Abstract
Objectives
Paraneoplastic neurological syndromes (PNS) constitute a challenging diagnostic problem, as the underlying tumour often remains unidentified for a long time, even with frequent conventional diagnostic procedures. For appropriate patient management timely identification of the tumour is critical. We evaluated the value of 18F-FDG-PET/CT in the investigation of PNS.
Methods
The case notes of 46 consecutive patients with clinically suspected PNS who underwent 18F-FDG-PET/CT were reviewed retrospectively and the performance of PET/CT for detecting underlying tumours was assessed.
Results
PET/CT detected foci of increased 18F-FDG uptake in 10 out of 46 patients. In six of these 10 patients combined PET/CT identified the underlying disease: four patients suffered from PNS; vasculitic and local metastatic disease was detected in two other patients.
Conclusions
Based on our results, we believe that the role of positron emission tomography in the detection of occult neoplasms in patients with PNS has been overestimated in the past. In clinical practice, PNS is far more often suspected than proven. In our study combined PET/CT identified malignancy as the underlying cause of suspected PNS in only 8.7% (4/46). We believe that combined PET/CT should be reserved for stringently selected patients with a high clinical index of suspicion for PNS and after conventional imaging techniques fail to detect a tumour.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Paraneoplastic neurological syndromes (PNS) are a rare group of autoimmune central nervous system disorders triggered by an immune response to a malignant tumour [1]. This response leads to generation of well-characterised paraneoplastic antibodies that include anti-Hu, anti-Yo, anti-CV2/CRMP5, anti-Ri, anti-Ma2 and anti-amphiphysin [2]. Classic neurological syndromes in PNS include subacute encephalomyelitis, limbic encephalitis, cerebellar degeneration, opsoclonus myoclonus and sensory neuronopathy. PNS is classified as either definite or possible PNS, based on the clinical syndrome and detection of paraneoplastic antibodies or detection of a tumour within 5 years [3, 4]. The neurological syndromes often precede the clinical manifestation of the underlying tumour by up to several years [3, 4]. Removal of the tumour as the antigenic source remains the only proven treatment to stabilise or improve the neurological dysfunction, which is frequently the cause of death in this group of patients [5–7]. Therefore, early localisation of cancer is of crucial therapeutic importance. As most of these tumours are rather small or limited to metastatic lymph nodes [8, 9], morphological imaging often fails [10].
Compared with morphological imaging, metabolic imaging like 18F-FDG-PET is highly efficient in mapping the increased glucose utilisation of tumours. In the past two decades FDG-PET has emerged as a practical and cost-effective imaging method in patients with various tumour entities [11–13]. The potential of FDG-PET was emphasised in preliminary retrospective reports dealing with PNS. The reported sensitivities in relatively small numbers of patients were up to 80%, suggesting a clear contribution of PET to patient management [14–18].
In the last decade combined PET/CT has become commercially available and has been demonstrated to have an impact on diagnostic performance in oncological patients [19–22]. Co-registered PET and CT images provide complementary information within a single study: the presence of a viable tumour and its accurate location. However, up to now only limited data have existed concerning the value of combined PET/CT in patients with PNS [23]. The aim of the present study was to retrospectively review the value of combined PET/CT using 18F-FDG in the investigation of patients with clinically suspected PNS.
Materials and methods
Patient population and inclusion criteria
The case notes of 46 patients (24 female, 22 male; range 21–81 years; median 65) with suspected PNS who had been referred for FDG-PET/CT at our University Hospital from 2006 to 2008 were reviewed retrospectively.
PNS was defined as the presence of signs and symptoms caused by damage to any part of the nervous system not due to a primary tumour, metastasis or any other brain, spinal cord or nerve disorder [1, 4]. The clinical diagnosis of PNS was based on clinical evaluation by consultant neurologists, and following exclusion of other possible causes. Consultant neurologists made all requests for PET/CT. Paraneoplastic antibodies, anti-Hu, anti-Yo, anti-CV2/CRMP5, anti-Ri, anti-Ma2 and anti-amphiphysin, were evaluated in 40 out of the 46 patients.
PET/CT acquisition and image processing
Patients were instructed to fast, except for oral glucose-free oral hydration, for 4–6 h before the injection of 350 MBq of 18F-FDG. Blood glucose level was measured before injection and 18F-FDG was administered at glucose levels below 200 mg/dl. Oral contrast (20 ml Gastrolux CT in 1 l water; 7.4 mg iodine/ml; Sanochemia Diagnostics, Neuss, Germany) was administered to the patients during the uptake time; no intravenous contrast material was administered for CT. At 90 min after the injection of 18F-FDG, PET and CT images were acquired using a hybrid PET/CT (Gemini GXL10, Philips, Best, the Netherlands). Whole body images were acquired from head to feet in a craniocaudal direction. All PET was performed in 3D mode. Emission images were acquired for 1.5 min per bed position (effective axial field of view 90 cm). Subsequent to the PET acquisition CT was performed. The acquisition parameters were as follows: 120 kV, 150 mAs, slice thickness 5 mm, no gap, pitch 0.9, rotation time 0.74 s, matrix 512 × 512. CT data were used for low noise attenuation correction of PET data and for co-registration with attenuation corrected PET images. PET, CT and fused PET/CT images were available for review and were displayed in axial, coronal and sagittal planes. The PET data were displayed as non-corrected and attenuation-corrected images and also in a rotating maximum intensity projection.
Interpretation and analysis of PET/CT images
An interdisciplinary team, including two nuclear medicine physicians and two radiologists, with knowledge of the patient’s clinical history evaluated all studies. Primarily, PET and CT images were read independently: PET images were evaluated by two nuclear medicine physicians (one with 12 years’ experience and one with 2 years’ experience) and CT images were evaluated by two radiologists (one with 15 years’ experience and one with 2 years’ experience). Finally studies were judged by consensus of physicians from both fields.
Foci of increased 18F-FDG uptake were recorded. Studies showing at least one area of increased 18F-FDG uptake with intensity higher than that of surrounding tissues, localised by hybrid images to an area that did not correspond to the physiological biodistribution of the radiotracer, were defined as positive. If subsequent imaging or biopsies confirmed the positive results from PET/CT, the studies were judged to be true positive. If subsequent investigations were negative, results from PET/CT were judged to be false positive. Studies showing 18F-FDG uptake only in areas of physiological tracer biodistribution or no sites of increased uptake were considered negative.
Results
Patient characteristics
Of 46 patients referred for PET/CT 17 patients (37%) had a classic paraneoplastic neurological syndrome [3]. The remaining patients had neurological symptoms compatible with PNS and extensive exclusion of relevant differential diagnoses (Table 1). The predominant neurological manifestations were encephalitis in 13 patients (28%) and polyneuropathy in 9 patients (20%).
PET/CT
All 46 patients underwent combined PET/CT: PET results were positive in 10 patients (22%); results from CT were positive in 8 patients (17%) (Table 2). The hybrid combination of PET/CT detected the underlying disease for the clinical syndrome in six patients, corresponding to 13% (6/46) of all investigated cases. Of these six patients, tumours distant from the CNS and biopsy-proven tumours were detected in four patients (cases 1–4), representing the verified PNS cases and corresponding to 8.7% (4/46) of the study population. PET was able to detect all four patients with confirmed PNS (Table 2, Figs. 1, 2, 3). CT, by comparison, detected three of the four histologically proven cases with PNS. The false-negative CT result was gastric cancer (Fig. 3) in case 3. In the remaining two of the six patients, neurological syndromes were due to brain metastasis (case 5) or were of vasculitic origin (case 6). Four patients (cases 7–10) had false-positive PET/CT results (Table 2).
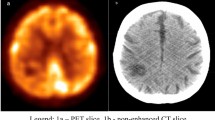
Case 1, a 61-year-old woman presented with anti-Hu antibodies and sensory neuropathy as well as limbic encephalitis. Transaxial FDG-PET demonstrates focus of increased tracer uptake in the right retroclavicular region (left, arrow), localised by PET/CT image (centre) to the right retroclavicular lymph nodes (right, arrow). Neuroendocrine carcinoma was diagnosed on lymph node biopsy
Case 2, a 48-year-old woman presented with anti-Hu antibodies and motor neuropathy. Transaxial FDG-PET (left) demonstrates intense tracer uptake in the left lung (not shown) and left hilus (arrow). PET/CT image (centre) localised this to a space-occupying lesion in the inferior lobe of the left lung (not shown) and ipsilateral hilar lymph nodes (right, arrow). After subsequent bronchoscopy small cell lung cancer was diagnosed on biopsy
Case 3, a 55-year-old woman presented with polyneuropathy. Paraneoplastic antibodies were not assessed. All previous radiological investigations had been negative. Transaxial FDG-PET (left) demonstrates focus of increased tracer uptake in the upper abdomen (arrow). The CT remains without pathological correlation (right); only combined PET/CT (centre) localises the tracer uptake to the gastric wall (right, arrow). Diagnosis of gastric cancer was estimated after gastroscopy and histological examination
Paraneoplastic antibodies
Well-established paraneoplastic antibodies were positive in four patients (4/40, 10%). Two patients had anti-Hu antibodies (cases 1 and 2), one had anti-Ri and one had CV2/CRMP5 antibodies, all classified as being definite PNS by consensus criteria [2]. In two patients of the four antibody-positive cases the underlying tumour was detected by PET/CT (cases 1 and 2); in the other two patients no tumour was detected. Of the four patients with verified PNS (cases 1–4) two had confirmed paraneoplastic antibodies (definite PNS, cases 1 and 2), one was negative (definite PNS, case 4) and one patient was not tested (possible PNS, case 3).
The clinical details and imaging findings of the 10 patients with abnormal FDG uptake are listed in detail in Table 2 and discussed below.
Patients with confirmed PNS and positive PET/CT (cases 1–4)
Patient 1 suffered from subacute sensory neuronopathy and limbic encephalitis and had elevated values of anti-Hu antibodies. In a previous CT performed 3 months before, retroclavicular lymph nodes had been described as not suspicious. FDG-PET revealed increased tracer uptake in the right retroclavicular region; based on the CT this focal uptake was anatomically assigned to retroclavicular lymph nodes (Fig. 1). Surgical exploration of retroclavicular lymph nodes led to the diagnosis of neuroendocrine carcinoma.
Patient 2 suffered from subacute, asymmetric, axonal purely motor neuropathy of the upper limbs and respiratory failure. She was positive for anti-Hu antibodies. FDG-PET showed increased tracer uptake of the inferior lobe of the left lung (not shown) and of the left hilus (Fig. 2), with corresponding lesions in the CT. Subsequent bronchoscopy and biopsy confirmed small cell lung cancer (SCLC) with hilar lymph node metastases. The CT alone sufficed to clearly identify the primary tumour, but could not definitively differentiate between metastatic and non-metastatic lymph nodes. FDG-PET allowed discrimination between the two entities and proved that the left hilar lymph nodes were involved.
Patient 3 suffered from subacute, severe, predominately motor, mixed axonal and demyelinating, distal-symmetric polyneuropathy. Paraneoplastic antibodies were not recorded. FDG-PET revealed focal increased glucose metabolism of the gastric wall (Fig. 3). The CT remained without pathological findings. Subsequent exploration of this focus by gastroscopy and histological examination confirmed gastric cancer (adenocarcinoma).
Patient 4 suffered from subacute generalised encephalomyelitis and axonal polyneuropathy. Relevant differential diagnoses were excluded. No paraneoplastic antibody was detected. FDG-PET/CT demonstrated a suspect retrosternal mass and suspect parasternal lymph nodes with increased tracer uptake. Surgical exploration showed thymoma and purely inflammatory lymphadenopathy.
Patients with confirmed non-PNS aetiologies and positive PET/CT (cases 5 and 6)
Patient 5 suffered from encephalitis of unknown origin. FDG-PET showed abnormal tracer uptake in the upper lobe of the right lung and in the right temporoparietal lobe of the brain, with corresponding lesions in the CT, leading to the diagnosis of lung cancer with cerebral metastases. Subsequent bronchoscopy and biopsy demonstrated the presence of adenocarcinoma of the lung and surgery of the brain confirmed metastasis. In retrospect, the symptoms of the patient were caused by the brain metastasis and were not related to PNS.
Patient 6 suffered from axonal, sensorimotor and distal-symmetric polyneuropathy. PET showed increased tracer uptake along of the thoracic aorta, which was interpreted as typical for a vasculitis. Biopsy of the sural nerve confirmed a microvasculitis and led to the diagnosis of idiopathic vasculitic polyneuropathy.
Patients with possible PNS and false-positive PET/CT (cases 7–10)
In four other cases (cases 7–10) abnormal FDG uptake was demonstrated, but the CT and subsequent investigations were negative for malignancy (Table 2). All of these were tested for paraneoplastic antibodies, and all of them were negative (Table 2). In these patients no definite diagnosis was reached. As all of them fulfilled the criteria for possible PNS and follow-up is less than 5 years (classic syndrome cases 8 and 9) or 2 years (non-classic syndrome cases 7 and 10), underlying PNS cannot be excluded.
Discussion
PET is more and more routinely used in oncology and has been shown to provide relevant information in the detection, staging and therapy monitoring of many tumours, such as bronchogenic carcinoma, colorectal cancer, lymphoma, gynaecological cancers, melanoma and many others [11–13]. The addition of CT to PET has been proven to improve specificity and sensitivity [21, 22, 24]. This is the first report of a series of patients who all have had 18F-FDG PET/CT as part of their diagnostic work-up for clinically suspected PNS.
The potential of FDG-PET was emphasized in some previous reports dealing with PNS [14–16, 25]. Rees et al. retrospectively investigated the role of FDG-PET in 43 unselected patients with suspected PNS. The authors reported 37% positive FDG-PET scans suggestive of malignancy [14]. Thus, based on their findings it was concluded that FDG-PET appears to play a pivotal role in detecting the offending malignancy in patients with PNS.
These results were later extended by Linke et al. who compared CT and FDG-PET for the detection of the primary tumour in 13 patients with antibody-positive paraneoplastic neurological symptoms [26]. They reported a sensitivity of 90% for FDG-PET and 30% for CT, and the combination of both methods showed a sensitivity of 100%. The authors concluded that the best diagnostic strategy in this group of patients is FDG-PET imaging complemented by CT. However, in our case series of unselected patients, combined PET/CT detected only four patients (8.7%, 4/46) with a histopathologically proven tumour responsible for PNS. This number is considerably lower than those reported previously.
Excluding the cases with vasculitic and metastatic aetiology, 50% of our positive findings were false positive (4/8). Of the false-positive patients, especially in cases 8 and 9, wrong classifications such as false positive are still possible, as tumours often escape imaging or histological detection due to the small size and special immunogenic situation in PNS [8, 27]. However, in the four false-positive patients the 18F-FDG accumulation was observed in maxilla, vagina, uterus or urinary bladder, which is known to occur often unspecifically in these organs [28, 29]. In these cases, as well as in the other patients with positive PET, it is most likely that a contrast-enhanced CT would have improved lesion localization and eased the right diagnosis [30].
Paraneoplastic antibodies are known to increase the likelihood of a positive PET [15–17]. In our study, only four patients tested positive for paraneoplastic antibodies. Two of them exhibited true-positive PET/CT results, consistent with a significantly higher detection rate of a tumour, which is in accordance with previous reports [15, 17]. Antibody-negative case 3 demonstrates the limitations of this approach. In this case only PET/CT helped in classifying the non-classic clinical syndrome as caused by a PNS. Furthermore, more stringent classification of PNS could also increase the probability of positive findings [2]. Five out of all 46 patients were classified as having definite PNS by consensus criteria [3]; three of these had an underlying tumour detectable by combined PET/CT.
Occasionally in patients with paraneoplastic antibodies no tumour is found even at autopsy [9, 27]. The time between onset of PNS and detection of an underlying malignancy in paraneoplastic antibody-positive patients has been reported to be as long as 8 years [9]. Consistent with this, in our study in 2 out of 4 patients with positive paraneoplastic antibodies no tumour was detected with PET/CT. It is well agreed that a negative PET or CT in a patient with a high clinical index of suspicion for PNS should not prevent ongoing follow-up. These cases have to be regarded as false negative, as the tumour may be missed if its size is too small for detection with FDG-PET or CT. Concerning the cases with a negative FDG-PET result it also has to be mentioned that PNS may also be caused by neuroendocrine tumours. Novel PET tracers like18F-DOPA, 11C-HTP and 68GA-DOTA-TOC are probably more sensitive in detecting the underlying neuroendocrine tumours in paraneoplastic syndromes [31, 32]. However, repeated FDG-PET is regarded as the best follow-up imaging technique currently available [15].
Up to now the use of co-registered PET/CT in the diagnosis of PNS has been reported only in one patient with positive anti-Yo antibodies and an underlying breast cancer [23]. In our four true-positive PET/CT, the underlying tumour was detected in three patients by the CT alone, whereas PET alone detected areas of increased glucose metabolism in all four cases, however, without accurate localisation of the lesions. By combining function and anatomy, hybrid PET/CT was able to establish the correct diagnosis in all four cases.
Our study has two major limitations. First, the number of patients is moderate, because of the low incidence of PNS. Second, the study is retrospective. For these reasons it is not possible to give an accurate indication of the sensitivity or specificity and accurate numbers of true- or false-negative results, which would require more consistent inclusion criteria, a larger number of patients and a prolonged follow-up [3].
However, in our study combined PET/CT detected the underlying disease for the clinical syndrome in six patients, corresponding to 13% (6/46). PNS was confirmed only in four patients, corresponding to 8.7% (4/46) of all investigated cases. Based on our results, we believe that the role of positron emission tomography in the detection of occult neoplasms in patients with PNS has been rather overestimated in the past. In clinical practice PNS is suspected far more often than proven and, therefore, combined PET/CT should be reserved for patients with a high probability of having PNS, and when conventional imaging fails to identify a tumour.
References
Darnell RB, Posner JB (2003) Paraneoplastic syndromes involving the nervous system. N Engl J Med 349:1543–1554
Voltz R (2002) Paraneoplastic neurological syndromes: an update on diagnosis, pathogenesis, and therapy. Lancet Neurol 1:294–305
Graus F, Delattre JY, Antoine JC, Dalmau J, Giometto B, Grisold W, Honnorat J, Smitt PS, Vedeler C, Verschuuren JJ, Vincent A, Voltz R (2004) Recommended diagnostic criteria for paraneoplastic neurological syndromes. J Neurol Neurosurg Psychiatry 75:1135–1140
Voltz R, Graus F (2004) Diagnosis and treatment of paraneoplastic neurological disorders. Onkologie 27:253–258
Graus F, Keime-Guibert F, Rene R, Benyahia B, Ribalta T, Ascaso C, Escaramis G, Delattre JY (2001) Anti-Hu-associated paraneoplastic encephalomyelitis: analysis of 200 patients. Brain 124:1138–1148
Keime-Guibert F, Graus F, Fleury A, Rene R, Honnorat J, Broet P, Delattre JY (2000) Treatment of paraneoplastic neurological syndromes with antineuronal antibodies (anti-Hu, anti-Yo) with a combination of immunoglobulins, cyclophosphamide, and methylprednisolone. J Neurol Neurosurg Psychiatry 68:479–482
Mason WP, Graus F, Lang B, Honnorat J, Delattre JY, Valldeoriola F, Antoine JC, Rosenblum MK, Rosenfeld MR, Newsom-Davis J, Posner JB, Dalmau J (1997) Small-cell lung cancer, paraneoplastic cerebellar degeneration and the Lambert–Eaton myasthenic syndrome. Brain 120:1279–1300
Dalmau J, Graus F, Rosenblum MK, Posner JB (1992) Anti-Hu-associated paraneoplastic encephalomyelitis/sensory neuronopathy. A clinical study of 71 patients. Medicine (Baltimore) 71:59–72
Lucchinetti CF, Kimmel DW, Lennon VA (1998) Paraneoplastic and oncologic profiles of patients seropositive for type 1 antineuronal nuclear autoantibodies. Neurology 50:652–657
Chartrand-Lefebvre C, Howarth N, Grenier P, Keime F, Orcel B, Beigelman C (1998) Association of small cell lung cancer and the anti-Hu paraneoplastic syndrome: radiographic and CT findings. AJR Am J Roentgenol 170:1513–1517
Basu S, Alavi A (2008) Unparalleled contribution of 18F-FDG PET to medicine over 3 decades. J Nucl Med 49:17N–21N 37N
Bomanji JB, Costa DC, Ell PJ (2001) Clinical role of positron emission tomography in oncology. Lancet Oncol 2:157–164
Hillner BE, Siegel BA, Liu D, Shields AF, Gareen IF, Hanna L, Stine SH, Coleman RE (2008) Impact of positron emission tomography/computed tomography and positron emission tomography (PET) alone on expected management of patients with cancer: initial results from the National Oncologic PET Registry. J Clin Oncol 26:2155–2161
Rees JH, Hain SF, Johnson MR, Hughes RA, Costa DC, Ell PJ, Keir G, Rudge P (2001) The role of [18F]fluoro-2-deoxyglucose-PET scanning in the diagnosis of paraneoplastic neurological disorders. Brain 124:2223–2231
Younes-Mhenni S, Janier MF, Cinotti L, Antoine JC, Tronc F, Cottin V, Ternamian PJ, Trouillas P, Honnorat J (2004) FDG-PET improves tumour detection in patients with paraneoplastic neurological syndromes. Brain 127:2331–2338
Antoine JC, Cinotti L, Tilikete C, Bouhour F, Camdessanche JP, Confavreux C, Vighetto A, Renault-Mannel V, Michel D, Honnorat J (2000) [18F]fluorodeoxyglucose positron emission tomography in the diagnosis of cancer in patients with paraneoplastic neurological syndrome and anti-Hu antibodies. Ann Neurol 48:105–108
Patel RR, Subramaniam RM, Mandrekar JN, Hammack JE, Lowe VJ, Jett JR (2008) Occult malignancy in patients with suspected paraneoplastic neurologic syndromes: value of positron emission tomography in diagnosis. Mayo Clin Proc 83:917–922
Berner U, Menzel C, Rinne D, Kriener S, Hamscho N, Dobert N, Diehl M, Kaufmann R, Grunwald F (2003) Paraneoplastic syndromes: detection of malignant tumors using [(18)F]FDG-PET. Q J Nucl Med 47:85–89
Antoch G, Stattaus J, Nemat AT, Marnitz S, Beyer T, Kuehl H, Bockisch A, Debatin JF, Freudenberg LS (2003) Non-small cell lung cancer: dual-modality PET/CT in preoperative staging. Radiology 229:526–533
Bar-Shalom R, Yefremov N, Guralnik L, Gaitini D, Frenkel A, Kuten A, Altman H, Keidar Z, Israel O (2003) Clinical performance of PET/CT in evaluation of cancer: additional value for diagnostic imaging and patient management. J Nucl Med 44:1200–1209
Townsend DW (2008) Dual-modality imaging: combining anatomy and function. J Nucl Med 49:938–955
Von Schulthess GK, Hany TF (2008) Imaging and PET-PET/CT imaging. J Radiol 89:438–447 quiz 448
Frings M, Antoch G, Knorn P, Freudenberg L, Bier U, Timmann D, Maschke M (2005) Strategies in detection of the primary tumour in anti-Yo associated paraneoplastic cerebellar degeneration. J Neurol 252:197–201
Miles KA (2008) PET-CT in oncology: making the most of CT. Cancer Imaging 8(Suppl):AS87–93
Hadjivassiliou M, Alder SJ, Van Beek EJ, Hanney MB, Lorenz E, Rao DG, Sharrack B, Tindale WB (2009) PET scan in clinically suspected paraneoplastic neurological syndromes: a 6-year prospective study in a regional neuroscience unit. Acta Neurol Scand 119:186–193
Linke R, Schroeder M, Helmberger T, Voltz R (2004) Antibody-positive paraneoplastic neurologic syndromes: value of CT and PET for tumor diagnosis. Neurology 63:282–286
Darnell RB, DeAngelis LM (1993) Regression of small-cell lung carcinoma in patients with paraneoplastic neuronal antibodies. Lancet 341:21–22
Cook GJ, Wegner EA, Fogelman I (2004) Pitfalls and artifacts in 18FDG PET and PET/CT oncologic imaging. Semin Nucl Med 34:122–133
Abouzied MM, Crawford ES, Nabi HA (2005) 18F-FDG imaging: pitfalls and artifacts. J Nucl Med Technol 33:145–155 quiz 162–143
Antoch G, Freudenberg LS, Beyer T, Bockisch A, Debatin JF (2004) To enhance or not to enhance? 18F-FDG and CT contrast agents in dual-modality 18F-FDG PET/CT. J Nucl Med 45(Suppl):156S–165S
Rufini V, Calcagni ML, Baum RP (2006) Imaging of neuroendocrine tumors. Semin Nucl Med 36:228–247
Goldsmith SJ (2009) Update on nuclear medicine imaging of neuroendocrine tumors. Future Oncol 5:75–84
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bannas, P., Weber, C., Derlin, T. et al. 18F-FDG-PET/CT in the diagnosis of paraneoplastic neurological syndromes: a retrospective analysis. Eur Radiol 20, 923–930 (2010). https://doi.org/10.1007/s00330-009-1606-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-009-1606-0