Abstract
To retrospectively evaluate the use of Micronester coils in port-catheter implantation with the fixed catheter tip method in comparison with other previously used coils. The cohort of this study was 143 consecutive patients with unresectable advanced liver cancer for whom a port-catheter system was percutaneously implanted. In the most recent 32 patients, Micronester coils were used for catheter tip fixation. Details of embolic agents for fixation, persistent blood flow beyond the distal end of the indwelling catheter, and complications were compared between cases without and with Micronester coils. In all, percutaneous port-catheter placement was successful. Mean number of coils used for fixation was 4.2 without Micronester coils vs. 2.5 with Micronester coils. N-butyl cyanoacrylate (NBCA)-Lipiodol was additionally used for catheter tip fixation in 85.6% of 111 procedures without Micronester coils and in 50% of 32 using Micronester coils. The gastroduodenal artery beyond the distal end was not detected at the final examination after any procedure. Catheter dislocation occurred in five and hepatic arterial obstruction or severe stenosis in eight. The number of coils used and necessity of NBCA-Lipiodol could be decreased with usage of Micronester coils without decreasing fixation ability compared to other coils.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Long-term hepatic arterial infusion chemotherapy (HAIC) via an implanted port-catheter system is well known as a last-resort treatment for unresectable advanced liver cancer [1, 2]. In the past, such catheter placement was done by surgical laparotomy under general anesthesia [3–6], making this an invasive procedure. However, recent advancements in interventional radiological techniques allow the less invasive implantation of port-catheter systems, that is, percutaneous implantation under local anesthesia [7–14].
Among various methods to achieve such implantation using interventional radiological techniques [7–19], implantation with the fixed catheter tip method [7, 8, 15–21] is advantageous from the standpoint of preventing catheter dislocation and hepatic arterial obstruction [7, 8, 16, 17, 21].
Most recently in our institution, after Micronester coils (Cook, Bloomington, IN) that provide immediate cross-sectional vessel occlusion when “nested” into a densely packed coil mass [22] became commercially available, we have used these coils in addition to other coils to fix the catheter tip to the gastroduodenal artery. This strategy was employed to simplify the procedure, to reduce the number of used coils, and to fix the catheter tip more strongly.
In the present study, we retrospectively evaluated the feasibility and usefulness of Micronester coils in port-catheter placement with the fixed catheter tip method in comparison with other previously used coils.
Subjects and methods
Patients
The cohort of this study was 143 consecutive patients (88 men and 55 women; mean age, 64.5 years; range, 23–84 years) with unresectable advanced liver cancer for whom a percutaneously implantable port-catheter system with the catheter tip fixed at the gastroduodenal artery was placed between February 2002 and February 2006. Sixty-four had primary liver cancer, and 79 had metastatic liver cancer that originated from colo-rectal (n = 53), breast (n = 9), gastric (n = 4), gallbladder (n = 3), pancreatic (n = 3), lung (n = 2), duodenal (n = 1), anal (n = 1), esophageal (n = 1), uteral (n = 1), and jejunal cancer (n = 1). All patients had diffuse or multiple (more than five) malignant lesions or a few huge malignant lesions in both the right and left lobes of the liver, making surgical resection impossible. Most of these patients had been administered systemic chemotherapy or other interventional treatments, only to develop intractable disease.
Approval was obtained from the institutional ethics committee before the start of this study. All procedures were performed after the patient provided written informed consent. The consent form included permission to use records, images, and data for research purposes.
Preparation prior to implantation of the port-catheter system
As the first step in port-catheter implantation, or the day before port-catheter implantation, arteries were embolized for two purposes with micro-coils (Diamond Coil, Boston Scientific, Watertown, MA; Trufill, Cordis, Miami, FL, or Tornado Coil, Cook, Bloomington, IN). One purpose was to prevent infusion of anticancer drugs into extra-hepatic adjacent organs during HAIC and the other was to allow perfusion of the entire tumor-bearing region from one catheter. Arteries embolized for these two purposes are described below.
For the first purpose, in all patients, all angiographically demonstrated branches that supplied blood to extra-hepatic adjacent organs (i.e., stomach, duodenum, and pancreas) and that arose from any segment in the hepatic artery beyond the projected side hole opening in the common hepatic artery were embolized with the exception of the gastroduodenal artery and the right gastroepiploic artery into which the tip of the indwelling catheter would be inserted. Examples of such arteries are the right gastric or the dorsal pancreatic arteries (Fig. 1a).
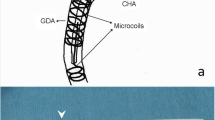
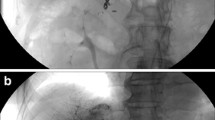
A 70-year-old woman with advanced hepatocellular carcinoma in whom port-catheter was placed with fixed catheter tip technique using Micronester coils. a Celiac arteriography shows sufficient embolization of the right gastric artery (arrow) and supraduodenal artery (arrowhead). b Roentgenogram shows that a micro-catheter (arrows) is being advanced over a micro-guide wire (arrowhead) already positioned with its tip in the right gastroepiploic artery. Note that the micro-catheter is coaxially advanced from a 5-French catheter (small arrowheads) inserted from the left subclavian artery. c Arteriography obtained from the indwelling catheter with a side hole, which was advanced to the planned position after exchanging previously inserted 5-French catheter and micro-catheter with an indwelling catheter, confirms that the side hole (arrow) opens to the common hepatic artery. All intrahepatic arterial branches are visualized. d Roentgenogram shows that the tip of the micro-catheter (arrow) and micro-guide wire is advanced through the second 5-French catheter inserted from the left femoral artery with its tip in the common hepatic artery (arrowhead). e Arteriography via the port obtained just after putting micro-coils in the gastroduodenal artery outside the indwelling catheter shows hepatofugal blood flow beyond the distal end of the indwelling catheter (thick arrows) at this stage. Note that three Micronester coils (arrows) and the NBCA-Lipiodol mixture were put in the gastroduodenal artery. f Arteriography via the port obtained just after implanting the port catheter system shows that the indwelling port-catheter system has been precisely implanted percutaneously and hepatofugal blood flow beyond the indwelling catheter tip has disappeared
With regard to the second purpose, in patients with two or more hepatic arteries, these arteries were converted into one hepatic artery. To accomplish this, all hepatic arteries were embolized with the exception of the hepatic artery from which the gastroduodenal artery diverged. Care was taken to embolize vessels distal to any extrahepatic branches of the aberrant hepatic artery and proximal to the first bifurcation of the intrahepatic segment of the aberrant hepatic artery. Hepatic arterial flow would then be redistributed to allow complete hepatic coverage via a single infusion catheter that was inserted into the hepatic artery from which the gastroduodenal artery arises. The most common example of such cases was a replaced hepatic artery arising from the superior mesenteric artery and the common hepatic artery arising from the celiac artery. In such a situation, the replaced right hepatic artery was embolized to convert two hepatic arteries into one common hepatic artery.
Implantation of port-catheter system
Percutaneous placement of long-term indwelling catheters was performed for frequent HAIC. Indwelling catheter was inserted from a branch of the left subclavian artery (n = 131), commonly the thoracic-acromial artery, or the femoral artery (n = 12) under local anesthesia. With catheter implantation, the tip of the catheter was positioned in the gastroduodenal artery, and embolic agents were placed on the outside of the catheter in the gastroduodenal artery to fix the catheter tip. A side hole was created to open into the common hepatic artery.
Specifically, implantation took place in the following order. Step 1: A cobra-shaped 5-French catheter (Clinical Supply, Gifu, Japan) was inserted from a branch of the left subclavian artery or the femoral artery and advanced to the common hepatic artery via the celiac artery. Step 2: A micro-catheter (Renegade-18; Boston Scientific) was inserted coaxially, after which a 0.016-inch micro guide-wire (GT wire; Terumo, Tokyo, Japan) was advanced far into the right gastroepiploic or pancreaticoduodenal artery (Fig. 1b). Step 3: Using the catheter exchange method, an indwelling catheter with a side hole was inserted so that the tip reached into the gastroduodenal artery approximately 5–10 cm from the bifurcation. The side hole was placed into the common hepatic artery at a site just proximal to the bifurcation where the gastroduodenal artery diverged (Fig. 1c). Step 4: A second 5-French catheter that was either hook- or cobra-shaped (Clinical Supply, Gifu, Japan) was inserted via the femoral artery, and its tip was positioned at the celiac or the common hepatic artery. Step 5: The gastroduodenal artery and the right gastroepiploic artery outside of the indwelling catheter were embolized with micro-coils through a micro-catheter inserted coaxially from the second 5-French catheter into the gastroduodenal artery outside of the indwelling catheter (Fig. 1d and e). The first inserted micro-coil was positioned at the gastroduodenal artery from the point beyond the distal end of the indwelling catheter (Fig. 1e). When stoppage of blood flow in the gastroduodenal artery was not sufficient by embolization with micro-coils alone, n-butyl cyanoacrylate (Histoacryl-Blue; Braun, Melsungen, Germany) (NBCA) mixed with Lipiodol (Laboratoire Guerbet, Roissy, France) was added as an embolic agent (Fig. 1e). Step 6: The proximal end of the indwelling catheter was connected to a port (Celsite Port; Toray Medical, Tokyo, Japan). The port was placed subcutaneously in a pocket created at the area from which the indwelling catheter had been inserted.
A catheter, which is made of polyurethane and coated with heparin, with a tapered tip (Anthron P-U catheter; Toray Medical, Tokyo, Japan) was used as the indwelling catheter. The outer diameter of the 50-cm proximal shaft was 5 French and that of the 20-cm distal shaft was 2.7 French, while the inner diameter of the proximal shaft was 0.035 inch and that of the distal shaft was 0.018 inch. A side hole was created by clipping with small scissors the distal shaft at a point 3–10 cm from the end of the indwelling catheter. The distance between the side hole and the distal end of the indwelling catheter was decided based on a celiac arteriogram.
In the first 111 patients in whom a catheter was implanted before Micronester coils became commercially available, the micro-coils used to occlude the gastroduodenal artery and the right gastroepiploic artery outside of the indwelling catheter included Diamond Coils (Boston Scientific, Watertown, MA), Trufill (Cordis, Miami, FL) or Tornado Coils (Cook, Bloomington, IN). Because Micronester coils had become available, in the last 32 cases, 3-mm, 4-mm or 6-mm 0.018-inch Micronester coils with an extended embolus length of 14 cm (Cook, Bloomington, IN) were utilized. In some cases, other micro-coils in addition to Micronester coils were used.
To prevent thrombosis of the port-catheter system, the catheter was flushed with saline and heparin after every chemotherapeutic drug administration.
Follow-up
All patients were examined by digital subtraction angiography obtained after contrast material was infused via the port to confirm patency of the hepatic artery or to identify any other dysfunction in the port-catheter system. This angiographic study was done just after (Fig. 1f) and within 2–10 days (mean±SD, 4.46 ± 2.06 days) of implantation, and every 1–4 months thereafter, while maintenance of the port-catheter system was required for HAIC therapy. Such intervals depended on the clinical circumstances of the patient.
Parameters investigated
The following were investigated: rate of success in port-catheter placement, details of embolic agents used to fix the indwelling catheter tip to the gastroduodenal artery, distance between the side hole and distal end of the indwelling catheter, existence of persistent blood flow beyond the tip of the indwelling catheter as shown on arteriography, and occurrence of complications closely related to indwelling catheter placement for HAIC.
For statistical analysis, quantitative variables were compared using the Student’s t-test. Qualitative variables were compared using the chi-square test or Fisher’s exact probability test. Differences were considered significant when the P value was less than 0.05.
Results
In all 143 cases, all branches of the hepatic arteries beyond the proper hepatic artery were well visualized on arteriography performed after contrast material was infused via the port just after placement, which showed technical success in implantation.
Overall, the mean distance of the side hole from the end hole of the indwelling catheter was 6.02 ± 1.05 cm (range, 3–10 cm). The mean number of coils used to fix the catheter tip in the gastroduodenal artery was 3.9 ± 1.8 (±SD; range, 1–12). NBCA-Lipiodol was additionally used to fix the catheter tip in 111 procedures. Mean time required for catheter implantation was 92.6 ± 30.3 min (±SD; range, 45–200 min). Difference in distance of the side hole from the end hole, number of coils, frequency of usage of NBCA-Lipiodol, and time required for catheter implantation between cases without and with Micronester coils to fix the catheter tip to the gastroduodenal artery are summarized in Table 1. For all of these parameters, the difference was statistically significant.
In 44 of 111 (39.6%) procedures without Micronester coils for catheter tip fixation, the gastroduodenal artery and the right gastroepiploic artery beyond the distal end of the indwelling catheter were detected on arteriography via the port just after implantation, while these arteries were detected in 15 of 32 (46.9%) procedures with Micronester coils (Table 1). The difference between the two methods was not statistically significant (p = 0.4639, chi-square test). However, when such arteriography was obtained 2–10 days after implantation, the gastroduodenal and/or right gastroepiploic artery was detected in only 2 of the 143 patients; the Micronester coil had been used in both cases. However, the gastroduodenal and/or right gastroepiploic artery were no longer detected on arteriography via the port 12 and 15 days after implantation, respectively.
Regarding difficulties that would prohibit continuation of HAIC if not corrected (Table 2), catheter dislocation occurred in 5 of 143 (3.5%) procedures. All five were patients in whom Micronester coils were not used, with one dislocation occurring within 10 days after implantation and four thereafter. Hepatic arterial obstruction or severe stenosis was seen in 8 of 143 (5.6%) procedures [occurring 1–25 months after implantation (mean, 11.4 months after implantation)]. All were cases without Micronester coils. No statistically significant difference was found in either the rate of dislocation or the frequency of hepatic arterial obstruction or stenosis between cases in which Micronester coils were and were not used (5/111 versus 0/32, p = 0.5872, Fisher’s exact probability test and 8/111 versus 0/32, p = 0.1991, Fisher’s exact probability test).
Other such complications were gastrointestinal mucosal lesions caused by distribution of anticancer drugs due to insufficient embolization of a branch of the pancreaticoduodenal arcade in one, large subcutaneous hematoma in one, necrosis of the skin covering the subcutaneous port in one, infection from the port-catheter system in two, and kinking of the catheter in the subcutaneous space in five (Table 2).
Discussion
According to a review of large published studies of percutaneous port-catheter placement for HAIC, indwelling catheter dislocation occurs at a relatively high rate, that is, from 5.6%–43.8% [9–14]. The rate of hepatic arterial obstruction is also relatively high, ranging from 0%–22.2% [9–14]. In these studies, the most common interventional radiological procedure involved positioning the indwelling catheter in the proper or common hepatic artery. In comparison, data from studies of implanted port-catheter systems with catheter tip fixation [7, 8, 16, 19–21] showed lower rates: 2.2% [8] to 4.4% [16] for catheter dislocation and 5.3% [19] to 6.8% [21] for hepatic arterial obstruction.
A problem with the catheter tip fixation method is that to obtain sufficient fixation of the catheter tip, a large number of coils is necessary. Additional use of an NBCA-Lipiodol mixture with the micro-coils for a stronger fixation has been recommended [8, 16, 21]. Yamagami et al. [21] reported in their study of port-catheter implantation with catheter tip fixation in 128 subjects that the mean number of micro-coils used for catheter tip fixation was 4.6 and that in 93% of these procedures the addition of NBCA-Lipiodol was necessary to obtain sufficient fixation. The present study showed similar results in those cases that did not receive Micronester coils for catheter tip fixation; the mean number of used micro-coils was 4.2 (range, 1–12), and in 86% NBCA-Lipiodol was required for sufficient fixation.
On the other hand, in the present study when Micronester coils were used for fixation, the mean number of micro-coils used was reduced to 2.5 (range, 1–4). NBCA-Lipiodol was needed in only 50%. The rate of catheter dislocation, hepatic arterial occlusion, and frequency of encountering other difficulties that would prohibit continuation of HAIC was equal in patients who did and did not receive Micronester coils. In addition, the period for the implantation procedure was significantly shorter with use of Micronester coils. These results show the superiority of the use of Micronester coils for fixation of the catheter tip compared to use of other coils alone.
Persistent hepatofugal blood flow in the gastroduodenal artery via the end hole, which might cause gastroduodenal mucosal lesions as a result of inflow of anticancer drugs into the gastroduodenal artery or the right gastroepiploic artery if continued [20], disappeared within 10 days in a majority (93.8%) of the 143 cases evaluated in the present study and in all cases finally (15 days after implantation at the latest). This result suggests equality of fixation ability with and without Micronester coils to fix the catheter tip.
Although this is a non-randomized, retrospective, and observational study, the number of subjects for such an evaluation is relatively large, and only consecutively treated patients were evaluated. Hence, results of this study might be valuable, reliable, and exemplify the potential advantage of using Micronester coils for this purpose. From the present results, it can be considered that usage of Micronester coils would simplify the procedure and reduce the cost of port-catheter implantation without decreasing the effectiveness of fixation.
References
Lorenz M, Muller HH (2000) Randomized, multicenter trial of fluorouracil plus leucovorin administered either via hepatic arterial or intravenous infusion versus fluorodeoxyuridine administered via hepatic arterial infusion in patients with nonresectable liver metastases from colorectal carcinoma. J Clin Oncol 18:243–254
Allen-Mersh TG, Earlam S, Fordy C, Abrams K, Houghton J (1994) Quality of life and survival with continuous hepatic-artery floxuridine infusion for colorectal liver metastasis. Lancet 344:1255–1260
Valeri A, Mini E, Tonelli P et al (1994) Intra-arterial hepatic chemotherapy with 5-Fluorouracil and 5-Methyltetrahydrofolate in the treatment of unresectable liver metastases from colorectal cancer. Anticancer Res 14:2215–2220
Rougier P, Laplanche A, Huguier M et al (1992) Hepatic arterial infusion of floxuridine in patients with liver metastases from colorectal carcinoma: long-term results of a prospective randomized trial. J Clin Oncol 10:1112–1118
Niederhuber JE, Ensminger WD (1983) Surgical considerations in the management of hepatic neoplasia. Semin Oncol 10:135–147
Huk I, Entscheff P, Prager M, Schulz F, Polterauer P, Funovics J (1990) Patency rate of implantable devices during long-term intraarterial chemotherapy. Angiology 41:936–941
Tanaka T, Arai Y, Inaba Y et al (2003) Radiologic placement of side-hole catheter with tip fixation for hepatic arterial infusion chemotherapy. J Vasc Interv Radiol 14:63–68
Yamagami T, Iida S, Kato T, Tanaka O, Hirota T, Nakamura T, Nishimura T (2002) Using n-butyl cyanoacrylate and the fixed-catheter-tip technique in percutaneous implantation of a port-catheter system in patients undergoing repeated hepatic arterial chemotherapy. AJR 179:1611–1617
Wacker FK, Boese-Landgraf J, Wagner A, Albrecht D, Wolf K-J, Fobbe F (1997) Minimally invasive catheter implantation for regional chemotherapy of the liver: a new percutaneous transsubclavian approach. Cardiovasc Intervent Radiol 20:128–132
Clouse ME, Ahmed R, Ryan RB, Oberfield RA, McCaffrey JA (1977) Complications of long term transbrachial hepatic arterial infusion chemotherapy. AJR 129:799–803
Oberfield RA, McCaffrey JA, Polio J, Clouse ME, Hamilton T (1979) Prolonged and continuous percutaneous intra-arterial hepatic infusion chemotherapy in advanced metastatic liver adenocarcinoma from colorectal primary. Cancer 44:414–423
Herrmann KA, Waggershauser T, Sittek H, Reiser MF (2000) Liver intraarterial chemotherapy: use of the femoral artery for percutaneous implantation of catheter-port systems. Radiology 215:294–299
Germer CT, Boese-Landgraf J, Albrecht D, Wagner A, Wolf KJ, Buhr HJ (1996) The totally implantable minimally invasive hepatic arterial catheter for intraarterial chemotherapy of unresectable liver metastasis in cases of dysfunction of arterial access devices. Chirung 67:458–462
Strecker E-PK, Boos IBL, Ostheim-Dzerowycz W, Heber R, Vetter SC (1997) Percutaneously implantable catheter-port system: preliminary technical results. Radiology 202:574–577
Arai Y, Inaba Y, Takeuchi Y (1997) Interventional techniques for hepatic arterial infusion chemotherapy. In: Castaneda-Zuniga WR (ed) Interventional radiology, 3rd edn. Williams & Wilkins, Baltimore, pp 192–205
Yamagami T, Nakamura T, Yamazaki S, Iida S, Kato T, Nishimura T (2002) Catheter-tip fixation of a percutaneously implanted port-catheter system to prevent dislocation. Eur Radiol 12:443–449
Seki H, Kimura M, Yoshimura N, Yamamoto S, Ozaki T, Sakai K (1999) Hepatic arterial infusion chemotherapy using percutaneous catheter placement with an implantable port: assessment of factors affecting patency of the hepatic artery. Clin Radiol 54:221–227
Irie T (2001) Intraarterial chemotherapy of liver metastases: implantation of a microcatheter-port system with use of modified catheter tip technique. J Vasc Interv Radiol 12:1215–1218
Takeuchi Y, Arai Y, Inaba Y et al (1996) A new percutaneous catheterization ‘side-holed catheter placement with fixation’ for a long term arterial chemotherapeutic infusion: Its effectiveness to prevent hepatic arterial occlusion. J Jpn Soc Angiography Interv Radiol 11:471–476
Yamagami T, Kato T, Iida S, Hirota T, Nishimura T (2005) Managemant of end hole in placement of port-catheter system for continuous hepatic arterial infusion chemotherapy using the fixed catheter tip method. AJR 184:1332–1339
Yamagami T, Kato T, Iida S, Tanaka O, Nishimura T (2004) Value of transcatheter arterial embolization with coils and n-butyl cyanoacrylate for long-term hepatic arterial infusion chemotherapy. Radiology 230:792–802
Osuga K, White RI Jr (2003) Micronester: a new pushable fibered microcoil for embolotherapy. Cardiovasc Interv Radiol 26:554–556
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamagami, T., Kato, T., Hirota, T. et al. Value of Micronester coils in port-catheter implantation for continuous hepatic arterial infusion chemotherapy with fixed catheter tip method. Eur Radiol 18, 152–157 (2008). https://doi.org/10.1007/s00330-007-0706-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-007-0706-y