that finding a significant signal consistent with a putative functional relationship becomes more statistically probable. Across smaller spatial scales, or along natural, non-manipulated gradients, environmental variability may be less extreme and effect sizes may be more modest. Further, with smaller effect sizes, spatial correlations can become obscured quantitatively by nonlinear dynamics, covariation among many factors, interaction among processes, lag-periods, and interference of processes that were not measured (Blonder et al. 2017; Yates et al. 2018; McGill 2019). To assess the degree to which spatial patterns in community structure can be successfully reconstructed from assumed functional relationships, high resolution data are required from across relatively large spatial scales or time series. While ecological coupling may be found through targeted assessment of subsets of taxa (e.g., corallivorous butterflyfish and stony corals; Graham et al. 2009), challenges remain in predicting the emergent patterns of marine species assemblage, especially at spatial scales between that of survey sites (10–100 s of m) and islands or regions (100–1000 s of km).
Although conducting studies across extreme gradients creates scientific value by elucidating ecological principles of cause and effect, comparable studies conducted across more modest gradients provide complementary value by quantifying the realized predictive power of our collective ecological understanding. For practical applications, the level of organization at intermediate spatial scales (1–10 s of km) is relevant as it reflects the scale at which human impacts (e.g., coastal development and fishing) can be expected to interfere with the natural dynamics of coastal reef communities. The ability to predict successfully patterns of reef change based on the known spatial variation in ecological, physical, and anthropogenic drivers is especially relevant where coastal communities seek to understand and minimize factors contributing to reef decline. Thus, an important goal for marine conservation is to understand how spatial patterns of functional groups such as reef builder cover and fish biomass are linked, and at what scales we can accurately predict the abundance of these groups using spatial variation in ecological and environmental conditions.
Here we investigate the variability of coral reef community structure across a well-studied Caribbean island, Curaçao. Survey efforts were focused within one coral reef habitat type, the leeward forereef habitat in the 8–12 m depth range. The observed variability in the structure of both fish and benthic assemblages was then considered in the context of mechanistic hypotheses of environmental forcing and community-level ecological interactions. The community data were used to address two fundamental questions: (i) Are there clear ecological, physical, or anthropogenic predictors of total fish biomass and reef builder cover? (ii) If so, are there shared patterns and spatial scales of predictability across the fish and the benthic assemblages?
Methods
Study sites
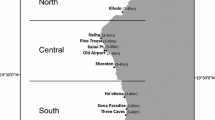
Curaçao (12.2°N, 69.0°W; land area of 444 km2) is an island in the Leeward Antilles of the southern Caribbean Sea, with the major axis oriented northwest to southeast. The principal currents and swell arrive from the east-northeast, thus the southwestern facing shores, spanning approximately 70 km, are more protected and are considered leeward shores (van Duyl 1985). The island is surrounded by a continuous fringing reef that, along the leeward shores, begins within meters of shore and slopes down steeply (slopes ranging from 30–60°) to a depth of 80 m, and continuing seaward with a series of terraced deep-water reefs. Notably, in recent decades the reefs of Curaçao have experienced only limited impacts of storms, marine heat waves, coral diseases, and other large-scale disturbances. In November 2015, we surveyed a total of 122 sites along this leeward shore. Sites were chosen by selecting coordinates along the 10 m isobath with an approximate spacing of 700 m.
Assemblage surveys—fish and benthos
Coral reef community surveys were conducted following the consensus methodological guidelines from the Global Coral Reef Monitoring Network (GCRMN) Caribbean node (GCRMN 2016). The Level 3 methods of the GCRMN Caribbean protocols were employed, providing detailed taxonomic and compositional data of the fish and benthic assemblages. These methods build from the Atlantic and Gulf Rapid Reef Assessment (AGRRA) protocols (Lang et al. 2010), with some modifications. An overview of the relevant survey protocol follows, with complete details outlined in GCRMN Caribbean documentation (GCRMN 2016). Only data describing the fish and non-mobile benthic assemblages are presented here.
Fish and benthic assemblages were surveyed during daylight (non-crepuscular) hours at each site along five 30 m-long transects by a team of divers (AJE, DG, SLH, HK, SDM, SFR, SAS, and BJZ). Within each site, transect lines were laid out sequentially, each parallel to shore between 8 and 12 m depth with sequential transect lines separated by approximately 5 m. Transect data were averaged to produce a site-specific mean, and site was used as the unit of replication in all analyses.
The fish assemblage was assessed by one diver on each transect. Every fish observed within a 2 m-wide swath was recorded, noting species and size (reported as total length using 5 cm bins). Survey times were limited to approximately 6 min per transect in order to minimize observer bias across levels of fish density. Lengths of individual fish were converted into biomass estimates based on published life historical data and assigned to trophic groups based on general feeding modes (Froese and Pauly 2015). Table S2 provides a list of species observed with associated trophic designation. Data are reported here as the mean biomass density estimated from the 300 m2 survey area per site (5 transects, each 30 m × 2 m).
To characterize benthic assemblages, a second diver on each transect collected benthic imagery. The percent cover of sessile organisms was quantified from quadrats (0.9 m × 0.6 m) photographed at 2 m intervals along each transect, collected from alternating sides of the transect. Photographs were analyzed by scoring the identity of each organism to the finest taxonomic resolution possible under 25 points placed over each photograph in a stratified random distribution (i.e., one randomly assigned point within each cell of an evenly spaced 5 × 5 grid) using the image analysis software Photogrid 1.0 (Smith et al. 2016). Data were then grouped into benthic functional groups—hard coral, crustose coralline algae (CCA), calcified macroalgae, gorgonian, fleshy invertebrates, turf algae, fleshy macroalgae, cyanobacteria, and non-biological. The mean percent cover of benthic taxa across all photographs from the five transects at each site was calculated. For the analyses presented here, we focused primarily on exploring potential predictors of the relative cover of reef-building taxa (defined here as the sum of hard coral and CCA) and total fish biomass across the 122 sites surveyed.
Estimation of site-level predictors
Potential predictors of observed variability of fish and benthic assemblages across the island were clustered into three categories—anthropogenic, physical, and ecological. An overview of each predictor is presented here, with the detailed methodology presented in the Electronic Supplementary Material and the data presented spatially in Figure S2.
Anthropogenic predictors
The direct effects of human activities on the reef environment can include resource use, pollution, and habitat modification. We estimated the site-specific magnitude of these effects through five variables, as follows: (i) Fishing pressure was estimated as a proxy of nearshore fisheries extraction, which has been associated with shifts in fish assemblage structure and potential indirect effects on benthic community structure (e.g., Sandin et al. 2008; Cinner et al. 2016). (ii) Diving pressure was estimated as a proxy for the density of SCUBA diving tourism, which can cause direct and indirect damage to reef resources through the actions of divers (e.g., kicking corals, increased sedimentation, boat noise) (Hawkins and Roberts 1992). The variables (iii) inhabited surface and (iv) altered surface represent the proportions of the onshore watershed that is occupied by (iii) human habitations or (iv) any human-modified surfaces (including habitations, commercial developments, and roads); both variables can be associated with patterns of urban runoff and pollution. (v) Nutrient pollution (measured using δ 15 N levels in macroalgae) was quantified as a proxy of human-derived nutrients available to reef organisms; this variable has been linked to shifts in competitive dominance of certain benthic taxa such as turf algae and macroalgae (Littler and Littler 1985; Smith et al. 2001).
Physical predictors
The physical environment can have effects on the dynamics and emergent community structure of coral reefs. We considered three site-specific physical variables, as follows: (i) Wave exposure estimates were used as a proxy of physical energy in the reef environment, which has been linked to the structure of fish and benthic assemblages (Bradbury and Young 1981). (ii) Primary productivity measurements were used as a proxy of pelagic nutrient availability (reported as estimated concentration of chlorophyll a in the surface waters), which can influence relative rates of productivity of fishes and benthic taxa (Williams et al. 2015b; Johnson et al. 2020). (iii) Topographical relief was estimated through in situ measurements of reef structural complexity, which has been associated with shifts in fish assemblages (Graham and Nash 2013).
Ecological predictors
The ecological environment is structured at multiple scales (Levin 1992) and can influence community structure. We considered two site-specific measurements of emergent ecological context, as follows: (i) Coral recruit density was estimated as a potential proxy of larval connectivity and local retention. Although the local production of coral larvae can be linked to local coral community composition (and thus could be viewed as a response variable; Hartmann et al. 2018), much of the realized pattern of coral recruitment is a consequence of environmental factors that move, retain, attract, or kill coral larvae rather than a direct consequence of site-specific adult coral density or larval production (Edmunds et al. 2010). For this study, we include coral recruitment as a potential proxy of recruitment limitation at a site scale, consistent with demographically open models of recruitment (Caley et al. 1996). (ii) Turf algal height was estimated as a proxy for site-specific intensity of herbivory (Flower et al. 2017), affecting the outcome of competitive interactions among benthic taxa, especially those among corals, turf algae, and macroalgae (Littler and Littler 1985).
Statistical analyses
Descriptions of the fish and benthic assemblages are presented as the density of major functional groups within each assemblage. Fish data are presented in units of estimated biomass per unit area, and benthic data are presented in units of percent benthic cover. For all analyses, fish biomass data were log-transformed to reduce the skewness that is typical of distributions of fish biomass estimates; benthic data were analyzed without transformation as they did not violate assumptions of normality. Using site-level means, we calculated Pearson’s correlation coefficients between all pairs of fish trophic group biomass estimates and benthic functional group cover to identify magnitudes of covariation.
A general linear modeling framework was used to explore which combination of potential predictors best described the observed spatial variation in fish and benthic community composition. For modeled descriptions of total fish biomass, the site-specific values of benthic cover by functional group were used as additional potential predictors. For modeled descriptions of the benthic cover of reef builders, the site-specific values of fish biomass by trophic group were used as additional predictors. To assess the potential of non-independence among predictors, pairwise Pearson correlation analyses were performed on all combinations of predictors. If any pair of predictors exceeded a threshold of correlation (Pearson \(\rho\) > 0.5), one predictor from the pair was removed from subsequent analyses. Only one potential predictor was dropped universally from subsequent analyses; given the essential equivalence of “altered surface” and “inhabited surface,” we arbitrarily decided to omit the latter. For other pairs of predictors that exceeded the stated threshold for collinearity, the individual predictors were retained in the model-fitting procedure but no models were considered that included both predictors. Note that all pairs of collinear predictors are reported in Tables 1 and 2, with the exception of reef relief and cover of each hard coral and CCA (\(\rho\) of 0.592 and 0.547, respectively). We performed a principal components analysis on all potential predictors, revealing no strong evidence of multi-collinearity (Electronic Supplementary Material). To account for patterns of positive covariation, as well as to focus on general metrics of community structure, all subsequent analyses were then focused on two response variables—total fish biomass (site-specific sum across all trophic groups) and percent cover of benthic reef builders (site-specific sum of hard coral and crustose coralline algae cover).
Linear models were fit for each response variable (total fish biomass and cover of reef builders) by exploring all combinations of potential predictors (12 and 16 potential predictors for fish and reef builders, respectively). We constrained models to include only additive, first-order (linear) terms for each predictor, with no interaction terms, and assumed a Gaussian error structure, comparable to the common implementation of multiple regression procedures. Models were compared based upon Akaike Information Criterion (AIC) values of best-fit models for each combination of predictors, as estimated with the bestglm() function in R (R Development Core Team 2020). Inspection of variance inflation factors of predictors, in the fully parameterized models and the best-fit models, indicated little evidence of collinearity affecting statistical power (Electronic Supplementary Material). For the model with the lowest AIC for each response variable, the site-specific model residuals were extracted for visual exploration. Patterns of spatial autocorrelation among residuals were analyzed using Moran’s I, using an inverse distance weighting of spatial effect.
Results
The coral reefs in the shallow (8–12 m depth) forereef habitats of Curaçao demonstrated substantial spatial variability in both fish and benthic composition (Fig. 1). Across sites, the total biomass density of the fish assemblage ranged from 14.0 to 773.0 g m−2 (n = 121), with a mean of 151.7 (10.9; standard error [SE]) g m−2. Among the four fish trophic groups, herbivorous fishes were the most abundant, composing over one third of the total biomass (Fig. 1B). Across sites, the percentage of total benthic cover of reef builders (i.e., percent cover of hard corals plus CCA) ranged from 0.3 to 45.8% (n = 121), with a mean of 15.7% (1.0%; SE). Within this functional group, hard corals composed the majority of reef builder cover (> 85%; Fig. 1C).
Summary of coral reef community composition across the leeward reefs of Curaçao. A Map of the island showing location of 122 survey sites (note map rotation, with survey sites spread across the majority of the southwest coastline). Shading on land represents areas of human development (i.e., “altered surface”). B Estimates of biomass density of the fish assemblage, presented by trophic group. C Estimates of benthic coverage, presented by functional group. Sites are organized in order along the coastline from the most northwest (left side of panels B and C) to the most southeast (right side of panels B and C); lines are provided as visual guides connecting panels
Pairwise correlation analyses revealed patterns of association within assemblages. All fish trophic groups were positively correlated with one another, with the biomass of planktivores and omnivores least associated (\(\rho\) = 0.246) and those of herbivores and omnivores most associated (\(\rho\) = 0.651; Table 1). The benthic functional groups revealed more variability in pairwise correlation, including both positive and negative values exceeding a threshold for statistical significance (|\(\rho\)|> 0.189, p < 0.05, n = 108). (Due to occasional operational errors, only 108 of the 122 survey sites had complete data describing both fish and benthic communities, as well as estimates of all potential predictors.) Relatively strong positive correlations were found among all pairs of cover values of hard corals, CCA, and calcified macroalgae (\(\rho\) > 0.5 for each pair). Sites with high coral cover were also characterized by high percent cover of other calcifiers. The strongest negative correlations occurred between turf algal cover and the cover of hard coral, gorgonians, and cyanobacteria (\(\rho\) < −0.3 for each pair; Table 1).
Correlation analyses revealed evidence of non-independence of potential predictors. The strongest patterns of covariation occurred among three anthropogenic predictors across survey sites, with inhabited surface, altered surface, and the pollution proxy all strongly positively correlated with one another (\(\rho\) > 0.75 for each; Table 2). Strong patterns of correlation, both negative, were observed between fishing pressure and each wave exposure (\(\rho\) = −0.543) and the pollution proxy (\(\rho\) = −0.407).
The best-fit model describing variability in benthic cover of reef builders (Bi) across survey locations (i) contained four parameters—wave exposure (X1), reef relief (X2), coral recruit density (X3), and turf height (X4)—as follows:
with an R2 value of 0.636 (and an adjusted R2 value of 0.622). The top 10 best-fit models were statistically indistinguishable (all AIC values within 1.6 of one another) and varied in the specific terms included. There was consistent support for inclusion of three parameters in all ten models (wave exposure [+], reef relief [+], and coral recruit density [+], with sign of relationship in brackets; Table 3), such that sites with high cover of reef builders occurred in areas of high wave exposure, reef relief, and coral recruit density. One additional predictor, turf height, was represented in 8 of these 10 models ([−]; Table 3), with high cover of reef builders at sites with low turf height.
The best-fit model describing variability in total fish biomass (Fi) across survey locations (i) contained seven parameters—fishing pressure (X1), diving pressure (X2), reef relief (X3), turf height (X4), and benthic cover values of fleshy invertebrates (X5), fleshy macroalgae (X6), and turf algae (X7)—as follows:
with an R2 value of 0.248 (and an adjusted R2 value of 0.196). The top 10 best-fit models were statistically indistinguishable (all AIC values within 1.0 of one another) and varied in the specific terms included. However, there was consistent support for inclusion of three parameters in all ten models (fishing pressure [–] and cover of fleshy macroalgae [–] and turf algae [ +]; Table 4), such that sites with high fish biomass tended to have low fishing pressure, low macroalgae cover, and high cover of turf algae. Among these models, there was relatively consistent support for the influence of diving pressure ([+]; 9 models) and cover of fleshy invertebrates ([–]; 7 models for each); sites with high fish biomass had relatively higher levels of recreational diving and lower percent cover of fleshy invertebrates. Notably, no appreciable difference in predictive power was found when considering fish biomass individually by trophic group. Best-fit models for herbivores and carnivores were most similar to that of total fish biomass, consistent with the high proportional representation of these groups to the total biomass (Supplementary Information).
Visual inspection of the residuals from the best-fit models reinforces the observation that the model predicting cover of reef builders describes more relative variability than the model describing fish biomass (Fig. 2). Consistent with the low R2 values of the best-fit model, the residuals of fish biomass have similar spatial patterns as the deviation of site-specific values from the island-wide mean (i.e., a zeroth-order approximation; Fig. 2A). Estimates of total fish biomass are spatially autocorrelated (p < 0.001) across the coastline, as are the residuals of the best-fit model (p < 0.05). In contrast, the spatial pattern of residuals from the best-fit model for the cover of reef builders is distinct from that of the original data; estimates of the cover of reef builders are spatially autocorrelated (p < 0.001), but the residuals of the best-fit model have no spatial autocorrelation (p > 0.05).
Summary of residuals. For each (A) total fish biomass and (B) coverage of calcifiers at each survey site, two types of residuals are presented. The colored bars (without outlines) represent the difference of the site-specific value from the mean of all values; the open bars (with black outlines) represent the residuals from the best-fit model for the group (Eqs. 1 and 2)
Discussion
Our survey across 122 coral reef sites along the ~ 70 km of Curaçao’s leeward coastline revealed substantial levels of variability in the community structure of both fish and reef builders (i.e., benthic calcifiers). The fish assemblage was characterized by high spatial variability, with 23 survey sites supporting over 200 g m−2 of total biomass and 47 sites supporting less than 100 g m−2 (from a total of 121 sites with fish biomass data; Fig. 1). The distribution of biomass density values, both in terms of mean and range, is comparable to that reported from studies across multiple Caribbean islands (Newman et al. 2006; Valdivia et al. 2017). The benthic assemblage also varied widely, with 33 sites supporting over 20% cover of hard coral and 59 supporting less than 10% cover (from a total of 121 sites with benthic data; Fig. 1). A recent report supported by the GCRMN showed a comparable mean and range of values for coral cover on reefs across 34 Caribbean countries, states, and territories (see Fig. 9 of Jackson et al. 2014). The coral reefs of Curaçao thus offer a template for considering potential correlates of reef structure, paralleling ranges observed in cross-island studies from the region.
Despite large amounts of spatial variability measured in both fish and benthic assemblages, the ability of predictive models to account for this variability through putative predictors differed dramatically between these two functional groups. We investigated a collection of 10 environmental predictors, complemented by predictors derived from the reef community data themselves. The best-fit model describing variability in the cover of reef-building organisms (summed hard coral and CCA) performed much better than the best-fit model describing variability of total fish biomass (R2 of 0.64 vs 0.25, respectively). Notably, the best-fit model describing variability of reef builder cover contained 4 parameters while the best-fit model describing fish biomass contained 7 parameters; increased descriptive power for the model describing reef builders was not linked to increased parameterization. Thus, when considering variability along an island coastline, we find that a collection of commonly considered anthropogenic, physical, and ecological drivers can provide strong predictive insights into the expected cover of reef builders but appreciably less predictive strength regarding the total fish biomass.
The relative cover of reef builders across Curaçao showed a consistent relationship with elements of the physical and ecological context of the site, but with no obvious linkage to the anthropogenic context. Specifically, sites with higher wave exposure—which, on the southern-facing shores of Curaçao, are found often on promontories—supported higher cover of corals and CCA, consistent with studies revealing the importance of water flow in the growth of reef-building taxa (Dennison and Barnes 1988). Three other variables showed a consistent ability to predict the spatial variability of benthic calcifiers, and each of these observed relationships was consistent with known models of benthic competition and life history. Sites with higher densities of coral recruits or higher relief tended to support higher cover of reef builders and sites with higher turf height tended to support lower cover of reef builders (Table 3). While the patterns of cause-and-effect are not discernable here, given the observational nature of this study, these data are consistent with observations of benthic competitive feedbacks. Sites with more heavily-cropped turf algae (lower turf height) may experience less competition between fast-growing turf and slow-growing reef builders, resulting in higher coral recruitment, higher growth rates of corals and CCA, and ultimately greater carbonate accretion (thus creating more reef relief). We found a strong and consistent signal linking the cover of reef builders, reef relief, and coral recruitment, each of which have been linked to persistence and growth of coral communities.
The total biomass of reef fishes across Curaçao showed more limited predictability relative to cover of reef builders. Two predictors of anthropogenic activity, fishing pressure and diving pressure, were included in most of the top 10 best-fit models describing fish biomass. Estimates of fishing and diving pressure were gathered through user interviews, with the metric linked with aggregate votes among users regarding the importance of sections of coastlines (Electronic Supplementary Material). Although the total predictive power of the model was low, the qualitative observation is consistent with some expectations. The observation that nearshore fishing reduces the standing stock biomass of reef fishes is found reliably across the global tropics (DeMartini et al. 2008; Williams et al. 2008; Cinner et al. 2016), and divers have shown an increased willingness to pay to visit reef areas with more and bigger fish (Gill et al. 2015). Interestingly, such a pair of hypotheses shows inverse directions of causation, with areas that fishers prefer being driven to lower fish biomass and areas with higher fish biomass driving diver preference to visit; this study offers only weak correlational evidence consistent with these hypotheses and importantly no direct evidence supporting such cause and effect. The other parameters that were consistently included in the top models describing fish biomass at a site scale were coverage of benthic types, with a negative relationship with macroalgae and a positive relationship with turf algae. While many studies have explored cause-and-effect relationships between fish biomass and specific algal groups (Ogden and Lobel 1978; Smith et al. 2001; Williams and Polunin 2001), we found little capacity to support hypotheses of fish-induced macroalgal control, especially given the weak statistical fit of the linear model and the lack of pairwise correlations.
In sum, these data from across the forereef habitats of Curaçao reveal only limited associations of total fish biomass with site-specific characteristics. Although we included some predictors linked with well-studied mechanisms affecting fish biomass (e.g., fishing, pollution, habitat availability), the list was not exhaustive. For example, proxies for fish recruitment were not considered, such as metrics of likely pelagic larval supply (Harborne et al. 2018) or proximity to nursery habitats (Nagelkerken et al. 2017). Further, it is possible that methods of fish enumeration themselves may introduce appreciable amounts of noise thus obscuring evidence of spatial patterning (McClanahan et al. 2007). Note that the regression-based approach used here homogenizes such effects of methodological noise.
Visual inspection of the spatial patterns of the total fish biomass and benthic cover of reef builders across Curaçao reveals two strong features: fish biomass is consistently lower at western sites (Fig. 1B) and benthic cover of reef builders is consistently higher at eastern sites (Fig. 1C). The inspection of patterns of spatial autocorrelation offers further insights into the power of environmental predictors to describe community variability across sites. Although the members of the fishing community of Curaçao report a preference for fishing in the western leeward areas of the island (Figure S2A), the spatial patterns of fishing pressure and fish biomass were only weakly aligned (Fig. 2A). When accounting for each potential predictor in the best-fit models of total fish biomass, the residuals from the model maintained a level of spatial autocorrelation consistent with the original fish data. Therefore, none of the environmental predictors, each with spatial signatures (e.g., regions of high value to fishers, proximity to anthropogenic pollution sources near more densely populated areas), held sufficient statistical power to account for spatial autocorrelation in the fish biomass signal. In contrast, the signal of elevated cover of reef builders on the eastern reefs is consistent with reports of high wave energy and complex reef structure and the spatial pattern of these predictors described much of the spatial patterning of benthic calcifiers. Overall, the best-fit models described over 60% of the spatial variability in reef builder cover and accounted for the majority of the spatial autocorrelation in the benthic signal (Fig. 2B).
Management implications of predictable benthic cover and less-predicable fish biomass
The ability to predict the composition of a coral reef community from contextual parameters is a key goal for many ecological studies and resource management efforts. We found that benthic community composition was more amenable to prediction using specifics of the environmental context than was fish biomass. Indeed, the sessile nature of benthic organisms relative to the mobility of fishes may contribute to this pattern at the single-island scale as benthic organisms cannot move in response to changes in local conditions or exogenous disturbances. Interestingly, many past studies have shown a strong ability to predict the spatial variability of fish assemblage structure across broader geographic scales (e.g., inter-region or inter-island comparisons; Friedlander and DeMartini 2002; Cinner et al. 2016; Zgliczynski and Sandin 2017). In contrast, comparable studies of benthic composition, especially when considering effects of local human activities, tend to have more limited statistical support (Williams et al. 2015a). Collating these observations, we find support for a model in which the scale of predictability of fish biomass differs from that of benthic reef builder cover. Whereas fish assemblages show more predictable patterns when site-level data are integrated across islands or regions, benthic assemblages appear to show more predictable patterns when investigated at smaller scales and when linked to specific environmental variables at the site level. Considering, acknowledging, and accounting for these distinct differences in the scales of observation, of human interest, and of ecological predictability is essential in order to maximize the value of survey-based data for scientific and management applications.
We conclude by bounding our interpretation of the data reported here. In contrast to many studies in coral reef ecology, this study was designed to explore the ecological covariation of relatively coarse-scale metrics of coral reef community structure across the island of Curaçao. Given the high level of replication, this dataset provided an unusual opportunity to explore statistical patterns of association at the scale of individual sites and to generate hypotheses regarding the relative strength of potential functional relationships among key environmental and ecological variables at this scale. A common temptation in marine conservation is to focus upon pairwise associations in the development of management assumptions, predictions, and recommendations but we advise that our results not be used in this manner. Correlational studies across locations may offer evidence consistent with current hypotheses of cause-and-effect, but these studies cannot be relied upon to provide robust predictions regarding patterns of change through time. For example, we show only limited evidence that current levels of pollution are linked with lower cover of reef builders (only 3 of the top 10 models report a negative effect of our pollution proxy). These data, however, offer only a temporal snapshot of both pollution and coral cover using observational data from one time point; it is impossible to reject the hypothesis that there has been a strong effect of pollution in the past, for example, sites currently exposed to high anthropogenic nutrient inputs could have supported even higher coral cover in years past. Thus, caution must be taken when interpreting data from one time point, particularly regarding patterns that are likely subject to rapid temporal change.
Our finding that coral reef benthic assemblages show more reliable and more quantifiable patterns of spatial variability than fish assemblages offers valuable insight for the resource management community. For example, our data have direct application to the essential coral reef conservation goal of effectively selecting and delineating reef management zones. For benthic coral reef communities, our results are consistent with the ecological understanding that promontories with higher flow and wave energy can serve as important local refugia for calcifying, reef-building benthic species, which generally suffer from lower rates of survival and recruitment at sites with lower overall water flow, flushing, and oxygen exchange. In contrast, the fish assemblages considered in this study showed only low degrees of predictability across space. We suggest that this finding simply reflects the fact that the variability of fish community structure is not strongly driven by the variables measured here at the spatial scale of data collection (i.e., fine-scale survey data conducted at one point in time). In the case of quantifying fish assemblages for use in adaptive management, there is particular value in maintaining reliable monitoring programs that capture data at temporal and spatial scales consistent with the scale of management goals. The snapshot data presented here highlight the importance of time-series data for accurately evaluating and predicting management efficacy on coral reefs.
References
Almany GR (2004) Differential effects of habitat complexity, predators and competitors on abundance of juvenile and adult coral reef fishes. Oecologia 141:105–113
Arkema KK, Reed DC, Schroeter SC (2009) Direct and indirect effects of giant kelp determine benthic community structure and dynamics. Ecology 90:3126–3137
Blonder B, Moulton DE, Blois J, Enquist BJ, Graae BJ, Macias-Fauria M, McGill B, Nogué S, Ordonez A, Sandel B (2017) Predictability in community dynamics. Ecol Lett 20:293–306
Bouma TJ, Olenin S, Reise K, Ysebaert T (2009) Ecosystem engineering and biodiversity in coastal sediments: posing hypotheses. Helgol Mar Res 63:95–106
Bradbury RH, Young PC (1981) The effects of a major forcing function, wave energy, on a coral reef ecosystem. Mar Ecol Prog Ser 5:229–241
Burkepile DE, Allgeier JE, Shantz AA, Pritchard CE, Lemoine NP, Bhatti LH, Layman CA (2013) Nutrient supply from fishes facilitates macroalgae and suppresses corals in a Caribbean coral reef ecosystem. Sci Rep 3:1–9
Caley MJ, Carr MH, Hixon MA, Hughes TP, Jones GP, Menge BA (1996) Recruitment and the local dynamics of open marine populations. Annu Rev Ecol Syst 27:477–500
Cinner JE, Huchery C, MacNeil MA, Graham NA, McClanahan TR, Maina J, Maire E, Kittinger JN, Hicks CC, Mora C, Allison EH, D’Agata S, Hoey A, Feary DA, Crowder L, Williams ID, Kulbicki M, Viliola L, Wantiez L, Edgar G, Stuart-Smith RD, Sandin SA, Green AL, Hardt MJ, Beger M, Friedlander A, Campbell SJ, Holmes KE, Wilson SK, Brokovich E, Brooks AJ, Cruz-Motta JJ, Booth DJ, Chabanet P, Gough C, Tupper M, Ferse SCA, Sumaila UR, Mouillot D (2016) Bright spots among the world’s coral reefs. Nature 535:416–419
Darling ES, Graham NA, Januchowski-Hartley FA, Nash KL, Pratchett MS, Wilson SK (2017) Relationships between structural complexity, coral traits, and reef fish assemblages. Coral Reefs 36:561–575
DeMartini EE, Roberts DA (1990) Effects of giant kelp (Macrocystis) on the density and abundance of fishes in a cobble-Bottom kelp forest. Bull Mar Sci 46:287–300
DeMartini EE, Friedlander AM, Sandin SA, Sala E (2008) Differences in fish-assemblage structure between fished and unfished atolls in the northern Line Islands, central Pacific. Mar Ecol Prog Ser 365:190–215
Dennison WC, Barnes DJ (1988) Effect of water motion on coral photosynthesis and calcification. J Exp Mar Biol Ecol 115:67–77
Edmunds PJ, Leichter JJ, Adjeroud M (2010) Landscape-scale variation in coral recruitment in Moorea, French Polynesia. Mar Ecol Prog Ser 414:75–89
Flower J, Ortiz JC, Chollett I, Abdullah S, Castro-Sanguino C, Hock K, Lam V, Mumby PJ (2017) Interpreting coral reef monitoring data: A guide for improved management decisions. Ecol Indicators 72:848–869
Friedlander AM, DeMartini EE (2002) Contrasts in density, size, and biomass of reef fishes between the northwestern and the main Hawaiian islands: the effects of fishing down apex predators. Mar Ecol-Prog Ser 230:253–264
Froese R, Pauly D (eds) (2015) FishBase. World Wide Web electronic publication
GCRMN (2016) GCRMN-Caribbean guidelines for coral reef biophysical monitoring Seventh Meeting of the Scientific and Technical Advisory Committee to the Protocol Concerning Specially Protected Areas and Wildlife in the Wider Caribean Region. United Nations Environment Programme, Miami, FL, USA
Gill DA, Schuhmann PW, Oxenford HA (2015) Recreational diver preferences for reef fish attributes: Economic implications of future change. Ecol Econ 111:48–57
Graham N, Nash K (2013) The importance of structural complexity in coral reef ecosystems. Coral Reefs 32:315–326
Graham N, Wilson S, Pratchett MS, Polunin NV, Spalding M (2009) Coral mortality versus structural collapse as drivers of corallivorous butterflyfish decline. Biodivers Conserv 18:3325–3336
Harborne AR, Mumby PJ, Micheli F, Perry CT, Dahlgren CP, Holmes KE, Brumbaugh DR (2006) The functional value of Caribbean coral reef, seagrass and mangrove habitats to ecosystem processes. Adv Mar Biol 50:57–189
Harborne AR, Green AL, Peterson NA, Beger M, Golbuu Y, Houk P, Spalding MD, Taylor BM, Terk E, Treml EA (2018) Modelling and mapping regional-scale patterns of fishing impact and fish stocks to support coral-reef management in Micronesia. Divers Distrib 24:1729–1743
Hartmann AC, Marhaver KL, Vermeij MJ (2018) Corals in healthy populations produce more larvae per unit cover. Conserv Lett 11:e12410
Hawkins JP, Roberts CM (1992) Effects of recreational SCUBA diving on fore-reef slope communities of coral reefs. Biol Conserv 62:171–178
Jackson JBC, Donovan MK, Cramer KL, Lam W (eds) (2014) Status and trends of Caribbean coral reefs: 1970–2012. Global Coral Reef Monitoring Network, IUCN, Gland, Switzerland
Johnson MD, Fox MD, Kelly EL, Zgliczynski BJ, Sandin SA, Smith JE (2020) Ecophysiology of coral reef primary producers across an upwelling gradient in the tropical central Pacific. PLoS ONE 15:e0228448
Knowlton N, Jackson JBC (2008) Shifting baselines, local Impacts, and global change on coral reefs. PLoS Biol 6:e54
Lang JC, Marks KW, Kramer PA, Kramer PR, Ginsburg RN (2010) AGRRA protocols version 5.4. Atlantic and Gulf Rapid Reef Assessment Program, Florida, USA
Lester SE, Rassweiler A, McCoy SJ, Dubel AK, Donovan MK, Miller MW, Miller SD, Ruttenberg BI, Samhouri JF, Hay ME (2020) Caribbean reefs of the Anthropocene: variance in ecosystem metrics indicates bright spots on coral depauperate reefs. Global Change Biol 26:4785–4799
Levin SA (1992) The problem of pattern and scale in ecology. Ecology 73:1943–1967
Littler MM, Littler DS (1985) Factors controlling relative dominance of primary producers on biotic reefs. Proceedings of the 5th International Coral Reef Symposium, Tahiti 4:35–39
Madin JS, Connolly SR (2006) Ecological consequences of major hydrodynamic disturbances on coral reefs. Nature 444:477–480
McClanahan T, Graham N, Maina J, Chabanet P, Bruggemann JH, Polunin N (2007) Influence of instantaneous variation on estimates of coral reef fish populations and communities. Mar Ecol Prog Ser 340:221–234
McGill BJ (2019) The what, how and why of doing macroecology. Global Ecol Biogeogr 28:6–17
Meyer JL, Schultz ET, Helfman GS (1983) Fish schools: an asset to corals. Science 220:1047–1049
Nagelkerken I (2009) Evaluation of nursery function of mangroves and seagrass beds for tropical decapods and reef fishes: patterns and underlying mechanisms. In: Nagelkerken I (ed) Ecological connectivity among tropical coastal ecosystems. Springer, Netherlands, Dordrecht, pp 357–399
Nagelkerken I, Huebert K, Serafy J, Grol M, Dorenbosch M, Bradshaw C (2017) Highly localized replenishment of coral reef fish populations near nursery habitats. Mar Ecol Prog Ser 568:137–150
Nakaoka M (2005) Plant–animal interactions in seagrass beds: ongoing and future challenges for understanding population and community dynamics. Popul Ecol 47:167–177
Newman MJH, Paredes GA, Sala E, Jackson JBC (2006) Structure of Caribbean coral reef communities across a large gradient of fish biomass. Ecol Lett 9:1216–1227
Ogden JC, Lobel PS (1978) The role of herbivorous fishes and urchins in coral reef communities. Environ Biol Fishes 3:49–63
R Development Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Roberts CM, Ormond RF (1987) Habitat complexity and coral reef fish diversity and abundance on Red Sea fringing reefs. Mar Ecol Prog Ser 1:1–8
Sandin SA, Smith JE, DeMartini EE, Dinsdale EA, Donner SD, Friedlander AM, Konotchick T, Malay M, Maragos JE, Obura D, Paulay G, Richie M, Rohwer F, Schroeder RE, Walsh SM, Jackson JBC, Knowlton N, Sala E (2008) Baselines and degradation of coral reefs in the northern Line Islands. PLoS ONE 3:e1548
Smith JE, Smith CM, Hunter CL (2001) An experimental analysis of the effects of herbivory and nutrient enrichment on benthic community dynamics on a Hawaiian reef. Coral Reefs 19:332–342
Smith JE, Brainard R, Carter A, Grillo S, Edwards C, Harris J, Lewis L, Obura D, Rohwer F, Sala E, Vroom PS, Sandin SA (2016) Re-evaluating the health of coral reef communities: baselines and evidence for human impacts across the central Pacific. P Roy Soc B-Biol Sci 283:20151985
Valdivia A, Cox CE, Bruno JF (2017) Predatory fish depletion and recovery potential on Caribbean reefs. Sci Adv 3:e1601303
van Duyl FC (1985) Atlas of the living reefs of Curaçao and Bonaire, Netherlands Antilles. Ph.D. Ph.D. thesis, Vrije Universiteit, p37
Vermeij MJ, Van Moorselaar I, Engelhard S, Hörnlein C, Vonk SM, Visser PM (2010) The effects of nutrient enrichment and herbivore abundance on the ability of turf algae to overgrow coral in the Caribbean. PLoS ONE 5:e14312
Whitfield AK (2017) The role of seagrass meadows, mangrove forests, salt marshes and reed beds as nursery areas and food sources for fishes in estuaries. Rev Fish Biol Fish 27:75–110
Williams GJ, Gove JM, Eynaud Y, Zgliczynski BJ, Sandin SA (2015a) Local human impacts decouple natural biophysical relationships on Pacific coral reefs. Ecography 38:751–761
Williams I, Walsh W, Schroeder R, Friedlander A, Richards B, Stamoulis K (2008) Assessing the importance of fishing impacts on Hawaiian coral reef fish assemblages along regional-scale human population gradients. Environ Conserv 1:261–272
Williams ID, Polunin NVC (2001) Large-scale associations between macroalgal cover and grazer biomass on mid-depth reefs in the Caribbean. Coral Reefs 19:358–366
Williams ID, Baum JK, Heenan A, Hanson KM, Nadon MO, Brainard RE (2015b) Human, oceanographic and habitat drivers of central and western Pacific coral reef fish assemblages. PLoS ONE 10:e0120516
Wilson SK, Graham NAJ, Pratchett MS, Jones GP, Polunin NVC (2006) Multiple disturbances and the global degradation of coral reefs: are reef fishes at risk or resilient? Global Change Biol 12:2220–2234
Yates KL, Bouchet PJ, Caley MJ, Mengersen K, Randin CF, Parnell S, Fielding AH, Bamford AJ, Ban S, Barbosa AM (2018) Outstanding challenges in the transferability of ecological models. Trends Ecol Evol 33:790–802
Zaneveld JR, Burkepile DE, Shantz AA, Pritchard CE, McMinds R, Payet JP, Welsh R, Correa AM, Lemoine NP, Rosales S (2016) Overfishing and nutrient pollution interact with temperature to disrupt coral reefs down to microbial scales. Nat Commun 7:11833
Zgliczynski BJ, Sandin SA (2017) Size-structural shifts reveal intensity of exploitation in coral reef fisheries. Ecol Indicators 73:411–421
Acknowledgements
We thank the collaborative community of Blue Halo Curaçao, who serve to integrate science, economics, and public opinion into effective marine management plans for tomorrow, and who provided expert support for the research described here. We thank Forest Rohwer for valuable discussions regarding this study and the thoughtful commentary and suggestions of two anonymous reviewers. This research and the larger efforts of Blue Halo Curacao were supported by funding from the Waitt Institute and with permissions from the Government of Curacao, Ministry of Health, Environment, and Nature. Field logistics were further supported by the Waitt Institute vessel crew, CARMABI Foundation, The Dive Shop Curacao, and Dive Charter Curacao.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Topic Editor Alastair Harborne
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sandin, S.A., Alcantar, E., Clark, R. et al. Benthic assemblages are more predictable than fish assemblages at an island scale. Coral Reefs 41, 1031–1043 (2022). https://doi.org/10.1007/s00338-022-02272-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-022-02272-5