Abstract.
Transient focal lesions in the splenium of the corpus callosum have been reported in epileptic patients receiving antiepileptic drugs. The characteristic imaging features included an oval high signal lesion on T2-weighted images in the central part of the splenium, no enhancement on post-contrast MR images, and complete reversibility without specific treatment. We report identical MR imaging findings in a depressive patient who had received antiepileptic drugs. In addition, diffusion-weighted MR imaging findings are described in our case, which is the first report on this unique lesion associated with antiepileptic drugs. Although this lesion has been assumed to be vasogenic edema in the previous reports, diffusion-weighted MR imaging showed markedly restricted diffusion of the lesion in the present case, suggesting that cytotoxic edema was the main pathophysiological abnormality.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Since the advent of MR imaging, focal lesions in the splenium of the corpus callosum have been described in patients with a wide variety of central nervous system disorders [1]. Among these lesions, transient lesions limited to the central area of the splenium have been reported in epileptic patients receiving antiepileptic drugs [2, 3]. The imaging findings are of clinical interest because such lesions share common MR features, including the presence of an oval lesion located in the central area of the splenium, no enhancement on post-contrast MR images, and complete reversibility without specific treatment [2, 3].
Although several hypotheses have been proposed concerning the possible causes of this unique lesion, little is known about its etiology. More recently, Polster et al. suggested that such a transient splenial lesion appears to be a non-specific end point of different disease processes leading to vasogenic edema [3]. We have observed identical MR imaging findings in a depressive patient in whom antiepileptic drugs had been used as mood stabilizers for the treatment of emotional lability. Diffusion-weighted MR imaging was performed in this patient. To the best of our knowledge, this is the first report of a patient with a splenial lesion associated with antiepileptic drugs evaluated by diffusion-weighted MR imaging.
Case report
A 27-year-old woman had been treated for depression for 2 years. She had been suffering from depressed mood and emotional lability. Social phobia and binge eating had also been identified as clinical problems. Although the patient often complained of anxiety and occasionally experienced drowsiness (possibly as a side effect of the antidepressant medications), no neurological findings were observed. Although electroencephalographic examinations revealed epileptiform findings, such as positive spikes at six per second in the temporal lobes, she had never experienced an epileptic seizure. She had been treated with antiepileptic drugs (valproate, carbamazepine, or clonazepam) combined with anxiolytics (etizolam or oxazolam) for depression for 6 months before the initial MR examination. During the 10 days preceding the initial MR examination, the patient had received a combination of clonazepam and oxazolam.
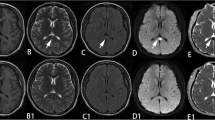
Magnetic resonance examination was performed to rule out pathological brain abnormalities such as brain tumor. Magnetic resonance imaging revealed a discrete oval lesion in the central part of the splenium of the corpus callosum. The lesion showed low signal intensity on a T1-weighted image (Fig. 1a) and high signal intensity on a T2-weighted image (Fig. 1b). The lesion was 15×10 mm in size. No definite enhancement was seen on a post-contrast image (Fig. 1c). No other abnormalities were observed on brain MR images.
The MR findings in a 27-year-old woman with depression. a Sagittal T1-weighted image shows a well-defined, focal, hypointense lesion (arrow) in the splenium of the corpus callosum. b Axial T2-weighted image shows an ovoid hyperintense lesion (arrow) in the splenium of the corpus callosum. c Contrast-enhanced T1-weighted image shows no enhancement in the splenial lesion (arrow). d Axial echo-planar diffusion-weighted image shows markedly increased signal intensity in the splenial lesion (arrow)
Single-shot spin-echo echo-planar diffusion-weighted MR imaging was performed with the following parameters: TR 9999 ms, TE 89 ms, no. of excitations 1, acquisition matrix 128×96, and a field of view of 34×17 cm. Section thickness was 6 mm with intersection gap of 2 mm. Diffusion gradients were sequentially activated in each of the three principal anatomic axes in order to obtain diffusion-weighted images sensitive to diffusion in the x, y, and z planes. Gradient strength corresponding to a b value of 1000 s/mm2 was used. Trace diffusion-weighted images were created and used for image interpretation. Average apparent diffusion coefficient (ADC) was calculated as ADC=1/3(ADCx+ADCy+ADCz). A diffusion-weighted image showed markedly increased signal intensity in the splenial lesion (Fig. 1d). The ADC values (mean±SD) were 0.33±0.04×10–3 mm2/s within the lesion in the splenium, 0.76±0.02×10–3 mm2/s in normal areas in the lateral splenium, and 0.74±0.06×10–3 mm2/s in normal areas of the white matter.
No signs or symptoms suggestive of callosal disconnection were detected by neurological examination. Based on the MR imaging findings of the isolated splenial lesion, the differential diagnoses included multiple sclerosis, Marchiafava-Bignami disease, a brain tumor such as glioma or lymphoma, and antiepileptic drug toxicity. However, since the patient was not a heavy drinker and serum levels of vitamins B1, B2, and B12 were within normal limits, Marchiafava-Bignami disease was considered unlikely. No oligoclonal band was observed in the cerebrospinal fluid; however, the patient was subsequently followed without specific treatment because multiple sclerosis, brain tumor, and antiepileptic drug toxicity could not be ruled out based on this finding alone.
Three months later, follow-up MR imaging for brain and spine was performed. The MR images showed complete disappearance of the splenial lesion (Fig. 2), and showed no abnormal lesions in brain and spine. The ADC values were 0.82±0.01×10–3 mm2/s in the central part of the splenium and 0.77±0.01×10–3 mm2/s in normal areas in the lateral splenium. These MR findings did not fulfill diagnostic criteria recommended by the international panel on the diagnosis of multiple sclerosis [4]. Brain tumor was also considered unlikely.
Between the initial and follow-up MR examinations, antiepileptic drugs were discontinued due to drowsiness, and an antidepressant (fluvoxamine) was administered to the patient. The patient was subsequently managed without the use of antiepileptic drugs, and no neurological abnormalities were observed during this period. A second series of follow-up MR images were obtained 1 year later and showed no abnormalities in the brain. Based on such clinical findings and serial MR imaging findings, the splenial lesion in the current case was probably due to antiepileptic drugs.
Discussion
Transient focal lesions limited to the splenium of the corpus callosum have been observed in patients with epilepsy [2, 3]. These patients had been treated with antiepileptic drugs and showed no clinical signs or symptoms related to the splenial lesion when the lesion was found on MR images. The characteristic imaging features of such lesions are the presence of an oval lesion in the central part of the splenium, no enhancement on post-contrast MR images, and complete reversibility without specific treatment. These MR imaging findings are identical to those observed in our patient.
It is noted that splenial lesions have been observed not only in epileptic patients but also in depressive patients such as in the present case. The effectiveness of mood-stabilizing drugs, including lithium, antiepileptic drugs, and antipsychotics, in the management of depressive patients, has been established [5]. Antiepileptic drugs, such as carbamazepine and valproate, have also been used and are considered to be good alternatives, especially in patients with emotional lability. Monotherapy with a mood stabilizer is the first option, but the limited efficacy of monotherapy in many patients may necessitate combined treatment with two or even three mood stabilizers or with a mood stabilizer and other psychoactive agents. In our patient, combination therapy with anxiolytics and antiepileptic drugs had been continued for 6 months before the initial MR examination. Between the initial and first follow-up MR examinations, the antiepileptic drug was withdrawn and follow-up MR examination showed complete resolution of the splenial lesion. Based on the patient's clinical course and MR findings, we assume that the administration of the antiepileptic drug was causally related to the splenial lesion.
The initial report describing such lesions suggested that focal splenial lesions are associated with seizure activity and represent an area of transient edema due to a transcallosal seizure spreading to secondary generalized seizures [6]; however, our patient had been suffering not from epilepsy but from depression, although some abnormal electroencephalographic findings were observed in the temporal lobes. Polster et al. also reported that none of their patients with epilepsy experienced secondary generalized seizures in the relevant period before imaging [3]. In addition, the role of the corpus callosum in secondary generalized seizures with an epileptic focus in the temporal lobe is controversial [7]. The common clinical feature shared by our case and the other cases reported in the literature is that all received antiepileptic drugs before MR examination. In addition, some of the patients described in the literature showed complete resolution of the splenial lesion following the withdrawal or rapid reduction of the antiepileptic drugs [2, 3], as was also observed in our patient.
The pathophysiology of this splenial lesion remains unknown. Polster et al. suggested that it is a non-specific end point of different disease processes leading to vasogenic edema [3]. They assumed that this edema was due to the influence of antiepileptic drugs on fluid-balance systems, namely, arginine–vasopressin, although there have been no definite data explaining a special vulnerability of the splenium towards focal edema. On the contrary, in our patient, the splenial lesion showed markedly restricted diffusion in diffusion MR imaging, which appears not to be consistent with the changes in diffusion seen in vasogenic edema. In vasogenic edema, it is known that the ADC is increased compared with that in normal brain parenchyma [8]. Hypertensive encephalopathy is a well-known example of vasogenic edema observed by diffusion-weighted MR imaging [9]. On the other hand, reduced or restricted diffusion may be seen in various brain diseases, including acute infarction [10], diffuse axonal injury [11], and status epilepticus [12]. Cytotoxic edema has been suggested to be the probable cause of restricted diffusion in such conditions; therefore, we hypothesize that cytotoxic edema was the major pathophysiological abnormality in our patient, although an element of vasogenic edema cannot be completely ruled out.
However, it remains unknown why the splenial lesion of our case exhibited restricted diffusion. Several hypotheses have been proposed to explain decreased diffusion of brain lesions in various central nervous system diseases. In central pontine myelinolysis, relative intracellular hypotonicity was hypothesized as a key event in decreased diffusion of pontine lesions [13]. In carmofur-induced leukoencephalopathy, intramyelinic edema or myelin vacuolation observed in animal experiments may be explainable for decreased diffusion [14]. Despite hypotheses in such diseases, we have not found evident data to explain decreased diffusion in antiepileptic toxicity.
Based on MR imaging findings alone, the differential diagnosis may include multiple sclerosis, infarction, a brain tumor (particularly glioma or lymphoma), post-infectious encephalopathy, diffuse axonal injury, acute mountain sickness, and Marchiafava-Bignami disease, although the latter four diseases may easily be ruled out by observing the patient's clinical course. The fact that complete resolution was observed in the follow-up MR examination allowed us to rule out a brain tumor. Infarction restricted to the corpus callosum is rare, especially in young adults without any risk factors for stroke. In addition, focal infarction in the corpus callosum is usually confined to one side because of the bilateral arterial supply [1]. Multiple sclerosis may be difficult to rule out in the clinical setting, and clinical follow-up and follow-up MR imaging are necessary [4]. It has been reported that ADC values in acute and chronic multiple sclerosis lesions are commonly elevated due to axonal demyelination, an increase in water, and inflammatory cell swelling [15]. The markedly reduced ADC values in our patient do not suggest the pathophysiological changes in a multiple sclerosis plaque.
Although the MR findings seen in splenial lesions associated with antiepileptic drugs are unique, transient focal splenial lesions may not be specific to antiepileptic drug toxicity. Ogura et al. reported a reversible splenial lesion in a girl with mild encephalopathy caused by O-157 Escherichia coli [16]. They speculated that verotoxin from O-157 E. coli may induce localized microvascular angiopathy and causes diffuse axonal damage in the splenium. Several other diseases or conditions may show a predilection for the central part of the splenium; however, minor splenial lesions may be asymptomatic and MR examinations may not be performed frequently in the clinical setting. More clinical data are required to test this hypothesis.
Conclusion
In conclusion, we have presented the MR findings of a transient focal splenial lesion associated with the administration of antiepileptic agents in a depressive patient. The lesion showed markedly restricted diffusion in diffusion MR imaging, indicating cytotoxic edema. Unlike acute infarction with similar restricted diffusion abnormality, the splenial lesion caused by antiepileptic drugs can be reversible. Although it is not clear why transient splenial lesions are induced by the use of antiepileptic drugs, the complete reversibility of such lesions without specific treatment suggests that unnecessary invasive examinations and therapeutic intervention should be avoided.
References
Friese SA, Bitzer M, Freudenstein D, Voigt K, Küker W (2000) Classification of acquired lesions of the corpus callosum with MRI. Neuroradiology 42:795–802
Kim SS, Chang KH, Kim ST et al. (1999) Focal lesion in the splenium of the corpus callosum in epileptic patients: antiepileptic drug toxicity? Am J Neuroradiol 20:125–129
Polster T, Hoppe M, Ebner A (2001) Transient lesion in the splenium of the corpus callosum: three further cases in epileptic patients and a pathophysiological hypothesis. J Neurol Neurosurg Psychiatry 70:459–463
McDonald WI, Compston A, Edan G et al. (2001) Recommended diagnostic criteria for multiple sclerosis: guidelines from the international panel on the diagnosis of multiple sclerosis. Ann Neurol 50:121–127
Shelton RC (1999) Mood-stabilizing drugs in depression. J Clin Psychiatry 60:37–40
Chason DP, Fleckenstein JL, Ginsburg MI, Mendelsohn DB, Mathews D (1996) Transient splenial edema in epilepsy: MR imaging evaluation. Annual Meeting of the American Society of Neuroradiology, 21–27 June, Seattle
Gotman J (1991) Seizure spread. In: Luders H (ed) Epilepsy surgery. Raven, New York, pp 349–354
Schaefer PW (2000) Diffusion-weighted imaging as a problem-solving tool in the evaluation of patients with acute strokelike syndromes. Top Magn Reson Imaging 11:300–309
Schwarz RB, Mulkern RV, Gudbjartsson H, Jolesz F (1998) Diffusion-weighted MR imaging in hypertensive encephalopathy: clues to pathogenesis. Am J Neuroradiol 19:859–862
Warach S, Gaa J, Siewert B, Wielopolski P, Edelman RR (1995) Acute human stroke studied by whole brain echo planar diffusion-weighted magnetic resonance imaging. Ann Neurol 37:231–241
Liu AY, Maldjian JA, Bagley LJ, Sinson GP, Grossman RI (1999) Traumatic brain injury: diffusion-weighted MR imaging findings. Am J Neuroradiol 20:1636–1641
Lansberg MG, O'Brien MW, Norbash AM, Moseley ME, Morrell M, Albers GW (1999) MRI abnormalities associated with partial status epilepticus. Neurology 52:1021–1027
Cramer SC, Stegbauer KC, Schneider A, Mukai J, Maravilla KR (2001) Decreased diffusion in central pontine myelinolysis. Am J Neuroradiol 22:1476–1479
Fujikawa A, Tsuchiya K, Katase S, Kurosaki Y, Hachiya J (2001) Diffusion-weighted MR imaging of carmofur-induced leukoencephalopathy. Eur Radiol 11:2602–2606
Tievsky AL, Ptak T, Farkas J (1999) Investigation of apparent diffusion coefficient and diffusion tensor anisotropy in acute and chronic multiple sclerosis lesions. Am J Neuroradiol 20:1491–1499
Ogura H, Takaoka M, Kishi M et al. (1998) Reversible MR findings of hemolytic uremic syndrome with mild encephalopathy. Am J Neuroradiol 19:1144–1145
Acknowledgement.
The authors thank Y. Miki for his invaluable assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maeda, M., Shiroyama, T., Tsukahara, H. et al. Transient splenial lesion of the corpus callosum associated with antiepileptic drugs: evaluation by diffusion-weighted MR imaging. Eur Radiol 13, 1902–1906 (2003). https://doi.org/10.1007/s00330-002-1679-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-002-1679-5