Abstract
Increasing abundance of arborescent octocorals (often referred to as gorgonians) on Caribbean reefs raises the question of whether habitat structure provided by octocorals can mediate a transition between coral- and algal- dominated states by increasing fish abundance and herbivory. This study tested the hypotheses that feeding rates and densities of demersal reef fishes are affected by the habitat structure provided by dense octocoral communities. Surveys of fishes on coral reefs in St John, US Virgin Islands, found 1.7-fold higher densities, and 2.4-fold higher feeding rates within versus outside of dense octocoral canopies. This difference, however, was only seen at sites with octocoral densities > 8 colonies m−2. Furthemore, the proximity of octocoral colonies to fish had an effect on the grazing rate of key herbivores (surgeonfishes and parrotfishes), with a 53% higher feeding rate (1.90 ± 0.11 bites min−1 m−2) near octocorals (< 20 or 30 cm, depending on the site) versus farther from them (1.24 ± 0.09 bites min−1 m−2). Finally, within the canopy of dense octocoral communities (17 colonies m−2), reef fishes fed at a rate that was 2.2-fold higher within the community than at the edge of the community that faced an adjacent sand patch. Fish abundance, however, was not uniformly higher within versus at the edge of the octocoral community, as ecotone specialists such as gobiids, blennioids, ostraciids, holocentrids, labrids, and pomacentrids were 1.3—2.3 times more abundant at the edge. In contrast, other taxa of demersal fishes, notably herbivores, were twice as abundant within octocoral communities than at the edges. Together, these results reveal an association between habitat structure created by octocorals on shallow reefs and increased feeding rates of demersal fishes (including those of herbivores). The potential of octocorals to increase herbivory that could mediate stony coral recovery is therefore worthy of further study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Arborescent benthic organisms (sensu Jackson 1979) such as corals are among the most effective of ecosystem engineers (sensu Jones et al. 1994) due to the large size and great quantity of the physical habitat structure they potentially can provide. Habitat structure affects the behavior of fishes on coral reefs as it provides refuges from predators, and habitats where suitable refuges for fish are present, often have higher fish abundance than relatively barren habitats (Hixon and Beets 1989). Shelter from predators can be provided to reef fishes by a variety of rigid physical structures (Dahlgren and Eggleston 2000; Pollux et al. 2007), which create cavities and spaces among coral branches (Holbrook et al. 2000, 2002; Holbrook and Schmitt 2002; Coni et al. 2013), within crevices in the reef matrix, and among rocks or boulders of igneous and biogenic origins.
In addition to hard structures, soft, three-dimensional structure provided by ecosystem engineers such as seagrass, kelp, and octocorals can also provide protection to fish by reducing the chance of visual detection by predators (e.g., Dahlgren and Eggleston 2000; Anderson 2001a; Walter and Haynes 2006; Pollux et al. 2007). While providing reduced physical protection to organisms that associate with them, these ecosystem engineers can nevertheless hide fish behind their swaying branches, thus facilitating their camouflage, which is an important survival strategy among reef-associated fishes (Randall and Randall 1960; Endler 1981). In this way, benthic organisms providing soft structure can ultimately fulfill a similar role for fishes as that provided by rigid physical shelter (i.e., protection from predation).
Octocorals (often referred to as gorgonians) are one such taxon of ecosystem engineers that can provide soft physical structure. Analogous to terrestrial forests, where the above ground portion of plant communities creates a structurally complex canopy, communities of arborescent octocorals (hereafter, “octocorals”) provide a canopy, which serves as habitat to associated organisms (Parrish et al. 2002; Carvalho et al. 2014), and can be found across a variety of depths and latitudes. For example, octocoral canopies can provide habitat to demersal fishes, as evidenced by fisheries expanding into octocoral habitat (Wolff et al. 1999) referred to as “gorgonian plains” (sensu Mumby 2016). Given that octocorals have become more common over the last decades on at least some shallow reefs in the Caribbean (Ruzicka et al. 2013; Tsounis and Edmunds 2017; Tsounis et al. 2018; Johnson and Hallock 2020), the question arises of how these octocoral communities affect ecological processes. Here we focus on the effects of habitat structure created by octocorals on the abundance and feeding rates of fishes, with an emphasis on herbivorous fishes because of their role in determining benthic community structure (Adam et al. 2015).
On tropical coral reefs, herbivory can structure shallow benthic communities, as it affects the relative abundance of coral and macroalgae (Aronson and Precht 2000). Herbivory by fishes can play a key role in regime shifts between coral- and macroalgal-dominated phase states (Mumby 2006; Hughes et al. 2007; Adam et al. 2015; Plass-Johnson et al. 2015). This has become dramatically evident when overfishing of large herbivorous fishes reduced the resilience of Jamaican reefs against the impact of hurricane Allen in 1980, thus contributing to the subsequent phase shift toward an algal-dominated state (Hughes 1994). Furthermore, reefs with low stony coral cover offer less shelter for fish, and thus insufficient herbivorous control of macroalgal cover (Williams et al. 2001). Therefore, in light of decreasing stony coral cover ("reef flattening", sensu Newman et al. 2015) and increasing abundance of octocorals on Caribbean reefs, the potential influence of octocorals on top-down control by herbivores on macroalgae emerges as a timely question with relevance to conservation and restoration.
The potential role of the habitat created by dense octocoral canopies in mediating ecological processes on Caribbean reefs motivated our field research to test three hypotheses. (1) Fish abundance and their feeding rates are higher within the canopy of dense octocoral communities than outside, as a result of octocorals providing visual cover. (2) Individual fish feed more intensely near octocoral colonies than away from them, because they are safer due to visual cover. And (3) the abundance of fishes and their feeding rates are higher within dense octocoral communities than at their edge, due to a preference for microhabitats that are safer from visually guided predators. Together, these hypotheses elucidate the role of thriving octocoral communities on fish feeding rates, which could mediate competition between macroalgae and scleractinian corals.
Materials and methods
Study area
Surveys were conducted in St John, US Virgin Islands, in February and March 2016. Octocoral communities are common on hard substrata in shallow water in St. John (Edmunds et al. 2015) and abundance of reef fishes is high (Monaco et al. 2007). Building on our previous studies on the environmental impact of octocoral community structure (Tsounis and Edmunds 2017; Tsounis et al. 2018), the present study was conducted at East Cabritte, Europa Bay, and East Tektite, within the Virgin Islands National Park, along the south shore of St. John (Fig. 1). At these sites, the fringing reefs at 5–9 m depth are populated by diverse octocoral communities growing on igneous rock and biogenic reef substrata (Tsounis et al. 2018). These sites differ from nearby sites in slightly deeper water that historically have been dominated by Orbicella annularis, and in one case (Tektite) remain dominated by this species (Edmunds 2015). Europa Bay is close to a “gorgonian plain” sensu Mumby (2016), while East Cabritte is a fringing reef that is exposed to prevailing waves (Tsounis et al. 2018). East Tektite is the most sheltered site and the horizontal hard substratum in this location is partially and ephemerally covered by sand that limits colonization by benthic organisms.
Study design
Because our study was conducted in a national park where manipulative experiments are not encouraged, a mensurative approach was taken to explore the influence of octocorals on reef fishes. Fish abundances and behavior were quantified at sites differing in population densities of octocorals. Since octocoral communities are spatially heterogeneous on a scale of 100′s of meters along the south coast of St. John (Edmunds et al. 2015; Tsounis et al. 2018), sites (and locations within sites) of contrasting octocoral density were censused to test for associations between octocoral density and densities of herbivorous fishes, and between octocoral density and the feeding rate of fishes with which they are associated. Surveys were conducted at the three aforementioned sites (Fig. 1), which provided a range of octocoral densities, enabling a broader view of the potential impacts of octocorals on fishes. The hypotheses were tested through a three-tiered approach.
Effects of the octocoral canopy on fish abundance and feeding rate
The objective of this hypothesis was to test for variation in the rate of fish foraging and fish abundance within, versus outside of dense octocoral canopies. Our sampling regime was designed to quantify these factors in a manner that reduced the likelihood that the observation procedure would affect the behavior of the fishes. Some of the features (octocoral density and colony height) defining these habitats were quantified in 2015 (Tsounis et al. 2018), and we assumed these did not differ between 2015 and when surveys of fishes were conducted in 2016, at least not in biologically meaningful ways relative to the hypotheses we wished to test. In order to provide a broader characterization the habitat, other features of the benthos were measured using quadrats (1 × 2 m) that were surveyed for fish foraging behavior. Each of these quadrats was censused for the number of scleractinian corals, octocorals, and demospongia (pooled among species within each broad taxon). Scleractinians were counted regardless of colony size, and a colony was defined by an autonomous area of tissue. Colonies of octocorals > 5 cm tall were counted if their holdfasts were in the quadrat, and demospongia were counted based on the presence of discrete areas of biomass that were unconnected to other sponges.
The key contrast used to test our hypothesis was the distinction between locations categorized as within vs outside octocoral canopies, which we based on the presence or absence of tall octocorals. Arborescent octocorals are often the most conspicuous biogenic features on the shallow reefs of St. John, with adult colonies of many species taller than other topographic features created by stony corals, sponges, and igneous rocks (Tsounis et al. 2018). Locations within each of the three sites were defined as “within canopy” if at least two large octocoral colonies were present in the quadrat. Where this criterion was not met, the area was categorized as “outside of canopy”. "Large colonies" were defined by the estimated mean height of arborescent octocorals at each site, and thus differed among sites. Large colonies were > 20 cm tall at Europa Bay and East Tektite, and > 30 cm tall at East Cabritte. This definition of "within canopy" and "outside of canopy" was used because octocorals were so prevalent at our study sites that finding large areas on comparable substrata (i.e., hard surfaces at the same depth and same exposure to waves) without octocorals was not possible. Hard substrata at East Cabritte and Europa Bay were densely populated by octocorals, but colonies were patchily distributed. Sand patches with no octocorals were not sampled to avoid confounding effects of octocorals with effects of physical substratum. The surveyed hard substrata appeared to have fairly uniformly cover of macroalgae and turf, so that the availability of food resource to fishes was unlikely to influence the results. At East Tektite, arborescent octocorals tended to form small patches on exposed hard substrata within surrounding areas covered with sand and lacking octocorals. Furthermore, while macroalgae cover was not quantified, they were present in all quadrats (at an estimate cover of > 70% for turf and macroalgae combined). The criteria defining octocoral canopies were refined based on observations of how visual detection of fish within octocoral canopies is impacted by the relationship between octocoral size and observer position above the canopy. The outcome of these observations led us to hypothesize that octocoral communities with colonies ≥ 20 cm in height at densities allowing nearest neighbor distances approximately equal to their height, to effectively hide small demersal reef fishes (Fig. 2). Online Appendix 1 characterizes how habitat features differ between areas classified as within vs outside of octocoral.
Diagram illustrating the relationship between the height of octocoral colonies and the capacity of predatory fish (X) that hunt through visual identification of prey to locate prey among octocoral colonies. Based on the visual angle of fish predators above the canopy, prey items have a potential refuge behind the colonies, the size of which is a function of their height. The density of the octocoral community determines the proportion of areas serving as effective shelter (marked as shaded area "2"), and areas where fish are visible a 45° angle or steeper (area 1)
Point census surveys (Thresher 1986) in 1 × 2 m quadrats placed within canopies and outside of canopies were used to quantify the abundances of fishes by family, and in these quadrats the foraging rates of fishes were estimated from the rate at which they took bites from benthic surfaces. An L-shaped PVC frame (1 × 2 m, hereafter referred to as “quadrat”) was placed haphazardly on the reef at each site, and then the observer retreated 5–7 m away and let 2–3 min pass to allow fishes to acclimate to the presence of the PVC frame. This interval was chosen based on preliminary surveys in which fish approached and fed within the quadrat within 2 min of its placement on the reef. All fish within 1 m of the bottom were counted (i.e., not within the entire water column above the quadrat). Quadrats were categorized as either within versus outside of the octocoral canopy, as described above, after being placed. Each quadrat was observed for 5 min, during which the total number of bites on benthic surfaces by fishes (pooled among taxa) was counted. Quadrats in which fish reacted to the presence of the observer, for example, by seeking refuge in response to the diver, were excluded from the analyses.
Fish abundance was defined as the number of fish (by family) within the quadrat and not above the octocoral canopy during the last minute of each observation period, with this approach avoiding overestimating fish abundances (Ward-Paige et al. 2010). If the same fish entered the quadrat repeatedly they were scored as one individual. The abundance of fishes was quantified by taxon, mainly at the family level: Chaetodontidae, Ostracidae, Synodontidae, Carangidae, non-parrotfish labrids (Labridae), parrotfishes (Labridae, former Scaridae), Scorpanidae, Acanthuridae, Lutjanidae, Aulostomidae, Monacanthidae, Pomacanthidae, Pomacentridae. Another category included all blennioids and gobiids, and a final category included all other fishes (which were < 3-cm long and could not easily be identified). Since some groups (especially Pomacanthidae, Pomacentridae, and Blennioidei/Gobiidae) contained both herbivores and carnivores, we could not evaluate herbivores and carnivores separately.
To test for differences in fish abundance and foraging rates within versus outside the canopy and among study sites, two-way fixed effects univariate PERMANOVA was used. This non-parametric approach was used instead of parametric analysis of variance (ANOVA) because the data were not normally distributed due to an abundance of quadrats in which fishes were absent. Data were square root transformed to ensure they met the homogeneity of variance/dispersion assumption (p > 0.05), to which PERMANOVA is sensitive (Anderson and Walsh 2013). These and all other PERMANOVA analyses were conducted in PRIMER v7 with PERMANOVA + and the permutational P values were based on 9999 permutations of the data. All univariate PERMANOVA analyses used a Euclidean distance matrix (Anderson 2001b).
To explore association of benthic habitat features with fish assemblage and feeding rate, multivariate analyses were performed using PRIMER v7. These analyses tested for association between multivariate benthic features and multivariate fish abundance in a permutational framework. Prior to computing resemblance matrices based on Bray–Curtis dissimilarity indices, data were square-root transformed and a dummy variable (1) was added. The resemblance matrix describing benthic features included the density of octocorals (colonies m−2), scleractinians (colonies m−2), and sponges (individuals m−2). Statistical significance of the separation was tested using a two-way PERMANOVA in which sites and canopy categories were fixed effects. A similarity percentage analysis (SIMPER, Clarke 1993) was performed to evaluate the contribution of each substratum category to the dissimilarity in multivariate communities between the canopy categories (pooled). Two-dimensional ordination by NMDS was used to illustrate separation among sites and canopy categories of benthic features defined by the three variables using the Vegan package for R.
A second resemblance matrix summarized the abundance of fishes by family, and it was prepared using the same methods as described above. It was used to illustrate separation of the multivariate fish abundances by family into the canopy categories using NMDS, and to test for differences between canopy categories and sites using PERMANOVA. SIMPER was used to determine the contribution of individual fish species to the dissimilarity between canopy categories. The role of benthic community structure in determining the rate of fish foraging (Hypothesis 1) was explored using multiple linear regression to test the effects of density of octocorals, scleractinians and sponges on bites rate (pooled among fish taxa), using quadrats as statistical replicates.
Effects of proximity to octocorals on feeding by herbivorous fishes
A “focal individual” sampling technique (sensu Altman 1974) was used to quantify the bite rate of individual herbivorous fishes as a function of proximity to octocoral colonies. In this approach, the behavior of a haphazardly selected focal fish (belonging to one of two important and abundant groups, surgeonfishes and parrotfishes) was recorded until either it was lost from view or the observation period ended (5 min duration). During the observation period, the number of bites on benthic surfaces by the focal fish was counted and summed by location relative to the nearest octocoral colony of at least average size at each site (> 19.5 cm at Europa Bay and East Tektite; > 31.8 cm at East Cabritte, hereafter referred to as “large” colonies). Sizes of colonies were estimated by eye. Proximity to the nearest octocoral colony was categorically distinguished as “close” or “far” based on distances estimated by eye to the nearest 0.1 m, with these categories determined by the average colony height for the site (i.e., close was within ~ 20 cm at Europa Bay and East Tektite; and ~ 32 cm at East Cabritte). Consequently, distances between fish and large octocorals were considered far if they were > 20 cm at Europa Bay and East Tektite; and > 32 cm at East Cabritte. This categorization of proximity of the focal fish to the nearest octocoral colony does not apply the definition of canopy, and was instead based on the assumption that fish near the seafloor would be obscured from view of fish predators in the water above the octocoral canopy as a function of their proximity to octocoral colonies and the height of the colony (Fig. 2). To fish predators looking down at a viewing angle of < 45° to the horizontal, potential fish prey in an octocoral community would be obscured when they are 20–30 cm from an octocoral colony of 20–30 cm height. Such prey should only be visible from a viewing angle of > 45°, or when the predator is close enough to the prey that no octocoral is blocking its view. Because the data did not meet the assumption of normality (with or without transformation) univariate PERMANOVA on square-root transformed data was used to test the effects of site and proximity to octocoral colonies as fixed factors, and fish individual as a random factor (included because each individual had a bite rate measured both close and far from large octocorals).
Fish abundance and feeding within the octocoral canopy versus at its edge
If fish seek octocoral canopies for visual shelter, then the edge of the canopy should be a location less frequently selected by fishes compared to within the canopy. This test was made possible by the spatial configuration of octocoral communities at Europa Bay and East Cabritte, both of which were > 10 m wide, and characterized by a well-defined seaward edge adjacent to sand at 11-m depth. Our third site, East Tektite could not be used to test this hypothesis because there were no well-defined edges to the octocoral communities.
We conducted point census surveys to quantify fish abundance and feeding rates within octocoral communities and at their edges. We expected that fishes at the edges of octocoral communities would forage less frequently than those within octocoral communities, because at the edges they would be exposed to higher risk of predation. Predators (serranids, sphyraenids, and lutjanids) were observed roaming the study sites. Serranids were quantified if present on the quadrats, but other piscivores (e.g., sphyraenids and lutjanids) were too sparse to be quantified reliably with our sample size. To compare demersal fish feeding behavior within octocoral communities versus at the edges of these communities, locations “within” octocoral communities were defined as > 5 m from the edge. Abundance and feeding rate of demersal fishes (Ostraciidae, Mullidae, Holocentridae, non-parrotfish Labridae, Chaetodontidae, Tetraodontidae, Serranidae, Acanthuridae, parrotfishes, Pomacentidae, Blennioid/Gobiidae) were compared between these two locations using point census quadrats as described above to test hypothesis 1. Two-way fixed effects PERMANOVA in PRIMER v7 was used on square-root transformed data to test whether the multivariate fish assemblage differed in abundance relative to the canopy (within vs at the edge), as well as between sites. Additionally, two univariate PERMANOVAs using Euclidean distance matrices were used to test differences in overall feeding rate and total fish abundance (all families pooled).
Results
Overview
Most fish in the survey areas belonged to families characterized by small (< ~ 30 cm long) demersal species. The best represented taxa were non-parrotfish wrasses (Labridae) and surgeonfish (Acanthuridae), although damselfish (Pomacentridae), parrotfish (Labridae), and angelfish (Pomacanthidae) were also common (Table 2). Overall, mean (± SE) fish abundance at each site (averaging between inside and outside the canopy) was 2.3 ± 0.4 individuals m−2 (n = 35) at Europa Bay, 1.1 ± 0.2 individuals m−2 (n = 34) at East Cabritte, and 0.5 ± 0.1 individuals m−2 (n = 36) at East Tektite.
Effects of the octocoral canopy on fish abundance and feeding rate
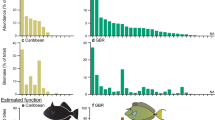
Overall feeding rate (total bites per minute by all fishes) was higher inside octocoral canopies than outside of them (pseudo-F1,99 = 7.44, Pperm = 0.006), and differed among sites (pseudo-F2,99 = 4.37, Pperm = 0.013), but there was no interaction between the two (pseudo-F2,99 = 1.15, Pperm = 0.316). Overall mean (± SE) feeding rate at Europa Bay was 2.41 ± 0.63 bites m−2 min−1 (inside the canopy) versus 0.95 ± 0.43 bites m−2 min−1 (outside the canopy), and at East Cabritte 1.73 ± 0.84 bites m−2 min−1 (inside the canopy) versus 0.47 ± 0.11 bites m−2 min−1 (outside the canopy) (average ± SE; n = 93). At East Tektite mean feeding rate was lower than at the other two sites and was similar inside vs. outside the canopy, amounting to 0.50 ± 0.17 bites m−2 min−1 inside the canopy, and 0.52 ± 0.32 bites m−2 min−1 outside the canopy (Tables 1, 2; Fig. 3a).
a Feeding rate of fishes (Mean ± SE shown bites min−1 m−2) in areas of reef categorized as located within the canopy versus outside the canopy at three sites on the south shore of St. John. If density of large (> 30 cm tall) octocorals was ≥ 2 colonies m−2 the quadrat was categorized as “within the canopy”, and "outside the canopy" if lower. Data are separated into 3 sites (no significant difference between sites). b Total fish abundance (pooled among taxa) per square meter in 1 × 2 m quadrats. The number of fish observed is shown above each bar. The number of quadrats (n) observed were: 21 (Europa, within), 14 (Europa, outside), 18 (East Cabritte, within), 16 (East Cabritte outside), 17 Tektite, within), 19 (Tektite outside)
Fish abundance varied among sites and canopy categories in a pattern similar to that of feeding rate. Fish abundance was higher inside the octocoral canopy than outside (pseudo-F1,99 = 5.5, Pperm < 0.023) (Fig. 3b), and differed among sites (pseudo-F2,99 = 10.6, Pperm < 0.001), but there was no interaction between the two (pseudo-F2,99 = 1.9, Pperm = 0.165). At East Tektite, fish abundances inside the canopy were lower than at the other sites, and were also similar inside the canopy (0.41 ± 0.12 individuals m−2 min−1) and outside the canopy (0.53 ± 0.23 individuals m−2 min−1) (Table 1). Multivariate fish community structure statistically differed between canopy categories (pseudo-F1,99 = 2.6, Pperm = 0.047), despite broad overlap between the canopy categories in NMDS ordination (online Appendix 2). Multivariate fish community structure also differed among study sites (pseudo-F1,99 = 5.2, Pperm = 0.003), with no interaction between canopy categories and sites (pseudo-F1,99 = 1.3, Pperm = 0.246).
Effects of proximity to octocorals on rate of feeding by herbivorous fishes
Among the 89 fish that were observed for 1–5 min at the three sites, 66 were surgeonfishes (Acanthuridae), and 23 parrofishes (Labridae). These fishes all roamed through the study area and grazed on benthic algae both close to and far from octocorals. Feeding rate (both families combined) was 1.56-fold higher close to octocoral colonies (i.e., within < 20 cm at Europa Bay and East Tektite, and < 30 cm at East Cabritte), with mean (± SE) feeding rates of 1.7 ± 0.25 to 2.0 ± 0.15 bites min−1 m−2 close to octocorals, and 1.1 ± 0.20 to 1.4 ± 0.13 bites min−1 m−2 far from octocorals (Fig. 4). These differences in feeding rate were statistically significant (pseudo-F1,86 = 13.9, Pperm = 0.001), and there were also differences among sites (pseudo-F2,86 = 5.7, Pperm = 0.004), but no interaction between the two (pseudo-F2,86 = 0.1, Pperm = 0.98). Overall, 60.8 ± 2.4% of feeding occurred near octocorals.
Feeding rate of herbivorous fishes (acanthurids and scarids combined; bites min−1 m−2) as a function of proximity to octocorals (“near” versus “far”) at three sites along the south shore of St. John. Proximity to the nearest octocoral colony was categorically distinguished as “close” or “far” based on estimated distance, with these categories determined by the average colony height for the site (i.e., close was within 19.5 cm at Europa Bay and East Tektite; and 31.8 cm at East Cabritte). Mean ± SE shown, with number (n) of observed fish = 24 (East Cabritte), 61 (Europa Bay), 19 (East Tektite)
Fish abundance and feeding within the octocoral canopy vs at its edge
Overall feeding rate (all fish taxa pooled) was higher in quadrats within versus at the edge of octocoral communities (pseudo-F1,96 = 8.3, Pperm < 0.008), but did not differ between Europa Bay and East Cabritte (F1,96 = 0.1, Pperm = 0.859), with no interaction between the two (F1,96 = 0.02, Pperm = 0.890) (Europa Bay, n = 52; East Cabritte, n = 47) (Fig. 5). The feeding rate within canopies was 2.40 ± 0.38 bites min−1 m−2 versus 1.09 ± 0.26 bites min−1 m−2 at the edge of canopies (within and edge, n = 50).
Total fish abundance (Individuals m−2, black bars) and their feeding rate (Mean ± SE shown bites min−1 m2, grey bars) at locations categorized as within octocoral communities or at their edges for data pooled between East Cabritte and Europa Bay (based on no significant differences between the two). Right hand columns show data for 1 × 2 m quadrats placed within the octocoral community while right hand columns show data for quadrats placed at the edge of the community, facing a sand patch. Feeding rates are based on 25 observations for each category. The number of fish observed is shown above each bar. The number of quadrats (n) observed were 50 (edge), 50 (center)
Overall fish abundance (pooled among taxa) displayed the opposite trend as feeding rate, with fewer fish within canopies (2.54 ± 0.28 fish m−2) versus at the edge of canopies (3.72 ± 0.21 fish m−2) (pseudo-F1,96 = 8.3, Pperm = 0.006). Fish abundance did not differ between the two sites (pseudo-F1,96 = 0.1, Pperm = 0.763), with no significant interaction between “canopy” and “site” (pseudo-F1,96 = 0.02, Pperm = 0.896). The higher density of fishes at the edge of octocoral community vs. inside them was driven by gobiids, blennioids, ostraciids, holocentrids, carnivorous labrids, and pomacentrids, which were 1.3–2.3 times more abundant at the edge of octocoral community than within the canopy. In contrast, some other taxa of fishes, notably the herbivorous parrotfishes and surgeonfishes were twice as abundant within the octocoral canopy than at the edge (Table 3). The multivariate fish assemblage differed between the canopy edge and within the canopy (pseudo-F1,96 = 3.1, Pperm = 0.010) and between sites (pseudo-F1,96 = 7.2, Pperm < 0.001), with no interaction between the two (pseudo-F1,96 = 1.7, Pperm = 0.148).
Discussion
Our study was motivated by environmental changes over recent decades that have resulted in modifications to the community structure of present day Caribbean coral reefs (Hughes 1994; Jackson et al. 2014). While these changes are best known in the benthic realm for changes that have favored macroalgae over scleractinians (Pandolfi et al. 2005), on some reefs they have favored octocorals (gorgonians) relative to scleractinians (Ruzicka et al. 2013; Tsounis and Edmunds 2017). Our mensurative approach reveals that many reef fishes associate with octocorals, and highlights their higher feeding rates near octocoral colonies.
Effects of the octocoral canopy on fish abundance and feeding rate.
Overall our study showed that fish abundance and foraging rates were higher inside octocoral canopies than outside of them, but this pattern was absent at East Tektite where fish abundances, and feeding rates were lower than at the other two sites. In fact, the feeding rates of fishes inside canopies at East Tektite were similar to the rates recorded outside of canopies at the other two sites, suggesting that the lack of a continuous canopy and low densities of octocorals at East Tektite (i.e., 3.3 colonies m−2) compared to the other two sites (i.e., 16.8 and 8.2 colonies m−2) may not provide a canopy effect for demersal fishes. Densities of octocorals in areas categorized as “inside canopies” at East Tektite were equivalent to the densities recorded in areas of reef categorized as outside canopies at the other two sites. This finding suggests that there may be a threshold density of octocorals necessary to attract fish and facilitate their foraging. Differences in octocoral height, rather than their density, seem unlikely to explain this pattern given that mean octocoral height was similar at East Tektite and Europa Bay. A threshold octocoral density for a canopy effect (i.e., effective in shielding small demersal fishes from predators) between 3.3–8.2 colonies m−2 would imply a degree of resistance of the canopy effect to disturbance by hurricanes, as octocoral communities in St. John were found to maintain such densities after hurricanes between 1987 and 2013 (Tsounis and Edmunds 2017; Tsounis et al. 2018). Furthermore, the present data suggest that octocorals may now be providing at least some environmental services (i.e., habitat provisioning for demersal fishes) that have been lost on Caribbean reefs due to region-wide decline of architectural complexity (sensu Alvarez-Filip et al. 2009) attributed to the decline in abundance of scleractinian corals.
The highest fish abundances and feeding intensities were recorded at Europa Bay, where the octocoral canopy had a lower mean height than at East Cabritte, suggesting there may be an optimum canopy height with respect to canopy effects on fishes, or that fish behavior was driven by other habitat features. The benthic community at our sites included not only octocorals, but also sponges and scleractinians, which provide physical habitat structure. However, the biggest contributor to differences between our octocoral canopy categories was octocoral height and abundance. The abundance and type of fish predators present can also influence fish behavior, including the time that herbivores spend grazing (Heithaus et al. 2008; Rizzari et al. 2014). Large predators such as barracudas were regularly observed in open water at East Cabritte, but rarely at Europa Bay (G. Tsounis, pers. obs.). Hydrodynamic conditions such as exposure to weather may also affect habitat preferences of fishes (Fulton and Bellwood 2005), and higher abundance in Europa is consistent with East Cabritte being more exposed to weather (Tsounis et al. 2018). Benthic rugosity on the scale of decimeters to meters can create physical habitat structure, potentially affecting site preferences similarly to benthic invertebrates. Small-scale substratum rugosity quantified at the decimeter scale did not differ between East Cabritte and Europa Bay (Tsounis et al. 2018). However, differences at larger scale (meter-scale) have not been quantified in the study areas, so that dissimilar fish abundance and behavior between sites cannot be fully explained by rugosity with our present data set. The quadrat size (1 × 2 m) furthermore appears to be smaller than the home range sizes of the observed fishes, and thus to not bias the results. Although the exact sizes of the home ranges for all the species encountered are not known, they are likely on the meter-scale for blennioids and gobiids, and on the scale of 10’ s or 100’ s of meters for most other demersal fish (Tables 2, 3). Overall, higher fish abundance in complex habitat structure is consistent with other studies (Holbrook et al. 2002; Walter and Haynes 2006; Huntington et al. 2017).
Effects of proximity to octocorals on rate of feeding by herbivorous fishes
Based on focal individual sampling (sensu Altman 1974), individual fish foraged 1.4–1.8-fold faster near octocorals. This finding suggests that fish use the octocoral canopy as visual cover against roving predators, and thus spend more time foraging and less time on being vigilant to the presence of potential predators. Visual cover relies on visual confusion in a similar way as group living (shoaling in open water fish), reducing hunting success of predators and consequently the per capita predation risk of prey (Krause and Godin 1995). Analogous to predation risk decreasing with prey group size (Pitcher and Parrish 1993), it is likely that predation risk in demersal fishes decreases with increasing octocoral density and patch sizes, as in both examples, individual fish being visually obscured reduces their risk of predation. The octocoral communities at our two sites with dense canopies probably provided effective visual cover from most viewing positions from which predators would stage their attacks (Fig. 2). Non-visual hunting predators (e.g., using electroreception or chemoreception) would not be affected by this visual obstruction of prey by octocorals, but such predators (e.g., sharks and moray eels) were uncommon and inactive during the day at our study sites.
Fish abundance and feeding within the octocoral canopy vs at its edge
Edges of octocoral canopies were expected to provide less effective visual shelter from predators than their interiors. The feeding rate of fishes was 2.2-fold higher within the canopy than at the edge, thereby demonstrating an “edge effect” with respect to the intensity of fish feeding (sensu Leopold 1932). This effect has been observed in seagrass meadows as well (Smith et al. 2011). Fish abundance, however, was 0.68-fold lower within the canopy than at the edge, seemingly contradicting the concept that the edge is a less desirable zone than more central parts of the canopy. This contradiction can be resolved by evaluating which kinds of fishes were more abundant at the edge versus inside the octocoral canopy. Certain fishes are ecotone specialists, being more abundant where sandy bottom meets hard bottom, such as gobies that shelter and nest in reef rubble but forage on invertebrates that live in sand (e.g., Forrester and Steele 2004). Our data indicate that several taxa fall into this group. Gobiids, blennioids, ostraciids, holocentrids, carnivorous labrids, and pomacentrids were 1.3–2.3 times more abundant at the edge of octocoral patches than within the canopy, whereas other demersal fish families, notably herbivores (mainly surgeonfishes and parrotfishes), were twice as abundant within the octocoral canopy than at the edge (Table 3). Similar patterns have been observed at the edges of seagrass meadows, where some fish species are more abundant near the edge, whereas others are more abundant within the seagrass canopy (Smith et al. 2008). These patterns have been linked to predation as the underlying cause for some species (Smith et al. 2011). The cause of different habitat preferences regarding the canopy edge is likely also driven by differential distribution of their prey, as for example, in seagrass meadows, where crustaceans are more abundant at the edges, with polychaetes showing the opposite trend (Tanner 2005).
Conclusions and implications
Overall, our results suggest that communities of arborescent octocorals can provide habitat structure used by demersal fishes on shallow Caribbean reefs, at least where such octocorals are abundant. The results highlight the ecological role that may be played by an increasingly common type of benthic habitat on shallow Caribbean reefs that is characterized by low scleractinian cover. Such areas have been described as gorgonian plains (Mumby 2016), and are common throughout the Caribbean (Williams et al. 2015). Habitats dominated by octocorals can support productive fisheries, and there is at least one documented case of human fishing efforts shifting towards gorgonian habitats as the abundance of reef fishes in Orbicella reefs has declined (Wolf et al. 1999). This trend highlights the merits of targeted conservation of gorgonian plains (Mumby 2016), and we recommend restricting fishing gear that damages these benthic organisms. The data presented here suggest that conservation of gorgonian plains should target maintaining octocoral densities of > 8 colonies m−2 with heights of ≥ 20 cm, which were found to provide habitat structure used by foraging demersal reef fishes.
As macroalgae compete with scleractinians for settlement space, more intense grazing by herbivores within areas dominated by octocorals could facilitate increases in population sizes of scleractinian corals (Mumby and Harborne 2010; Lirman 2001; Nugues and Bak 2006). Shading by octocoral canopies may further reduce algal cover. Together, these considerations raise the question of what role octocorals play in successional processes on Caribbean coral reefs. In the short term, on the scale of few decades, octocorals can proliferate on reefs with declining scleractinian cover by occupying space liberated by bleaching, hurricanes, and diseases that have killed scleractinians (Tsounis and Edmunds 2017). Nevertheless, over longer time scales, octocorals might facilitate a phase shift back towards scleractinian dominance by shading and enhancing herbivory on macroalgae, provided other environmental conditions are appropriate for scleractinians. A hypothesis for future work is therefore that octocorals function as “early succession species” (in terms of succession theory in natural communities, see: Connell and Slatyer 1977), where they would modify the environment so that it becomes less suitable for subsequent recruitment of early successional species such as macroalgae, but more suitable for subsequent recruitment of late successional species such as scleractinians. Given the recent proliferation of shallow water octocorals in Caribbean reefs under environmental stress (Tsounis and Edmunds 2017), future studies testing this hypothesis would improve our understanding of coral reef recovery and succession.
References
Adam TC, Burkepile DE, Ruttenberg BI, Paddack MJ (2015) Herbivory and the resilience of Caribbean coral reefs: knowledge gaps and implications for management. Mar Ecol Prog Ser 520:1–20
Altmann J (1974) Observational study of behavior: sampling methods. Behavior 49:227–266
Alvarez-Filip L, Dulvy NK, Gill JA, Côté IM, Watkinson AR (2009) Flattening of Caribbean coral reefs: region-wide declines in architectural complexity. Proc R Soc B 276:3019–3025
Anderson TW (2001a) Predator responses, prey refuges, and density-dependent mortality of a marine fish. Ecology 82:245–257
Anderson MJ (2001) A new method for non–parametric multivariate analysis of variance. Austral Ecol 26:32–46
Aronson RB, Precht WF (2000) Herbivory and algal dynamics on the coral reef at Discovery Bay, Jamaica. Limnol Oceanogr 45:251–255
Bellwood DR, Hughes TP, Folke C, Nyström M (2004) Confronting the coral reef crisis. Nature 429:827
Brickhill MJ, Lee SY, Connolly RM (2005) Fishes associated with artificial reefs: attributing changes to attraction or production using novel approaches. J Fish Biol 67:53–71
Carvalho S, Cúrdia J, Pereira F, Guerra-García JM, Santos MN, Cunha MR (2014) Biodiversity patterns of epifaunal assemblages associated with the gorgonians Eunicella gazella and Leptogorgia lusitanica in response to host, space and time. J Sea Res 85:37–47
Clarke KR, Ainsworth M (1993) A method of linking multivariate community structure to environmental variables. Mar Ecol Prog Ser 92:205–205
Connell JH, Slatyer RO (1977) Mechanisms of succession in natural communities and their role in community stability and organization. Am Nat 111:1119–1144
Coni EO, Ferreira CM, de Moura RL, Meirelles PM, Kaufman L, Francini-Filho RB (2013) An evaluation of the use of branching fire-corals (Millepora spp.) as refuge by reef fish in the Abrolhos Bank, eastern Brazil. Environ Biol Fishes 96:45–55
Dahlgren CP, Eggleston DB (2000) Ecological processes underlying ontogenetic habitat shifts in a coral reef fish. Ecology 81:2227–2240
Edmunds PJ (2015) A quarter-century demographic analysis of the Caribbean coral, Orbicella annularis, and projections of population size over the next century. Limnol Oceanogr 60:840–855
Endler JA (1981) An overview of the relationships between mimicry and crypsis. Biol J Linn Soc 16:25–31
Fox J (2008) Applied regression analysis and generalized linear models. Sage, London
Fulton CJ, Bellwood DR (2005) Wave-induced water motion and the functional implications for coral reef fish assemblages. Limnol Oceanogr 50:255–264
Green AL, Maypa AP, Almany GR, Rhodes KL, Weeks R, Abesamis RA, Gleason MG, Mumby PJ, White AT (2015) Larval dispersal and movement patterns of coral reef fishes, and implications for marine reserve network design. Biol Rev 90:1215–1247
Heithaus MR, Frid A, Wirsing A, Worm B (2008) Predicting ecological consequences of marine top predator declines. Trends Ecol Evol 23:202–210
Hixon MA, Beets JP (1989) Shelter characteristics and Caribbean fish assemblages: Experiments with artificial reefs. Bull Mar Sci 44:666–680
Holbrook SJ, Schmitt RJ (2002) Competition for shelter space causes density dependence predation mortality in damselfishes. Ecology 83:2855–2868
Holbrook SJ, Forrester GE, Schmitt RJ (2000) Spatial patterns in abundance of a damselfish reflect availability of suitable habitat. Oecologia 122:109–120
Holbrook SJ, Brooks AJ, Schmitt RJ (2002) Variation in structural attributes of patch-forming corals and in patters of abundance of associated fishes. Mar Freshw Res 53:1045–1053
Hughes TP (1994) Catastrophes, phase shifts, and large-scale degradation of a Caribbean coral reef. Science 265:1547–1551
Hughes TP, Rodrigues MJ, Bellwood DR, Ceccarelli D, Hoegh-Guldberg O, McCook L, Moltschaniwskyj N, Pratchett MS, Steneck RS, Willis B (2007) Phase shifts, herbivory, and the resilience of coral reefs to climate change. Curr Biol 17:360–365
Huntington BE, Miller MW, Pausch R, Richter L (2017) Facilitation in Caribbean coral reefs: high densities of staghorn coral foster greater coral condition and reef fish composition. Oecologia 184:247–257
Jackson J (1979) Morphological strategies of sessile animals. In: Larwood G, Rosen B (eds) Biology and systematics of colonial organisms. Academic Press, London, pp 499–555
Jackson JBC (1997) Reefs since Columbus. Coral Reefs 16:S23–S32
Jackson J, Donovan M, Cramer K, Lam V (2014) Status and trends of Caribbean coral reefs: 1970–2012 Global Coral Reef Monitoring Network. IUCN, Gland
Johnson SK, Hallock P (2020) A review of symbiotic gorgonian research in the western Atlantic and Caribbean with recommendations for future work. Coral Reefs Jan 31:1–20.
Jones CJ, Lawton JH, Shachak M (1994) Organisms as ecosystem engineers. Oikos 69:373–386
Jones KM (2007) Distribution of behaviours and species interactions within home range contours in five Caribbean reef fish species (Family Labridae). Environ Biol Fishes 80:35–49
Kramer DL, Chapman MR (1999) Implications of fish home range size and relocation for marine reserve function. Environ Biol Fishes 55:65–79
Krause J, Godin JGJ (1995) Predator preferences for attacking particular prey group sizes: consequences for predator hunting success and prey predation risk. Anim Behav 50:465–473
Legendre P, Legendre L (1998) Numerical Ecology. Elsevier, Amsterdam
Leopold A (1932) Game management. Charles Scribner's Sons, New York
Lirman D (2001) Competition between macroalgae and corals: effects of herbivore exclusion and increased algal biomass on coral survivorship and growth. Coral Reefs 19:392–399
Monaco ME, Friedlander AM, Caldow C, Christensen JD, Rogers C, Beets J, Miller J, Boulon R (2007) Characterising reef fish populations and habitats within and outside the US Virgin Islands Coral Reef National Monument: a lesson in marine protected area design. Fish Manag Ecol 14:33–40
Mumby PJ (2006) The impact of exploiting grazers (Scaridae) on the dynamics of Caribbean coral reefs. Ecol Appl 16:747–769
Mumby PJ (2016) Stratifying herbivore fisheries by habitat to avoid ecosystem overfishing of coral reefs. Fish Fish 17:266–278
Mumby PJ, Harborne AR (1999) Development of a systematic classification scheme of marine habitats to facilitate regional management and mapping of Caribbean coral reefs. Biol Cons 88:155–163
Newman SP, Meesters EH, Dryden CS, Williams SM, Sanchez C, Mumby PJ, Polunin NV (2015) Reef flattening effects on total richness and species responses in the Caribbean. J Anim Ecol 84:1678–1689
Nugues MM, Bak RPM (2006) Differential competitive abilities between Caribbean coral species and a brown alga: a year of experiments and a long-term perspective. Mar Ecol Prog Ser 315:75–86
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Wagner H (2015) Vegan: community ecology package. R package version 2.3–0. https://CRAN.R-project.org/package=vegan
Pandolfi JM, Jackson JBC, Baron N, Bradbury RH, Guzman HM, Hughes TP, Kappel CV, Micheli F, Ogden JC, Possingham HP, Sala E (2005) Are the US coral reefs on the slippery slope to slime? Science 307:1725–1726
Parrish FA, Abernathy K, Marshall GJ, Buhleier BM (2002) Hawaiian monk seals (Monachus schauinslandi) foraging in deep-water coral beds. Mar Mamm Sci 18:244–258
Pitcher TJ, Parrish JK (1993) Functions of shoaling behaviour in teleosts. In: Pitcher TJ (ed) Behaviour of teleost fishes. Chapman & Hall, London, pp 363–439
Plass-Johnson JG, Ferse SC, Jompa J, Wild C, Teichberg M (2015) Fish herbivory as key ecological function in a heavily degraded coral reef system. Limnol Oceanogr 60:1382–1391
Pollux BJA, Verberk WCEP, Dorenbosch M, Cocheret de la Morinière E, Nagelkerken I, van der Velde G (2007) Habitat selection during settlement of three Caribbean coral reef fishes: indications for directed settlement to seagrass beds and mangroves. Limnol Oceanogr 52:903–907
Randall JE, Randall HA (1960) Examples of mimicry and protective resemblance in tropical marine fishes. Bull Mar Sci 10:444–480
R Development Core Team (2008) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3–900051–07–0, https://www.R-project.org
Rizzari JR, Frisch AJ, Hoey AS, McCormick MI (2014) Not worth the risk: apex predators suppress herbivory on coral reefs. Oikos 123:829–836
Roberts CM, Ormond RF (1987) Habitat complexity and coral reef fish diversity and abundance on Red Sea fringing reefs. Mar Ecol Prog Ser 41:1–8
Ruzicka RR, Colella MA, Porter JW (2013) Temporal changes in benthic assemblages of Florida Keys reefs 11 years after the 1997/1998 El Niño. Mar Ecol Prog Ser 489:125–141
Smith TM, Hindell JS, Jenkins GP, Connolly RM (2008) Edge effects on fish associated with seagrass and sand patches. Mar Ecol Prog Ser 359:203–213
Smith TM, Hindell JS, Jenkins GP, Connolly RM, Keough MJ (2011) Edge effects in patchy seagrass landscapes: the role of predation in determining fish distribution. J Exp Mar Bio Ecol 399:8–16
Tanner JE (2005) Edge effects on fauna in fragmented seagrass meadows. Austral Ecol 30:210–218
Thresher RE, Gunn JS (1986) Comparative analysis of visual census techniques for highly mobile, reef-associated piscivores (Carangidae). Environ Biol Fishes 17:93–116
Tsounis G, Edmunds PJ (2017) Three decades of coral reef community dynamics in St. John, USVI: a contrast of scleractinians and octocorals. Ecosphere 8(1):e01646
Tsounis G, Edmunds PJ, Bramanti L, Gambrel B, Lasker HR (2018) Variability of size structure and species composition in Caribbean octocoral communities under contrasting environmental conditions. Mar Biol 165:29
Walter RP, Haynes JM (2006) Fish and coral community structure are related on shallow water patch reefs near San Salvador, Bahamas. Bull Mar Sci 79:365–374
Ward-Paige C, Flemming JM, Lotze HK (2010) Overestimating fish counts by non-instantaneous visual censuses: consequences for population and community descriptions. PLoS ONE 5:e11722
Wild C, Hoegh-Guldberg O, Naumann MS, Colombo-Pallotta MF, Ateweberhan M, Fitt WK, Iglesias-Prieto R, Palmer C, Bythell JC, Ortiz JC, Yossi L, van Woesik R (2011) Climate change impedes scleractinian corals as primary reef ecosystem engineers. Mar Freshwater Research 62:205–215
Williams ID, Polunin NV, Hendrick VJ (2001) Limits to grazing by herbivorous fishes and the impact of low coral cover on macroalgal abundance on a coral reef in Belize. Mar Ecol Prog Ser 222:187–196
Williams SM, Mumby PJ, Chollett I, Cortes J (2015) Importance of differentiating Orbicella reefs from gorgonian plains for ecological assessments of Caribbean reefs. Mar Ecol Prog Ser 530:93–101
Wolff N, Grober-Dunsmore R, Rogers CS, Beets J (1999) Management implications of fish trap effectiveness in adjacent coral reef and gorgonian habitats. Environ Biol Fishes 55:81–90
Acknowledgements
This research was funded through the US National Science Foundation through their programs in Long-Term Research in Environmental Biology (DEB 08–41441 and DEB 13–50146) and Biological Oceanography (OCE 13–32915), and was completed under a permit issued by the VI National Park (VIIS-2011-SCI-0016). We are grateful to H. Lasker and A. Martinez for octocoral community data at East Tektite. Special thanks to S. Prosterman for local support, and the staff of the Virgin Islands Environmental Resource Station for making our visits productive and enjoyable. This is contribution number 309 of the Marine Biology Program of California State University, Northridge.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Topic Editor Andrew Hoey
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tsounis, G., Steele, M.A. & Edmunds, P.J. Elevated feeding rates of fishes within octocoral canopies on Caribbean reefs. Coral Reefs 39, 1299–1311 (2020). https://doi.org/10.1007/s00338-020-01963-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-020-01963-1