Abstract
While links between heat stress and coral bleaching are clear and predictive tools for bleaching risk are well advanced, links between heat stress and outbreaks of coral diseases are less well understood. In this study, the effects of accumulated heat stress on tagged colonies of tabular Acropora were monitored over the 2017 austral summer at Beaver Reef, which is located in the central region of the Great Barrier Reef. The initial surveys in midsummer (21 February) coincided with an accumulated heat stress metric of 4.5 °C-weeks, and documented high coral cover (74.0 ± 6.5%), extensive bleaching (71% of all corals displayed bleaching signs) and an outbreak of white syndromes (WSs) (31% of tabular acroporid corals displayed white syndrome signs). Repeat assessments of the impacts of bleaching and disease on these corals provided real-time information to reef managers by tracking the unfolding reef health incident on 100 colonies of Acropora hyacinthus (Dana, 1846), tagged in mid-March and surveyed intermittently until late October 2017. Heat stress increased rapidly on Beaver Reef, peaking at 8.3 °C-weeks on 31 March, which coincided with the highest prevalence of WS recorded in the study. Of the 85 tagged colonies surviving on 31 March, 41 (~ 48%) displayed WS signs, indicating a link between heat stress and WS. When re-surveyed at eight months (24 October), 68 of 100 tagged colonies had suffered whole-colony mortality and only four colonies had not displayed signs of bleaching or disease (WS) in any of our surveys. Overall, coral cover on Beaver Reef was reduced by more than half to 31.0 ± 11.2%. Significant tissue loss due to severe bleaching was observed with up to 20 times greater tissue loss on severely bleached colonies (i.e. categorised as > 50% bleached) compared to mildly/moderately bleached colonies (< 50% bleached) at the heat stress peak (31 March). This suggests that for Acropora hyacinthus, a threshold of 50% colony bleaching is a good indicator that substantial mortality at both the colony and population level is likely to follow a heat stress event. Across all levels of bleaching, colonies displaying WS signs exhibited up to seven times greater tissue loss than bleached-only colonies. WS caused a threefold increase in accumulated tissue loss (69.6 ± 10.5% tissue lost) in the mildly bleached category, suggesting that disease exacerbated mortality in bleached corals and contributed significantly to the substantial loss of corals on the GBR in 2017.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Unprecedented back-to-back thermal anomalies in the Australian summers of 2016 and 2017 resulted in mass coral bleaching affecting two-thirds of the Great Barrier Reef (GBR) and caused extensive reductions in coral cover across the northern and central regions (GBRMPA 2017a; Hughes et al. 2018b; Sweatman 2018). A shift in community composition was subsequently observed during detailed in situ monitoring in 2016 (Hughes et al. 2018b), as high inter-specific variation in bleaching resistance caused disproportionately higher mortality of heat-sensitive species (cf. Marshall and Baird 2000; Baker et al. 2008). The potential contributions that disease might have made to such coral community shifts are not well understood, and consequently, this source of mortality has largely been ignored in studies of large-scale heat stress events on the GBR. Heat stress from current and projected increases in sea surface temperatures (SST) is generally identified as the primary threat to coral reefs over the next century (Hoegh-Guldberg 1999; van Hooidonk et al. 2016; Hughes et al. 2017a, b) and is also predicted to increase disease occurrences in many cases (Selig et al. 2006; Maynard et al. 2015). Given the likelihood that reefs will be exposed to more frequent and severe bleaching events (van Hooidonk et al. 2016; Hughes et al. 2017b, 2018a) and disease outbreaks (Maynard et al. 2015), disentangling the effects of bleaching and thermally induced disease outbreaks on coral communities is becoming increasingly important.
In addition to bleaching, warm thermal anomalies have been linked to a number of coral diseases that can result in partial or whole-colony mortality and ultimately reduced abundance at the population level (Green and Bruckner 2000; Willis et al. 2004; Work et al. 2012; Peters 2015). Although increasing reports of coral disease outbreaks on the GBR and in the Caribbean have been linked to a range of environmental stressors, anomalously high seawater temperatures have been identified as a major driver (Selig et al. 2006; Bruno et al. 2007; Harvell et al. 2007; Sweatman et al. 2008; Heron et al. 2010; Randall et al. 2014). On the GBR, white syndromes (WSs) are a prevalent disease affecting a broad range of coral species, particularly in conjunction with heat stress events (Willis et al. 2004; Hobbs et al. 2015).
WSs are characterised by a narrow white band, representing recently exposed white skeleton that advances as a regular front across the coral colony as tissue at the lesion front undergoes necrosis (Bourne et al. 2015). WS outbreaks can have devastating effects, e.g. 36% of Acropora spp. colonies suffered total mortality at Christmas Island in 2008 (Hobbs et al. 2015). Mortality occurred in the absence of thermal stress and signs of bleaching and caused coral cover on Christmas Island to decline from 7.0 to 0.8% over an 8-month period (Hobbs et al. 2015). Coral cover, an indicator of host density, also affects WS abundance and, in combination with sea surface temperature, has been used as a co-predictor for disease outbreak modelling (Bruno et al. 2007; Heron et al. 2010; Maynard et al. 2011). There are suggestions that the role of diseases in causing mortality on reefs under future scenarios of elevated seawater temperatures has been underestimated (Miller et al. 2009; Maynard et al. 2015), highlighting the need for more detailed studies of the links between heat stress, disease and coral mortality.
The co-occurrence of coral disease and bleaching following accumulated heat stress has been noted in a number of studies (Selig et al. 2006; Brandt and McManus 2009; Miller et al. 2009). For example, the above-average seawater temperatures on the GBR in 2002–2003 triggered mass bleaching and WS outbreaks, with a 20-fold increase in WS abundance on outer-shelf reefs in the northern region (Willis et al. 2004; Sweatman et al. 2008). A study of the additive impacts of bleaching and disease on coral populations in the USA Virgin Islands in 2005 found a 13-fold increase in mortality associated with white plague disease when co-occurring with bleaching versus when bleaching was absent (Miller et al. 2009). At other Caribbean sites, the additive effect of bleaching and disease on coral resulted in 50% mortality at some sites during and after a major heat stress event in 2005 (Eakin et al. 2010; see also Miller et al. 2009). In another example, a recent study in Florida recorded reductions in coral colony abundance of more than 97% in several species as a result of both mass bleaching and diseases following a oceanic heatwave in 2014–2015 (Precht et al. 2016). Given the potential for bleaching and disease events to reduce the diversity and productivity of coral assemblages on reefs (Hughes et al. 2017a; Neal et al. 2017), it becomes increasingly important to validate the relationship between these factors (Miller et al. 2009; Heron et al. 2010; Precht et al. 2016). However, empirical evidence for such links at the population level is lacking (Ban et al. 2013). Documenting in situ interactive effects of bleaching and disease on corals at the population and colony level is important for understanding the high mortality often observed on reefs during and following marine heatwaves (Brandt and McManus 2009; Precht et al. 2016; Hughes et al. 2018a).
This study investigated the impact of the 2017 heat stress event on populations of tabular species of Acropora by monitoring 100 tagged colonies of A. hyacinthus on Beaver Reef, in the central region of the GBR, during and subsequent to a combined mass bleaching event and WS outbreak. Documentation of the cumulative impacts of bleaching and WSs on this population and the timing of these impacts in relation to summer peaks in SSTs provide insights into disease-bleaching dynamics at both a population and colony level. Our study also provides important estimates rates of lesion progression and tissue loss associated with WS on bleached versus unbleached corals and highlights how thermal stress accelerates tissue loss associated with WSs. Outcomes from our study improve our capacity to predict the impacts of future heat stress events on coral populations.
Methods
Study site and survey protocols
This study was conducted in partnership with the Great Barrier Reef Marine Park Authority (GBRMPA) as part of the Reef Health Incident Response System (GBRMPA 2013), following numerous reports of coral disease in the Mission Beach area of the central GBR region submitted in December 2016 via the Eye on the Reef program. Prior to 2016, no evidence of bleaching or disease was found in occasional surveys of coral health at Beaver Reef by the AIMS long-term monitoring program (Sweatman et al. 2008). In addition, there are no records of bleaching or disease at this site in the GBRMPA Eye on the Reef Program prior to the 2016–2017 summer (pers. comm. J. Stella). By late January 2017, bleaching was occurring on reefs in the central and northern sections of the GBR and the number of reports of disease and the severity of their impacts had been increasing on reefs throughout the region, but particularly on reefs off Mission Beach. These reports were corroborated on reefs around Mission Beach, including Beaver Reef (17°50′49″S 146°29′53″E; Fig. 1), in late February by the GBRMPA employing reef health and impact surveys (RHIS) (GBRMPA 2017b) and by an independent disease expert (BLW). Total live coral cover, bleaching extent and observations of any other reef health impacts, such as predation, were recorded by RHISs (protocol detailed in Beeden et al. 2014). In this initial survey, disease and bleaching prevalence were estimated using a rapid snorkel survey, whereby the presence of bleaching, disease or normal pigmentation was recorded for all corals within an approximately 2 m belt directly under the surveyor on a 20-min swim. This included tabular species of Acropora (n = 125 colonies), which were the dominant group of corals on these reefs. The WS outbreak confirmed on Beaver Reef on 21 February constituted a unique opportunity to study the interactive effects of WS and bleaching.
Subsequent coral cover surveys were undertaken by triplicate 15 m line intercept transects (LIT) on 27 April and 24 October 2017 (as in Hill & Wilkinson 2004). In order to inform management decisions and guide further survey efforts, real-time reef health information was provided to GBRMPA after each survey. Impacts of bleaching and disease were communicated to the public via the GBRMPA website (http://www.gbrmpa.gov.au/about-the-reef/reef-health/timeline-and-actions).
Monitoring coral health on tagged colonies
The tabular coral Acropora hyacinthus was selected as the target species for a tagging program at Beaver Reef, based on the high prevalence of WSs affecting this species in the pilot study and the known susceptibility of this species to both bleaching and disease (Marshall and Baird 2000; Willis et al. 2004; Harvell et al. 2007; Hobbs et al. 2015). On 16 March, 100 colonies of A. hyacinthus (1–4 m depth) were tagged with numbered plastic cattle tags and their locations recorded on an underwater map for ease of relocation in subsequent surveys. The status of each tagged colony was recorded at the onset of this study (16 March 2017) and again at each of four subsequent time points: twice during the heat stress event (31 March and 27 April) and twice at later time points to follow their longer term fate (17 June, 24 October 2017). At each time point, the bleaching state of each colony was visually assessed and scored (Fig. 2) in one of three categories (modified from Baird and Marshall 2002): normal pigmentation (no signs of bleaching), moderately bleached (1–50% of the colony bleached) and severely bleached (51–100% of the colony bleached). The presence of WS signs was recorded in each category and distinguished from bleaching by the presence of tissue loss and a distinct lesion front exposing a band of white skeleton, which was verified in close-up photographs (Bourne et al. 2015).
Examples of bleaching categories recorded on tagged colonies of Acropora hyacinthus. a Normal pigmentation, b mildly-to-moderately bleached: 1–50% bleached, and c severely bleached: 51–100% bleached. White Syndrome (WSs) on colonies with: d normal pigmentation, and e moderate bleaching. f Close-up of a WS lesion showing white skeleton devoid of polyps. Green areas on d, e and f behind the WS front are algal overgrowth on skeleton recently exposed following tissue loss
All colonies were tabular and photographed (Canon G16) parallel to the plane of the colony with a 10-cm scale placed in the colony plane. Colonies were photographed at the five time points, except in cases where colonies had been dislodged and could not be located. The total surface area of live tissue (cm2) on each colony was calculated from photographs using ImageJ (version 1.48), calibrated with the 10-cm scale bar in each photo. At each survey time point (starting 31 March), partial mortality was estimated as the area that had died since the previous survey, as indicated by the presence of recently exposed white skeleton or non-eroded skeleton covered by a light fouling community. The partial mortality metric was standardised to the area of living tissue measured in the prior survey to account for difference in colony size. Hence, the metric represents the percent of tissue lost in the interval between two surveys and is not representative of accumulated tissue loss over the whole study period. Because partial mortality is most likely not linear, rate of tissue loss would not be ecologically representative of mortality dynamics. For example, partial mortality in the interval between 16 and 31 March on the colony illustrated in Fig. 2d was calculated as follows: the area of recent tissue loss [i.e. white area representing recently exposed skeleton plus light green area representing skeleton recently overgrown by algae in the 31 March image (outlined in red)], was divided by the total area of tissue alive on 16 March (areas of normally pigmented brown tissue, plus areas of recently exposed white skeleton and light green algal overgrowth in the 31 March image (outlined in yellow)). At time 0 (16 March), tissue loss was estimated from the 16 March photograph based on this interpretation of skeleton appearance. Areas of old mortality (old dead) were distinguished by deteriorating skeleton overgrown by a more mature, grey or dark green fouling community and excluded from colony area calculations. Bleached tissue was included in the live tissue category and readily distinguished from areas of bare white skeleton representing tissue loss. In summary, 50% partial mortality represents loss of half of the live tissue on a colony in the interval from one survey time point to the next survey time point. If whole-colony mortality occurred between surveys, the colony was classed in the same bleaching category as in the survey immediately prior.
Following the conclusion of the heat stress event and immediately prior to the predicted mass spawning, the remaining live colonies within the tagged population (n = 32) were sampled to assess their fertility on 24 October 2017. A small fragment within the fertile zone (greater than 2 cm from the tip of a branchlet) was collected from each tagged colony, placed in a numbered plastic bag underwater, fixed in 10% formalin seawater immediately upon surfacing and then transferred to 70% ethanol in the laboratory. Fragments were decalcified in 3% formic acid. Five decalcified polyps were haphazardly selected from each fragment, dissected under a stereo microscope (Olympus CX31RBSF), and the reproductive status of the colony was characterised by either the presence or absence of eggs and sperm.
Accumulated heat stress at Beaver Reef
Satellite-derived sea surface temperatures (SST) from the National Oceanic and Atmospheric Administration (NOAA) Coral Reef Watch (CRW) program’s CoralTemp data product (www.coralreefwatch.noaa.gov/satellite/coraltemp.php) were used to calculate accumulated heat stress for the period 1 January 2016 to 31 December 2017. Heat stress was evaluated as Degree Heating Weeks (DHW), an established predictor for coral bleaching, for which thresholds of 4 and 8 °C-weeks are associated with significant bleaching and mortality, respectively (Eakin et al. 2010; Liu et al. 2013, 2014; Heron et al. 2016a). The DHW metrics were calculated using a SST climatology calculated from an initial release of CoralTemp, as described in Liu et al. (2017).
Statistical analysis
To determine the effects of bleaching and WS on tissue loss, we used a linear-mixed effects model, fit by restricted maximum likelihood (REML), with WS (presence/absence) and bleaching categories (normally pigmented, mild and severe) as interactive fixed effects, and colony as a random factor to account for repeated measures. Therefore, the model explains survey-specific partial mortality as a function of bleaching category and the presence/absence of WS, while the temporal component was integrated through the repeat measure for each colony. All analyses were performed in R (R Core Team 2017) using the nlme package (Pinheiro et al. 2017). Assumptions were checked, and the best model of several iterations was selected based on Akaike information criterion, adjusted for small sample size. Additionally, total accumulated tissue loss (as a percentage of the original colony size) over the course of the survey was compared with colonies grouped into maximum observed bleaching and disease severity. Natural pigmentation with WS was excluded as a category due to insufficient sample size (n < 3). Maximum severity groups were compared individually by the Wilcoxon rank sum test as the accumulated tissue loss data were heteroscedastic and could not be alleviated by transformation. Results are reported as mean ± SE.
Results
Heat stress exposure
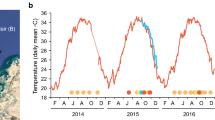
Sea surface temperatures (SST) in 2016 and 2017 indicated corals at Beaver Reef experienced heat stress in both years, although accumulated heat stress (DHW) in 2016 was below a level at which significant bleaching would be expected, peaking at 1.9 °C-week (Fig. 3). In contrast, heat stress in 2017 was more than fourfold greater, peaking at 8.3 °C-week on 31 March, a level at which bleaching and mortality are expected. The 2017 heat stress began in early January, nearly 6 weeks earlier than the 2016 heat stress onset, and peaked 4 weeks later than the timing of the 2016 peak. The winter months between these events were exceptionally warm; July 2016 (mean = 25.23 °C) was, on average, 2 °C above the mean climatological temperature for July (23.16 °C).
Satellite-derived sea surface temperatures (SST) at Beaver Reef (5 km resolution) (purple line) prior to and throughout survey dates. Heat stress is measured as Degree Heating Weeks (DHW, red) and accumulated for SST at or above the bleaching threshold (solid blue line), which is 1 °C greater than the maximum of the monthly mean SST climatology (dashed blue line). Climatological monthly means (blue crosses) indicate that the recorded SSTs are above climatological averages throughout the period. DHW peaked at 8.3 °C-weeks on 31 March 2017. Grey lines indicate survey dates
Changes in coral cover
At the beginning of the monitoring period (21 February), overall coral cover was 74.0 ± 6.5%, with a high proportion of corals surveyed already bleached (71.0 ± 8.7%; Fig. 4), along with signs of recent whole-colony mortality and recent partial colony mortality. Following the peak in heat stress (31 March), the survey on 27 April revealed coral cover had dropped to 43.1 ± 3.9%, with 55.0 ± 9.1% of surviving corals bleached. Notably, the overall percentage of normally pigmented colonies did not change substantially between these surveys. Six months after the climax of the bleaching event, when heat stress was no longer present (24 October), coral cover had dropped further to 31.0 ± 11.2%. No bleaching was observed at this time. The higher variance in coral cover recorded in the October survey reflected the patchy distribution of the surviving corals.
Patterns in the percentage of live coral cover over 8 months on Beaver Reef, established by GBRMPA’s reef health and impact surveys during the initial reef survey (21 February) and subsequently by line intersect transect surveys (17 April, 24 October). Bleached category includes any degree of bleaching observed, and error bars are SE of the total cover
Population-level response to heat stress
The initial survey at Beaver Reef (21 February) found that 32% of tabular acroporids were bleached (n = 125 colonies surveyed), 31% (i.e. 39 colonies) had characteristic WS lesions, and 19% (i.e. 24 colonies) experienced both bleaching and WS. Other reef impacts, such as crown-of-thorns starfish predation, were not observed in RHIS surveys. At the first survey of tagged colonies (16 March; DHW = 6.2 °C-weeks), 15 of the 100 tagged, tabular acroporid colonies had normal pigmentation with no WS signs (Fig. 5). Of the 100 tagged colonies, 81 were bleached and most of these (55 colonies or 68%) were categorised as severely bleached (i.e. 51–100% bleached). Disease lesions characteristic of WSs was prevalent within the population, with 38 colonies with active lesions, 28 of which were in the highest bleaching category. Two weeks later at the DHW peak (31 March, 8.3 °C-weeks), only five colonies were visually healthy. Fifteen colonies had suffered complete mortality, and most of the remaining live colonies were severely bleached (i.e. 51 colonies), with more than half of these (34 colonies) also having WS lesions. On 31 March, WS prevalence was the highest observed, affecting 41 of the 85 surviving colonies (48%). On 27 April, after the DHW peak, 53 colonies had died, and 40 of the 47 surviving colonies still displayed bleaching signs. WS signs were also observed on 24 colonies, with most again in the highest bleaching category (17 of 23 severely bleached colonies). Of the 17 moderately bleached colonies (i.e. 1–50% bleached), only four had signs of WSs. By 17 June, only five of the surviving 35 colonies still displayed moderate bleaching signs, while the remaining survivors had regained normal pigmentation (i.e. 30 colonies). WSs were noted in only two colonies at this time: one moderately bleached and one normally pigmented. At the time of the final survey (24 October), a further three colonies had suffered whole-colony mortality, but the remaining surviving colonies (32) were observed to have regained their pigmentation; only one colony had signs of WS.
Number of live colonies on five survey dates at Beaver Reef in each of three colony colour categories: normal pigmentation (no bleaching signs), mildly-to-moderately bleached (1–50% of colony bleached) and severely bleached (51–100% of colony bleached). The fraction of colonies displaying white syndrome signs in each bleaching category is displayed in white. Dead colonies (cross-hatched) are the cumulative number of dead colonies through time
Of the 100 tagged colonies, 68 died during the survey period. Of the surviving 32 colonies at the end of the study (24 October), 13 had substantial (> 30% tissue loss) partial mortality (i.e. 66.7 ± 6.6% tissue loss on average), 16 had low-to-moderate partial mortality (< 30% tissue loss), losing, on average, 11.1 ± 1.8% of their tissues, and three colonies had no tissue loss. Whole-colony mortality was observed for corals that displayed bleaching signs in each category, although highest mortality occurred for colonies that had been severely bleached. For example, 15 severely bleached colonies tagged on 16 March died within 2 weeks. Despite thermal stress peaking at the end of March, coral health continued to decline, with a further 38 colonies (32 of which had been severely bleached on 31 March) suffering complete mortality in the following months. From 27 April to 17 June, whole-colony mortality continued, with 12 of the surviving 47 colonies dying. A further three colonies died between 17 June and 24 October. WS lesions had been present on the majority (60–84%) of colonies that died in each survey interval, except the final interval. When WSs had been present, mortality was greater, with up to five times more colonies dying when WS was present versus absent in the first three survey intervals when heat stress was present (16 March to 17 June).
Colony-level response to heat stress
Partial mortality on colonies in the tagged population of acroporids differed widely across the three bleaching categories (normally pigmented, moderately bleached and severely bleached). However, the highest percentage tissue loss consistently occurred on severely bleached colonies that were also affected by WSs (Fig. 6). On 16 March, mean area of tissue loss on colonies that were both bleached and diseased was 4–7 times greater than on colonies in the same bleaching categories without WS (i.e. 19.4 ± 5.4% versus 2.7 ± 1.6% for moderately corals with versus without WS signs; 27.8 ± 4.5% versus 6.9 ± 2.7% partial mortality for severely bleached corals). At the peak of the heat stress (31 March), mean tissue loss on severely bleached corals with WSs reached 61.9 ± 5.0% and represented recent partial mortality on more than half of the live colonies in that category. In comparison, mean tissue loss on severely bleached colonies with no disease signs was substantially lower (46.6 ± 10.2%). In the absence of WSs, tissue loss on severely bleached corals was 20-fold greater than the minor tissue loss recorded on normally pigmented and moderately bleached colonies (0.6 ± 0.6% and 1.9 ± 0.5%, respectively). On 27 April, tissue loss on tagged corals had increased further, with severely bleached colonies with and without WSs averaging 83.2 ± 4.5% and 58.5 ± 13.3% tissue loss, respectively. On 17 June, well past the DHW peak, further tissue regression was observed in all bleaching categories; however, only a few colonies remained bleached (i.e. five colonies, all moderately bleached). At the end of the survey period, accumulated tissue loss was highest in the severely bleached category with WS, indicating that the cumulative effects of disease and bleaching have the highest impact on the population. However, accumulated tissue loss was not statistically different in severely bleached corals without WS, suggesting that for the most severe forms of bleaching, mortality is likely either with or without the presence of WSs. In contrast, amongst colonies with mild bleaching, the added presence of WS exacerbated total tissue loss; in the absence of disease, mildly bleached colonies sustained lower partial mortality. Increased mortality amongst mildly bleached colonies with WS suggests that at low levels of bleaching, colonies may have the capacity to endure the event in the absence of WSs, but loss of photosymbionts increases their disease susceptibility. The further partial mortality recorded on 17 June indicated knock-on effects of heat stress and ongoing declines in coral health well past the period of heat stress. Of the moderately bleached colonies alive on 17 June, the two with WS experienced 100% tissue loss, while the three without disease had ~ 60% lower tissue loss (39.3 ± 15.2%). On the last survey (24 October), all 32 colonies were normally pigmented, of which only one had WS lesions. Mean tissue loss on the normally pigmented colonies without WSs was 17.3 ± 4.9%, suggesting lingering mortality post-heat stress. Finally, the proportion of gravid colonies in the surviving population (approximately 80%; n = 32 colonies) immediately prior to spawning was lower than proportions found in a study on A. hyacinthus reproduction during non-bleaching years (Baird and Marshall 2002). The same study documented fewer gravid colonies (45%) after bleaching. All six of the non-gravid colonies had suffered some degree of bleaching. In contrast, all colonies that sustained normal pigmentation throughout the study were gravid.
Tissue loss (partial mortality) on tagged colonies of Acropora hyacinthus in three colony bleaching categories, each with and without white syndromes (WSs), on the five survey dates. Partial mortality is the proportion of live tissue that had recently died. At the last survey date, only normal pigmentation (no bleaching) with and without WSs was observed. When n = 1, no error bars were plotted, i.e. for normally pigmented colonies with WS on 27 April and 24 October, and for severely bleached colonies without WSs on 17 June. Error bars are SE
Over the course of the study, total accumulated tissue loss was highest in the severely bleached category when WSs were present (97.1 ± 1.6%), although it did not differ significantly from tissue loss on severely bleached colonies without WSs (85.4 ± 7.1%) (Wilcoxon ranked: W = 377.5, p = 0.052). Tissue loss was greater on severely bleached corals than on mildly bleached corals (19.3 ± 8.2%; W = 17.5, p < 0.001), while the ‘mild bleaching with WS’ category had threefold higher accumulated tissue loss (69.6 ± 10.5%) than mild bleaching only corals (W = 20.5, p = 0.003). Finally, tissue loss was similar on mildly bleached and normally pigmented corals (5.4 ± 2.6%) (W = 33, p = 0.46).
The linear-mixed effects model applied to tease apart the synergistic effects of bleaching and WSs confirmed that patterns in tissue loss on colonies with and without WSs were consistent for the two bleaching categories, i.e. no interactive effects detected between the presence of WSs and either moderate bleaching (t = 0.842, df = 355, p = 0.400) or severe bleaching (t = 0.311, df = 355, p = 0756). Overall, the presence of WSs and severe bleaching each significantly increased tissue loss (t = 2.281, df = 355, p = 0.0231; and t = 5.681, df = 355, p < 0.001, respectively). In contrast, the presence of moderate bleaching did not affect patterns in tissue loss significantly. The model explained 50.9% of the variance in the data (conditional r-squared).
Discussion
Impact of the marine heatwave
Our study confirms that the presence of severe bleaching combined with an outbreak of WSs caused extensive mortality of tabular acroporid corals at Beaver Reef in the summer of 2017. The 71.0 ± 8.7% of tabular acroporids found to be bleached on 21 February provides corroborative evidence that an accumulated heat stress metric of 4.5 °C-week exceeds the threshold for bleaching, and the subsequent mortality documented in the following 10 weeks (peak heat stress of 8.3 °C-week) similarly supports the established mortality threshold (Heron et al. 2016b; Hughes et al. 2018b). The presence of accompanying disease signs, particularly on severely bleached colonies, highlights the reduced disease resistance of corals during warm thermal anomalies. It is likely that similar disease outbreaks contributed to coral mortality in the central and northern regions of the Great Barrier Reef, where heat stress was generally more severe in the summer of 2017 (Hughes et al. 2018b; Sweatman 2018).
Our finding that 81% of colonies (n = 100) were bleached in our first survey of tagged Acropora on 16 March is testament to the severity of the 2017 thermal stress event at Beaver Reef, as well as at other sites in the northern and central regions of the GBR (Hughes et al. 2018b; Sweatman 2018). At the peak of the DHW metric (8.3 °C-week on 31 March), severely bleached colonies suffered 20-fold greater levels of tissue loss than colonies assessed as moderately bleached (1–50%), highlighting the large difference in mortality levels sustained by corals in the two bleaching categories. The significance of this difference in partial and whole-colony mortality for corals above and below a 50% visual bleaching severity threshold was supported by the mixed effect model, which confirmed a significant (p < 0.01) association between tissue loss and severely bleached corals, but not with moderately bleached corals. Collectively, this suggests that a 50% colony bleaching threshold is a useful indicator for predicting extensive bleaching- and disease-associated mortality.
Impacts to coral health after exposure to high heat stress can take weeks to months (and longer) to fully unfold, often long after water temperatures return to normal (Baker et al. 2008; Miller et al. 2009). This study documented progressive tissue loss in all colonies with bleaching and WSs well after the DHW peak, indicating that health impacts continue despite SSTs returning to levels below thermal thresholds. Peaks in tissue loss on both moderately and severely bleached colonies (39.3 ± 15.2% and 100%, respectively) approximately 4–6 weeks after the DHW peak indicate a lag effect in the final extent of partial and whole-colony mortality following a thermal stress event. It is likely that depleted energy reserves following reductions in Symbiodiniaceae communities affected both disease resistance (Maynard et al. 2015; Muller et al. 2018) and the capacity of corals to maintain tissue integrity and repair tissue loss in response to daily interactions, such as predation (cf. Shaver et al. 2018) and competition. This lingering impact on colonies unable to respond swiftly to favourable water temperature changes because of depleted energy reserves (Marshall and Baird 2000; Anthony et al. 2007) is likely to cause ongoing partial mortality in most coral species following severe bleaching. Prolonged impacts of bleaching on coral health were also highlighted in a Caribbean study, in which bleaching persisted in populations despite temperatures returning to levels below thermal threshold maxima (Miller et al. 2009). The study also reported high levels of mortality and increased disease susceptibility post-heat stress, even though colonies regained their pigmentation and SSTs had decreased (Miller et al. 2009; Muller et al. 2018). Similarly, surviving colonies at Beaver Reef that had returned to normal visual appearance, associated with an increase in Symbiodiniaceae populations within coral tissues (Muller-Parker et al. 2015), still sustained further partial mortality. This highlights the vulnerability of corals following heat stress events, with ongoing partial mortality of compromised colonies compounded by sources other than bleaching. This was observed in our study as the total tissue loss observed increased threefold in mildly bleached colonies with WS, which otherwise would potentially have had the energy reserves to fully recover (Anthony et al. 2009). While heat stress events cannot be locally managed, intervention to reduce additional stressors (e.g. through culling of predatory crown-of-thorns starfish) could increase coral survival following these events, and reductions in sources of chronic stress, such as nutrient loading or turbidity, could support coral recovery (Hughes et al. 2010; MacNeil et al. 2019).
The four colonies that retained their normal pigmentation throughout the study probably had greater resistance to heat stress, maintaining symbioses with their Symbiodiniaceae communities during the entire period of anomalous heat. Intraspecific differences in survival during bleaching events were also observed for species of Acropora on the GBR in 2002 (Jones 2008). As all colonies in our study were located at similar depths and exposure aspects, the increased resistance of these four colonies was likely not due to shelter from additional stressors such as solar irradiance or water current (McClanahan et al. 2005; Anthony et al. 2007), or potentially other microenvironmental factors that may have ameliorated impacts of the heat stress (e.g. Page et al. 2019). Possibly, these colonies had higher energy reserves, enabling them to sustain symbiosis through the heat stress event (Wooldridge 2014). Alternatively, they may have hosted a more temperature tolerant Symbiodiniaceae clade, which potentially could have increased their thermal resistance by up to 1–1.5 °C (Berkelmans and van Oppen 2006). These heat tolerant colonies constitute a selective brood stock for stress tolerant genotypes, crucial for restoration efforts in warmer oceans forecast for the near future (Heron et al. 2016b; van Oppen et al. 2017).
In October, 6 months after the heat stress event, none of the surviving colonies had re-grown tissue over their denuded skeletons, described as the “Phoenix effect” in a study of Porites—an effect that can substantially facilitate reef recovery after mass bleaching events (Roff et al. 2014; Holbrook et al. 2018). It is possible that either the duration of our study was too short to detect re-growth or A. hyacinthus does not recover by re-growing over old skeleton and instead prioritises new growth (Roff et al. 2014).
The cumulative impacts of bleaching and white syndromes
The presence of WSs significantly exacerbated the impact of bleaching. On average, corals with WS signs in addition to bleaching-related stress suffered four to sevenfold greater tissue loss than colonies in the same bleaching categories without WSs. As an example of the severity of the cumulative effects of WSs and bleaching, moderately bleached colonies had more than 60% greater tissue loss when WS signs were present versus absent by the end of the heat stress event (based on 17 June survey). Additionally, WS signs occurred on the majority of colonies that subsequently suffered complete mortality. Overall, whole-colony mortality was up to fivefold greater by the June survey for corals displaying combined bleaching and WSs than for those that were bleached but not diseased. Similarly, a mass bleaching event followed by a disease outbreak resulted in a 60% reduction in coral cover in the Caribbean in 2005 (Miller et al. 2009). Subsequently, an outbreak of white plague disease following the 2015 mass bleaching event further reduced populations of some coral species to less than 3% of their prior density (Precht et al. 2016). Corals that had bleached were more susceptible to disease (Brandt and McManus 2009; Muller et al. 2018), and thus, bleaching was argued to be the precursor for the 2015 disease outbreak in the Caribbean (Precht et al. 2016). Although disease was observed prior to the extensive bleaching on Beaver Reef, bleaching most likely further reduced disease resistance in the tagged population of A. hyacinthus. Our finding that WSs significantly exacerbated tissue loss, increasing accumulated tissue loss by threefold in mildly bleached colonies (to 69.6 ± 10.5% loss), underlines the devastating cumulative effects of bleaching and disease and highlights the importance of including disease impacts when surveying bleaching, as the co-occurrence of disease may be an important factor in predicting mortality following mass bleaching events. Although WSs were observed on moderately bleached colonies in our study, they were most prevalent on severely bleached colonies, further highlighting the role that compromised health (i.e. bleaching) plays in lowering resistance to disease (Maynard et al. 2015). WS prevalence was at its maximum (41 of 85 colonies) at the heat stress peak (DHW = 8.3 °C-weeks) on 31 March, supporting previous reports of WS prevalence being temperature driven (Selig et al. 2006; Bruno et al. 2007; Harvell et al. 2007; Heron et al. 2010).
The drivers of WS outbreaks are likely several. Bruno et al. (2007) linked increased disease prevalence with higher sea surface temperatures; a conclusion that was further supported in modelling studies by Heron et al. (2010) and Maynard et al. (2011). However, the occurrence of bleaching, on its own, has been suggested to be a poor predictor of WS outbreaks (Ban et al. 2013). It is possible that warm winter temperatures (i.e. + 2 °C above winter averages) in 2016 sustained a higher than normal baseline prevalence of WSs (Harvell et al. 2009; Heron et al. 2010; Randall and van Woesik 2015), facilitating an outbreak in the 2017 summer. This may have been because of either reduced host resistance caused by chronic heat stress and/or maintenance of pathogen loads through winter months. Declines in WS abundance by the end of the study, with only two colonies displaying WS signs on 17 June and one colony at the end of the study period (24 October), were most likely linked with decreasing SSTs, but were also potentially due to reductions in the population density of the coral host (Bruno et al. 2007; Heron et al. 2010). Previously, high host density (> 50% coral cover) has been correlated with high prevalence of WSs (Bruno et al. 2007), and thus reductions in host density by more than half (to 31.0 ± 11.2% cover) by the end of the study accord with host density being lower than the suggested threshold for sustaining an outbreak. Finally, the high mortality of heat-stressed colonies, many of which were diseased, also reduced disease prevalence in the population, as dead colonies do not have disease signs. This added to the overall reduction in relative disease prevalence observed in this study.
Long-term implications of heat stress on the coral population
The 2017 extreme heat stress event, which peaked at 8.3 °C-weeks on Beaver Reef, caused extensive reef-wide and population-level mortality of corals, as demonstrated by the 68% mortality and sharp reduction in coral cover of tagged corals at Beaver Reef. Furthermore, extensive tissue loss on the 32 surviving colonies confirmed that virtually all corals were affected by heat stress. This extensive loss of the dominant tabular coral and reduced structural complexity of the coral community is predicted to have severe effects on reef-associated fish and invertebrate communities and overall ecosystem functioning (Rogers et al. 2014; Kerry and Bellwood 2015; Darling et al. 2017). Furthermore, disease and bleaching substantially reduced population-level reproductive output, as 68 colonies suffered complete mortality and many (13) of the surviving colonies had suffered considerable partial mortality. Additionally, six of the surviving colonies were not gravid pre-spawning in October. Overall, reproductive potential of the A. hyacinthus population was reduced by ~ 75%, significantly reducing the potential for local recruitment. Low local recruitment potential, combined with the absence of a “Phoenix effect”, suggests that coral recovery at Beaver Reef will be slow and dependent on nearby source populations (Done et al. 2010; Holbrook et al. 2018). Similarly, the 2016–2017 back-to-back bleaching events were found to reduce overall coral recruitment by 89% along the whole Great Barrier Reef in a recent large-scale study (Hughes et al. 2019). This finding leaves little hope that source populations will be available to repopulate Beaver Reef and other denuded reefs, which has significant implications for the resilience of the whole reef system (Hughes et al. 2019). Ongoing monitoring of sites like ours, where the history of bleaching and disease is known, would yield important insights into recovery trajectories on GBR reefs (cf. Graham et al. 2011; Neal et al. 2017). As marine heatwaves are forecast to increase in frequency and severity (Frölicher et al. 2018), recovery windows will shrink, increasing the likelihood of permanent changes in the ecosystem (Hughes et al. 2018a, b). Therefore, understanding recovery times after such events is of prime importance to estimate the likelihood and extent of such changes.
The high heat stress in 2017 together with high coral cover at the onset of bleaching fits with model predictions for projecting disease outbreaks and bleaching (Bruno et al. 2007; Heron et al. 2010; Maynard et al. 2011). Our data support the utility of these models and their usefulness for future projections of heat stress impacts on reefs. We documented strong cumulative effects of bleaching and WSs with good explanatory power, even though other stressors on coral populations, like irradiance or water quality, were not considered. In addition, our study highlights the importance of understanding the recent thermal history of reefs, beyond the immediate summer season (DHW), for predicting disease outbreaks. Overall, by following tagged colonies through time, we demonstrate cumulative impacts of disease and bleaching on a coral population and confirm that disease (at least locally) contributed to the substantial loss of corals on the GBR from heat stress in 2017. Future studies of the impacts of heat stress events should consider the role of diseases in coral mortality, as they magnify the impacts of stress events that cause bleaching.
References
Anthony KRN, Connolly SR, Hoegh-Guldberg O (2007) Bleaching, energetics, and coral mortality risk: Effects of temperature, light, and sediment regime. Limnol Oceanogr 52:716–726
Anthony KRN, Hoogenboom MO, Maynard JA, Grottoli AG, Middlebrook R (2009) Energetics approach to predicting mortality risk from environmental stress: A case study of coral bleaching. Funct Ecol 23:539–550
Baird AH, Marshall PA (2002) Mortality, growth and reproduction in scleractinian corals following bleaching on the Great Barrier Reef. Mar Ecol Prog Ser 237:133–141
Baker AC, Glynn PW, Riegl B (2008) Climate change and coral reef bleaching: An ecological assessment of long-term impacts, recovery trends and future outlook. Estuar Coast Shelf Sci 80:1–37
Ban SS, Graham NAJ, Connolly SR (2013) Relationships between temperature, bleaching and white syndrome on the Great Barrier Reef. Coral Reefs 32:1–12
Beeden RJ, Turner MA, Dryden J, Merida F, Goudkamp K, Malone C, Marshall PA, Birtles A, Maynard JA (2014) Rapid survey protocol that provides dynamic information on reef condition to managers of the Great Barrier Reef. Environ Monit Assess 186:8527–8540
Berkelmans R, van Oppen MJH (2006) The role of zooxanthellae in the thermal tolerance of corals: A “nugget of hope” for coral reefs in an era of climate change. Proc R Soc B Biol Sci 273:2305–2312
Bourne DG, Ainsworth TD, Pollock FJ, Willis BL (2015) Towards a better understanding of white syndromes and their causes on Indo-Pacific coral reefs. Coral Reefs 34:233–242
Brandt M, McManus JW (2009) Disease incidence is related to bleaching extent in reef-building corals. Ecology 90:2859–2867
Bruno JF, Selig ER, Casey KS, Page CA, Willis BL, Harvell CD, Sweatman H, Melendy AM (2007) Thermal stress and coral cover as drivers of coral disease outbreaks. PLoS Biol 5:e124
Darling ES, Graham NAJ, Januchowski-Hartley FA, Nash KL, Pratchett MS, Wilson SK (2017) Relationships between structural complexity, coral traits, and reef fish assemblages. Coral Reefs 36:561–575
Done TJ, DeVantier LM, Turak E, Fisk DA, Wakeford M, van Woesik R (2010) Coral growth on three reefs: development of recovery benchmarks using a space for time approach. Coral Reefs 29:815–833
Eakin CM, Morgan JA, Heron SF, Smith TB, Liu G, Alvarez-Filip L, Baca B, Bartels E, Bastidas C, Bouchon C, Brandt M, Bruckner AW, Bunkley-Williams L, Cameron A, Causey BD, Chiappone M, Christensen TRL, Crabbe MJC, Day O, de la Guardia E, Díaz-Pulido G, DiResta D, Gil-Agudelo DL, Gilliam DS, Ginsburg RN, Gore S, Guzmán HM, Hendee JC, Hernández-Delgado EA, Husain E, Jeffrey CFG, Jones RJ, Jordán-Dahlgren E, Kaufman LS, Kline DI, Kramer PA, Lang JC, Lirman D, Mallela J, Manfrino C, Maréchal J-P, Marks K, Mihaly J, Miller WJ, Mueller EM, Muller EM, Orozco Toro CA, Oxenford HA, Ponce-Taylor D, Quinn N, Ritchie KB, Rodríguez S, Ramírez AR, Romano S, Samhouri JF, Sánchez JA, Schmahl GP, Shank BV, Skirving WJ, Steiner SCC, Villamizar E, Walsh SM, Walter C, Weil E, Williams EH, Roberson KW, Yusuf Y (2010) Caribbean Corals in Crisis: Record Thermal Stress, Bleaching, and Mortality in 2005. PLoS One 5:e13969
Frölicher TL, Fischer EM, Gruber N (2018) Marine heatwaves under global warming. Nature 560:360–364
GBRMPA (2013) Reef health incident response system. Great Barrier Reef Marine Park Authority, Townsville
GBRMPA (2017a) Reef health update. Great Barrier Reef Marine Park Authority, Townsville
GBRMPA (2017b) Final report: 2016 coral bleaching event on the Great Barrier Reef. Great Barrier Reef Marine Park Authority, Townsville
Graham NAJ, Nash KL, Kool JT (2011) Coral reef recovery dynamics in a changing world. Coral Reefs 30:283–294
Green EP, Bruckner AW (2000) The significance of coral disease, epizootiology for coral reef conservation. Biol Conserv 96:347–361
Harvell D, Altizer S, Cattadori IM, Harrington L, Weil E (2009) Climate change and wildlife diseases: When does the host matter the most? Ecology 90:912–920
Harvell D, Jordán-Dahlgren E, Merkel S, Rosenberg E, Raymundo L, Smith G, Weil E, Willis B (2007) Coral disease, environmental drivers, and the balance between coral and microbial associates. Oceanography 20:172–195
Heron S, Johnston L, Liu G, Geiger E, Maynard J, De La Cour J, Johnson S, Okano R, Benavente D, Burgess T, Iguel J, Perez D, Skirving W, Strong A, Tirak K, Eakin C (2016a) Validation of reef-scale thermal stress satellite products for coral bleaching monitoring. Remote Sens 8:59
Heron SF, Maynard JA, van Hooidonk R, Eakin CM (2016b) Warming trends and bleaching stress of the world’s coral reefs 1985–2012. Sci Rep 6:38402
Heron SF, Willis BL, Skirving WJ, Eakin CM, Page CA, Miller IR (2010) Summer hot snaps and winter conditions: Modelling white syndrome outbreaks on Great Barrier Reef corals. PLoS One 5:e12210
Hill J, Wilkinson C (2004) Methods for ecological monitoring of coral reefs. Australian Institute of Marine Science, Townsville
Hobbs JA, Frisch AJ, Newman SJ, Wakefield CB (2015) Selective impact of disease on coral communities: Outbreak of white syndrome causes significant total mortality of Acropora plate corals. PLoS One 10:e0132528
Hoegh-Guldberg O (1999) Climate change, coral bleaching and the future of the world’s coral reefs. Mar Freshw Res 50:839–866
Holbrook SJ, Adam TC, Edmunds PJ, Schmitt RJ, Carpenter RC, Brooks AJ, Lenihan HS, Briggs CJ (2018) Recruitment drives spatial variation in recovery rates of resilient coral reefs. Sci Rep 8:1–11
Hughes TP, Graham NAJ, Jackson JBC, Mumby PJ, Steneck RS (2010) Rising to the challenge of sustaining coral reef resilience. Trends Ecol Evol 25:633–642
Hughes TP, Barnes ML, Bellwood DR, Cinner JE, Cumming GS, Jackson JBC, Kleypas J, van de Leemput IA, Lough JM, Morrison TH, Palumbi SR, Van Nes EH, Scheffer M (2017a) Coral reefs in the Anthropocene. Nature 546:82–90
Hughes TP, Kerry JT, Álvarez-Noriega M, Álvarez-Romero JG, Anderson KD, Baird AH, Babcock RC, Beger M, Bellwood DR, Berkelmans R, Bridge TC, Butler IR, Byrne M, Cantin NE, Comeau S, Connolly SR, Cumming GS, Dalton SJ, Diaz-Pulido G, Eakin CM, Figueira WF, Gilmour JP, Harrison HB, Heron SF, Hoey AS, Hobbs J-PA, Hoogenboom MO, Kennedy EV, Kuo C, Lough JM, Lowe RJ, Liu G, McCulloch MT, Malcolm HA, McWilliam MJ, Pandolfi JM, Pears RJ, Pratchett MS, Schoepf V, Simpson T, Skirving WJ, Sommer B, Torda G, Wachenfeld DR, Willis BL, Wilson SK (2017b) Global warming and recurrent mass bleaching of corals. Nature 543:373–377
Hughes TP, Anderson KD, Connolly SR, Heron SF, Kerry JT, Lough JM, Baird AH, Baum JK, Berumen ML, Bridge TC, Claar DC, Eakin CM, Gilmour JP, Graham NAJ, Harrison H, Hobbs JPA, Hoey AS, Hoogenboom M, Lowe RJ, McCulloch MT, Pandolfi JM, Pratchett M, Schoepf V, Torda G, Wilson SK (2018a) Spatial and temporal patterns of mass bleaching of corals in the Anthropocene. Science (80−) 359:80–83
Hughes TP, Kerry JT, Baird AH, Connolly SR, Dietzel A, Eakin CM, Heron SF, Hoey AS, Hoogenboom MO, Liu G, McWilliam MJ, Pears RJ, Pratchett MS, Skirving WJ, Stella JS, Torda G (2018b) Global warming transforms coral reef assemblages. Nature 556:492–496
Hughes TP, Kerry JT, Baird AH, Connolly SR, Chase TJ, Dietzel A, Hill T, Hoey AS, Hoogenboom MO, Jacobson M, Kerswell A, Madin JS, Mieog A, Paley AS, Pratchett MS, Torda G, Woods RM (2019) Global warming impairs stock–recruitment dynamics of corals. Nature 568:387–390
Jones R (2008) Coral bleaching, bleaching-induced mortality, and the adaptive significance of the bleaching response. Mar Biol 154:65–80
Kerry JT, Bellwood DR (2015) Do tabular corals constitute keystone structures for fishes on coral reefs? Coral Reefs 34:41–50
Liu G, Rauenxahn J, Heron S, Eakin C, Skirving WJ, Christensen T, Strong A (2013) NOAA Coral Reef Watch 50 km satellite sea surface temperature-based decision support system for coral bleaching management. 33
Liu G, Heron SF, Eakin CM, Muller-Karger FE, Vega-Rodriguez M, Guild LS, De La Cour JL, Geiger EF, Skirving WJ, Burgess TFR, Strong AE, Harris A, Maturi E, Ignatov A, Sapper J, Li J, Lynds S (2014) Reef-scale thermal stress monitoring of coral ecosystems: new 5-km global products from NOAA Coral Reef Watch. Remote Sens 6:11579–11606
Liu G, Skirving WJ, Geiger EF, De La Cour JL, Marsh BL, Heron SF, Tirak KV, Strong AE, Eakin CM (2017) NOAA coral reef watch’s 5 km satellite coral bleaching heat stress monitoring product suite version 3 and four-month outlook version 4. Reef Encount 32:39–45
MacNeil MA, Mellin C, Matthews S, Wolff NH, McClanahan TR, Devlin M, Drovandi C, Mengersen K, Graham NAJ (2019) Water quality mediates resilience on the Great Barrier Reef. Nat Ecol Evol
Marshall PA, Baird AH (2000) Bleaching of corals on the Great Barrier Reef: Differential susceptibilities among taxa. Coral Reefs 19:155–163
Maynard JA, Anthony KRN, Harvell CD, Burgman MA, Beeden R, Sweatman H, Heron SF, Lamb JB, Willis BL (2011) Predicting outbreaks of a climate-driven coral disease in the Great Barrier Reef. Coral Reefs 30:485–495
Maynard JA, Van Hooidonk R, Eakin CM, Puotinen M, Garren M, Williams G, Heron SF, Lamb J, Weil E, Willis B, Harvell CD (2015) Projections of climate conditions that increase coral susceptibility and pathogen abundance and virulence. Nat Clim Chang 5:688
McClanahan T, Maina J, Moothien-Pillay R, Baker A (2005) Effects of geography, taxa, water flow, and temperature variation on coral bleaching intensity in Mauritius. Mar Ecol Prog Ser 298:131–142
Miller J, Muller E, Rogers C, Waara R, Atkinson A, Whelan KRT, Patterson M, Witcher B (2009) Coral disease following massive bleaching in 2005 causes 60% decline in coral cover on reefs in the US Virgin Islands. Coral Reefs 28:925–937
Muller-Parker G, D’Elia CF, Cook CB (2015) Interactions between corals and their symbiotic algae. Coral Reefs in the Anthropocene. Springer, Netherlands, pp 99–116
Muller EM, Bartels E, Baums IB (2018) Bleaching causes loss of disease resistance within the threatened coral species Acropora cervicornis. Elife 7:1–20
Neal BP, Khen A, Treibitz T, Beijbom O, O’Connor G, Coffroth AM, Knowlton N, Kriegman D, Mitchell BG, Kline DI (2017) Caribbean massive corals not recovering from repeated thermal stress events during 2005–2013. Ecol Evol 7:1339–1353
Page C, Leggat W, Heron S, Choukroun S, Lloyd J, Ainsworth T (2019) Seeking resistance in coral reef ecosystems. The interplay of bio-physical factors and bleaching resistance under a changing climate. BioEssays In print:
Peters EC (2015) Diseases of coral reef organisms. In: Birkeland C (ed) Coral Reefs in the Anthropocene. Springer, Netherlands, Dordrecht, pp 147–178
Pinheiro J, Bates D, DebRoy S, Sarkar D, Team RC (2017) Nlme: Linear and Nonlinear Mixed Effects Models. R package version 3.1-128. 2016. R Softw
Precht WF, Gintert BE, Robbart ML, Fura R, Van Woesik R (2016) Unprecedented disease-related coral mortality in southeastern Florida. Sci Rep 6:31374
R Core Team (2017) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Randall CJ, van Woesik R (2015) Contemporary white-band disease in Caribbean corals driven by climate change. Nat Clim Chang 5:375–379
Randall CJ, Jordan-Garza AG, Muller EM, van Woesik R (2014) Relationships between the history of thermal stress and the relative risk of diseases of Caribbean corals. Ecology 95:1981–1994
Roff G, Bejarano S, Bozec YM, Nugues M, Steneck RS, Mumby PJ (2014) Porites and the Phoenix effect: Unprecedented recovery after a mass coral bleaching event at Rangiroa Atoll, French Polynesia. Mar Biol 161:1385–1393
Rogers A, Blanchard JL, Mumby PJ (2014) Vulnerability of coral reef fisheries to a loss of structural complexity. Curr Biol 24:1000–1005
Selig ER, Harvell CD, Bruno JF, Willis BL, Page CA, Casey KS, Sweatman H (2006) Analyzing the relationship between ocean temperature anomalies and coral disease outbreaks at broad spatial scales. Coral reefs Clim Chang Sci Manag 111–128
Shaver EC, Burkepile DE, Silliman BR (2018) Local management actions can increase coral resilience to thermally-induced bleaching. Nat Ecol Evol 2:1075–1079
Sweatman H (2018) Annual summary report on coral reef condition for 2017/18
Sweatman H, Cheal A, Coleman G, Emslie M, Johns K, Jonker M, Miller I, Osborne K (2008) Long-term monitoring of the Great Barrier reef. Status Rep 8:1–379
Willis BL, Page CA, Dinsdale EA (2004) Coral disease on the Great Barrier Reef. In: Rosenberg E, Loya Y (eds) Coral health and disease. Springer, Berlin Heidelberg, pp 69–104
van Hooidonk R, Maynard J, Tamelander J, Jamison G, Ahmadia G, Raymundo L, Williams G, Heron SF, Planes S (2016) Local-scale projections of coral reef futures and implications of the Paris Agreement. Sci Rep 6:e39666
van Oppen MJH, Gates RD, Blackall LL, Cantin N, Chakravarti LJ, Chan WY, Cormick C, Crean A, Damjanovic K, Epstein H, Harrison PL, Jones TA, Miller M, Pears RJ, Peplow LM, Raftos DA, Schaffelke B, Stewart K, Torda G, Wachenfeld D, Weeks AR, Putnam HM (2017) Shifting paradigms in restoration of the world’s coral reefs. Glob Chang Biol 23:3437–3448
Wooldridge SA (2014) Assessing coral health and resilience in a warming ocean: Why looks can be deceptive. Bioessays 36:1041–1049
Work TM, Russell R, Aeby GS (2012) Tissue loss (white syndrome) in the coral Montipora capitata is a dynamic disease with multiple host responses and potential causes. Proc R Soc B Biol Sci 279:4334–4341
Acknowledgements
This study was made possible by financial support from the Great Barrier Reef Marine Park Authority, and funding from the ARC CoE for Coral Reef Studies to B. Willis. Kerryn Bell (Reef Express) is thanked for facilitating the survey trips to Beaver Reef and her infectious passion for corals reefs. Abby Fatland, Allison Paley, Carly Hayk, Carine Lefevre, Kate Quigley, Margaux Hein, Phil Osmond, Saskia McDonald and Svenja Müller are all thanked for their laboratory and fieldwork assistance. SFH was supported by NASA ROSES Ecological Forecasting Grant #16-eco4cast-0032 to the University of Hawaii. The scientific results and conclusions, as well as any views or opinions expressed herein, are those of the author(s) and do not necessarily reflect the views of NOAA or the Department of Commerce. All tagging, surveys and sampling were performed under the auspice of GBRMPA permit G16/38009.1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Topic Editor Morgan S. Pratchett
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Brodnicke, O.B., Bourne, D.G., Heron, S.F. et al. Unravelling the links between heat stress, bleaching and disease: fate of tabular corals following a combined disease and bleaching event. Coral Reefs 38, 591–603 (2019). https://doi.org/10.1007/s00338-019-01813-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-019-01813-9