Abstract
Purpose
To study the long-term evolution of the bone marrow burden (BMB) score at MRI in patients with Gaucher disease (GD) under enzyme replacement therapy (ERT).
Material and methods
Forty patients treated for GD were retrospectively studied in a referral centre. BMB scores were assessed on spine and femur MR examinations performed between January 2003 and June 2014. The long-term evolution of the BMB scores was analyzed using a linear mixed model.
Results
A total of 121 MRI examinations were performed during the study period with a mean follow-up of 7.1 years ± 5.6, an average rate of 3.1 MR examinations ± 1.7 per patient and an interval of 2.3 years ± 1.1 between examinations. Patients had received ERT during 12 years on average ± 6.7. The trend of BMB scores with time decreased significantly by 15 % (P = 0.008) during the total study period and 39 % (P = 0.01) during the first 5 years of treatment. No changes in BMB scores were observed after five years of treatment.
Conclusion
In Gaucher patients, the trend of MRI BMB scores with time decreased significantly under ERT the first 5 years of treatment before a long-term stabilization.
Key points
• Bone marrow infiltration of Gaucher patients responds to enzyme replacement therapy
• MRI BMB score decreases mainly during the first five years of treatment
• MRI BMB score tends to stabilize after five years of treatment
• MR examinations could be limited after five years of treatment
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Gaucher disease (GD) is the most frequent lysosomal storage disorder [1, 2]. It is an autosomal recessively inherited condition, resulting in the deficiency of the lysosomal enzyme glucocerebrosidase (or acid β-glucosidase) [3]. The enzyme is encoded on chromosome 1 (1q21). Its deficiency results in the storage of the substrate glucocerebroside in macrophages. The lipid-laden cells are named Gaucher cells and are primarily found in the liver, spleen and bone marrow. Three types of GD have been described: type I is the most common phenotype. Types II and III are more severe and involve neurological manifestations. Type I GD is most common in the Ashkenazi Jewish population, with an estimated prevalence of 1 per 855 people [4, 5]. The overall prevalence in the general population is 1 per 60,000 people, but this prevalence may be underestimated [6]. In addition to its social and psychological impact, the musculoskeletal manifestations are the main cause of patient disability and complaints in type I GD [7]. Skeletal manifestations involve abnormal bone remodelling and limb deformity, osteopenia, osteonecrosis, osteomyelitis, and acute episodes of severe pain with bone infarct known as “Gaucher crisis” [8–10].
Since the early 1990s and the development of enzyme replacement therapy (ERT), the outcomes of Gaucher disease have considerably improved [11, 12]. ERT proved to be highly effective in reversing cytopenia and reducing organ volumes [13]. However, the skeletal disease may be slower to respond or even unresponsive, particularly in patients with extensive lesions and delayed treatment [14]. MRI has become a reference for the assessment and monitoring of bone marrow infiltration under ERT [15–17] and for the detection of early complications [18]. As highlighted by Hermann in his review [19] and in the study by Terk and colleagues [20], various MR imaging methods have been used to track the skeletal changes under therapy. The evaluation of the bone marrow infiltration may be quantitative, based on T1 relaxation [21], or using the chemical shift imaging [17, 22, 23]. Several authors have even suggested that the measurements of bone marrow fat fraction using Dixon quantitative chemical shift imaging (QCSI) could be used as an imaging biomarker for Gaucher patients [24]. Nevertheless, although MR imaging is widely available, the QCSI technique is not always provided in the standard packages of sequences on MR imagers and may, thus, limit its use worldwide. The semi-quantitative assessment of the bone marrow infiltration is also a reliable method for scoring both signal intensity changes and the involvement of bone sites [25–27]. The Bone Marrow Burden (BMB) score [25, 26], based on the visual interpretation of signal intensity and the geographic distribution of the disease on conventional MR images, proved to be a reliable and reproducible semi-quantitative scoring system. It is more available and easier to use in comparison with other quantitative methods, such as fat fraction measurement using the Dixon chemical shift imaging technique [25]. Unlike previous studies using qualitative or semi-quantitative scores, which were restricted to the assessment of the peripheral skeleton [28, 29], the BMB score assessed bone infiltration of the spine in addition to the femur. It is, thus, more representative of the status of bone marrow infiltration in Gaucher patients [25].
In our study, we aimed to assess the trend of BMB scores with time in Gaucher patients under ERT, on both spine and femur MR examinations, to better define the long-term evolution of skeletal involvement and to optimize the surveillance of Gaucher patients using MRI.
Material and methods
Institutional review board approval was obtained for the anonymous retrospective use of the clinical and imaging data of patients. The informed consent of patients was waived.
Patients
From January 2003 to June 2014, 58 patients were referred to a single institution (AP-HP, Referral Center for Lysosomal Diseases, Hôpital Beaujon, Clichy, France) for the management of proven (biologically and genetically) Gaucher disease. Thirteen patients were excluded due to incomplete or unavailable imaging data. Five patients were excluded because they were never treated. A total of forty patients with MR examinations of both the spine and femur, were included in the study. Nine patients out of 40 (23 %) were placed under ERT during the study period. The majority of patients (31/40, 77 %) were already under ERT at the beginning of the study period. Gender, age at diagnosis, age at first symptoms, duration of symptoms until the end of the study period, presence of bone manifestations (“Gaucher crisis”, chronic bone/articular pain), presence of organomegaly, type, and duration of treatment were recorded.
Data collection, MR protocol and image analysis
From January 2003 to June 2014, 121 MR scans of both the spine and femur were performed on 40 patients in two locations using three MRI machines: Hôpital Beaujon (Department of musculoskeletal imaging, Clichy, France) with Philips Intera 1.5 T and Philips Ingenia 3 T (from January 2013), MR machines (Philips Healthcare, MA, USA), and in the Musculoskeletal Imaging Center Bachaumont with Siemens Magnetom Espree 1.5 T MR machine (Siemens Healthcare, Erlangen, Germany). The intervals between the MR examinations were recorded as well as the date of introduction of the ERT for patients who were not previously treated. The average follow-up per patient under ERT was computed. The imaging protocol included T1 and T2 ± fat saturation/suppression weighted images in sagittal view for the spine, coronal, and axial views for the pelvis and femurs. The BMB scores (total of BMB scores of lumbar spine and femurs) were assessed according to the study of Maas et al. [25] (Tables 1 and 2) by two readers (BF and DP) with five and 15 years of experience in reading MR scans, respectively. The readers were blinded to the clinical data. MR images were analyzed in random order using the same workstation (Vue PACS, version 11:3; Carestream Health, Rochester, NY, USA).
Statistical analysis
The mean and standard deviation, range and median were computed for the age, age at diagnosis, duration of symptoms until the first available MR, duration of treatment, number of MR examinations performed during the study period, intervals between MR examinations for each patient, and BMB score (spine + femurs) for each MR examination.
The intraclass correlation coefficient was computed to test the interrater agreement for the assessment of the BMB scores by the two readers.
To investigate the trend of the BMB scores with time under ERT, we used a linear mixed model analysis of repeated measures (R statistical software, lme4 package) with the BMB scores as continuous outcome variables. The Restricted Maximum Likelihood (REML) estimation method and type 3 tests of fixed effects were performed [30, 31]. First, we used the linear mixed model to analyze the trend of the BMB scores for all periods of treatment. Next, we used the same model, but the analysis focused on the trend of BMB scores during the first five years of treatment. Finally, the analysis was performed using the same statistical model from the 6th year of treatment until the end of the study period. The analysis was also performed on a subgroup of nine patients whose treatment was introduced during the study period and for whom MR scans and BMB scores were assessed before and after ERT.
P-values less than 0.05 were considered to indicate a significant difference. Confidence intervals were established at 95 %.
Results
Among the 40 patients, there were 22 females (55 %) and 18 males (45 %) (sex ratio of female to male patients, 1.22:1). The type I GD was observed in 38/40 (95 %) and type III GD in 2/40 (5 %). The age of the patients at the end of the study period was 46.1 ± 14 (range, 26-87). The age at diagnosis was 21 ± 16.2 (1-67). The age at first symptoms was 18.7 ± 15.8 (1-67). The duration of symptoms at the end of the study period was 27.3 years ± 14.3 (4-69). Bone and articular pain was experienced by 37/40 (93 %) patients. Hepatomegaly was reported in 32/40 (80 %) patients and splenomegaly in 36/40 (90 %). Nine patients (23 %) underwent splenectomy before the study period. Patients were placed under ERT for an average period of 12 years ± 6.7 (range, 1-22). In 9/40 (23 %) cases, ERT was introduced during the study period and MR scans were performed before and after treatment. The ERT was consistent with Imiglucerase (Cerezyme® Genzyme Therpautics, Cambridge, MA, USA), or Velaglucerase alfa (VPIRV® Shire Human Genetic Therapies, Lexington, MA, USA), or Taliglucerase alfa (Elelyso™ Genzyme Therapeutics, Cambridge, MA, USA).
A total of 121 MR examinations were performed and analyzed. The average follow-up per patient was 7.1 years ± 5.6 (range, 1-10). The number of MR examinations per patient during the follow-up was 3 ± 1.7 (1-7). The interval between MR examinations was 2.3 years ± 1.1 (1-6).
The evolution of the BMB score for the whole cohort is shown in Fig. 1. The statistical results are provided in Table 3. According to the linear mixed model, the trend of the BMB scores with time decreased significantly during the study period (Fig. 2). This evolution was nonlinear with an important decrease during the first five years of treatment (-39 %) and then a stabilization of the BMB scores. The analysis of the subgroup of nine patients for whom the treatment was introduced during the study period showed an important decrease of 37 %, almost marginally significant (P = 0.051). After five years of treatment, because of no changes in the BMB scores, the linear mixed model failed to converge, meaning that there was no statistical result to express the non-evolution of the BMB score in Gaucher patients.
Evolution of the Bone Marrow Burden (BMB) score with time at MRI for each patient during follow-up. Note − Each dot represents the BMB score related to a MR examination. Patients’ evolution are represented by curves joining the dots. Using a linear mixed model fit by the restricted maximum likelihood method, the trend of BMB scores with time decreased of 15 % (P = 0.008) on the all study period and of 39 % (P = 0.01) when considering only the first five years under enzyme replacement therapy. No significant changes of BMB scores were observed after five years of treatment
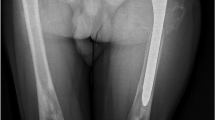
Spine and femur MRI of a patient with type 1 Gaucher disease before (a-c) and one year after introduction of enzyme replacement therapy (ERT) (b-d). (a-b) Sagittal T1-weighted images of the lumbar spine show a diffuse infiltration of the bone marrow of vertebrae in low signal intensity (a), decreasing under ERT (b). (c-d) Coronal T1-weighted images of the femurs show a patchy bone marrow infiltration in epiphyses (a, arrows) that decrease under ERT (b, arrows). Sequelae of bone infarcts are visible in femoral and tibial diaphyses (a, stars)
The intraclass correlation coefficient for the BMB scores was excellent (0.98 [0.97-0.99]).
Discussion
The characteristics of our study sample were similar to those reported in the French register of the comity of Gaucher disease evaluation [32] as well as in the international collaborative Gaucher group [6], with regards to the sex ratio, age of patients, prevalence of organomegaly, and bone/articular pain.
MRI is the gold standard to assess and monitor bone involvement in Gaucher patients. Quantitative or semi-quantitative methods were used to assess the infiltration of bone marrow by storage cells. Several studies [17, 25] have shown that monitoring bone involvement using the semi-quantitative BMB score was equivalent to quantitative analysis using the measurement of the bone marrow fat fraction by QCSI. The BMB score has proven to be easy to assess in clinical routine and is reproducible [25]. Moreover, we report an excellent interrater agreement.
Our study showed that the trend of BMB scores with time decreased significantly, which is consistent with previous studies using either the QCSI or the BMB score [17, 29, 26, 33, 34]. Hollak et al. [17] examined 12 patients under ERT and observed a normalized fat fraction at QCSI in 11/12 (92 %) patients with a significant decrease from the very first year of treatment. However, their long-term results were insufficient to characterize the long-term evolution. More specifically, we observed a nonlinear evolution of the BMB score under ERT with a higher rate of decrease during the first five years of treatment and a stabilization after five years under ERT. This evolution was suggested in a study by Elstein et al. [34] on a series of 11 patients at a 9-month follow-up and was limited to eight patients after a 5-year follow-up. Moreover, our analysis quantified the decrease in the BMB score in providing a rate of decrease per year at approximately 39 % for the first five years of treatment. However, this rate should be considered with caution due to the effect of patients characteristics (duration of symptoms before treatment, baseline severity of the GD, and splenomegaly or history of splenectomy), which we did not study because the subgroups were too small for statistics. These characteristics may modulate the response to treatment as Demayo et al. showed in their study of the correlation of BMB scores with the GD genotype and spleen status [35]. Such characteristics may be the subject of further studies. The stabilization of BMB scores that we observed after five years of treatment is also an important characteristic of the evolution. Only a few studies in the English literature [29, 33, 36] have suggested a non-evolution of the QCSI or BMB score under ERT, but monitoring of patients in these studies did not exceed 36 months. Our results may prompt clinicians to limit the MRI surveillance by increasing the interval between MR scans after five years of treatment in asymptomatic patients or patients without significant changes in their clinical status.
We acknowledge several limitations to our work. First, our retrospective study may have been affected by selection biases. The retrospective format of this study was also responsible for the variability in intervals between MR examinations. Because Gaucher disease is a very rare condition, it is very difficult to include consecutive patients and to follow a large sample of patients. Moreover, these patients may have suffered from a variable delay prior to diagnosis and may be referred to specific centres at a variable time of the evolution of the disease. In this retrospective study, we did not obtain more accurate information about the episodes of clinical and imaging bone complications. More specifically, the bone complications visible on MR, such as bone infarct or sequelae of fracture, were not retrospectively recorded, which may affect the BMB scoring. Despite this variability in patient characteristics, the use of a linear mixed model enabled the demonstration of a significant decrease followed by a stabilization of the BMB scores. Second, our analysis on the subgroup of patients for whom treatment was introduced during the study period, approached but fell short of significance, which was likely due to the small sample size. Nevertheless, our global analysis strengthened the findings of previous studies with a comparable sample size and a similar trend of bone involvement with time [26, 34]. Third, the long-term follow-up of patients was performed using different MRI devices. Given a study period of over ten years, the technical development and changes in imaging devices were mandatory. More precisely, from January 2013, some patients were examined with 3 T MRI machines while their previous examinations were performed with 1.5 T MRI devices. This difference in field strengths may affect the quality of the images and BMB scoring. However, as a semi-quantitative analysis, we think that the evaluation of bone involvement of the GD using the BMB score may be less influenced by technical developments than the direct measurement of the fat fraction in bone marrow using the Dixon QCSI. With new MRI developments, the diffusion weighted imaging and whole body MRI may prove useful, but could not be employed in our study.
In conclusion, we showed that, in Gaucher patients, the trend of bone marrow burden scores with time decreased significantly under enzyme replacement therapy during the five first years of treatment. No changes in BMB scores were observed after five years of ERT. This stabilization may prompt clinicians and radiologists to limit MR examinations in asymptomatic patients after five years of ERT.
References
Meikle PJ, Hopwood JJ, Clague AE, Carey WF (1999) Prevalence of lysosomal storage disorders. JAMA 281:249–254
Fuller M, Meikle PJ, Hopwood JJ (2006) Epidemiology of lysosomal storage diseases: an overview. In: Mehta A, Beck M, Sunder-Plassmann G (eds) Fabry Dis. Perspect. 5 Years FOS. Oxford PharmaGenesis, Oxford
Brady RO, Kanfer JN, Bradley RM, Shapiro D (1966) Demonstration of a deficiency of glucocerebroside-cleaving enzyme in Gaucher’s disease. J Clin Invest 45:1112–1115
Beutler E, Nguyen NJ, Henneberger MW, Smolec JM, McPherson RA, West C (1993) Gaucher disease: gene frequencies in the Ashkenazi Jewish population. Am J Hum Genet 52:85–88
Weinreb NJ, Andersson HC, Banikazemi M, Barranger J, Beutler E, Charrow J et al (2008) Prevalence of type 1 Gaucher disease in the United States. Arch Intern Med 168:326–327, author reply 327–328
Charrow J, Andersson HC, Kaplan P, Kolodny EH, Mistry P, Pastores G et al (2000) The Gaucher registry: demographics and disease characteristics of 1698 patients with Gaucher disease. Arch Intern Med 160:2835–2843
Masek BJ, Sims KB, Bove CM, Korson MS, Short P, Norman DK (1999) Quality of life assessment in adults with type 1 Gaucher disease. Qual Life Res Int J Qual Life Asp Treat Care Rehab 8:263–268
Yossipovitch ZH, Herman G, Makin M (1965) Aseptic osteomyelitis in Gaucher’s disease. Isr J Med Sci 1:531–536
Weisstein JS, Steinbach LS, Diamond CA, Huang SJ, O’Donnell RJ (2001) Pseudo-osteomyelitic crisis upon presentation of Gaucher disease. Skelet Radiol 30:407–410
Lutsky KF, Tejwani NC (2007) Orthopaedic manifestations of Gaucher disease. Bull NYU Hosp Jt Dis 65:37–42
Brady RO, Barton NW (1996) Enzyme replacement and gene therapy for Gaucher’s disease. Lipids 31:S137–S139
Grabowski GA (2008) Phenotype, diagnosis, and treatment of Gaucher’s disease. Lancet 372:1263–1271
Barton NW, Brady RO, Dambrosia JM, Di Bisceglie AM, Doppelt SH, Hill SC et al (1991) Replacement therapy for inherited enzyme deficiency–macrophage-targeted glucocerebrosidase for Gaucher’s disease. N Engl J Med 324:1464–1470
De Fost M, van Noesel CJM, Aerts JMFG, Maas M, Pöll RG, Hollak CEM (2008) Persistent bone disease in adult type 1 Gaucher disease despite increasing doses of enzyme replacement therapy. Haematologica 93:1119–1120
Lanir A, Hadar H, Cohen I, Tal Y, Benmair J, Schreiber R et al (1986) Gaucher disease: assessment with MR imaging. Radiology 161:239–244
Rosenthal DI, Scott JA, Barranger J, Mankin HJ, Saini S, Brady TJ et al (1986) Evaluation of Gaucher disease using magnetic resonance imaging. J Bone Joint Surg Am 68:802–808
Hollak C, Maas M, Akkerman E, den Heeten A, Aerts H (2001) Dixon quantitative chemical shift imaging is a sensitive tool for the evaluation of bone marrow responses to individualized doses of enzyme supplementation therapy in type 1 Gaucher disease. Blood Cells Mol Dis 27:1005–1012
Pastores GM, Patel MJ, Firooznia H (2000) Bone and joint complications related to Gaucher disease. Curr Rheumatol Rep 2:175–180
Hermann G, Pastores GM, Abdelwahab IF, Lorberboym AM (1997) Gaucher disease: assessment of skeletal involvement and therapeutic responses to enzyme replacement. Skelet Radiol 26:687–696
Maas M, Poll LW, Terk MR (2002) Imaging and quantifying skeletal involvement in Gaucher disease. Br J Radiol 75:A13–A24
Magnaldi S, Longo R, Ukmar M, Zanatta M, Bottega M, Sottocasa GL (1997) Bone marrow relaxation times in Gaucher disease before and after enzyme replacement therapy. Eur Radiol 7:486–491
Johnson LA, Hoppel BE, Gerard EL, Miller SP, Doppelt SH, Zirzow GC et al (1992) Quantitative chemical shift imaging of vertebral bone marrow in patients with Gaucher disease. Radiology 182:451–455
Poll LW, Maas M, Terk MR, Roca-Espiau M, Bembi B, Ciana G et al (2002) Response of Gaucher bone disease to enzyme replacement therapy. Br J Radiol 75:A25–A36
Van Dussen L, Akkerman EM, Hollak CE, Nederveen AJ, Maas M (2014) Evaluation of an imaging biomarker, Dixon quantitative chemical shift imaging, in Gaucher disease: lessons learned. J Inherit Metab Dis 37(6):1003–1011
Maas M, van Kuijk C, Stoker J, Hollak CEM, Akkerman EM, Aerts JFMG et al (2003) Quantification of bone involvement in Gaucher disease: MR imaging bone marrow burden score as an alternative to Dixon quantitative chemical shift MR imaging–initial experience. Radiology 229:554–561
Robertson PL, Maas M, Goldblatt J (2007) Semiquantitative assessment of skeletal response to enzyme replacement therapy for Gaucher’s disease using the bone marrow burden score. AJR Am J Roentgenol 188:1521–1528
Roca M, Mota J, Alfonso P, Pocoví M, Giraldo P (2007) S-MRI score: a simple method for assessing bone marrow involvement in Gaucher disease. Eur J Radiol 62:132–137
Terk MR, Esplin J, Lee K, Magre G, Colletti PM (1995) MR imaging of patients with type 1 Gaucher’s disease: relationship between bone and visceral changes. AJR Am J Roentgenol 165:599–604
Poll LW, Koch JA, vom Dahl S, Willers R, Scherer A, Boerner D et al (2001) Magnetic resonance imaging of bone marrow changes in Gaucher disease during enzyme replacement therapy: first German long-term results. Skelet Radiol 30:496–503
Gad AM, Youssif NA (2006) Linear mixed models for longitudinal data with nonrandom dropouts. J Data Sci 4:447–460
Vazquez AI, Bates DM, Rosa GJM, Gianola D, Weigel KA (2010) Technical note: an R package for fitting generalized linear mixed models in animal breeding. J Anim Sci 88:497–504
Stirnemann J, Vigan M, Hamroun D, Heraoui D, Rossi-Semerano L, Berger MG et al (2012) The French Gaucher’s disease registry: clinical characteristics, complications and treatment of 562 patients. Orphanet J Rare Dis 7:77
Van Dussen L, Zimran A, Akkerman EM, Aerts JMFG, Petakov M, Elstein D et al (2013) Taliglucerase alfa leads to favorable bone marrow responses in patients with type I Gaucher disease. Blood Cells Mol Dis 50:206–211
Elstein D, Haims AH, Zahrieh D, Cohn GM, Zimran A (2014) Impact of velaglucerase alfa on bone marrow burden score in adult patients with type 1 Gaucher disease: 7-year follow-up. Blood Cells Mol Dis 53:56–60
DeMayo RF, Haims AH, McRae MC, Yang R, Mistry PK (2008) Correlation of MRI-Based bone marrow burden score with genotype and spleen status in Gaucher’s disease. AJR Am J Roentgenol 191:115–123
Kamath RS, Lukina E, Watman N, Dragosky M, Pastores GM, Arreguin EA et al (2014) Skeletal improvement in patients with Gaucher disease type 1: a phase 2 trial of oral eliglustat. Skelet Radiol 43:1353–1360
Acknowledgments
The scientific guarantor of this publication is: David PETROVER. The authors of this manuscript declare relationships with the following companies: David Petrover, Jerome Stirnemann and Nadia Belmatoug have received research funding and consultant honoraria from Shire, and Genzyme in the past. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed. The authors state that this work has not received any funding. One of the authors has significant statistical expertise: Jerome STIRNEMANN. Institutional review Board approval was obtained. Written informed consent from patients was waived by the Institutional review Board. No subjects or cohorts of our study have previously been reported. Methodology and design: retrospective, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fedida, B., Touraine, S., Stirnemann, J. et al. Bone marrow involvement in Gaucher disease at MRI : what long-term evolution can we expect under enzyme replacement therapy?. Eur Radiol 25, 2969–2975 (2015). https://doi.org/10.1007/s00330-015-3715-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-015-3715-2